Abstract
Background:
Tuberculosis (TB) remains a significant global health challenge, especially among people living with HIV. Drug-resistant TB (DR-TB) complicates treatment outcomes in high-burden countries like Uganda, particularly for adolescents and young adults living with HIV (AYALH).
Objectives:
We described the characteristics, treatment outcomes, and factors associated with treatment success among AYALH and DR-TB at a TB treatment unit in Mulago National Referral Hospital, Kampala, Uganda.
Design:
A retrospective cohort study was conducted.
Methods:
Medical records of AYALH treated for DR-TB between January 2013 and December 2021 were reviewed. Descriptive statistics and multivariable logistic regression were used to analyze treatment outcomes and associated factors.
Results:
Among 327 participants (mean age: 28.2 years, SD: 4.75; 52.6% male), the treatment success rate was 65.7%. A body mass index (BMI) ⩾ 18.5 kg/m2 (adjusted odds ratio [aOR]: 0.53, 95% CI: 0.33–0.83, p = 0.005), Efavirenz-based antiretroviral therapy (ART) regimens (aOR: 0.56, 95% CI: 0.35–0.89, p = 0.014), and primary DR-TB (aOR: 0.42, 95% CI: 0.28–0.64, p < 0.001) were significantly associated with treatment success.
Conclusion:
The study revealed a treatment success in only two-thirds of participants emphasizing persistent challenge of achieving optimal treatment outcomes for AYALH. The findings highlight that a higher BMI and Efavirenz-based ART regimens are significantly associated with improved treatment success pointing to the necessity for addressing nutritional needs and optimizing ART regimens to improve the management of DR-TB among AYALH.
Plain language summary
This study examined adolescents and young adults living with HIV in Uganda who had drug-resistant tuberculosis. Among 327 participants treated at Mulago National Referral Hospital from 2013 to 2022, about two-thirds had treatment success. The study found that having a healthier weight and being on an HIV regimen that included Efavirenz were associated with better treatment outcomes. Additionally, those with primary DR-TB had better outcomes than those who aquired drug resistance during treatment. The findings highlight the need to address nutritional needs and optimize HIV treatments to improve outcomes in this group.
Introduction
Drug-resistant tuberculosis (DR-TB) remains a significant challenge, particularly among people living with HIV (PLWH). Tuberculosis (TB) has regained its position as the leading cause of death globally from a single infectious agent, overtaking COVID-19 after a 3-year span. TB currently accounts for nearly twice as many fatalities as HIV/AIDS. Annually, more than 10 million individuals are diagnosed with TB, with incidence rates showing an upward trend since 2021. 1 The burden of TB is disproportionately distributed, with over 95% of cases and deaths occurring in low- and middle-income countries, predominantly in Africa and Asia.2,3 Moreover, the emergence of DR-TB, including multidrug-resistant TB (MDR-TB) and extensively drug-resistant TB (XDR-TB), has further complicated TB management and control efforts, posing a significant threat to global health. 4
About one in four TB-related deaths occur in the African region, and more than 60% of 15 million cases of HIV/TB co-infection have been reported in the region.5,6 Studies have demonstrated an increased risk of DR-TB among PLWH, likely due to factors such as inadequate TB treatment, poor adherence to anti-TB medications, poor adherence to antiretroviral therapy (ART), and the high prevalence of HIV-associated immunosuppression. 7 Uganda is a high-burden TB/HIV country, with an estimated annual TB incidence of 253 cases per 100,000 population and an HIV prevalence of 6.2% with up to 45% among all reported TB cases. 8 Furthermore, PLWH have a 20-fold higher risk of developing TB compared to their peers without HIV. 9 The emergence of DR-TB in Uganda is a growing concern, with an estimated 1.4% of new TB cases and 12.1% of previously treated cases being classified as MDR-TB or rifampicin-resistant TB (RR-TB) in 2020. 10 The prevalence of rifampicin resistance was significantly higher in PLWH with TB (32%) than in HIV-negative individuals with TB (12%) at the National Referral Hospital, suggesting an increased risk of DR-TB among PLWH. 11 Over the study period, significant advances were made in DR-TB management, including the adoption of shorter, more effective treatment regimens in 2017, such as the Bedaquiline-based 9-month regimen. 12 Similarly, ART transitioned to Dolutegravir (DTG)-based regimens starting in 2018, replacing older Efavirenz (EFV)-based regimens. 13 These changes reflect ongoing efforts to optimize treatment outcomes for PLWH co-infected with DR-TB.
