Abstract
Adipose tissue-derived mesenchymal stem cells (ADSCs) are derived from adipose tissue and can be induced in vitro to differentiate into osteoblasts, chondroblasts, myocytes, neurons, and other cell types. Cisplatin is a commonly used chemotherapy drug for cancer patients. However, the effects of cisplatin on ADSCs remain elusive. This study found that a high concentration of cisplatin affects the viability of ADSCs. First, the IC50 concentration of cisplatin was evaluated. Proliferation of ADSCs, as assessed by the XTT method, decreased immediately after treatment with various concentrations of cisplatin. ADSCs maintained mesenchymal stem cell surface markers after cisplatin treatment, as determined by flow cytometry. Upon differentiation by adding specific reagents, a significant decrease in adipogenic differentiation (by Oil red O staining) and osteogenic differentiation (by Alizarin red staining), and significant chondrogenic differentiation (by Alcian blue staining) were found after cisplatin treatment. Quantitative RT-PCR was also used in evaluating expression of specific genes to confirm differentiation. Finally, ADSCs from one donor who had received cisplatin showed significantly decreased adipogenic differentiation but increased osteogenic differentiation compared with ADSCs derived from one healthy donor. In conclusion, cisplatin affects the viability, proliferation, and differentiation of ADSCs both in vitro and in vivo via certain signaling pathways, such as p53 and Fas/FasL. The differentiation abilities of ADSCs should be evaluated before their transplantation for repairing cisplatin-induced tissue damage.
Introduction
With cancers remaining as the leading cause of death, chemotherapies are frequently applied for treating cancer patients 1 . Platinum-based chemotherapy is the most widely used chemotherapeutic for cancers. Cisplatin is one of the platinum compounds used for the treatment of head and neck, lung, breast, ovarian, and cervical cancer 2 . However, cisplatin has side effects such as renal toxicity and peripheral neuropathy 3 . It may also cause harm to stem cells residing in the body.
Adipose tissue has proven to be an abundant, accessible, and rich source of adult stem cells with multipotent properties suitable for tissue engineering and regenerative medical applications 4 . Like other mesenchymal stem cells (MSCs), adipose tissue-derived mesenchymal stem cells (ADSCs) can be induced in vitro to differentiate into osteoblasts, chondroblasts, myocytes, neurons, and other cell types 5 . ADSCs can also be reprogrammed to induced pluripotent stem cells (iPSCs) more efficiently than other cell types 6 . Furthermore, ADSCs are also immune privileged cells and secrete immunomodulatory, angiogenic, antiapoptotic, and hematopoietic factors that facilitate tissue repair7,8. Therefore, ADSCs play important roles in regenerative medicine, especially for adults.
MSC-based treatment for repairing tissue damage either by differentiating into functional cells or providing a protective microenvironment has been discussed 9 . MSCs have recently been used for repair of cisplatin-caused kidney damage in an animal model10–14. Nevertheless, the effects of cisplatin on stem cells remain elusive.
The aims of this study were to investigate the effects of cisplatin on the survival, proliferation, and differentiation capabilities of ADSCs derived from healthy patients and cancer patients and discuss the possible mechanisms.
Materials and Methods
Isolation and Cell Culture
Human ADSCs were obtained from subcutaneous adipose tissue of four patients undergoing scheduled surgery. All four patients participating in this study provided informed consent, and the study was approved by the research ethics committee of Hualien Tzu Chi General Hospital (IRB 100–107). In this study, we selected three samples of ADSCs derived from patients without chemotherapy [ASC01: 79 years old (y.o.), female; ASC02: 20 y.o., male; ASC03: 53 y.o., male) and one sample of ADSCs (ASC04: 38 y.o., female) cultivated from a cancer patient who had completed platinum-based chemotherapy for 4 months.
