Abstract
Background:
Patients with multidrug-resistant tuberculosis (MDR-TB) receive treatment that is at least 20% less effective than drug-susceptible cases globally.
Objectives:
The study evaluated the diagnostic accuracy of mfloDx MDR-TB assay (EMPE Diagnostics AB, Solna, Sweden) for simultaneous detection of resistance to isoniazid (INH) and rifampicin (RIF) compared to culture-based drug susceptibility test (DST).
Design:
A prospective diagnostic accuracy study.
Methods:
Clinical samples from 287 patients (mean age 45.3 (95% CI 43.3–47.2) years; 50 (17.0%) were female) from TB facilities in Ukraine (n = 82, 28.6%), Moldova (n = 37, 12.9%) and Spain (168, 58.5%) with bacteriologically confirmed TB and persons evaluated for non-tuberculous mycobacteria (NTM) were consecutively tested. The results of the mfloDx MDR-TB assay were compared with culture-based DST.
Results:
A total of 186 out of 287 sputum specimens (64.8%) yielded conclusive results that allowed a definitive interpretation of resistance to at least one drug. The mfloDx MDR-TB assay demonstrated sensitivity at 86.9 (95% CI 80.6–91.7) and specificity at 100 (95% CI 79.4–100) correctly differentiating mycobacterium tuberculosis complex from NTM in smear positive sputum samples. Considering only conclusive results in sputum samples, the mfloDx MDR-TB assay simultaneously predicted resistance to INH and RIF in TB patients showing high sensitivity 100 (95% CI 93.0–100) and specificity 98.7 (95% CI 92.8–100) compared to MDR-TB detection by culture-based DST.
Conclusion:
While approximately one-third of tests yielded inconclusive results, the mfloDx™ MDR-TB assay demonstrated potential as a rapid screening tool for INH and RIF resistance, offering a time advantage over conventional culture-based DST.
Plain language summary
Why was the study done: Tuberculosis (TB) is an infectious disease that mainly affects the lungs. Some forms of TB no longer respond well to standard medicines. These cases are called multidrug-resistant tuberculosis (MDR-TB) when resistance to the most effective drugs, such as isoniazid (INH) and rifampicin (RIF), is documented. There are only a limited number of technologies that are able to identify this resistance in a single test. People with MDR-TB have a much lower chance of being cured than those with drug-sensitive TB. How we did it: This study tested a new laboratory method, called the mfloDx MDR-TB assay, developed by EMPE Diagnostics (Sweden). The goal was to see how accurately this test could detect whether TB bacteria are resistant to INH and RIF. The results were compared with the standard, slower culture-based drug susceptibility test (DST). Clinical samples from 287 individuals were tested from February to August 2024. What we found: The mfloDx test produced clear results in about two-thirds (65%) of the samples. Among these, the test correctly identified resistance to INH and RIF with high accuracy: Sensitivity: 100% — how often the test detected MDR-TB resistance when it was truly present. Specificity: 98.7% — how often the test correctly showed no MDR-TB resistance when the bacteria were sensitive to both medicines. The test also accurately distinguished TB bacteria from other similar organisms, known as non-tuberculous mycobacteria (NTM). What do the findings mean: Although one-third of the samples did not produce clear results, the mfloDx MDR-TB test performed well in identifying TB drug resistance quickly and accurately. This rapid testing method could help doctors begin the correct treatment much sooner than with traditional culture-based methods, which can take several weeks.
Keywords
Introduction
Antimicrobial resistance (AMR), particularly in patients with tuberculosis (TB), has been identified at the United Nations High-Level Meeting in 2024 as a one of the most urgent worldwide health challenges. 1 Multidrug-resistant tuberculosis (MDR-TB) is defined as a disease caused by strain of Mycobacterium tuberculosis complex (MTC) that is resistant to the key first-line anti-TB drugs isoniazid (INH) and rifampicin (RIF). 2 Globally, the estimated rate of INH-monoresistant (Hr)-TB is almost equivalent (8.4%) 3 to the proportion of MDR-TB and/or rifampicin-resistant (RR)-TB—11.1%. 4 This issue is particularly relevant for Europe, where the proportion of people with TB who had MDR/RR-TB is the highest in the world, estimated at 37%, which necessitates more complex and expensive treatment options. 4 Thus, the molecular detection of resistance to both INH and RIF at the time of diagnosis would be the most cost-effective testing strategy for any smear-positive TB patient in countries with at least 5.8% of Hr-TB and 2.1% of MDR-TB cases. 5
Conventional culture-based detection of resistance is lengthy and complicated process, 6 especially in conflict-affected areas like Ukraine where due to the permanent attacks on the healthcare and energy infrastracture 7 the cost of phenotypic drug susceptibility test (pDST) significantly increased by the need of fuel electricity generation. Molecular diagnostics of MDR-TB is a rapid alternative for pDST allowing appropriate treatment decisions. However, commonly used initial tests XpertMTB/RIF or Xpert Ultra (Cepheid, Sunnyvale, CA, USA) are limited to detect only resistance to RIF and only if RR-TB is documented—INH resistance is evaluated by reflex test Xpert MTB/XDR. 8 This approach leaves a critical diagnostic gap in the rapid Hr-TB detection among patients susceptible to RIF corresponding to the delay in the selection of recommended treatment regimens and TB prevention in contact persons.
