Abstract
Successful subretinal transplantation is limited by considerable early graft loss despite pharmacological suppression of adaptive immunity. We postulated that early innate immune activity is a dominant factor in determining graft survival and chose a nonimmunosuppressed mouse model of retinal pigment epithelial (RPE) cell transplantation to explore this. Expression of almost all measured cytokines by DH01 RPE cells increased significantly following graft preparation, and the neutrophil chemoattractant KC/GRO/CINC was most significantly increased. Subretinal allografts of DH01 cells (C57BL/10 origin) into healthy, non immunosuppressed C57BL/6 murine eyes were harvested and fixed at 1, 3, 7, and 28 days postoperatively and subsequently cryosectioned and stained. Graft cells were detected using SV40 large T antigen (SV40T) immunolabeling and apoptosis/necrosis by terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL). Sections were also immunolabeled for macrophage (CD11b and F4/80), neutrophil (Gr1 Ly-6G), and T-lymphocyte (CD3-∊) infiltration. Images captured with an Olympus FV1000 confocal microscope were analyzed using the Imaris software. The proportion of the subretinal bolus comprising graft cells (SV40T+) was significantly (
Keywords
Introduction
Retinal degenerative diseases, including neovascular and nonneovascular age-related macular degeneration, are the most common causes of blindness in the developed world 1 . Anti-vascular endothelial growth factor (anti-VEGF) agents prevent loss of vision and improve visual acuity in patients with neovascular degeneration2–4. However, no effective treatment exists for nonneovascular degeneration, which is more common 5 . Transplantation of cells into the subretinal space (SRS) to replace degenerating or dysfunctional cells has been proposed as a treatment strategy 6 and rescues anatomical features and vision in animal models of retinal degenerative disease7–22 as well as affected human patients23–25.
Rapid loss of grafted cells and decreases in anatomical and functional benefits following transplantation26,27 remain major challenges. This occurs despite suppression of the T-lymphocyte-mediated adaptive immune response with drugs such as cyclosporine and azathioprine. These immunosuppressive regimes still have graft survival rates of just 11% at 4 weeks28,29 and 0.2% at 28 weeks 28 . Indeed, rabbit allograft failure is not associated with lymphocyte infiltration and is not altered by cyclosporine immunosuppression30,31.
Poor graft survival in the SRS is paradoxical, as this site is characterized by a suppressed antigen-specific adaptive immune response32–34. Retinal pigment epithelial (RPE) cells express the CD95 ligand that induces T-cell apoptosis, secrete transforming growth factor-β (TGF-β), which is important in the induction of tolerance 35 , and can also phagocytose T lymphocytes 36 . We hypothesized that cells transplanted into the immune-deviant SRS are destroyed by an alternate immune mechanism rather than via the classic T-lymphocyte-mediated immunological rejection.
Innate immune responses are characterized by upregulation of proinflammatory cytokines 37 and a coordinated local and systemic inflammatory response 38 . A critical role for innate immunity in acute allograft rejection is now emerging39–41 and has been shown to be crucial in skin 42 and pancreatic islet 43 allograft rejection. However, the role of innate immunity in subretinal transplantation has not been studied. We hypothesized that subretinal graft failure is a consequence of innate immune mechanisms and used a mouse RPE allograft model to investigate this. The purpose of this study was to examine the mechanisms underlying early allograft cell loss in the SRS rather than to achieve a structural or functional benefit following transplantation. Accordingly, we used healthy nonimmunosuppressed hosts to investigate our hypothesis.
Materials and Methods
Ethics Statement
Ethical approval for this research was obtained from Animal Research Ethics Committee (AREC-P-07–09-Keegan). All procedures involving the use of mice were performed in accordance with the Association for Research in Vision and Ophthalmology (ARVO) Statement for the Use and Care of Animals in Ophthalmic and Vision Research, and all efforts were made to minimize suffering.
Cell Line Derivation
The DH01 RPE line was prepared from RPE cultured from a healthy C57BL/10.RIII-H-2r mouse as previously described 44 and immortalized using supernatant from the SVU 19.5 cell line 45 secreting retrovirus encoding a temperature-sensitive non-SV40 origin-binding U19 mutant of the SV40T antigen and the neomycin resistance gene 46 .
Cell Culture
DH01 cells were routinely cultured in high-glucose Dulbecco's modified Eagle's medium (DMEM) (Sigma-Aldrich Ireland, Wicklow, Ireland) supplemented with 1% fetal calf serum (FCS; Sigma-Aldrich Ireland), 200 mM L-glutamine (Sigma-Aldrich Ireland), and 5 mg/ml penicillin/streptomycin (Sigma-Aldrich Ireland). As these cells harbor SV40T, they were grown at 33°C in 5% CO2.
Transplant Conditions (TC) and Baseline Conditions (BC)
TC cells were prepared as a highly concentrated cell suspension (50,000 cells/μl), deprived of serum (suspended in serum-free medium), and kept on ice for the maximum period that typically exists between harvesting and transplanting the graft (4 h). To determine the effect of graft cell suspension preparation on cytokine production, we compared cytokine expression by DH01 cells immediately following resuspension in full media (BC) and after being prepared for subretinal transplantation (TC).
Preparation of Samples for Cytokine Quantification
Cytokine production by DH01 was quantified under BC and following the stress of the graft cell suspension preparation procedure (TC). Cells were cultured in full medium, trypsinized (Sigma-Aldrich Ireland), and seeded in triplicate in 24-well culture plates with 2×105 cells/well to yield confluent cultures. BC cells were seeded in 0.5 ml of serum-free medium. TC cells were subjected to the process involved in the preparation of a graft cell suspension as described above and, after 4 h on ice, were seeded in 0.5 ml of serum-free medium. Following culture for 24 h, the medium was removed and stored at −80°C for subsequent analysis.