Previous studies among patients with DR-TB/HIV examined gender differences, noting higher mortality rates among men, 14 while also highlighting high treatment success among pregnant women with DR-TB despite a high prevalence of HIV co-infection. 15 Similarly, a study by Bayowa et al. 16 focused solely on mortality as an outcome, reporting a mortality rate of 33%, but included a broad age range of 1–80 years with a mean age of 35. However, these studies did not specifically address the unique age group of adolescents and young adults living with HIV (AYALH) who face distinct biological, psychological, and social challenges that differ significantly from those of younger children or older adults living with HIV. These gaps in existing literature indicate that characteristics and treatment outcomes of adolescents and young adults living with DR-TB/HIV are not well documented, stressing the need for a study in this specific population as this group represents a vulnerable population with unique challenges. The purpose of this study was to describe the characteristics, treatment outcomes, and factors associated with treatment success among AYALH and DR-TB at a TB treatment unit in Mulago National Referral Hospital, Kampala, Uganda.
Methods
Study design
We conducted a retrospective cohort study reviewing medical records of patients who initiated DR-TB treatment or received care at Mulago National Referral Hospital (MNRH) during their treatment course between January 2013 and December 2021 with a minimum follow up period of 12 months, ensuring comprehensive capture of treatment outcomes. The reporting of this study was in accordance with the STROBE guidelines. 17
Study area
The study was conducted at the TB unit of MNRH, Uganda. MNRH is a tertiary care referral center providing comprehensive healthcare services, including specialized care for TB and PLWH. It is also Uganda’s largest TB Treatment Center, managing an estimated 200 patients per quarter and offering care to referrals (about 40%) and non-referral cases. MNRH primarily manages DR-TB cases referred from across Uganda. These referrals often include patients with severe disease, failed previous DR-TB regimens, or those requiring specialized care unavailable at lower-level facilities.
Study population
Our target population was AYALH aged 10–35 years with a diagnosis of DR-TB, focusing on those who attended the TB unit of MNRH between January 2013 and December 2021.
Selection criteria
We included AYALH who had a confirmed diagnosis of DR-TB and excluded those with missing data on age and sex.
Study variables
Dependent variables
According to the standard definitions of the WHO Definitions and reporting framework for tuberculosis 2013 revision, 18 the following treatment outcome definitions were used:
Cured: Treatment completed as recommended by the national policy without evidence of failure AND three or more consecutive cultures taken at least 30 days apart are negative after the intensive phase.
Treatment completed: Treatment completed as recommended by the national policy without evidence of failure BUT no record that three or more consecutive cultures taken at least 30 days apart are negative after the intensive phase.
Treatment failed: Treatment terminated or need for permanent regimen change of at least two anti-TB drugs because of: lack of conversion by the end of the intensive phase, or bacteriological reversion in the continuation phase after conversion to negative, or evidence of additional acquired resistance to fluoroquinolones or second-line injectable drugs, or adverse drug reactions.
Died: A patient who dies for any reason during the course of treatment.
Lost to follow-up: A patient whose treatment was interrupted for two consecutive months or more.
Treatment success: The sum of cured and treatment completed
Unsuccessful treatment: The sum of loss to follow-up, death and treatment failure.