Various multiplex molecular TB diagnostic platforms are represented in current World Health Organization (WHO)-consolidated guidelines, 8 while other innovative tools under different stages of development are listed in the test directory of the Foundation for Innovative New Diagnostics. 9 In recent years, several commercially available molecular technologies have been evaluated for the simultaneous detection of RIF and INH resistance. These include automated real-time nucleic acid amplification assays that use specialized reagents, 10 as well as PCR–multiple-probe melting curve analysis assays. 11 Beyond canonical target-gene mutations, other mechanisms contributing to antibiotic resistance—such as overexpression of efflux pumps that reduce intracellular drug concentrations—may also play a significant role in the development of MDR-TB. 12 In addition to these approaches, targeted next-generation sequencing (tNGS) has been explored for sputum-based detection as a follow-on test, 13 as it offers broad mutation coverage; however, its widespread implementation is currently limited by the need for expensive sequencing equipment and its lower diagnostic accuracy compared with conventional DST. 14 In this context, a novel and promising technology for confirming TB diagnosis and predicting drug resistance has been developed by EMPE Diagnostics AB, Solna, Sweden, and called mfloDx™ MDR-TB assay. This test is based on nucleotide-specific padlock probe-dependent rolling circle amplification and lateral flow biosensors for visual assessment of signals. 15
The objective of the study was to evaluate the diagnostic accuracy of mfloDx MDR-TB assay for simultaneous detection of resistance to INH and RIF in clinical samples compared with pDST in the cohort of patients with bacteriologically confirmed TB.
Methods
Study design and setting
This is a prospective diagnostic accuracy study that evaluated the mfloDx assay in clinical samples from bacteriologically confirmed patients with TB and persons evaluated for nontuberculous mycobacterial (NTM) lung disease. We consecutively enrolled patients at the Regional Phthisiopulmonology Center (Vinnytsia, Ukraine), Regional Center of Socially Significant Diseases (Odesa, Ukraine), PMSI Institute of Pneumology “Chiril Draganiuc” (Chişinău, Moldova) from May 2019 to June 2020. We also included in the study clinical samples collected for a previous study among the Serveis Clinics (Barcelona, Spain) and the Germans Trias i Pujol Research Institute (IGTP). These samples were properly stored at −80°C.
Study populations
Only patients with bacteriologically confirmed TB and laboratory isolated NTM who provided informed consent were included into the study. The main purpose of involvement individuals with laboratory confirmed NTM isolation was the need of TB negative control group for evaluation of accuracy in MTC detection. From February to August 2024, we consecutively tested clinical samples by mfloDx MDR-TB assay at the IGTP, Badalona, Barcelona, Spain, from 287 patients who had been treated in TB facilities in Ukraine (n = 82, 28.6%), Moldova (n = 37, 12.9%) and Spain (n = 168, 58.5%).
Study procedures
All samples were collected at the moment of diagnosis or in the first 2 weeks after initiating of treatment. Only a single sample from each patient was selected and tested. Early morning spontaneusly expectorated sputum sample minimum 1 mL in volume was obtained from each patient. Other than sputum samples were obtained during standard diagnostics procedures according with national regulations. Smear microscopy, pDST (either on solid Lowenstein–Jensen medium or BACTEC MGIT 960 system (Becton Dickinson, Franklin Lakes, NJ, USA) and Xpert tests were performed for routine patient-care purposes using standard decontamination (N-acetyl-L-cysteine-sodium hydroxide) and homogenization (if required) steps. pDST was done using critical concentration (CC) method. 16 Assessors of the Xpert test and pDST were not informed about index test results. Each positive culture sample passed a confirmation test to detect the MPT64 antigen (Ag) according with national laboratory standards. In case of negative MPT64 Ag result, in Spain the culture isolate was tested by commercial GenoType Mycobacterium CM (Hain Lifescience GmbH, Nehren, Germany) allowing reliable differentiation of clinically relevant NTM species.