Multiplex Cytokine Assay
A nine-plex multispot T helper 1 (TH1)/TH2 cytokine assay kit [K15013B-1; Meso Scale Discovery (MSD), Rockville, MD, USA] was used according to the manufacturer's instructions to quantify the following cytokines: interferon-γ (IFN-γ), tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), IL-2, IL-4, IL-5, IL-10, IL-12, and keratinocyte chemoattractant/growth regulated oncogene-α/cytokine-induced neutrophil chemo attractant (KC/GRO/CINC). An 8-point calibration curve from 0 to 10,000 pg/ml was constructed using a plot of signal intensity from a series of known concentrations of the multiplex standard provided by the kit manufacturer. Cytokine levels were calculated using the MSD Sector Imager 2400 and Discovery Software 2.0 (MSD). Concentrations were determined in triplicate and expressed in pg/ml.
IL-6 Enzyme-Linked Immunosorbent Assay (ELISA)
IL-6 was not included on the MSD TH1/TH2 multiplex kit. Therefore, expression of this cytokine was measured separately using an IL-6 ELISA kit (KMC0061; Invitrogen, Carlsbad, CA, USA). A 6-point calibration curve from 0 to 500 pg/ml was generated from standard mouse IL-6 reconstituted in standard diluent buffer. ELISA was carried out as per the manufacturer's instructions. Absorbance at 450 nm was measured using a spectrophotometer (SpectraMax M2; Molecular Devices, Sunnyvale, CA, USA). Sample IL-6 concentrations were calculated against the graph of the standard curve. Concentrations were determined in triplicate and expressed in pg/ml.
Subretinal RPE Cell Transplantation
Male C57BL/6 mice were obtained from Harlan (Bicester, UK). All surgery was performed under ketamine (Ketalar; 75 mg/kg; C&M Vetlink, Limerick, Ireland) and medetomidine (Domitor; 0.5 mg/kg; C&M Vetlink) anesthesia, and the mice were recovered with atipamezole (Antisedan; 1 mg/kg; C&M Vetlink). Euthanasia was with sodium pentobarbitone (Euthatal; 140 mg/kg; C&M Vetlink) followed by cervical dislocation. All drugs were delivered by intraperitoneal (IP) injection, with the exception of atipamezole, which was delivered by subcutaneous injection.
DH01 (C57BL/10 origin) cell suspensions (50,000 cells/μl DMEM) were prepared from preconfluent cultures as described under TC above. We have previously demonstrated that preparing DH01 cell suspensions in this manner results in 95% graft cell viability at the time of transplantation
44
. Subretinal transplants were delivered transsclerally through glass microinjection pipettes (BioMedical Instruments, Zollnitz, Germany) to the dorsotemporal SRS of healthy nonimmunosuppressed C57BL/6 mice. Eyes received 2 μl of the graft cell suspension (
Graft Detection (SV40T), TUNEL Labeling, and Identification of the Host Immune Response to Subretinal RPE Transplants
To examine temporal graft survival, graft cells were identified using a specific primary antibody to SV40T (SC-20800; 1:100; Santa Cruz Biotechnology, Dallas, TX, USA) and goat anti-rabbit Texas Red-labeled secondary antibody (111-075-003; 1:100; Jackson Immuno Research Laboratories, West Grove, PA, USA). DNA strand breaks were detected by terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) as previously described 47 .
To examine the host immune response to subretinal DH01 allografts, cryosections were immunolabeled for the SV40T antigen (SC-20800; 1:100; Santa Cruz Biotechnology) using a donkey anti-rabbit fluorescein isothiocyanate (FITC)-labeled secondary antibody (711–095–152; 1:100; Jackson ImmunoResearch Laboratories). In addition, sections were immunolabeled to detect macrophages (CD11b and F4/80), neutrophils (Gr1 Ly-6G), or T lymphocytes (CD3-∊). Rat anti-mouse CD11b (MCA711; 1:100; AbD Serotec, Oxford, UK), rat anti-mouse F4/80 (MCA 497EL; 1:25; AbD Serotec), and rat anti-mouse Gr1 Ly-6G (MAB1037; 1:100; R&D Systems, Minneapolis, MN, USA) primary antibodies were secondarily immunolabeled using donkey anti-rat tetramethylrhodamine (TRITC)-labeled secondary antibody (712–025–150; 1:50; Jackson ImmunoResearch Laboratories). Goat anti-mouse CD3-∊ (sc-1127; 1:100; Santa Cruz Biotechnology) was secondarily immunolabeled using donkey anti-goat TRITC-labeled secondary antibody (705–025–003; 1:100; Jackson ImmunoResearch Laboratories).
Nonspecific secondary antibody binding was blocked using serum (1:50) from the host species of the secondary antibody. 4,6-Diamidino-2-phenylindole (DAPI; 10 ng/ml; D9542; Sigma-Aldrich Ireland) counterstaining was used to enable visualization of nuclei. Phosphate-buffered saline (PBS; Sigma-Aldrich Ireland) was used to dilute all reagents and for three 5-min washes between steps. After the final wash, sections were mounted using Vectashield® HardSet™ (Vector Laboratories, Peterborough, UK).
