Abstract
Introduction
Antigen-presenting cells of the primary and secondary follicles are termed follicular dendritic cells. These cells can be found in B cell follicle germinal centers, which can be found in lymph nodes and extranodal sites as acquired lymphoid tissue or as a component of the structured constitutive lymphoid tissue. 1 The term “follicular dendritic cell sarcoma” (FDCS) refers to a tumor that originates from follicular dendritic cells. Since follicular dendritic cells can be found in both nodal and extranodal regions, FDCS can develop there as well. These tumors are uncommon in lymph nodes and even less common in extranodal locations. Due to its rarity and diverse architecture, FDCS at an extranodal site is challenging to identify. In addition to lymph nodes, FDCS has been documented in the tonsils, palate, throat, spleen, mediastinum, liver, thyroid, lung, thigh soft tissue, and gastrointestinal tract. 2 With only four cases reported in the literature,3–6 primary FDCS originating in the stomach poses a great diagnostic challenge with broad differential diagnoses. In this study, we report the fifth case of an incidentally primary gastric FDCS presenting as a subserosa mass and aim to review clinical characteristics, pathologic features, emphasize differential diagnosis, and discuss therapeutic modalities and prognosis of this rare entity.
Case presentation
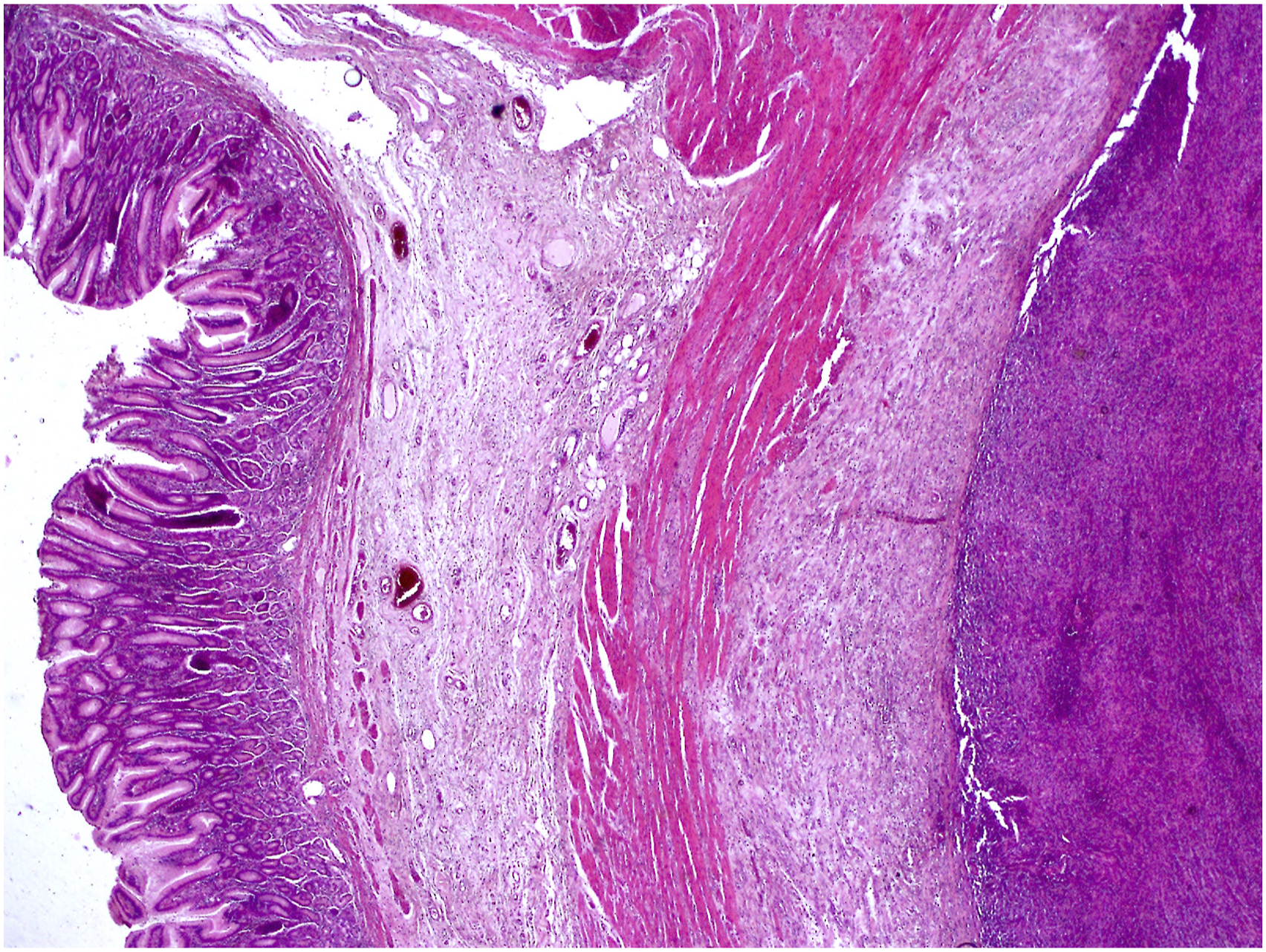
Our patient, a 36-year-old woman with no remarkable past medical history, originally presented for the management of an axillary furuncle. On investigation, she reported mild dyspnea and denied any abdominal pain. Except for a moderate level of anemia, her physical examination and laboratory testing were unremarkable; the level of blood carcinoembryonic antigen was normal. A subsequent abdominal ultrasonography was performed, revealing a 4 cm well-defined gastric mass associated to a moderate peritoneal effusion. The patient had been missing for 5 months. She presented afterwards for abdominal pain. A thoraco-abdomino-pelvic computed tomography (CT) scan was performed, showing an increase in tumor size to 9 cm with the presence of deep and retro-pancreatic enlarged lymph nodes. The mass was biopsied for histological examination. The biopsy showed a tumor composed of large, oval to spindle-shaped cells organized in sheets and interlacing fascicles. Numerous small lymphocytes frequently coexisted with the tumor cells. Immunohistochemically, tumor cells expressed CD21 and CD23. They were negative for AE1/AE3, CD34, c-kit, DOG-1, CD20, CD30, CD10, bcl-6, CD68, S-100, D2-40, CD45, or EBER. The diagnosis of FDCS was retained. A 4/5 subtotal gastrectomy with extended para-aortic and retro-pancreatic lymphadenectomy was performed. Grossly, the specimen