The study results were reported in accordance with the EQUATOR Network guideline, “Standards for Reporting Diagnostic Accuracy Studies” 17 (see Supplemental Material).
DNA isolation
Mycobacterial DNA extraction was done from 500 μL aliquot of each specimen using provided with the assay DNA extraction kit (EMPE Diagnostics Private Limited, Hyderabad, India) according to the mfloDx MDR-TB test manufacturer’s recommendations by using of the heat lysis at 95°C for 20 min by heat block. After incubation, the tubes were centrifuged at 12,000 rpm for 15 s at room temperature. The supernatant was used for performing the test directly after the lysis.
MfloDx MDR-TB test
The test was performed following the manufacturer’s instructions by a qualified lab technician who underwent specific training in molecular diagnostics. Clinical information and reference standard results were not available for the perfomer of the index test.
M. tuberculosis strain H37Rv was used as a positive control while we added PCR grade water instead of DNA to test whether we have contamination in our PCR workflow (negative control).
To perform the test, 5 μL of previously heat-lysed supernatant from clinical specimens was used to detect MTC and mutant or wild-type sequences in regions, responsible for INH and RIF resistance. Following DNA extraction, the test involves six steps: preamplification, target capture via probe ligation, magnetic bead-based purification of ligated circles, rolling circle amplification, restriction enzyme digestion, and visual signal development on lateral flow cassettes. The MDR-TB AMP Kit (primers, padlock probes and enzymes) was used for amplification, whereas the MDR-TB VIS Kit (primers) was used for visual signal development. During the pre-amplification stage, 15 µL of the PCR working solution (14.64 µL PCR mix + 0.36 µL enzyme) was added to each tube along with 5 µL of the DNA sample. Rolling circle amplification was performed with Applied Biosystems Veriti Dx 96-Well Thermal Cycler (Waltham, MA, USA) using standard PCR program. Then, 1 µL of the amplified PCR product was used for ligation, capture, rolling-circle amplification, and digestion, following the instructions provided. The mixture of specific primers VIS-1 (75 μL) and VIS-2 (40 μL) was prepared, and 115 μL of the aliquot was added to each sample. Subsequently, a total of 136 μL of aspirate was applied to the reaction window of each cassette. The whole process can be completed in 3–4 h, depending on the number of testing samples. The mfloDx MDR-TB test’s performance was evaluated against the phenotypic DST and WHO-recommended Xpert MTB/RIF molecular assay.
Interpretation of MfloDx MDR-TB test results
In the test cassette, the “WT” window identifies MTC and the presence of the wild-type alleles, while the “MUT” window detects mutations associated with drug resistance. Hybridization control (HC) demonstrates the effectiveness of the probe binding, and an internal control (IC) band indicates test completion. Visual signals, appearing as distinct pink bands, were evaluated in the corresponding reading window of the lateral flow cassettes. If a band was extremely faint, making visual interpretation difficult when compared to the intensity of any control band, the result was considered negative. However, this issue did not occur with any of the cassettes assessed.
Resistance to INH is based on detection of mutations in katG gene (codon 315) and inhA regulatory region (promoter region-15) while detection of mutations in rpoB (codons 516, 526, 531, 533) corresponds to RIF resistance. Categorization of the test results was performed following the manufacturer’s instruction for use. The result of test was classified either as a sensitive if only WT bands in the corresponding probes were present and no MUT band detected, as a resistant if any MUT band is present and indeterminate (if neither MUT nor WT band developed for a certain gene, while MTC control (and other controls) are positive). In the absence of confirmed MTC identification, resistance interpretation was not feasible due to overlapping mutation profiles with NTM species. The test result was considered invalid if at least 2 of 3 controls (HC, IC, MTC band) are not developed.
Statistical analysis
Sensitivity, specificity, and observed accuracy of the mfloDx MDR-TB test were calculated in comparison with the pDST. Indeterminate and invalid results of index test as well as missing data of reference standard were excluded from accuracy calculations. We used the term observed accuracy as we didn’t adjust the calculations for resistance prevalence due to different estimations in the paticipants’ countries of origin. Concordance between the evaluated test and the reference method was determined by the Cohen’s Kappa coefficient. The Cochran–Armitage test was used to assess the presence of a linear trend in the probability of obtaining certain resistance results across ordered acid-fast bacilli (AFB) smear microscopy categories. Statistical analysis and sample size estimation were performed with R Statistical Software. 18 Intended sample size estimation reflected national surveillance data on MDR-TB rate in Ukraine (31%) and in Moldova (40%) considering estimated proportion of Hr-TB at 15% 19 and expected test sensitivity of 0.95 and specificity of 0.95, n = 209 patients with bacteriologically confirmed TB diagnosis.