Confocal Microscopy
Immunolabeling was visualized using an Olympus (Tokyo, Japan) FluoView™ FV1000 confocal laser scanning microscope. Differential interference contrast microscopy (DIC) images were taken at the time of fluorescence confocal microscopy to more accurately identify the SRS. Z-stack images were taken through areas of interest to enable three-dimensional (3D) image reconstruction using an image analysis software as described below. Captured images were viewed using Olympus Fluoview Ver. 1.4a software.
Image Analysis
Four transplanted eyes and four sham-treated eyes were examined for each postoperative time point. Four unoperated eyes were also examined. In order to maintain consistency in analyses of transplanted eyes, cryosections through the center of the subretinal cell bolus where the greatest numbers of cells were present were used for all eyes. For sham-treated eyes, cryosections in the region of the injection site were used. All sections were immunolabeled for SV40T to identify transplanted cells and counterstained with DAPI to label all nuclei. Sections were also immunolabeled to detect one of the following: DNA nicks (TUNEL), macrophages (CD11b and F4/80), neutrophils (Gr1 Ly-6G), or T cells (CD3-∊).
Single optical sections from four transplanted eyes and four sham-treated eyes per time point, in addition to four unoperated control eyes, were analyzed using the Imaris image analysis software (Bitplane AG, Zurich, Switzerland). For sections fluorescently labeled to detect both SV40T and TUNEL, the following counts of cells in the SRS were performed: total number of cells (DAPI+), number of graft cells (DAPI+/SV40T+), and number of graft cells with DNA nicks (DAPI+/SV40T+/TUNEL+). As the total number of cells in the SRS varied between images, the proportion of the subretinal cell bolus comprising grafted cells was calculated as SV40T+ cells per 100 cells (DAPI+) in the SRS. The proportion of graft cells (SV40T+) that had DNA nicks (SV40T+/TUNEL+) was also calculated.
As immune cell infiltration of the SRS was identified by cell-specific surface markers (CD11b, F4/80, Gr1 Ly-6G, and CD3-∊), it was not possible to accurately estimate cell number. Therefore, sections immunolabeled for these markers in conjunction with the graft cell marker (SV40T) were analyzed by calculating the area of immunolabeling using Imaris. To correct for differing sizes of subretinal boluses, the numbers of nuclei (DAPI) in the SRS were also counted, and the areas of immunolabeling were expressed as μm2/100 nuclei in the SRS. This enabled determination of the temporal change in expression of the graft cell marker (SV40T) relative to markers of infiltrating immune cells.
The proportion of the area of SV40T+ immunolabeling that colabeled with immune cell markers (CD11b, F4/80, Gr1 Ly-6G, and CD3-∊) was also calculated using Imaris. This allowed determination of altered temporal colabeling of the graft cell marker with markers of infiltrating cells.
Z-stack confocal images of selected regions of SV40T colabeling with immune cell markers were processed using Imaris and reconstructed in 3D form using isosurface rendering to determine the nature of the relationship between colabeling markers at a cellular level.
Statistical Analysis
Data were analyzed using GraphPad Prism 5 statistical software (GraphPad, San Diego, CA, USA). Data are expressed as mean±standard deviation (SD), and a value of
Comparisons of Cytokine Levels.
Student's
Comparisons of Cell Numbers in the SRS at Different Postoperative Time Points.
At POD 1, 3, 7, and 28, counts of cells in the SRS were performed to determine the total number of cells, the proportion of cells expressing SV40T, and the proportion of SV40T+ cells that were also TUNEL+. One-way analysis of variance (ANOVA) followed by all pairwise post hoc
Analysis of Areas of Immunolabeling
The areas of immunolabeling and the total numbers of nuclei in the SRS were calculated for each immunolabeling combination at POD 1, 3, 7, and 28. Thus, areas of immunolabeling/100 nuclei were determined. To assess changes in marker expression between time points, areas of immunolabeling/100 nuclei were treated as normally distributed and analyzed with ANOVA and Tukey's post hoc test.
Analysis of Immunofluorescence Colabeling.
The proportions of SV40T immunolabeling that colabeled with immunolabeled infiltrating cells were calculated using Imaris. The colabeling proportions are likely not normally distributed and were analyzed with nonparametric methods, Kruskal-Wallis test, and Dunn's post hoc comparisons.
Results
Cytokine Expression Increases Significantly Following Graft Preparation
Cytokine expression by the mouse RPE cell line DH01 was quantified under BC and TC prior to transplantation (Table 1). With the exception of IL-4, expression of all cytokines increased significantly (
DH01 Cytokine Expression Under BC and TC

Values represent pg/ml and are expressed as mean ± standard deviation (SD).
Retinas from Unoperated and Sham-Treated Eyes of C57BL/6 Mice Contain F4/80+ Cells and Few TUNEL+ Cells, But do not Contain CD11b+, Gr1 Ly-6G+, or CD3-∊+ Cells
To establish the baseline expression of markers of interest in the C57BL/6 mouse retina, cryosections from unoperated eyes (

Retinas from unoperated and sham-treated eyes of C57BL/6 mice contain F4/80+ cells and few terminal deoxynucleotidyl transferase dUTP nick-end labeling-positive (TUNEL+) cells, but do not contain SV40T+, CD11b+, Gr1 Ly-6G+, or CD3-∊+ cells. Representative confocal microscopy images of sections from unoperated and sham-treated eyes immunolabeled for SV40 large T antigen (SV40T) (Texas Red: a—e; fluorescein isothiocyanate (FITC), green: f–y) to identify background expression of the graft cell marker and either TUNEL (FITC, green) to identify DNA strand nicks in apoptotic and necrotic cells (a–e) or immunolabeled for the immune cell surface markers CD11b (f–j), F4/80 (k–o), Gr1 Ly-6G (p–t), or CD3-∊ (u–y) [all tetramethylrhodamine (TRITC), red]. All nuclei were counterstained with 4,6-diamidino-2-phenylindole (DAPI, blue). Unoperated and sham-treated retinas had F4/80+ cells in the inner retina. Low background levels of TUNEL+ cells were evident in the inner and outer nuclear layers, particularly on postoperative days (POD) 1 and 3. SV40T+, CD11b+, Gr1 Ly-6G+, or CD3-∊+ cells were not observed. Scale bar: 50 μm.
