Abstract
Background
Four oral anti-tuberculosis drugs are conceived to be the most effective ones to eradicate Mycobacterium tuberculosis bacteria and to obviate the resistant organisms. However, the patients’ adherence and medication discrepancies are obstacles to achieving the goal. This study aimed to define the anti-tuberculosis drugs used in the hospitals and to detect the discrepancies in the continuity of the tuberculosis treatment.
Design and methods
This retrospective cross-sectional study was based on medical records of adult patients, and was conducted in two district tertiary care hospitals. Only 35 out of 136 patient records from Hospital A and 33 out of 85 records from Hospital B met the inclusion criteria.
Results
The most common systemic anti-infective drugs in the study were ceftriaxone (51.80 DDD/100 patient-days) used in Hospital A and isoniazid (59.53 DDD/100 patient-days) used in Hospital B. The number of rifampicin prescriptions was less than that of isoniazid. Each patient received an average of two DDD/100 patient-days, which is an under dosage for an effective treatment.
Conclusion
This study showed a medication discrepancy of tuberculosis therapy. Tuberculosis patients’ medical histories are not under the full attention of treating physicians wherever they are admitted. Thus, medication reconciliation is needed to accomplish the goal of a Tuberculosis-free world in 2050.
Keywords
Significance for public health
Among other infectious diseases, tuberculosis causes not only more death in all countries and age groups, but also threatens global health with multidrugresistant TB. Tuberculosis is curable but may have uncertain diagnosis and needs continuation treatment for a minimum of six months. Recently, there is some investigation of the patient pathway for tuberculosis care-seeking; this study showed that even though the patient goes to public health services, discontinuation of therapy happens. The unfulfilled medication needs of tuberculosis patients, should increase awareness about TB resistance hazards and encourage healthcare professionals, healthcare management, and government, particularly in Indonesia, to increase microbiology capacity and develop an information system to connect patient data in the primary care and secondary care.
Introduction
Tuberculosis is a global disease burden. The target of the Sustainable Development Goals (SDGs) for tuberculosis (TB) is to achieve 80% reduction in TB incidence by 2030 with the current success of curtailing the global TB incidence barely at 1.5% annually. 1 Thirty-eight percent of the TB global deaths were observed in the South Asian region. 2 The incidence of TB cases and the rifampicin-resistant (RR) or multidrug-resistant (MDR) TB has remained stable over time. 3 In 2018, Indonesia had 8% of tuberculosis cases worldwide, the third highest ranked country, after India (27%) and China (9%). Indonesia had a global warning because there was a 70% rise in new cases added from 2015-2018, 28% of it in period 2017-2018. In 2018, there was 1.020.000 incident. The incidence rate was 391 per 100.000 population. Indonesia's situation was distressing not only because of new tuberculosis cases but also there was a 10% gap between the number of new cases reported and the estimated incident cases, due to under-reporting of detected cases or under-diagnosis. Sometimes under-diagnosis occurs because of failure to test for TB or diagnostic tests that are not sufficiently sensitive or specific to ensure accurate identification of tuberculosis cases when people go to health facilities. 4 This situation arises not only because of the nonspecific TB signs and symptoms that were undiagnosed, 5 delayed, 6 or transmitted, but also because of unfinished,7-9 unrecorded, and abruptly abandoned treatments. All these may occur when the patients are seeking the care of other healthcare facilities. 4 Based on the patient pathway analysis (PPA) studies in 13 countries, less than 50% of the TB patients were permanently cured; the remainder had a possibility to relapse. 1 The PPA methodology was developed to better understand the alignment between care-seeking patients and TB service availability. Indonesia Ministry of Health targeted tuberculosis (TB) elimination by 2035 and TB free by the year 2050. To achieve it, the regulation stated that the Central Government, Local Government, and the community are responsible for organizing TB control. 10 Besides public hospitals and health centers (puskesmas), there are a lot of private hospitals and private medical practices. The government supports the government's health facilities financially; whereas the private hospital's income is from the patients’ out-ofpocket (OOP) payments.11,12 Among TB patients, in 2015, notified cases were primarily being treated in the public sector, which accounts for 91% of notified cases, whereas the private sector only notified 9% of cases. The low notification rate in the private sector leads to an underestimation of TB treatment in private facilities. A PPA aimed to identify the patient care-seeking direction and the availability of TB diagnostic and treatment services; to estimate the missing tuberculosis cases. 13 General practitioners and medical specialists who work at the government's health facilities had better knowledge of TB control regulation program and management than who work in the private sector.14,15 The levels of the public health system are classified into several types. These types are rural health care centers (primary care), district hospitals (secondary care), and referral hospitals (tertiary care). The PPA coverage in each of the levels of the public health system includes the availability of the TB screenings, diagnoses, and treatments at various levels to minimize delays in the experiences of care-seeking or treatment initiations, inappropriate care access, or missed follow- ups during one or more phases of medication.16-18