Results
Mean age was 45.3 years (95% CI 43.3–47.2), 50 (17.0%) individuals were assigned female at birth, representing predominant age category and sex ratio among TB patients in the European Region. 19 Two hundred and ninety patients were recruited initially. Figure 1 illustrates a flow diagram of participants representing enrolment into the study and those who have been excluded from the analysis.

Patient flow diagram of enrolled patients and samples assessed.
Types of specimens tested within the study and the distribution of pDST results in TB patients, as well as, NTM species isolated in patients who were evaluated for NTM lung disease with excluded TB diagnosis, are represented in Table 1.
Types of sample and resistance profile detected by culture-based DST.

Hr, isoniazid monoresistant tuberculosis; MDR-TB, multidrug resistant tuberculosis; NTM, nontuberculous mycobacteria; pDST, phenotypic drug susceptibility test; RR-TB, rifampicin mono-resistant tuberculosis; TB, tuberculosis.
Resistance detection by mfloDx MDR-TB results in sputum samples
A total of 154/226 (68.1%) tested sputum specimens from TB patients had valid results with certain interpretation of resistance to at least one drug while all 32/32 (100%) sputum specimens from patients evaluated for NTM had conclusive results. The overall results obtained by mfloDx MDR-TB test in sputum samples of TB patients are presented in Table 2. Considering only tests with certain results to INH, there were 97.2% (70/72) sensitivity, 96.2% (76/79) specificity, and 96.7% (146/151) agreement in sputum samples compared to pDST. Sensitivity, specificity, and agreement values for the detection of RIF resistance were 100.0% (53/53), 97.8% (88/90), and 98.6% (141/143), respectively (Table 3).
Distribution of the mfloDx MDR-TB test results according to the phenotypic DST results for 226 sputum samples.
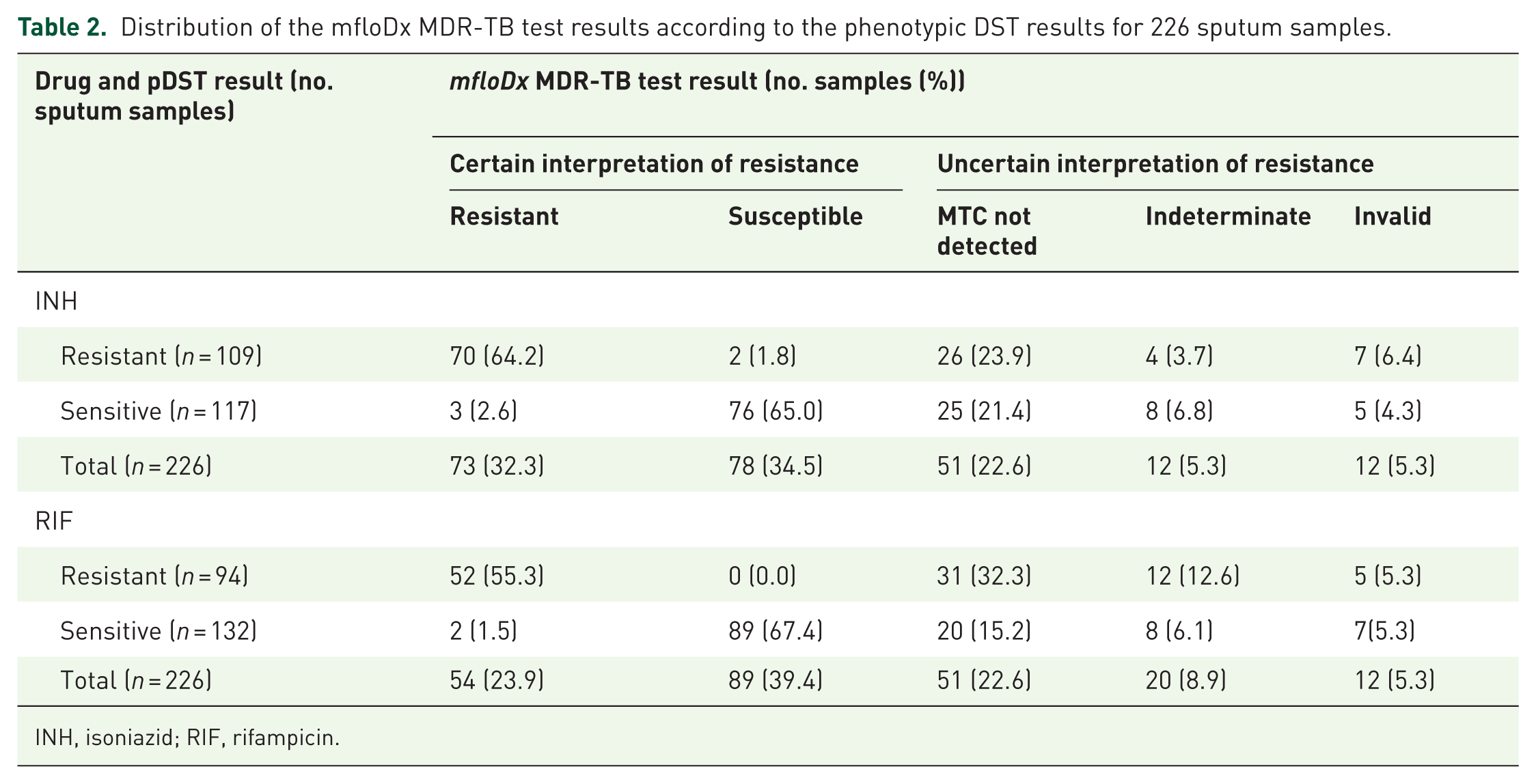
INH, isoniazid; RIF, rifampicin.
Evaluation of the mfloDx™ MDR-TB test compared to pDST for detecting resistance to INH and RIF in sputum samples with conclusive results.