ADSCs were isolated and cultured as previously described 4 . Briefly, the adipose tissue was washed with phosphate-buffered saline (PBS) and incubated with 0.075% type I collagenase (Sigma-Aldrich, St. Louis, MO, USA). Then mononuclear cells (MNCs) obtained from tissue digestion and centrifugation were suspended in Dulbecco's modified Eagle's medium (DMEM)-F12 (Caisson Laboratories, Inc., Smithfield, UT, USA) supplemented with 10% fetal bovine serum (FBS; Biological Industries, Kibbutz Beit Haemek, Israel), 0.2 mmol/L L-ascorbic acid 2-phosphate sesquimagnesium salt hydrate, 2 mmol/L N-acetyl-L-cysteine (Sigma-Aldrich), and 1% penicillin/streptomycin (P/S; Sigma-Aldrich). Cells were seeded at 1 × 104 cells/cm2 into tissue culture flasks and expanded at 37°C in a humidified culture chamber with a 5% CO2 atmosphere, and the culture medium was changed every 2–3 days. Upon reaching 90% confluence, cells were transferred to a new flask with 103 cells/cm2. Cells were used between passages 3 and 5 in all experiments.
Viability and Survival Assays
ADSCs were seeded in triplicate at a density of 2 × 103 cells/cm2 in a 96-well plate with DMEM-F12 (Caisson Laboratories, Inc.) supplemented with 10% FBS, 0.2 mmol/L L-ascorbic acid 2-phosphate, 2 mmol/L N-acetyl-L-cysteine, and 1% P/S (Sigma-Aldrich). For the dose-dependent study, two different ADSCs (ASC01 and ASC02) were treated with 2, 5, 10, and 20 μmol/L of cisplatin (Fresenius Kabi Oncology Limited, Solan, India) for 3 days. Then the half maximal inhibitory concentration (IC50) of both ADSCs was obtained. We used the 4PL method described in the previous literature
15
. The equation is expressed as follows:
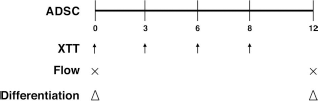
Schematic illustration of experimental design. Proliferation, flow cytometry, and differentiation of different adipose tissue-derived mesenchymal stem cells (ADSCs) before or after cisplatin treatment for 3 days were examined. Arrows, cell proliferation detected by XTT assay; ×, flow cytometry; triangles, differentiation.
To obtain the population doubling time (DT), 1 × 104 cells were seeded in a 10-cm Petri dish. The culture medium was changed every 3 days, and cells were harvested and counted on day 12. DT was calculated according to the following formula: DT = log (final number of cells) — log (initial number of cells) = K × T, where K is the generation constant (0.008963), and T is the time in hours 17 . The experiments were performed in triplicate.
Flow Cytometry of ADSCs with or without Cisplatin Treatment
Surface molecules of two different ADSC samples, treated with and without 2 μmol/L cisplatin for 3 days, were characterized by flow cytometry after 12 days of culture (Fig. 1). Cells were detached with 2 mM ethylenediaminetetraacetic acid (EDTA) in PBS and then washed with PBS containing 2% bovine serum albumin (BSA) and 0.1% sodium azide (Sigma-Aldrich). Cells were then incubated with their respective antibody conjugated with fluorescein isothiocyanate (FITC) or phycoerythrin (PE), including CD34, CD44, CD45, CD73, CD90, CD105, and human leukocyte antigen (HLA)-ABC and -antigen D related (DR) (BD Pharmingen, Franklin Lakes, NJ, USA). Cells were analyzed using a flow cytometer (BD Biosciences, San Jose, CA, USA).
Adipogenesis and Measurement
On days 0 and 12, the ADSCs of both groups were seeded in a 12-well plate at a density of 5 × 104 with adipogenic medium consisting of DMEM supplemented with 10% FBS, 1 μmol/L dexamethasone, 5 μg/ml insulin, 0.5 mmol/L isobutylmethylxanthine, and 60 μmol/L indomethacin (all purchased from Sigma-Aldrich). These ADSCs were allowed to grow for 14 days. The medium was changed every 3 days, after which the ADSCs were stained with Oil red O (Sigma-Aldrich). After staining, the samples were washed twice with PBS. The lipids were then extracted from the cells with 100% isopropanol and gentle shaking for 5 min. The concentrations of lipids were measured according to the absorbance at 510 nm. The quantity of lipid in each sample was measured in triplicate. The experiments were performed in triplicate.