There is a drug-related problem related to the transfer of patients within primary, secondary and tertiary care. One of the most common problems within health care institutions is that TB patients often miss follow-ups or skip treatments. These missed follow-ups may impede the successful treatments (the patients’ total recovery process from the disease), particularly if the reason for the patient's admittance to the hospital is not triggered by respiratory illness.19,20
The harmonization of the drug treatment is challenging when the data are not synchronized at all levels of care institutions and units. 21 Medication reconciliation is the process of identifying the most accurate list of a patient's current medicines, including their names, the dosage, the frequencies and the routes of administration; any medication discrepancies will increase medication error risk. 22 Thus, a complete list of medications that is accurately communicated particularly to TB patients is needed. 23 Accordingly, this study is aimed to define the anti-tuberculosis drug use for inpatients and to detect the drug use gap in the rural health care centers (primary care) and district hospitals (secondary care).
Design and methods
This research was an observational descriptive study with a cross-sectional design and a retrospective approach. This research material was drawn from medical record data of adult patients (17-65 years old) with diagnosis of pulmonary tuberculosis and hospitalized with the ICD-X code A15.0 (verified using sputum microscopy with or without culture), integrated patient care records, nurse/midwife observation records, and drug administration forms. Primary healthcare refers (the patient's tuberculosis) to the hospital for secondary care. Patient's data in two referral public hospitals, type B classification, in East Java province, were collected and analyzed. The distance between these two hospitals (Surabaya city and Pasuruan district) was 67 km. Data collection was performed in adult patients with pulmonary tuberculosis that were hospitalized from January through December 2018. These two hospitals were tertiary care hospitals with a total of 223 and 323 inpatient beds respectively.
In this study, antibiotic use profiles were defined as the number of anti-tuberculosis drug (OAT) administered to inpatients in one year expressed in DDD/100 patient-days units. 24 The calculation was expressed using the DDD (Defined Daily Dose) method where each antibiotic had DDD values determined by WHO based on its average and main indications in adults. 25
The DDD/100 patient-days was calculated for all the patients receiving antibiotic therapies using the following equation:

The start and stop date of oral anti-tuberculosis drug use was collected from the patient's medical record to calculate days of therapy, whereas the date of admission and date of discharge to calculate the length of stay. Delays in oral anti-tuberculosis (OAT) treatment were noted if the date of admission preceding the date of start OAT use.
Results
Baseline characteristics in both hospitals were similar except in the patient weight and categories of the TB (Table 1). The number of patients weighing 50-70 kg in Hospital A (71.4%) was more than that in Hospital B (30.3%). The category of relapse in Hospital A (20.0%) was less than that in Hospital B (42.5%). There were 206 available medical records of pulmonary tuberculosis patients. Only 68 of 206 records (33.0%) met the inclusion criteria. Patients excluded were twenty-three patients (11.2%) aged ≤17 years old, 54 patients (26.2%) aged >65 years old, 41 patients (19.9%) with other infectious diseases besides TB, and 20 patients (9.7%) with incomplete data. Most patients are male, aged 45-55 years old, with hospitalization paid using health insurance, category 1 TB (Table 1). Among all rapid molecular diagnostic test (41/68, 60%), only one of them was rifampicin-resistant.
Baseline characteristics of the patients.

Anti-tuberculosis drug dosage was defined by considering the patient's weight.10,26 This study shows the differences between the prescribed dosage and the WHO standard. The hospital prescribed dosage was lower than that the WHO standard dosage (Table 2). In Hospital A there were four patients (weighing 51 kg, 60 kg, 67kg, and 70 kg) who received 600 mg of rifampicin. For patients whose weight is 51kg, for the first consecutive five days, they were prescribed with the 600mg of rifampicin, and for the next consecutive two days they were prescribed with 450mg. In Hospital B, there were different care- or treatment-related factors where patients received 750 mg of ethambutol, had a history of TB treatment for one month, or continued with the hospital's previous treatment on the first day of admission. However, after TB Rapid Molecular Diagnostic test results were positive and rifampicin-resistant, the treatment with rifampicin was stopped.