Due to dichotomical categorization approach only samples from MDR-TB cases and individuals susceptible to both drugs were considered for this calculation with absolute agreement or disagreement due to inability of proper categorization in case of monoresistance discordance.
All combinations of observed agreements and disagrements were used for calculation.
INH, isoniazid; MDR-TB, multidrug resistant tuberculosis; RIF, rifampicin.
In Table 4, we presented the evaluation of the mfloDx MDR-TB test in comparison with XpertMTB/RIF assay. The mfloDx MDR-TB assay detected resistance in all 25 patients identified as rifampicin-resistant by Xpert; however, one discordant result indicating rifampicin susceptibility was observed in a patient classified as a resistant by Xpert.
Evaluation of the mfloDx™ MDR-TB test using the Xpert MTB/RIF a assay as a comparator in sputum samples yielding conclusive results for detecting resistance to rifampicin.

Only valid Xpert TB positive results with provided information about RIF resistance were reported in our patients. There were no indeterminate Xpert resistance results observed in our patients.
RIF, rifampicin.
mfloDx MDR-TB test results in other than sputum samples
We performed mfloDx MDR-TB in the 29 patients with samples different than sputum (see Table 1 for details). There was only one specimen phenotipically resistant to INH while the rest of clinical samples were fully susceptible by pDST. In all 11 (37.9%) cases with certain interpretation of results were obtained concordant results with pDST, in the remaining 18 (62.1%) the MTC control band was not detected. The mfloDx MDR-TB test demonstrated high accuracy in detecting resistance in nonsputum samples. RIF specificity was 100% (11/11), with a 95% CI 71.5–100, while sensitivity was not calculated due to the absence of samples with documented resistance. INH sensitivity was 100% (1/1) with a 95% CI 2.5–100, and specificity was 100% (10/10) with a 95% CI 69.2–100.
Additionally, the mfloDx MDR-TB test’s results were compared with the Ziehl–Neelsen stained microscopy (ZN) for a better understanding of the limit of detection and addressing potential issues with using of this test in clinical practice. ZN-smear positivity was significantly associated with the chances to obtain valid result, odds ratio (OR) 8.8 (95% CI 1.5–50.1), p = 0.0149.