Independent variables
These included demographic characteristics such as age, sex, marital status, and employment status, as well as clinical characteristics like body mass index (BMI), TB regimen (Table 1), smoking, alcohol use, ART regimen, and TB treatment adherence. The adherence was based on reports from clinic-based directly observed therapy (DOT), home-based DOT, and patient self-reports and was categorized as “good” or “poor” based on clinic- and home-based DOT reports combined with patient self-reports throughout the treatment period as noted in the patients’ records. Good adherence was defined as ⩾90% of prescribed doses taken, while poor adherence was <90%. Reports from clinic- and home-based DOT were treated as verified doses, while patient self-reports were included only when cross-validated using methods like pill counts, diaries, or follow-up interviews. All sources were integrated into a composite adherence percentage, prioritizing directly observed doses. This 90% threshold aligns with adherence standards in clinical research and therapeutic guidelines. Other factors considered were comorbid conditions such as diabetes mellitus and cancer, site of DR-TB (pulmonary tuberculosis or extra-pulmonary tuberculosis), and mode of HIV acquisition (vertical or horizontal transmission) based on past medical records, facility records, and self-reporting. Additionally, documentation of AIDS-defining illness was sought for (any of Pneumocystis jirovecii pneumonia, esophageal or tracheal candidiasis, toxoplasmosis, cryptococcosis, Kaposi’s sarcoma, non-Hodgkin lymphoma, invasive cervical cancer, and molluscum contagiosum), and the diagnostic category (primary or acquired DR-TB) was included. Also, ART regimen was noted based on whether it was DTG-based regimen (Tenofovir Disoproxil Fumarate + Lamivudine + DTG), EFV-based regimen (Cobicistat + EFV, Tenofovir Disoproxil Fumarate + Lamivudine + EFV), or other regimens such as Abacavir + Lamivudine + Lopinavir/ritonavir, Zidovudine + Lamivudine + Nevirapine, and Tenofovir Disoproxil Fumarate + Lamivudine + Nevirapine. The type of DR-TB was classified as RR-TB (resistance to rifampicin detected using phenotypic or genotypic methods, with or without resistance to other anti-TB drugs), MDR-TB (resistance to at least both Rifampicin and Isoniazid), or XDR-TB (TB caused by Mycobacterium tuberculosis strains that fulfill the definition of MDR/RR-TB and that are also resistant to any fluoroquinolone and at least one additional Group A drug, that is levofloxacin, moxifloxacin, bedaquiline, and linezolid). Uganda adopted WHO-recommended shorter treatment regimens for DR-TB in 2017, which include a 9-month treatment course involving Bedaquiline, Linezolid, Levofloxacin, Clofazimine, and Cycloserine. Prior to 2017, treatment regimens were individualized and often included injectable agents like kanamycin. Regimens were assigned based on the national guidelines active during each calendar year. Patients were started on a regimen following the national policy recommendations for their specific clinical presentation, drug susceptibility test results, and prior treatment history. The decision to initiate a particular regimen was determined by considering the availability of drugs, patient tolerability, and coexisting medical conditions, including HIV. The regimen described in the study was the starting regimen that patients were initiated on at the beginning of their DR-TB treatment. Changes to the initial regimen during follow-up, if any, were made in response to adverse drug reactions, lack of clinical improvement, or detection of additional drug resistance. While such changes were documented in individual patient records, our analysis focused on the initial regimens. The regimens obtained from the patient records are summarized in Table 1.
The TB treatment regimens.

Sample size
We reviewed all available medical records for AYALH with DR-TB co-infection who attended the TB unit of MNRH between January 2013 and December 2021.
Study procedures
Data were collected from routinely existing health facility records into structured forms by a team of trained research assistants. Sources of data included TB physical registers and patient files, in cases of missing data, the online register was accessed. Reported DR-TB cases in this population were diagnosed using GeneXpert MTB/RIF assay and confirmed through sputum culture where required. The data collected from these registers included information on patient demographics, clinical characteristics, and their treatment outcomes.
Data analysis
The demographic data were summarized using descriptive statistics, including frequencies, percentages, and means. The aim of the analysis was to identify independent predictors of treatment outcomes (treatment success vs unsuccessful treatment). Multivariable logistic regression was used to identify these predictors, incorporating those with p-values <0.20 in bivariate analysis. We considered clinically relevant interactions a priori, particularly the site of DR-TB and ART regimen, based on prior studies and expert opinion. Other interactions were assessed post-hoc. However, none of these interaction terms significantly improved model fit, suggesting that no key interactions were present. Adjusted odds ratios (aORs) were calculated to quantify the strength of these associations, allowing for a robust evaluation of the factors influencing the outcome. All statistical analyses were conducted using IBM SPSS Statistics v27 software. 19
Results
Characteristics of AYALH with DR-TB
Of the 340 participant data retrieved, 13 were excluded due to missing data on age (4) and sex (9). Of the 327 participants were included, a majority were male (n = 172) with over half of participants married (n = 180). The mean age was 28.2 years (range 10–35) with 150 (45.9%) having a BMI under 18.5 kg/m2 and majority having RR-TB (92.7%). 200 (61.2%) were on EFV-based ART regimens and 179 (54.7%) had primary DR-TB (Table 2).