The unoperated retina was characterized by the presence of F4/80+ cells, particularly in the ganglion cell layer (Fig. 1k). Low background levels of TUNEL+ cells were also present in the retina (Fig. 1a). SV40T, CD11b, Gr1 Ly-6G, or CD3-∊ labeling was not observed, and no cells were present in the SRS.
Sham-treated eyes did not demonstrate SV40T immunolabeling but had low levels of TUNEL+ cells in the inner and outer nuclear layers of the retina (Fig. 1b-e). At all postoperative time points, sham-operated eyes did not have cells in the SRS, and the retina did not stain for CD11b, Gr1 Ly-6G, or CD3-∊. However, F4/80+ cells were seen in retinas of sham-operated eyes, particularly in the ganglion cell layer in a pattern analogous to that observed in unoperated eyes (Fig. 1l–o).
The First Week Posttransplantation Is Characterized by Rapid Loss of DH01 RPE Allograft Cells But Low Levels of Graft Apoptosis and Necrosis
We sought to ascertain whether apoptosis or necrosis was a predominant mechanism of graft failure in the early postoperative period. DH01 (C57BL/10 origin) cell suspension allografts (in serum-free medium) were transplanted into the SRS of C57BL/6 mice. Subretinal DH01 allografts were examined at POD 1, 3, 7, and 28 (
Subretinal DH01 allograft cells were distinguishable from host cells by SV40T immunolabeling (Fig. 2A). At POD 1 and POD 3, most cells in the subretinal bolus were identified as graft cells by the expression of the SV40T in the nucleus (Fig. 2Aa and b). By POD 7, there was a dramatic reduction in the proportion of cells in the subretinal bolus expressing SV40T (Fig. 2Ac). TUNEL labeling demonstrated that few cells in the subretinal bolus had DNA strand nicks at each time point. No graft cells were identified at POD 28 (Fig. 2Ad). Few cells in the nuclear layers of the retina were TUNEL+ following subretinal cell transplantation (Fig. 2Aa–d). This observation relates to the retinal detachment induced to place the graft and was also evident in sham-treated eyes (Fig. 1b–e).
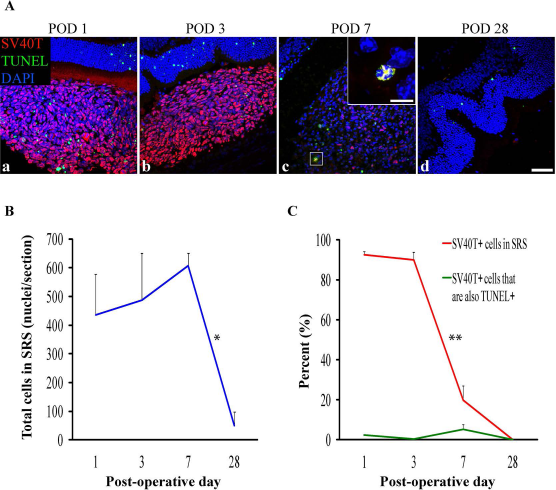
DH01 retinal pigment epithelial (RPE) cells allografted to the subretinal space (SRS) of healthy nonimmunosuppressed C57BL/6 mice are lost by POD 28. (A) Representative confocal microscopy images of sections immunolabeled for SV40T (Texas Red) to identify transplanted cells and TUNEL (FITC, green) to identify DNA strand nicks in apoptotic and necrotic cells. All nuclei were counterstained with DAPI (blue). At POD 1 (a) and POD 3 (b) most cells in the subretinal bolus are graft cells (SV40T+). However, few transplanted cells are seen at POD 7 (c), and no graft cells remain at POD 28 (d). TUNEL labeling in the subretinal cell bolus remains low at each time point. High-power inset at POD 7 (c) shows an example of an apoptotic/necrotic graft cell (DAPI+/SV40T+/TUNEL+). Scale bars: 50 μm, 10 μm [high-power inset in (c)]. (B) There was no significant change in the total number of cells in the SRS between POD 1 (435 ± 143), POD 3 (487 ± 164), and POD 7 (607 ± 45), but the total cell number decreased significantly (*
Cell counts were performed using Imaris image analysis software. The proportion of all cells (DAPI+) identifiable as graft cells (DAPI+/SV40T+) in the SRS was quantified. The proportion of graft cells (DAPI+/SV40T+) with DNA strand nicks (DAPI+/SV40T+/TUNEL+) was also quantified. There was no significant change in the total number of cells in the SRS between POD 1 (435 ± 143), POD 3 (487 ± 164), and POD 7 (607 ± 45), but total cell number decreased significantly (
The Period of Graft Loss Is Characterized by Infiltrating Cells Expressing Markers of Innate Immunity, But Not T lymphocytes
Figure 2 illustrates a significant (
In eyes that received subretinal transplants, the period of graft loss between POD 1 and POD 7 was characterized by infiltration of the SRS by cells expressing markers of innate immunity (Fig. 3A). CD11b and F4/80 are cell surface markers used to identify macrophages, and Gr1 Ly-6G is a cell surface marker used to identify neutrophils. Low levels of infiltrating CD11b+, F4/80+, and Gr1 Ly-6G+ cells were seen at POD 1 and POD 3, with a marked increase evident at POD 7. No graft cells were identified at POD 28. Only one of four eyes at POD 28 had cells remaining in the SRS, and these were F4/80+ (Fig. 3Ah). No T lymphocytes (CD3-∊+) were seen at each time point (Fig. 3Am–p). At POD 28, the neural retina overlying the graft site had folds in the outer nuclear layer but otherwise appeared undamaged (Fig. 3Ad, h, l, and p).