Osteogenesis and Measurement
On days 0 and 12, ADSCs of both groups were seeded in a 12-well plate at a density of 1 × 104 and grown with osteogenic medium [DMEM supplemented with 10% FBS, 0.1 μmol/L dexamethasone, 10 mmol/L β-glycerol phosphate (Sigma-Aldrich), and 50 μmol/L ascorbate] that was changed every 3 days. Cells were allowed to grow for 21 days and stained with Alizarin red (Sigma-Aldrich). For quantification of staining, 800 μl of 10% (v/v) acetic acid was added to each well, and the plate was incubated at room temperature for 30 min with shaking. The monolayer, now loosely attached to the plate, was then scraped from the plate using a cell scraper (Fisher Scientific, Hampton, NH, USA) and transferred with 10% (v/v) acetic acid to a 1.5-ml microcentrifuge tube with a wide-mouth pipette. After 30 s of vortexing, the slurry was overlaid with 500 μl of mineral oil (Sigma-Aldrich), heated to exactly 85°C for 10 min, and transferred to ice for 5 min. The slurry was then centrifuged at 20,000 × g for 15 min, and 500 μl of supernatant was removed and transferred to a new 1.5-ml microcentrifuge tube. Then 200 μl of 10% (v/v) ammonium hydroxide was added to neutralize the acid. Aliquots (150 μl) of the supernatant were read in triplicate at 405 nm in a 96-well format using opaque-walled, transparent-bottomed plates (Fisher Scientific). The experiments were performed in triplicate.
Chondrogenesis and Measurement
On days 0 and 12, 1 × 105 ADSCs from both groups were seeded in a 12-well plate and were grown in chondrogenic media consisting of DMEM, 10% FBS, 10 ng/ml transforming growth factor-β1 (TGF-β1), 50 μg/ml of ascorbic acid-2-phospate, and 6.25 μg/ml of insulin, and the media were changed every 3 days. Cells were incubated with the chondrogenic media at 37°C with 5% CO2 for 3 weeks. After fixing in paraformaldehyde (PFA), cells were mounted on slides and stained using standard Alcian blue (Fluka; Chemie GmbH, Buchs, Switzerland) protocols. To quantify incorporation of Alcian blue into the proteoglycan-rich extracellular matrix, cultures were incubated with 6 M guanidine hydrochloride (Sigma-Aldrich) overnight and subjected to photometric measurement at an OD of 595 nm 18 . The experiments were performed in triplicate.
Protein-Protein Interaction Network Analysis for the Effect of Cisplatin on ADSC Mesoderm Differentiation
To investigate whether cisplatin affects mesoderm differentiation of ADSCs, the protein-protein interaction network was evaluated using the STRING v9.1 program [European Molecular Biology Lab (EMBL), Heidelberg, Germany] 19 . The osteogenesis-related protein alkaline phosphatase (ALP), chondrogenesis-related protein aggrecan (ACAN), and adipogenesis-related protein, fatty acid-binding protein 4 (FABP4) were selected to be inputted into STRING v9.1. Additionally, cisplatin was reported to influence many apoptosis signaling pathway proteins, including tumor protein P53 (TP53), BCL2-αssociated X protein (BAX), BCL2, and FAS, which were also inputted into STRING v9.1 for establishing the interaction between cisplatin-influenced proteins and mesoderm differentiation-associated proteins.
Results
High-Concentration Cisplatin Affects ADSC Viability
For the dose-dependent study, the survival of ADSCs treated with 0, 2.5, 5, 10, and 20 μmol/L of cisplatin for 3 days was studied. The XTT assay showed reducing survival in both ASC01 and ASC02 only in a high concentration of cisplatin (10 μmol/L) (Fig. 2A), yielding IC50 of 17.32 and 14.53 μmol/L, respectively.