Characteristics of participants.

ART, antiretroviral therapy; BMI, body mass index; DM, diabetes mellitus.; DR-TB, drug-resistant tuberculosis; DTG, dolutegravir; EFV, efavirenz; EPTB, extra-pulmonary tuberculosis; IND, individualized; LTFU, loss to follow-up; LTR/STD, long-term regimen/standardized; MDR-TB, multidrug-resistant tuberculosis; mSTR, modified short-term regimen; PTB, pulmonary tuberculosis; RR, rifampicin-resistant; STR, short-term regimen; STR/STD, short-term regimen/standardized; TB, tuberculosis; XDR-TB, extensively drug-resistant tuberculosis.
Treatment outcomes of AYALH with DR-TB
Treatment completion was documented in 118 (36.1%), 56(17.1%) patients died and 46 (14.1%) were lost to follow-up. A total of 97 (29.7%) patients were cured, and only 10 (3.1%) were treatment failure. This translated to 215 (65.7%) patients with treatment success and 112 (34.3%) with unsuccessful treatment outcomes.
Factors associated with DR-TB treatment success in AYALH
Participants with a BMI ⩾ 18.5 kg/m2 (54.1%) had higher odds of treatment success compared to those with a BMI below 18.5 kg/m2 (aOR: 0.53, 95% CI: 0.33–0.83, p = 0.005). Similarly, individuals on EFV-based ART regimens (61.2%) demonstrated the greatest likelihood of successful outcomes, with lower odds observed for those on DTG-based (aOR: 0.56, 95% CI: 0.35–0.89, p = 0.014) or other regimens (aOR: 0.25, 95% CI: 0.11–0.55, p = 0.001). Participants with primary DR-TB (54.7%) had significantly higher odds of achieving treatment success compared to those with secondary DR-TB (aOR: 0.42, 95% CI: 0.28–0.64, p < 0.001; Table 3).
Factors associated with treatment success.
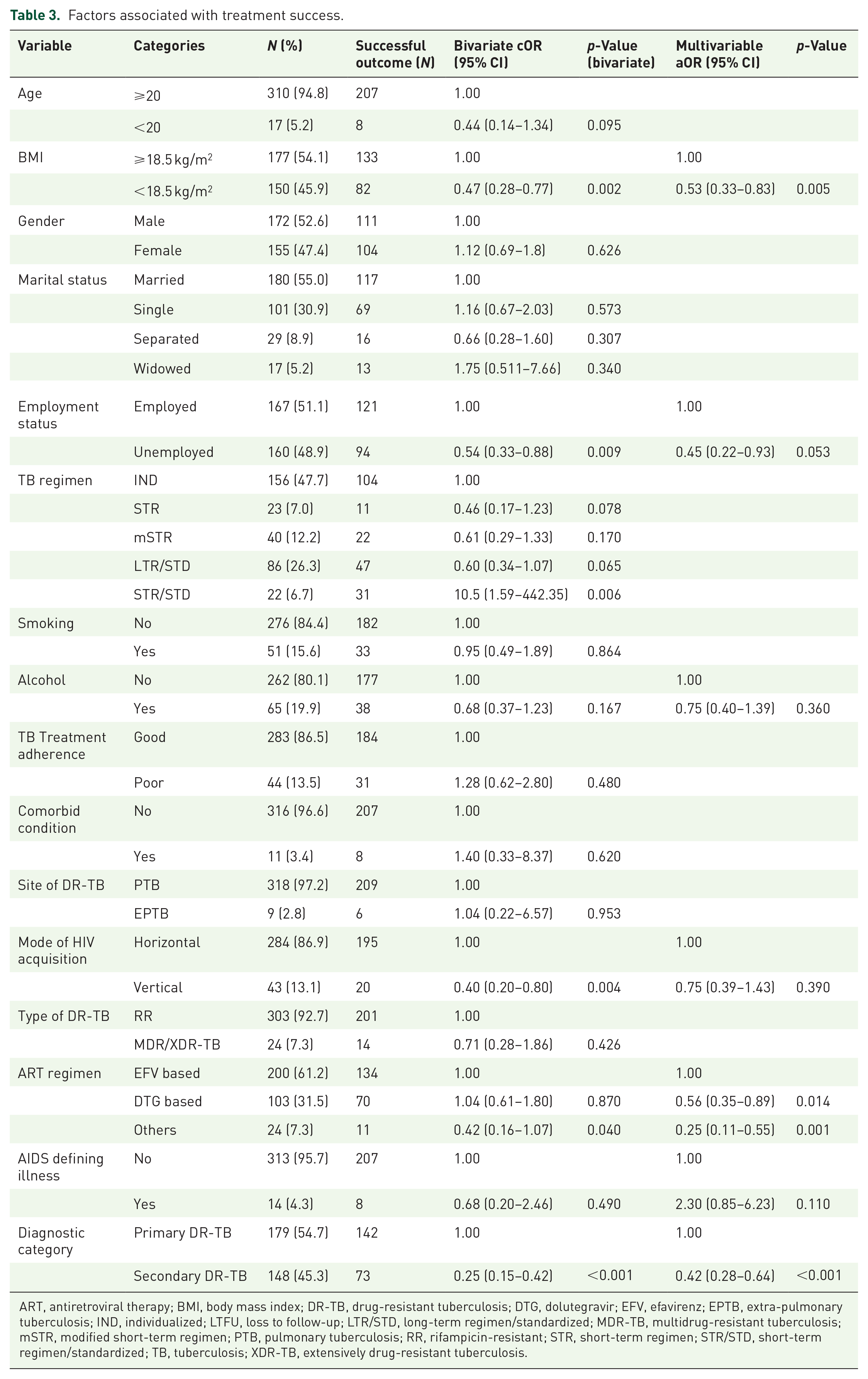
ART, antiretroviral therapy; BMI, body mass index; DR-TB, drug-resistant tuberculosis; DTG, dolutegravir; EFV, efavirenz; EPTB, extra-pulmonary tuberculosis; IND, individualized; LTFU, loss to follow-up; LTR/STD, long-term regimen/standardized; MDR-TB, multidrug-resistant tuberculosis; mSTR, modified short-term regimen; PTB, pulmonary tuberculosis; RR, rifampicin-resistant; STR, short-term regimen; STR/STD, short-term regimen/standardized; TB, tuberculosis; XDR-TB, extensively drug-resistant tuberculosis.
Discussion