The period of DH01 RPE allograft loss in the SRS of healthy nonimmunosuppressed C57BL/6 mice is characterized by infiltrating cells expressing markers of macrophages (CD11b and F4/80) and neutrophils (Gr1 Ly-6G), but not by T lymphocytes (CD3-∊). (A) Representative confocal microscopy images of sections immunolabeled for SV40T (FITC, green) to identify transplanted cells, and also immunolabeled for either CD11b (a–d), F4/80 (e–h), Gr1 Ly-6G (i–l), or CD3-∊ (m–p) to identify infiltrating cells (all TRITC, red). All nuclei were counterstained with DAPI (blue). At POD 1, the subretinal graft site is primarily composed of SV40T+ graft cells, but by POD 7 the proportion of cells in the subretinal bolus expressing this graft cell marker has considerably decreased. CD11b+ (a–c), F4/80+ (e–y), and Gr1 Ly-6G+ (i–k) cells infiltrate the graft in increasing numbers between POD 1 and POD 7. No graft cells are seen at POD 28 (d, h, l, p). One of four eyes assessed at POD 28 still had cells in the SRS, and these are F4/80+ (h). Graft cell loss is not associated with CD3-∊+ T-lymphocyte infiltration (m–p). Scale bars: 50 μm, 20 μm (high-power insets). (B) Quantitative analysis of the areas of immunolabeling confirms that the period of significant (*
The area of immunolabeling as a function of the number of nuclei present in the SRS was calculated and expressed as μm2/100 nuclei. A significant (
Macrophages and Neutrophils Colabel With, and Engulf, Graft Cells During the First Postoperative Week
Areas of colabeling of the graft cell marker (SV40T) with the innate immune cell markers (CD11b, F4/80, and Gr1 Ly-6G) were apparent on confocal microscopy images. The proportion of SV40T immunolabeling that colabeled with infiltrating immune cell markers on POD 1, 3, 7, and 28 was calculated. CD11b, F4/80, and Gr1 Ly-6G all colabeled with SV40T at significantly (

Colabeling of DH01 allografts with infiltrating CD11b+, F4/80+, and Gr1 Ly-6G+ cells peaks at POD 7 with evidence of graft engulfment by these innate immune cells. Large areas of SV40T were observed to colabel with CD11b+, F4/80+, and Gr1 Ly-6G+ on POD 7. Colabeling of SV40T with the T-lymphocyte marker (CD3-∊) remained low at all time points. Representative images of the immunofluorescence colabeling evident on POD 7 are shown. (A) The highlighted cell (white box) in the center of this image has positive cell membrane immunolabeling for the macrophage marker CD11b (TRITC, red) on POD 7. SV40T (FITC, green) immunolabeling can be seen within this CD11b+ cell, suggesting engulfment of graft by the macrophage. The differential interference contrast (DIC) image also identifies pigment granules within CD11b+ cells. Scale bar: 20 μm. (B) Multiple cells in this image have cell membrane immunolabeling for the macrophage marker F4/80 (TRITC, red) on POD 7. SV40T (FITC, green) immunolabeling can be seen within these F4/80+ cells. Areas of F4/80+/SV40T+ colabeling (orange) suggest graft engulfment by macrophages. The DIC image also identifies pigment granules within F4/80+ cells. Scale bar: 20 μm. (C) Many cells in this POD 7 subretinal graft have immunofluorescence colabeling (orange) for Gr1 Ly-6G (TRITC, red) and SV40T (FITC, green). Scale bar: 10 μm. (D) Sequential single optical sections from the z-stack confocal image through the highlighted (yellow box) Gr1 Ly-6G+ neutrophil on POD 7 is consistent with phagocytosis of SV40T from the neutrophil cell membrane. (E) The z-stack confocal image of the cell highlighted in (C) was also reconstructed in three-dimensional (3D) form, and the red channel was removed from the top portion of the cell to enable visualization inside the cell membrane. The reconstructed image confirms the presence of graft (SV40T, green) inside the neutrophil cell membrane (Gr1 Ly-6G, red). (F) The proportions of SV40T that colabeled with CD11b, F4/80, Gr1 Ly-6G, and CD3-∊ were analyzed. Colabeling of SV40T with all infiltrating immune cell markers was minimal on POD 1 and POD 3. However, there was a significant (*
To better discriminate areas of colabeling, high-power z-stack confocal images were taken through these areas. SV40T immunolabeling was apparent inside the cell membranes of CD11b+ (Fig. 4A) and F4/80+ (Fig. 4B) cells. DIC images also identified pigment granules inside the cell membranes of these cells. Similarly, Gr1 Ly-6G+/SV40T+ colabeling was apparent inside cell membranes of Gr1 Ly-6G+ cells (Fig. 4C–D). Reconstruction of z-stacked confocal images taken through such areas of colabeling confirms SV40T inside Gr1 Ly-6G+ cells (Fig. 4E). The images presented in Figure 4 are consistent with phagocytosis of graft cells by macrophages and neutrophils.