MTC detection in sputum samples
MTC detection by the mfloDx MDR-TB test had sensitivity at 76.2% (163/214) (95% CI 69.9–81.7), specificity 100% (32/32) (95% CI 89.1–100) and observed accuracy 79.3% (195/246) (95% CI 73.7–84.2) compared to the MGIT results. Bacterial load was determined semi-quantitatively using ZN smear microscopy grading, where AFB were categorized according to standard criteria (negative, scanty, 1+, 2+, 3+), with higher smear grades reflecting higher bacillary burden (Table 5). Among 214 TB patients with available smear microscopy results, 27 of 42 (64.3%) ZN-negative individuals had MTC not detected, compared with only 21 of 172 (12.2%) ZN-positive patients. Invalid results were most frequently observed due to the absence of both the MTC and IC bands (one smear-negative and seven smear-positive cases). In two additional cases, invalid results occurred because both the MTC and HC bands were absent (one smear-negative and one smear-positive individual). Regarding indeterminate results, nearly half of the cases occurred in smear-negative or paucibacillary samples (5 of 12 for INH and 9 of 20 for RIF). The Cochran–Armitage test demonstrated a significant positive linear trend (p < 0.01), indicating that as smear grade (bacillary load) increased, the probability of obtaining a definitive resistance result also increased. The mfloDx MDR-TB assay demonstrated a high sensitivity at 86.9 (95% CI 80.6–91.7) for TB detection in smear positive sputum samples and specificity at 100 (95% CI 79.4–100) correctly differentiating MTC from NTM.
Impact of positive smear microscopy status on the probability of cetain interpretation of results for the mfloDx MDR-TB test.

Fisher’s exact test was used to calculate the OR and corresponding 95% CIs.
For this analysis, the p-value was > 0.05, whereas for the remaining comparisons, p-values were < 0.0001.
AFB, acid-fast bacilli; CI, confidence intervals; INH, isoniazid; OR, odds ratio; RIF, rifampicin; ZN results N/A, Ziehl–Neelsen stained microscopy with not availble result.
There was a strong, negative correlation between sputum microscopy bacterial load and MTC negative result, which was statistically significant (Spearman’s rank-order correlation, rs = −0.90; p = 0.037). Higher bacterial load in positive smear microscopy sputum samples significantly improves the sensitivity of MTC detection by the mfloDx MDR-TB test against overall results regardless smear status (two-tailed z test, sensitivity comparison p = 0.00932). Furthermore, as shown in Table 5, smear-positive status was strongly associated with obtaining a definitive interpretation of INH and RIF resistance compared with smear-negative patients (INH: OR = 11.1, 95% CI: 4.8–27.8; RIF: OR = 12.2, 95% CI: 5.1–32.6; Fisher’s exact test, p < 0.05).
In 5 of the 32 sputum samples from patients with laboratory-confirmed NTM (4 - M. avium and 1 - M. fortuitum), the MTC band was absent but accompanied by WT or MUT bands. This finding highlights that, even in patients with bacteriologically confirmed TB, sputum samples may contain environmental mycobacteria, which could potentially affect resistance results.
Discussion
This study indicates that the mfloDx assay can detect molecular MDR-TB in sputum samples with a sensitivity of 100% (95% CI: 93.0–100) and a specificity of 98.7% (95% CI: 92.8–100) compared with culture-based DST, when restricted to cases with conclusive results. However, its overall capacity to detect resistance across all TB patients, particularly those with smear-negative pulmonary TB, remains limited. Moreover, the high rate of inconclusive resistance interpretation substantially reduces its potential clinical impact, underscoring the need for further refinement before broad implementation. In the participants provided other than sputum clinical samples, the mfloDx MDR-TB test correctly identified all susceptible and resistant cases to INH, and all susceptible to RIF cases (sensitivity was not calculated because there were no participants with RR-TB in this group).
The analysis of the mfloDx MDR-TB assay results in sputum samples from 226 TB patients demonstrated sensitivity in molecular detection of resistance to INH at 97.2% (95% CI 90.3–99.7) and RIF at 100% (95% CI 93.2–100) in comparison with pDST, while specificity was 96.2% (95% CI 89.3–99.2) and 97.8% (95% CI 92.3–99.7), respectively. These values are consistent with previous findings reported by Ramasubban G et al. in 72 samples from bacteriologically confirmed TB patients in Vellore, India. 20
Although the mfloDx MDR-TB assay showed high sensitivity, specificity and observed accuracy for detection of INH and RIF resistance, we have to mention that these values were calculated only for the samples with valid results. However, 27.9% (72/258) of tested sputum samples and 62.1% (18/29) of other than sputum specimens were associated with resistance inconclusive results. A significant number of uncetain resistance results observed in the mfloDx MDR-TB compared to pDST may underestimate the achieveing of minimum requirements for the target product profile for detection of drug resistance and appropriate triage of patients at the peripheral level. 21
Our findings confirmed that the selecion for testing only AFB positive samples could significantly increase the sensitivity of MTC detection by the mfloDx MDR-TB assay (two-tailed z test, p < 0.05). On the other hand, focusing on smear positive samples in diagnostic accuracy study may reduce the clinical utility of the test for routine practice and can affect estimation of sensitivity and specificity creating a potential bias in the evaluation. 22 Given that only one-fifth of smear-negative samples produced interpretable resistance results, further technical refinement of the assay is warranted to enhance its performance in this patient population. In addition to low bacterial load, factors such as suboptimal DNA extraction efficiency and the presence of amplification inhibitors may also contribute to invalid results, as observed in eight cases lacking both MTC and IC bands. Nevertheless, only two invalid results were associated with absence of the HC band, indicating that overall sample flow and visualization chemistry functioned appropriately in most cases.