In this study, we aimed at describing characteristics and treatment outcomes of AYALH with DR-TB. We found that 65.7% of patients achieved treatment success, while 34.3% had unsuccessful treatment outcomes. Our finding is almost identical to the nationwide data of treatment success in HIV/DR-TB reported at 66% 14 and also similar to those reported in other studies conducted in resource-constrained settings in sub-Saharan Africa, where treatment success rates often fall short of the WHO target of 90%.20,21
Our results indicated that a BMI ⩾ 18.5 kg/m2 was significantly associated with higher odds of treatment success compared to a BMI < 18.5 kg/m2. This finding conforms to existing knowledge that nutritional status plays a crucial role in TB treatment outcomes. Underweight patients are often at higher risk of adverse outcomes due to their compromised immune systems, which makes them less able to fight off infections effectively. 22 A low BMI at the start of treatment for RR-TB or MDR-TB is associated with poor treatment outcomes. 23 Looking at Bayowa et al. study, having a BMI < 18.5 kg/m2 was a risk factor to mortality. There was a 9% reduced likelihood in mortality among persons on treatment who had a BMI ⩾ 18.5 kg/m2 than those who had a BMI < 18.5 kg/m2. 16 Hanrahan et al. concluded that individuals living with HIV who are obese and overweight BMI have a significantly reduced risk of both mortality and TB, after adjusting for highly active antiretroviral therapy use and CD4 cell count whereas those with underweight BMI had an increased risk of mortality. 24 Shuter et al. identified an inverse link between BMI and mortality, regardless of baseline CD4 cell count, in a group of PLWH and using intravenous drug in Miami. 25 Shuter et al. 25 observed that overweight individuals experienced slower disease progression and lower viral loads among AIDS-free PLWH in New York City, after accounting for baseline CD4 cell count and the time to start antiretroviral therapy in a multisite US study of women living with HIV. The importance of addressing malnutrition in TB patients is emphasized by global health guidelines, which recommend nutritional support as part of comprehensive TB care. 26 There are still gaps in our knowledge of what specific supplementation macro and micronutrients would improve TB outcomes.