contained a 12 × 4.5 cm well-circumscribed fleshy mass with a pale-tan surface on sectioning arising in the subserosa of the stomach (Figure 1). Numerous lymph nodes along the major curve were metastatically enlarged. Microscopic examination revealed a multi-lobated mass centered in subserosa of the fundic wall (Figure 2). The lesion had the same features as in the biopsy specimen. The tumor exhibited spindle to epithelioid cells arranged in fascicles, storiform and whorling pattern. Tumor cells had plump eosinophilic cytoplasm with ill-defined cell borders generating a patternless diffuse growth, despite some places having short fascicles of spindle-shaped cells that produced a vague storiform pattern (Figure 3). They had hyperchromatic to vesicular nuclei and occasional prominent nucleoli. A few multinucleated tumor cells were also observed. Focal nuclear pleomorphism were seen (Figure 4). In background, sprinkling of small reactive lymphocytes was noted and in certain spots, a perivascular cuff of lymphocytes was also detected (Figure 5). Mitotic figures were scarce, found up in 3 per 10 high power fields. No necrosis was found. Due to the rarity of the tumor, its peculiar location, and the need to rule out numerous entities before considering FDCS diagnosis, an extensive panel of immunohistochemical stains was carried out. Tumor cells were positive for CD21 and CD23, while they were negative for C-kit, DOG1, Desmin, SMA, AE1/AE3, S100, CD34, CD30, HMB45, and EBER (Figure 6). A diagnosis of an extra-nodal gastric FDCS with metastasis to regional lymph nodes was rendered. No post-operative chemoradiation was administred. The postoperative course was uneventful, and the patient was discharged from the hospital after 15 days. A CT scan is scheduled in 4-weeks to detect distant metastases and the patient will be followed by complete clinical examination. After 5 months of follow-up, the patient is doing well and no recurrence was diagnosed. A 4/5 subtotal gastrectomy specimen showing the 12 × 4.5 cm fleshy mass with a pale-tan surface arising in the wall of the fundus. Hematoxylin-eosin, original magnification ×200. Low-power view of gastric follicular dendritic cell sarcoma with a well-defined subserosal tumor. The tumor is composed of plump spindle cells arranged in fascicular, storiform, and whorling patterns with lymphocytic infiltrate in the background (x200). Tumor cells had hyperchromatic to vesicular nuclei and occasional prominent nucleoli. A few multinucleated tumor cells were also observed (x400). FDCS with sprinkling of lymphocytes in the tumor background with a perivascular cuff of lymphocytes (x400). Immunohistochemical staining showing cytoplasmic and membranous positivity for CD21 (a) and CD23 (b). C-kit (c) and CD34 (d) are negative.