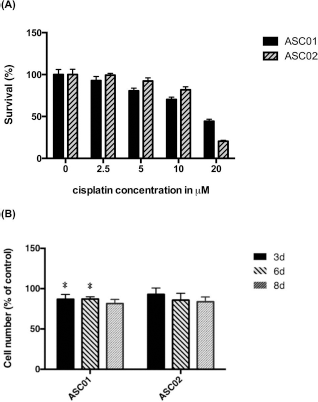
Survival and proliferation of two different ADSCs after cisplatin treatment. (A) With the increase in cisplatin concentrations, the percentage of viable cells decreased. The half maximal inhibitory concentration (IC50) was 17.53 μmol/L in ASC01 and 14.32 μmol/L in ASC02. (B) After treatment with 2 μmol/L cisplatin for 3 days, ADSCs showed decreased proliferation ability. Error bars represent standard deviation (n = 3). *p < 0.05 compared with control.
ADSC Proliferation Decreased Immediately After Cisplatin Treatment
To evaluate the effect of cisplatin treatment on ADSCs, the growth of both ADSCs treated with 2 μmol/L cisplatin for 3 days and then cultured for another 5 days was evaluated by the XTT assay. On days 3, 6, and 8, the numbers of ASC01 cells after cisplatin treatment were 86.9 ± 6.0%, 87.1 ± 2.8%, and 81.6 ± 5.1% of those without cisplatin treatment, and the numbers of ASC02 cells were 93.0 ± 7.8%, 85.7 ± 8.5%, and 83.9 ± 5.8% of those without cisplatin treatment, respectively (Fig. 2B). Compared with cells without cisplatin treatment, ASC01 showed a statistically significant decrease in proliferation ability on days 3 and 6 after cisplatin treatment.
ADSCs Maintained Their Surface Marker Expressions After Cisplatin Treatment
To investigate the effect of cisplatin on the surface marker expressions of ADSCs, the MSC phenotype of ADSCs after treatment with 2 μmol/L cisplatin for 3 days was examined. The flow cytometry study of ADSCs demonstrated an identical MSC phenotype of positive CD44, CD73, CD90, CD105, and HLA-ABC, but negative CD34, CD45, and HLA-DR, whether with or without cisplatin treatment (Fig. 3).

Immunophenotypic profile of ADSCs with and without cisplatin treatment. The ADSCs expressed CD44, CD73, CD90, CD105, and human leukocyte antigen (HLA)-ABC, but did not express CD34, CD45, and HLA-antigen D related (DR), regardless whether treated with or without 2 μmol/L cisplatin for 3 days.
Cisplatin Affects Differentiation Capability of ADSCs
To investigate the effect of cisplatin on the differentiation ability of ADSCs, two ADSC samples (ASC01 and ASC02) from donors without chemotherapy were induced for adipogenic, osteogenic, and chondrogenic differentiation after treatment with 2 μmol/L cisplatin for 3 days in vitro. The cisplatin-treated ADSCs had preserved abilities to differentiate into adipocytes, osteocytes, and chondrocytes. However, there were significant decreases in both adipogenic differentiation (p < 0.001) (Fig. 4) and osteogenic differentiation (p < 0.001) (Fig. 5), but a significant increase in chondrogenic differentiation (p < 0.001) (Fig. 6) after cisplatin treatment.
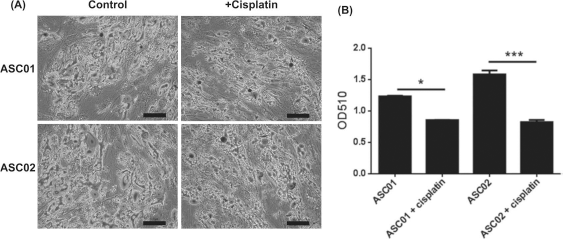
Adipogenic differentiation of ADSCs with and without cisplatin treatment. (A) After adipogenic differentiation, the cells were stained with Oil red O to show lipid droplets. Scale bars: 100 μm. (B) The quantification of adipogenic differentiation ability of both ADSCs significantly reduced after cisplatin treatment. Error bars represent standard deviations (n = 3). *p < 0.05, ***p < 0.001.