It is worth mentioning that insufficient probe coverage is a challenge in many visually assessed tests due to the limited space available for incorporating all probes. However, the capacity of the molecular test to identify resistance conferring mutations depends on their prevalence in the targeted population. For instance, mutations in codons 507–511, which are targeted by Probe A of the Xpert MTB/RIF assay, have been reported as uncommon in Ethiopia and southern India.23,24 In contrast, another study from a high-endemic region in Northeast India found these mutations in 25.9% of all RR-TB cases. 25 As for the INH resistance, the mfloDx MDR-TB test can detect mutations in one nucleic acid position for inhA gene (-15), while for the GenoType MTBDRplus assay the detection is also possible in other positions (-16 and -8). Perhaps, inclusion of a bigger number of probes in the mfloDx MDR-TB test could improve the accuracy of INH resistance detection, similarly to how it happened with the GenoType MTBDRplus assay compared to its earlier versions. 26
Among 88 samples with the available Xpert MTB/RIF assay results, 10 (11.4%) of the mfloDx MDR-TB test showed indeterminate RIF resistance results. However, even Xpert MTB/RIF that was recognized as the first test for TB diagnosis and antibiotic susceptibility testing that meets WHO’s prequalification standards, 27 could be associated with over 50% of false-positive results in samples with low bacterial load due to the probe mutation type and probe delay. 28
Among the key findings of the study is evaluation of the the mfloDx MDR-TB assay in the nonsputum samples from patients with different localization of TB disease. We have found a compatible sensitivity of the mfloDx MDR-TB assay detecting MTC in 37.9% of nonsputum samples in comparison with other commercially available molecular test Geno Type MTBDRplus assay version 2.0 (Hain Lifescience, Nehren, Germany) that was capable to detect MTC in 36.9% of extrapulmonary samples in the study of Ghanekar et al. 29 Furthermore, both technologies demonstrated comparable sensitivity and specificity for INH and RIF resistance detection when only conclusive results were considered. In comparison with another WHO-recommended technology for INH/RIF resistance testing, the Abbott RealTime MTB RIF/INH assay, mfloDx MDR-TB showed a higher rate of indeterminate results (31.9% vs 19.7%), higher sensitivity for INH resistance (97.2% vs 84.2%), and identical sensitivity for RIF resistance detection (100% for both assays). 30 However, specificity for both drugs was higher with the Abbott RealTime assay (96.2% and 97.8% vs 100% and 100%, respectively). Considering data from a recent study in South Korea that investigated a similar molecular assay, the BD MAX MDR-TB, we found that mfloDx MDR-TB showed a comparable rate of indeterminate results (31.9% vs 29.1%), lower sensitivity and specificity for INH detection (97.2% and 96.2% vs 100% and 100%), and twice the sensitivity for RIF resistance detection (100% vs 50%), while its specificity for RIF was slightly higher (97.8% vs 95.8%). 31
Due to high prevalence of Hr-TB among Xpert RIF-susceptible TB cases 32 it is crucial to perform initial detection of resistance to both INH and RIF. The mfloDx MDR-TB assay could facilitate in early treatment initiation reducing community and nosocomial TB transmission, and for correct selection of TB preventive treatment regimens in contact persons where both medicines commonly prescribed. 33
To the best of our knowledge, we report the first evaluation of the mfloDx MDR-TB assay in the multicenter cohort of patients including high MDR-TB countries such as Ukraine and Moldova. A major strengths of this study include enrollement of patients regardless their smear microscopy status, providing analyses of data for both sputum and nonsputum samples, clear description of methodology and results interpretation approach, and adding of the negative control group including patients evaluated for NTM lung disease.