The work has been reported in line with the SCARE 2020 criteria. 7
Discussion
Monda et al. 8 groundbreaking’s research from 1986 discovered the presence of a FDCS. Four patients with malignant tumors involving cervical lymph nodes were described by these authors. Gastrointestinal FDCS is exceedingly rare, accounting for 5.2% among 343 cases. 9 The prevalence of classic gastrointestinal FDCS is higher in middle-aged people without a gender bias (mean age = 45.3 years, M:F = 1:1). Both the upper and lower gastrointestinal tracts are equally affected. FDCS typically manifests as larger masses that measure 7.8 cm on average. 10 The tumor, in our case, was large measuring 12 × 4.5 cm.
The symptoms of FDCS in the gastrointestinal tract include weight loss and abdominal pain. 3 In the stomach, clinical symptoms consisted in asthenia, weight loss, 3 melena, dizziness 11 and epigastric pain. 5 Our patient had no abdominal signs, and the mass was discovered incidentally.
Microscopically, FDCS is characterized by a fascicular or storiform proliferation of spindle cells.11,12 Most tumors are made up of plump spindle cells that have been organized in fascicular, storiform, and whorling patterns with lymphocytic flecks in the background. Variations in morphological characteristics include necrotic foci, areas of significant atypia, numerous mitoses, an extensive lymphoid infiltrate, nested and sheet-like tumor cell organization, and epithelioid appearance. 2 FDCS stain diffusely with FDC markers: CD21, CD35, and CD23. CD21 and CD35 are the most commonly used markers. One or more FDC markers can, however, occasionally be lost in the gastrointestinal FDCS (24%, 8/33 cases). 10 Both morphological characteristics and immunohistochemistry are crucial in the diagnosis of FDCS since the histomorphology and immunohistochemistry of FDCS might be varied. 10 In the present case, CD21 and CD23 were expressed by tumor cells. Clusterin and podoplanin (D2–40) are other markers which also have high sensitivity for FDCS. 3 S-100 protein, EMA, CD68, SMA, desmoplakin, and CD45RB are all expressed in varying degrees in FDCSs. CD1a, CD20, Cytokeratin, Desmin, CD31, CD34, c-kit, and HMB-45 are all negative. 3
Morphological pattern of FDCS may overlap with a broad range of potential differential diagnoses. A distinction from better known mesenchymal sarcomas, such as the gastro-intestinal stromal tumours (GIST) must be considered. Typically, GIST tumor cells exhibit less eosinophilic cytoplasm than spindle cell tumor cells, which exhibit more focused differentiation, such as in smooth muscle and neurogenic tumors. However, GISTs can exhibit a variety of histologic characteristics, such as epithelioid and myxoid forms, making it sometimes difficult to distinguish between the two without the use of immunohistochemical and ultrastructural examinations. C-kit and CD34 are a helpful diagnostic marker of GIST. 13 We could rule out in the present case this diagnostic possibility as tumor cells were negative for c-kit, DOG-1 and CD34.