Chemotherapy Affects the Differentiation Ability of ADSCs
The therapeutic plan for cancer patients may include radiotherapy, chemotherapy, and surgical intervention. The chemotherapy regimens are usually a combination of several chemotherapy drugs targeting different pathways and are repeatedly given to patients over months and years 20 . Therefore, a single treatment of cisplatin to ADSCs in vitro may not represent the clinical condition. Instead, ADSCs (ASC04) from a donor who had received cisplatin-based chemotherapy 4 months ago were employed to investigate the effect of chemotherapy on the differentiation abilities of ADSCs in vivo. After adipogenic, osteogenic, and chondrogenic differentiation, ADSCs from the donor who had received chemotherapy demonstrated significantly decreased adipogenic differentiation (p < 0.001) but increased osteogenic differentiation compared with those from the donor who had not undergone chemotherapy (ASC03) (p < 0.001). No significant difference was found in chondrogenesis (Fig. 7).
Cisplatin Affected Mesoderm Differentiation
According to the analysis of protein–protein interaction, the adipogenesis-related protein, FABP4, only interacts with EP300 (Fig. 8A). A strong interaction of EP300 with TP53 indicated that the apoptosis-related protein TP53 may be involved in the regulation of adipogenesis. The osteogenesis-related protein ALP directly interacts with BAX, EP300, CDKN2A, and KAT2B (Fig. 8B). BAX, EP300, and CDKN2A showed strong interactions with TP53. This interaction prediction indicated that cisplatin-induced TP53 and BAX may influence the ALP expression to change osteogenesis differentiation. ACAN directly interacts with BCL2, TP53, and EP300 (Fig. 8C). The direct interaction of ACAN and TP53 showed that cisplatin-induced apoptosis may result in a change in chondrogenic differentiation of ADSCs. In addition, we found that three mesoderm differentiation-associated proteins have the same interaction with histone acetyltransferase P300 (EP300). Also, EP300 also shows a direct interaction with TP53 (Fig. 8). The interaction between TP53 and EP300 may play a crucial role in explaining how cisplatin treatment can affect mesoderm differentiation.

Osteogenic differentiation of ADSCs with and without cisplatin treatment. (A) After osteogenic differentiation, the cells were stained with Alizarin red to show calcific deposition. Scale bars: 100 μm. (B) The quantification of osteogenic differentiation ability of both ADSCs significantly reduced after cisplatin treatment. Error bars represent standard deviations (n = 3). ***p < 0.001.
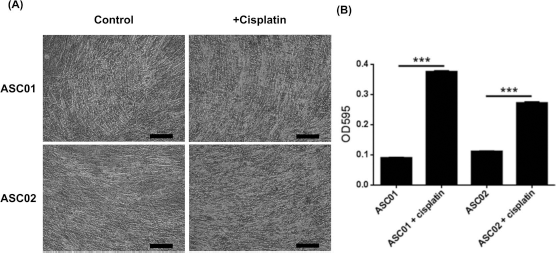
Chondrogenic differentiation of ADSCs with and without cisplatin treatment. (A) After chondrogenic differentiation, the samples were stained with Alcian blue to show glycosaminoglycans in cartilage. Scale bars: 100 μm. (B) The quantification of chondrogenic differentiation ability of both ADSCs significantly increased after cisplatin treatment. Error bars represent standard deviations (n = 3). ***p < 0.001.
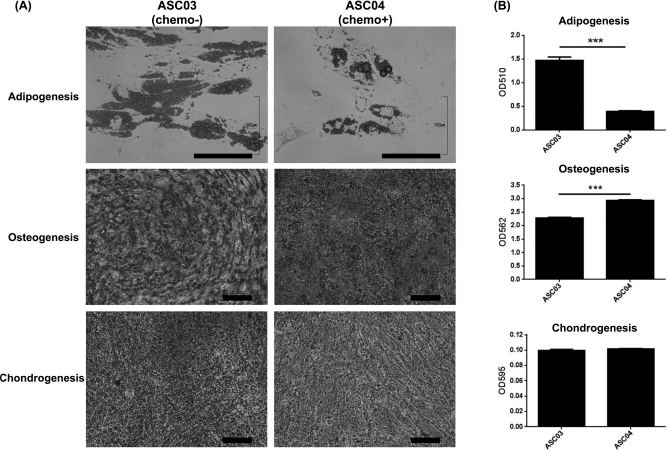
Adipogenic, osteogenic, and chondrogenic differentiation of ADSCs from donors who had or had not received chemotherapy (ASC03/ASC04). (A) After adipogenic, osteogenic, and chondrogenic differentiation, the samples were stained with Oil red O, Alizarin red, and Alcian blue, respectively. Scale bars: 100 μm. (B) The quantification of various differentiations of ASC03 and ASC04. ADSCs from the patient who had received chemotherapy demonstrated decreased adipogenic differentiation but increased osteogenic differentiation. Error bars represent standard deviations (n = 3). ***p < 0.001.