Discrepancies in dosage.
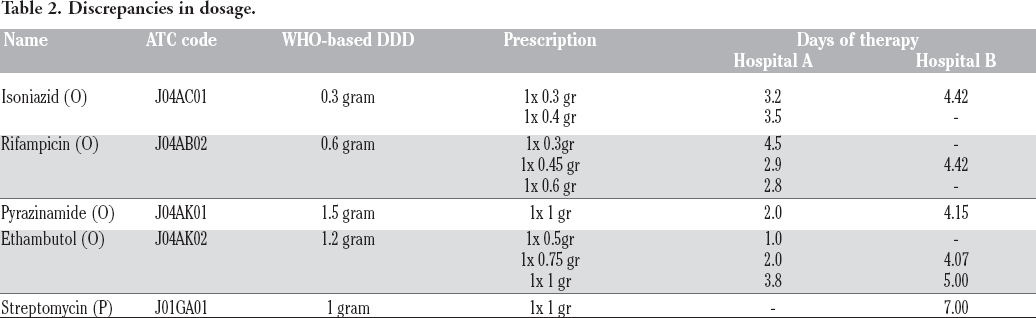
In Hospital A, 11 antibiotics were prescribed and the number of anti-TB drug was 108.8 DDD/100 patient-days (52.6%), whereas in Hospital B, only four antibiotics were administered and all of them were anti-TB drugs with 196.28 DDD/100 patient-days (Table 3). The average of antibiotic days of therapy is shorter than the average of the patient's length of stays (Table 1, Table 3). This drug-related problem will threaten the success of TB treatment. 27
Drugs used at hospitals A and B.

Most patients in the hospitals were delayed in receiving the anti-TB drugs since these patients needed to wait for the laboratory test results (Figure 1). Other TB patients had delayed treatments since they need to be hospitalized first for their cardiac problems. Two patients in hospital A needed to quit anti-TB treatment due to adverse drug events.
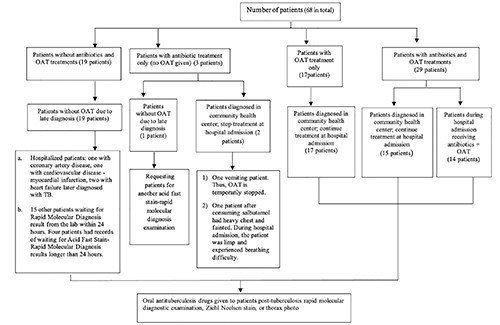
Patient medication profile.
Discussion
Older adults (aged ≥65 years) with tuberculosis were excluded from this study because they have unusual clinical manifestations, delayed diagnoses, and higher rates of adverse drug reactions and unfavorable outcomes, i.e., hepatitis and gastrointestinal discomfort. 28,29 Furthermore, older adults with physiological changes and co-morbidities need dosage adjustment according to weight, renal function, liver function, and other potentially complicating factors. These conditions will underestimate the defined daily dose per 100 patient-days value.