One category of inflammatory myofibroblastic tumors Eptein-Barr Virus (EBV) positive needs to be noted in terms of the differential diagnosis for EBV + inflammatory FDCS. FDC markers are negative, but EBV and SMA are positive. 10
Another reticular dendritic cell-derived tumor is interdigitating dendritic cell sarcoma, which share some histologic, immunophenotypic, and ultrastructural similarities to FDCS. Interdigitating dendritic cell sarcoma is less common than FDCS and it can also manifest as an extranodal mass, including one in the gastrointestinal tract. The presence of FDC markers and the absence of CD45 expression ruled out the diagnosis of interdigitating dendritic cell sarcoma, despite the immunophenotypic profiles having some overlap.
The etiology or predisposing factors for the vast majority of reported cases of FDCS are unknown. The single known risk factor for FDCS, which only occurs in a small percentage of cases, is hyaline-vascular Castleman’s illness. Epstein-Barr virus has been found in certain cases, although it doesn’t seem to be a major factor in the typical form of FDCS. 3
In the Perez et al. 14 analysis, 36% of participants experienced local recurrences and 28% had metastatic tumors. Lymph nodes, lungs, liver, and peritoneum are the most common sites for metastatic lesions to be described. A mortality rate of 14% was present at the time of the most recent follow-up. The 1997 study by Chan et al. 15 describes almost the same numbers. The intraabdominal locations are linked to a particularly aggressive course, according to both studies.14,15 Other studies claimed that the percentages of 2- and 5-year disease-free survival were only 57% and 32%, respectively. 13 An aggressive clinical behavior is associated with a high mitotic count (> 5 mitoses per 10 HPF), coagulative necrosis, nuclear pleomorphism, and significant cellular atypia. 15 Patients with extranodal FDCS have a worse prognosis than those with nodal FDCS. Additionally, compared to other sites, the intraabdominal tumors had a larger average size. It’s probable that the greater recurrence is caused less by a true difference in FDCS biology and more by the delay in diagnosis, which led to a larger lesion, similar to our case. 5
In the four reported cases of gastric FDCS involvement, the patients did not report recurrence of the disease after a follow-up of 1.15 and 10 months.3,11 In our case, lymph node metastases were noticed at moment of diagnosis.
The cornerstone of FDCS treatment is complete surgical excision. This is typically simple to accomplish in tumors involving the cervical or axillary lymph nodes. Because of their size or involvement with nearby structures, intraabdominal or mediastinal tumors are more challenging to remove. Due to the retrospective nature of published research and the small size of series, the role of adjuvant therapy, such as chemotherapy and/or radiotherapy, is yet unknown. 3 The effects of neoadjuvant and adjuvant chemotherapy have varied, with some studies demonstrating a benefit and others demonstrating no further benefit. 9 Our patient underwent a subtotal gastrectomy without adjuvant chemotherapy. She was doing well postoperatively. However, because she has already shown regional lymph node metastases, her clinical course is unknown. A close follow-up is imperative.
Conclusion
We describe the fourth occurrence of a stomach-based FDCS with metastatic lymph nodes at time of diagnosis. Particularly when there is no antibody found for epithelial and mesenchymal markers, one should consider this entity. Due to its rarity, a FDCS is rarely included in the differential diagnosis of gastrointestinal spindle cell tumors. Treatment for FDCS typically involves complete surgical excision.
Footnotes
Author contributions
All the authors read and approved the final version of the manuscript. Farah Sassi (MD): conception, acquisition of data, literature research and preparing the manuscript Ghada Sahraoui (MD): acquisition of clinical data, preparing and revising the manuscript Lamia Charfi (MD): conception, literature research supervision and revising the manuscript Leila Achouri (MD): clinical data and revising the manuscript Karima Mrad (MD): manuscript editing and revising the manuscript critically Raoudha Doghri (MD): final approval of the version to be published
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
(include full name of committee approving the research and if available mention reference number of that approval) *Salah Azaiez Institute* does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
Guarantor
Sassi Farah
Data availability
All data generated or analysed during this study are included in this published article (and its supplementary information files).