Discussion
We examined cytokine expression by DH01 RPE cells under BC and following preparation of a concentrated graft cell suspension (TC) to determine whether the graft preparation technique might influence the host immune response following transplantation. Graft preparation (TC) increased significantly the expression of almost all cytokines examined (Table 1). Notably, KC/GRO/CINC was expressed at by far the greatest levels and also demonstrated the greatest fold increase (sixfold) in expression following graft preparation. Mouse GRO and the chemoattractant KC are structural and functional homologs of the human chemokine IL-8 48 . IL-8 is a potent neutrophil chemoattractant and stimulates phagocytosis, superoxide radical production, and cytoplasmic degranulation49,50. Thus, production by graft cells of high levels of KC/GRO/CINC in the SRS in the period following transplantation would increase the risk of monocyte migration and inflammation at the graft site. KC/GRO/CINC, or IL-8, has not been identified previously as a potentially significant mediator of the immune response against subretinal grafts.
TC caused a threefold increase in expression of the inflammatory cytokines IL-1β, IL-5, IL-6, and IL-12. While increased expression of IL-1 and IL-6 at the graft site has been reported following human RPE xenografts to the SRS of nondystrophic Royal College of Surgeons (RCS) rats 51 , the effects of these cytokines in subretinal cell transplantation are unknown. IL-1 has many pro-inflammatory effects including mediating a neutrophilic inflammatory response to dying cells in vivo 52 . RPE allografts to rabbits cause elevated IL-6 in the vitreous in the first week postoperatively 53 . IL-6 is a key mediator of allograft rejection, and increased expression is highly correlated with human cardiac allograft rejection 54 . Graft-produced IL-6 promotes T-cell activation and cardiac allograft rejection in the mouse 55 .
DH01 expression of IL-10 increased significantly (2.1-fold increase;
IL-2 supports T-lymphocyte proliferation58–62 and survival
63
, as well as the differentiation of naive T lymphocytes into effector and memory T cells64–66. However, IL-2 also has an immunosuppressive function by promoting regulatory T cells (Tregs) production and homeostasis
67
and plays a fundamental role in immune regulation and tolerance in vivo68–73. While DH01 production of IL-2 increased significantly (2.7-fold increase,
In the context of consistently poor graft survival in the SRS, we sought to examine graft survival in the early postoperative period. We have previously demonstrated that our technique for preparing DH01 graft cell suspensions results in 95% cell viability at the time of transplantation 44 . Expression of SV40T by the DH01 cell line enabled examination of the fate of transplanted cells during the first postoperative month. The proportion of the subretinal bolus composed of grafted cells reduced from 90% on POD 3 to just 20% on POD 7, and no grafted cells survived to POD 28 (Fig. 2).
Our finding of rapid graft loss in the SRS is consistent with other studies. Human RPE cells xenografted into the SRS of cyclosporine-immunosuppressed RCS rats are quickly lost with just 27% survival at POD 7 and 11% at POD 28 28 . Similarly, just 3% of subretinal porcine fetal RPE cells xenografted into rabbits survived to 1 month in immunocompetent animals, and only 10.5% survived in rabbits receiving daily triple systemic immunosuppressive therapy with prednisone, cyclosporine, and azathioprine 29 .
In contrast to solid tissue or organ transplants, cell transplants are commonly derived from cell cultures and delivered as a cell suspension (e.g., stem cells 74 , Schwann cells75,76, pancreatic islets 43 , and RPE cells8,18,20). Transplantation paradigms involving cell suspension grafts have poor rates of graft survival. Human fetal dopaminergic neuronal xenografts to cyclosporine-immunosuppressed rats had only 5%–6% survival 77 , and poor graft survival is also a feature of fetal nigral allografts in humans78,79.
TUNEL labeling identifies apoptotic cells by detecting DNA fragmentation. However, necrosis is also accompanied by DNA breaks, and thus TUNEL labeling also identifies necrotic cells80–82. It is unlikely that the rapid graft cell loss observed in this study was due to apoptosis or necrosis, as rates of TUNEL labeling of grafted cells remained low at all time points (Fig. 2A). Graft cell apoptosis/necrosis was just 2% at POD 1, <1% at POD 3, and 5% at POD 7 (Fig. 2C). We hypothesized that the rapid graft cell loss observed during the first post operative week was a consequence of a host innate immune response against the subretinal allograft.
On POD 1 and POD 3, 90% of cells in the SRS were identifiable as graft cells (SV40T+). However, by POD 7 this figure reduced to just 20% (Fig. 2C). Thus, the remaining 80% of cells at this time must be host derived. We characterized the cellular composition of the subretinal bolus during the first postoperative month and found that macrophages (CD11b and F4/80) infiltrated subretinal grafts on POD 1 and POD 3 and predominated over transplanted cells by POD 7 (Fig. 3Aa–h and B).
There was also a significant increase (
The interaction of graft cells with F4/80+ macrophages in the SRS is particularly interesting because the mouse macrophage F4/80 receptor is known to play a critical role in the generation of antigen-specific efferent Tregs that suppress antigen-specific DTH responses 83 . However, while F4/80+ macrophages may inhibit a T-lymphocytic adaptive immune response, our observations reveal direct engulfment of transplanted cells by F4/80+ macrophages. Macrophages have been reported to associate with subretinal grafts in many host species including the mouse 84 , rat14,26,27, rabbit29–31,85–87, pig 88 , and primate 6 . While these studies reported macrophages associating with subretinal grafts, they did not correlate macrophage infiltration with graft cell loss. The study presented is the first investigation to make this connection.