Patients with primary DR-TB were more likely to achieve treatment success compared to those with acquired DR-TB. Primary resistance develops when patients are exposed to and infected with an already drug-resistant strain. Secondary resistance or acquired resistance develops due to poor adherence to medication, drug malabsorption, and inadequate regimen among patients taking TB medication. 27 Secondary DR-TB often involves patients with a history of TB treatment, potentially leading to more extensive drug resistance and compromised immune responses, especially in the context of HIV. 28 This could explain the poorer outcomes observed in secondary DR-TB cases. Early and accurate detection of drug resistance and personalized treatment regimens, particularly for those with a history of TB can improve treatment outcomes in this high-risk group.
EFV-based regimens were associated with higher odds of treatment success. Up to June 2018, the preferred first-line therapy for HIV-1 infection was an EFV-based regimen (with a 400 mg dose of EFV), 29 meaning that this regimen was most widely adopted in the first half of our study period. The superior outcomes observed with EFV-based regimens may be attributed to its minimal interaction with rifampin, a key anti-TB drug. Unlike DTG, which has been reported to reduce rifampin absorption, EFV maintains stable plasma levels, ensuring sustained therapeutic efficacy. These pharmacological differences likely explain the observed treatment success, highlighting the need for careful regimen selection in managing HIV/DR-TB co-infections. The observed advantage of EFV-based regimens in achieving higher treatment success conforms with other studies that showed its effectiveness in HIV/DR-TB management as EFV’s potent antiviral properties and favorable pharmacokinetics likely contribute to improved outcomes.30,31 Our findings, however, support the continued use of EFV-based regimens in case DTG-based regimens are unavailable as the NAMSAL study showed people with HIV on DTG- and EFV-based regimens showed long-term efficacy and safety of both ARTs. 32 Moving forward, focus should be on addressing barriers to adherence among AYALH with DR-TB to further enhance treatment success. Given the poorer outcomes associated with secondary resistance, we recommend implementation of robust adherence counseling strategies tailored to adolescents and young adults. This includes addressing stigma, enhancing social support, and utilizing digital tools to ensure consistent medication use.
Our study, however, had some limitations. The study is reliant on secondary data, which are limited by the availability and completeness of medical records. The varied durations of DR-TB regimens pose potential biases, as patients on longer regimens have greater risk exposure for death or loss to follow-up. Also, patients starting treatment in 2017 or later were more likely to receive shorter, Bedaquiline-based regimens, while earlier cases predominantly received individualized or standardized longer regimens hence interpretation of results should be done with caution. Grouping death and loss to follow-up as unsuccessful treatment may mask distinct risk factor patterns. Another limitation is in the analysis as there is variation in treatment durations across different ART regimens. The unequal follow-up times across participants could introduce bias, as individuals on longer regimens have more opportunities to experience the outcome (e.g., treatment failure, death, or lost to follow-up).
Conclusion
The study revealed a treatment success in only two-thirds of participants emphasizing persistent challenge of achieving optimal treatment outcomes for AYALH. The findings highlight that a higher BMI and EFV-based ART regimens are significantly associated with improved treatment success pointing to the necessity for addressing nutritional needs and optimizing ART regimens to improve the management of DR-TB among AYALH. Optimizing adherence through structured counseling programs is crucial for improving outcomes, particularly among patients with secondary DR-TB. Future studies should consider time-dependent variables or survival analysis techniques to better account for variation in treatment duration.