Fixed-dose combinations (FDCs) of drugs for TB treatment have been advocated internationally to prevent the emergence of drug resistance attributable to inappropriate drug intake or inappropriate drug choice problems.30,31 Use of the FDCs can reduce the risk of an incorrect dosage, simplify drug procurement, and aid in ensuring adherence without changing the drug dosage. 32 An adult with weight <50kgs needs three tablets of 4 fixed-dose combinations (rifampicin 150 mg, isoniazid 75 mg, pyrazinamide 400 mg, ethambutol 275 mg); and four tablets of 4 fixed-dose combination if their weight >50kgs. The DDD WHO for rifampicin, isoniazid, pyrazinamide, ethambutol is 0.6g, 0.3g, 1.5g, 1.2g, respectively; 26 therefore every patient had to have 3.0-3.9 DDD per day. The oral antituberculosis drug dosage was calculated based on mg/kg body weight and the number of fixed dosed combination (FDC) tablets. Three tablets of FDC equal to 450 mg rifampicin, 225 mg isoniazid, 1200 mg pyrazinamide, 825 mg ethambutol. Isoniazid and pyrazinamide are two OAT that are associated with hepatotoxicity. 33 Its severity was dose-dependent. Ethambutol was known as a bacteriostatic OAT, not bactericidal. Therefore, the prescribed dose for isoniazid, pyrazinamide, and ethambutol, was less than the standard dose. The change in antimicrobial usage is measured to evaluate antibiotic stewardship programs outcome. 34 The DDDs per 100 patient-days and days of therapy per patient-days were the most frequent metrics used to compare antibiotic consumption. 35
Twenty percent of the patients encountered diagnostic capacity at the location where they first sought care based on the results of Patient Pathway Analysis (PPA). Most initial care tests occur in the private sector, and case notification lags behind diagnostic confirmation in the public sector. 13 These so-called missing cases fall into three groups of patients, i.e. i) some patients never access care because of financial, geographic, or other barriers; ii) some patients seek care in the private (or non-state) sectors and are diagnosed and treated there, but are not reported to the National Tuberculosis Control Program (NTP); and iii) some patients were diagnosed and treated in the public sectors but are not reported to the NTP. Finding these “missing” patients is essential if the ultimate goal is to end TB. Patient-centered care is a core principle of the WHO End TB Strategy. 1
Medication reconciliation can mitigate discrepancies in patient care; however, it should be accompanied with effective implementation strategies. 21 Medication discrepancies happen due to an addition or withdrawal of a drug, a change to the dose or dosage, or an unintended error in prescribing admission medications. 36 Understanding a patient's journey from the emergence of the first symptoms to presentation and effective treatments is essential. For most patients, the journey can:
“involve a family member having TB, multiple presentations to health professionals, previous courses of antibiotic treatments or truncated courses of TB treatment, non-adherence or defaulting from treatment, premature discharge from a health facility and relapse with a more severe form of TB.” 37
Understanding the timeline and journey can help health professionals assess risk at the time of discharge planning, and understand families’ experiences of using health services. Risk assessment is required at specific milestones (every 6 months) in the treatment. 38 The risks are different in admission, in acute and chronic stages, and during discharge planning. One of the early risks may involve acute medical and nutritional complications, such as nosocomial infections. Chronic phase risks include the potential for non-adherence if discharged too early, or risks of chronic adverse health and developmental outcomes. Transition from hospital to outpatient clinics and community health centers requires that hospital services be intact;38,39 accordingly, good communication is essential to ensure the intact services.40,41 A systematic review reported that there is no issue of the gender equity for access to TB services quantitatively, but some barriers (financial, physical, stigma, health literacy, delay) are greater among women than men. 42 If the ultimate goal of controlling an infectious disease is to interrupt transmission, the current global tuberculosis strategy has not yet succeeded. 43
TB diagnosis has entered an era of molecular detection that provides faster and more cost-effective methods to diagnose and confirm drug resistance in TB cases whereas diagnosis using conventional culture systems requires several weeks to get the result. New advances in TB molecular detection, for example, faster and simpler nucleic acid amplification test (NAAT) and whole-genome sequencing (WGS), have resulted in shorter times for diagnosis and, thus, faster TB treatments. 44 Fundamental measures of TB control include early detection and timely treatment of the affected patient.
Tuberculosis control depends on early diagnosis and treatment at the primary health care level. However, many patients get a late TB diagnosis at hospitals. The delay in time between admission, diagnosis, treatment, and isolation 20 related to patients and health system factors. 45 Patient's delays are associated with knowledge, belief, and socio-economic factors. Health system factors are associated with negative sputum smear, age (older than 47 years old), and specialist consultation.20,39 A long delay in the diagnosis of pulmonary tuberculosis was also reported in Indonesia. 46 Adequate training of healthcare workers in diagnosing and notifying TB, and improvement in health care services may help to reduce the long healthcare delays.45-50
Conclusion
This study showed that the delay of medication and potential stopping of tuberculosis treatment could be reduced by medication reconciliation when the patient is transferred from a primary care to a secondary care facility or vice versa. The patients were seeking cure from TB and long-term tuberculosis treatment made it possible for these patients to completely miss treatments when they are being admitted or transferred to another healthcare institution. Thus, medication reconciliation or harmonization is an essential and urgent need to eradicate Mycobacterium tuberculosis and avert its resistance.
Footnotes
Acknowledgments
We are grateful to the management and staff of hospitals for allowing us to collect the data and to use them for the evaluation; and dr. Novita Maulidiyah, Sp.P to confirm the finding. We would like to thank David Scott, Pharmacy Department, Cardiff University, UK for reviewing, editing and proofreading the manuscript.