The observation of positive immunolabeling for F4/80, but not for CD11b, in the ganglion cell layer was unexpected and may be a consequence of the particularly high levels of expression of F4/80 by ameboid ganglion cell layer microglia compared to ramified parenchymal microglia 89 . Although the immunofluorescence techniques employed in this study were insufficient to detect the CD11b antigens expressed by ganglion cell layer microglia, they clearly identified CD11b cells in the SRS.
Zecher et al. described an innate immune response to allografts mediated by macrophages that is independent of natural killer cells and T lymphocytes 90 . Graft survival in the early postoperative period is promoted by macrophage inhibition in cellular transplants of pancreatic islet cells 91 and spleen cells 92 . Macrophage depletion via subconjunctival administration of clodronate liposomes also promotes long-term survival of high-risk corneal grafts 93 . We observed a significant macrophage response in the subretinal graft coinciding with the period of graft cell loss during the first postoperative week. It is likely that perioperative macrophage depletion or inhibition may also prove useful in promoting subretinal graft survival.
Having found that DH01 cells prepared for transplantation expressed large amounts of the neutrophil chemoattractant KC/GRO/CINC, we also investigated whether neutrophils infiltrated the subretinal graft. We used an antibody against Gr1 Ly-6G, which detects the myeloid differentiation antigen Gr1 94 and specifically identifies neutrophils as distinct from monocyte–macrophages via Ly-6G 95 . We found a significant increase in Gr1 Ly-6G+ cells infiltrating the subretinal cell bolus during the first postoperative week (Fig. 3Ai–k and B). Moreover, by POD 7 there was also a significant increase in immunofluorescence colabeling of transplanted cells with this neutrophil cell marker (Fig. 4F). The 3D image reconstruction of areas of colabeling demonstrated the presence of the graft cell marker internal to the neutrophil cell membrane confirming engulfment of graft cells by infiltrating neutrophils (Fig. 4C–E). Neutrophils have not previously been identified in subretinal grafts.
Neutrophils have recently been implicated as mediators of cardiac graft failure. They are the first leukocyte to infiltrate cardiac allografts, arriving within 1 h following transplantation 96 . Neutrophil infiltration also correlates with cardiac allograft rejection severity 97 , and cardiac allograft survival is promoted by neutrophil depletion 98 and inhibition of neutrophil infiltration 99 . Statins inhibit neutrophil transendothelial migration100,101, and when used in patients with cardiac allografts they lower the incidence of cardiac allograft vasculopathy and reduce the severity of allograft rejection102,103. Thus, the use of perioperative statins may also prove beneficial in suppressing neutrophil infiltration and in promoting subretinal graft survival.
T-lymphocyte (CD3-∊) infiltration was not a feature of this study (Fig. 3Am–p and B). T lymphocytes have also been notable by their absence in subretinal transplants to the rat26,27, rabbit29–31,85–87, and pig 88 . This is consistent with the deviant immune environment of the SRS32–34 characterized by a suppressed T-lymphocytic adaptive immune response35,36,104,105. Interestingly, after finding no significant differences between cyclosporine-immunosuppressed and control animals in RPE cell suspension allografts in the rabbit, Crafoord et al. concluded that graft failure was caused by either immunological mechanisms not inhibited by cyclosporine or by nonimmunologic events 31 . The study presented here describes the cellular infiltrate in the SRS at specific time points following subretinal allograft transplantation. It is possible that a T-lymphocyte response was occurring at the regional lymph nodes; however, this possibility was not investigated. Nevertheless, we found that the majority of graft cell loss had occurred by POD 7. This is much earlier than acute allograft rejection mediated by T lymphocytes, which occurs at 10–13 days following transplantation 106 .
Lopez et al. suggested that RPE cells transplanted to the SRS of the RCS rat could phagocytize shed outer segments 7 . This conclusion was made because histology showed pigmented cells incorporated into the host RPE layer, in the SRS, and also in between photoreceptor outer segments. These cells were assumed to be transplanted cells. However, the electron microscopy images revealed 10 times as many phagosomes in the putative transplanted RPE cells compared to normal rat RPE cells. The staining and imaging techniques were insufficient to determine if the phagosomes seen were truly in transplanted RPE cells or another cell type. In the present study, the DIC images clearly demonstrate pigment granules in macrophages (Fig. 4A and B) and neutrophils (Fig. 4C). We propose that infiltrating macrophages and neutrophils, rather than transplanted RPE cells, may be responsible for the hyper-phagocytosis observed by Lopez et al.
Retinal detachment activates retinal microglia 107 . We observed a considerable infiltrate of macrophages that appeared to arrive from the retinal side of the subretinal graft (Fig. 3Aa, b, and f). It is possible that the macrophage infiltration represented activated retinal microglia as CD11b 108 and F4/80 109 also identify retinal microglia. The macrophage or microglial response we observed was likely exaggerated due to the high-density cell suspension graft delivered. However, it is conceivable that a low-density cell suspension graft or even a simple subretinal injection of saline may result in lower levels of microglia activation and subretinal infiltration that proves beneficial to the host. Activated retinal microglia may assume a phagocytic phenotype and migrate to the SRS, thereby adopting the role of outer segment phagocytosis and enabling photoreceptor rescue. This explanation would theoretically explain the following phenomena of subretinal transplantation observed particularly, though not exclusively, in the RCS rat: (1) transplantation of many different cell types to the SRS results in photoreceptor rescue14–16,76,110; (2) sham surgery has anatomical76,111 and functional benefits18–21, and even subretinal saline alone promotes photoreceptor survival 112 ; (3) the area of rescue may extend beyond the area of the graft 17 ; (4) macrophages or macrophage-like cells have repeatedly been seen at the graft site6,14,26,27,29–31,84–88; and (5) temporal photoreceptor rescue may outlast graft survival 18 .