Interaction networks of mesoderm differentiation-related proteins from the STRING program. Interactions are determined by neighborhood, gene fusion, co-occurrence, coexpression, experiments, databases, text mining, and homology. (A) Fatty acid-binding protein 4 (FABP4) and the cisplatin-influenced proteins. (B) Alkaline phosphatase (ALP) and the cisplatin-influenced proteins. (C) ACAN and the cisplatin-influenced proteins. ACAN, chondrogenesis-related protein aggrecan; BAX, BCL2-associated X protein; KAT2B, K (lysine) acetyltransferase 2B; EP300, histone acetyltransferase P300; TP53, tumor protein P53; CDKN, cyclin-dependent kinase inhibitor; BCL2L11, BCL2 like 11; SIRT1, sirtuin 1; FADD, FAS associated via death domain.
Discussion
This study found that cisplatin in high concentration inhibited ADSC viability and proliferation, but ADSCs maintained their hallmark surface markers after treatment with a clinically relevant concentration of cisplatin 21 . Furthermore, after cisplatin treatment, ADSCs had decreased adipogenesis and osteogenesis but increased chondrogenesis in vitro. ADSCs derived from the patient who received chemotherapy with cisplatin still had decreased adipogenesis but had increased osteogenesis compared with ADSCs derived from the patient who had not received chemotherapy.
Upon entering the cell, cisplatin activates and binds to the DNA and induces apoptosis 20 . It also causes oxidative stress22,23 and modulates mitogen-activated protein kinase (MAPK), c-Jun N-terminal kinases (JNKs), and AKT (protein kinase B) signaling pathways24–28, which are involved in the differentiation of ADSCs29–32.
Nicolay et al. reported that cisplatin treatment does not abrogate the differentiation potential of ADSCs 21 . They found that adipogenic differentiation was inhibited after treatment with 1,000 ng/ml cisplatin for 4 h, but osteogenic differentiation was not affected. This study reported inhibition of adipogenic and osteogenic differentiation but enhancement of chondrogenic differentiation in vitro. The discrepancies in results may be attributable to the differences in ADSCs, dose, and duration of treatment. However, both studies showed inhibition of adipogenic differentiation.
The most important finding of this study was a significant reduction in the adipogenic differentiation of ADSCs from the patient who had received cisplatin-based chemotherapy. However, the osteogenic and chondrogenic differentiation were not comparable to the in vitro findings. This phenomenon may be due to the chemotherapy protocol being a combination of multiple chemotherapy drugs repeatedly given to the patient. Most importantly, chemotherapy was found to affect the functions of ADSCs in humans. As cancer progresses, the majority of cancer patients experience body weight loss and fat depletion, which may result from increased lipolysis and fat oxidation, decreased lipogenesis, impaired lipid deposition, and adipogenesis 33 . Chemotherapeutic drugs may further deteriorate the adipogenesis of fat tissue through inhibiting adipogenic differentiation of ADSCs.
Regarding the effect of cisplatin on osteogenesis, Nicolay et al. reported that there was no difference in MSCs after treatment with cisplatin 21 . Xu et al. also reported a case without interfering osteogenesis after cisplatin treatment 34 . Nevertheless, Stine et al. indicated that cisplatin inhibited bone healing during distraction osteogenesis 35 . Ehrhart et al. also reported a modest histomorphometric change after cisplatin treatment did not cause clinical complication in dogs 36 . Our study also demonstrated that two ADSC lines decreased osteogenesis after cisplatin treatment. Nevertheless, the ADSCs derived from a patient who received chemotherapy with cisplatin showed increased osteogenesis. This phenomenon of increased osteogenesis after chemotherapy may be attributable to the ability of ADSCs to recover osteogenic capability after a period of time after finishing chemotherapy with cisplatin.