One of the limitations of our study is a significant proportion of the results with inconclusive resistance interpretation in patients tested by this molecular assay. We did not repeat testing on a subset of samples with inconclusive results using an alternative DNA extraction method; however, we acknowledge that doing so could help determine whether the issue originates from the sample itself or from the detection workflow. Accordingly to the instruction for use, the results were considered indeterminate (if both WT and MUT bands are absent). Notably, 8.2% (11/134) of RIF and 5.2% (7/134) of INH indeterminate results occurred in highly smear-positive sputum samples (grades 1+ to 3+), where sufficient bacillary load would normally be expected to produce definitive banding patterns (Table 5). This finding suggests alternative explanations beyond low bacterial burden, including the possible presence of uncommon or rare mutations not covered by the assay’s mutation panel, which may result in failure of both WT and MUT probe hybridization. However, in the present study, we were unable to perform whole-genome or targeted sequencing to further investigate the underlying causes of discordant or indeterminate results between the mfloDx MDR-TB assay, pDST, and Xpert MTB/RIF.
There are two alternative approaches to evaluate absent WT band coexisting with no MUT band: (1) to consider all resistance inferred results as a resistant 34 or (2) according with revised Global Laboratory Initiative recommendations to count them as “resistance not detected.” 35 Both interpretation approaches may significantly impact on the calculations of diagnostic accuracy values in comparison with reference standard and clinical decision in TB management. Our calculations demonstrate that classifying inferred results for INH as resistant has minimal impact on sensitivity (from 97.2 (95% CI 90.3–99.7) to 97.4 (95% CI 90.8–99.7) but significantly reduces the test’s specificity (from 96.2 (95% CI 89.3–99.2) to 87.4 (95% CI 78.5–93.5); two-tailed z-test, p = 0.04136. A similar pattern was observed for RIF, with sensitivity remaining unchanged (from 100 (95% CI 93.2–100) to 100 (95% CI 94.3–100), while specificity again significantly decreased (from 97.8% (95% CI 92.3–99.7) to 89.9 (95% CI 82.4–94.4); two-tailed z-test, p = 0.0251. When results with absent WT and MUT bands were interpreted as “resistance not detected,” the sensitivity of the assay for INH resistance decreased from 97.2 (95% CI 90.3–99.7) to 92.1 (95% CI 83.6–97.1), two-tailed z-test, p = 0.17068, while specificity slightly increased from from 96.2 (95% CI 89.3–99.2) to 96.6 (95% CI 90.3–99.3). For RIF, sensitivity dropped significantly from 100 (95% CI 93.2–100) to 81.3 (95% CI 69.5–89.9); two-tailed z-test, p = 0.00096, whereas specificity showed only a marginal increase from 97.8 (95% CI 92.3–99.7) to 97.9 (95% CI 92.9–99.8).
Further study should be conducted to evaluate the role of NTM human respiratory tract colonization in the transmission of resistance-conferring mutations and interpretation of moleculr DST results in TB patients. 36
Conclusion
Approximately one-third of the results yielded an uncertain interpretation of resistance; however, this proportion could be reduced by prioritizing smear-positive samples for testing. When considering only results with a definitive resistance interpretation, the mfloDx MDR-TB assay demonstrated high diagnostic accuracy. Compared with culture-based DST, the assay detected INH resistance with 97.2% sensitivity and RIF resistance with 100% sensitivity, while specificity was 96.2% for INH and 97.8% for RIF. The ability of the test to simultaneously detect resistance to both drugs directly from clinical specimens in a single step has the potential to substantially shorten the time to appropriate treatment initiation, particularly in smear-positive samples and with further optimization of the technology. Nonetheless, further evaluation across diverse programmatic settings is needed to confirm these findings and support the integration of the mfloDx MDR-TB assay into routine diagnostic workflows.
Supplemental Material
sj-docx-1-tai-10.1177_20499361261436542 – Supplemental material for A new multiplex molecular assay based on padlock probes and rolling circle amplification for MDR-TB detection in clinical specimens: a prospective diagnostic accuracy study
Supplemental material, sj-docx-1-tai-10.1177_20499361261436542 for A new multiplex molecular assay based on padlock probes and rolling circle amplification for MDR-TB detection in clinical specimens: a prospective diagnostic accuracy study by Andrii Dudnyk, Nadiia Tytarenko, Oksana Lytvyniuk, Olena Tolstova, Joan-Pau Millet, Israel Molina, Adrián Antuori, Nelly Ciobanu, Valeriu Crudu and José Domínguez in Therapeutic Advances in Infectious Disease
Footnotes
Acknowledgements
We are grateful to the patients and their families, as well as the investigators and site staff, for their participation in this study. We would like to thank EMPE Diagnostics AB (Solna, Sweden) for kindly providing the kits for the study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