Regarding the effect of age on ADSCs, our previous study showed that donor age did not affect the proliferation and differentiation capacities of ADSCs 5 . However, Maredziak et al. reported that age had a negative influence on viability, proliferation, and the differentiation potential of ADSCs 37 . Therefore, the effect of age on ADSCs remains inconclusive 38 . With respect to gender, there are minimal observed changes in ADSC proliferation and differentiation 39 . Yang et al. found variable characteristics of ADSCs among donors of different ages and genders 40 . In this study, although ASC01 and ASC02 were from patients of different ages and sexes, their proliferation and differentiation capacities were comparable. In ADSCs derived from the young woman who had received chemotherapy (ASC04), the proliferation and differentiation were compromised compared with ASC03 derived from the older patient. Therefore, the present results suggested a minimal effect of age and gender on ADSC proliferation and differentiation.
There are many possible applications of MSCs in cancer therapy. Drug-targeting therapy is being developed according to the homing effect of MSCs on tumor niches41,42. On the other hand, MSCs can also migrate to the damaged tissues 43 . Therefore, transplantation of MSCs can help repair damaged organs to restore their functions 44 . ADSCs are ideal candidates for stem cell therapy because they are abundant in adipose tissue and can still be isolated from cancer patients receiving chemotherapy. Since cisplatin had a side effect of nephrotoxicity, ADSCs isolated from patients who received cisplatin could rescue the kidney injury by the differentiation45–47 or anti-inflammatory effects45,48.
Cisplatin is able to induce apoptosis via the p53-dependent pathway. The previous study reported that EP300/CREB-binding protein (CBP) could bind to p53 and modulate the p53 signaling pathway 49 . Thus, cisplatin may cause the interaction between EP300 and p53 to change the mesoderm differentiation abilities of ADSCs. There are many investigations in which the epigenetic mechanism involved in cartilage differentiation has been investigated using chondrocytes or relevant cell lines. Histone acetylation is among the epigenetic mechanisms that have been reported to be involved in cartilage-specific gene expression. EP300, an enzyme possessing histone acetyltransferase (HAT) activity, was observed in several studies. Tsuda et al. demonstrated that sex-determining region Y-box 9 (Sox9) associated with CBP/EP300 via its carboxyl termini activation domain and functions as an activator for cartilage tissue-specific gene expression during chondrocyte differentiation 50 . EP300/CBP acts as a coactivator to cartilage homeoprotein-1 (Cart1) through acetylation of the conserved lysine residue adjacent to the homeodomain 51 . Cart1 is induced selectively in chondrocyte lineage during embryonic development 52 . Additionally, EP300 and EP300/CBP-associated factor (PCAF) were reported to promote osteogenic differentiation of C2C12 and MC3T3 cells through acetylation of runt-related transcription factor 2 (Runx2) 53 . Moreover, Badri et al. demonstrated the importance of tension-induced/inhibited proteins (TIPs) in the recruitment of histone acetyltransferase (EP300) to specific promoters and the regulation of EP300 HAT activity through the involvement of the switching- defective protein 3, adaptor 2, nuclear receptor corepressor, and transcription factor IIIB (SANT) domain 54 . They also identified TIP-6 as a new member of the adipogenic cascade. Thus, cisplatin-induced p53 activation may interplay with EP300 to change adipogenic, osteogenic, and chondrocytic differentiation of ADSCs.
In conclusion, ADSCs maintain their MSC characteristics after cisplatin treatment, but their differentiation capabilities are affected in vitro and, possibly, in vivo. Hence, for cancer patients receiving chemotherapy, the differentiation abilities of ADSCs should be evaluated before ADSC transplantation for repairing cisplatin-induced tissue damage.
Footnotes
Acknowledgment
This work was supported by grants from the Intramural Research Project of Buddhist Tzu Chi General Hospital (TCRD 104–07). Y.-H.C.: conception of the study, experimental design and execution of experiments, and manuscript writing; H.-W.L., T.-Y.C., T.-Y.W., and R.-K.T.: critical discussion of the data and manuscript; D.-C.D.: experimental design and manuscript writing. The authors declare no conflicts of interests.