Our observation of infiltrating cells expressing the neutrophil marker Gr1 Ly-6G is novel. Neutrophils are distinguished from monocyte–macrophages via their expression of Ly-6G 95 . Neutrophils in the eye are CD11b+/Gr1 Ly-6G+113. However, neutrophils are negative for F4/80 114 . It follows therefore that the CD11b+ cellular infiltrate observed during the first week following transplantation in the present study may comprise a combination of CD11b+/F4/80+/Gr1 Ly-6G– microglia and CD11b+/F4/80–/Gr1 Ly-6G+ neutrophils. However, the immunohistochemical methods in our study could not subclassify the cellular infiltrate with certainty. Further studies such as multilabeled flow cytometric analysis of cells extracted from the SRS, or following whole-eye dissociation, would be helpful in more precisely phenotyping the infiltrate. Nevertheless, the data presented here reveal, for the first time, a critical role for the innate immune system early in subretinal graft rejection, while T lymphocytes did not feature at all during the main period of graft loss (PODs 3–7).
The rapid loss of graft cells observed in this study was accompanied by comparatively very low levels of graft cell death as examined by TUNEL labeling (Fig. 2A and C). TUNEL labeling identifies apoptotic and necrotic cells by detecting DNA strand breaks80–82. However, it is possible that the graft cell loss observed in the present study occurred via a process other than apoptosis or necrosis, such as by autophagy115,116. Autophagic cell death is characterized by increased autophagosomes/autolysosomes and extensive cytoplasmic vacuolization, but with relatively minor changes to the nucleus and chromatin 117 . Therefore, this mode of cell death could remain undetected by TUNEL labeling. Autophagic cell death is triggered by stresses including nutrient deprivation118–121 and hypoxia 119 . In order to limit potential immunological triggers against subretinal cell suspension transplants, such grafts are normally delivered suspended in serum-free medium18,20,76. For this reason, we also suspended DH01 cells in serum-free medium immediately prior to transplantation. It may be that the stresses of serum deprivation and the other processes involved in preparing a highly concentrated graft cell suspension (TC) trigger autophagic graft cell death. Cells that undergo autophagic cell death may be cleared by phagocytosis122–124 in a process that limits inflammatory and immunological responses 125 . Consistent with these features of autophagic cell death, there was clear evidence of graft engulfment by macrophages and neutrophils in this study (Fig. 4), and the retina overlying the graft site remained remarkably preserved with no substantial evidence of inflammatory scarring at POD 28 (Fig. 3Ad, h, l, and p).
Toll-like receptors (TLRs) are phylogenetically ancient mediators of innate immunity that detect microbes via pathogen-associated molecular patterns (PAMPs)38,126. Stressed and dying cells express endogenous TLR ligands termed damage-associated molecular patterns (DAMPs) 127 . Activation of the innate immune system via TLRs plays an important role in allograft rejection39,40. Blocking TLR4-mediated graft rejection prolongs pancreatic islet cell transplant survival 43 . TLR4 is constitutively expressed by RPE cells128,129, photoreceptors 130 , and resident antigen-presenting cells in the normal human uveal tract 131 . However, there have been no studies to date regarding the role of TLRs in subretinal cell transplantation. Cell suspension grafts are exposed to many potential stressors including enzymatic cleavage, centrifugation, resuspension in serum-free medium, and delivery through nonbiological injectors. It is likely that these processes not only incite increased inflammatory cytokine and chemokine production by graft cells as demonstrated in this study but may also provoke increased expression of DAMPs. Following transplantation to the SRS, excessive DAMP expression by transplanted cells could propagate an innate immune response by binding host TLRs, thereby triggering host cytokine and chemokine expression capable of recruiting microglia/macrophages from the overlying retina as well as neutrophils from the circulation. DAMPs may also trigger complement activation132,133 and the release of anaphylatoxins capable of mediating leukocyte chemotaxis134,135.
Future strategies to suppress the host immune response against cells transplanted to the SRS should focus more on limiting the effect of infiltrating macrophages and neutrophils by specifically targeting these mediators of the innate immune response. Potential agents include clodronate91,93, statins102,103, resveratrol136,137, and minocycline138,139. Controlling innate immune and inflammatory responses following transplantation will be particularly important in the context of treating conditions such as age-related macular degeneration, which is characterized by increased macrophage infiltration at the affected site140–142.
The rapid innate immune response observed in our study is likely a consequence of multiple inciting factors including increased expression of inflammatory cytokines and DAMPs by cells prepared for transplantation. Such factors are independent of genetic mismatch and would not be restricted to allogeneic cell transplants. Consequently, the innate immune response observed in our study is also likely to occur in response to other cell transplants including syngeneic, autologous, and stem cell transplants. The future success of subretinal transplantation will require more emphasis to be placed on optimizing techniques to prepare and deliver grafts of high quality that limit rapid innate immune-mediated clearance of transplanted cells. Furthermore, pharmacological strategies in the field of subretinal transplantation must go beyond merely suppressing adaptive immunity in the host and focus on methods of inhibiting the early innate immune response.
Footnotes
Acknowledgments
This work was funded by the Health Research Board, Ireland (project grant RP/2007/202), and by an Irish College of Ophthalmologists Fellowship awarded to Kevin Kennelly, Ph.D. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript. The authors are grateful for the assistance provided with the confocal microscopy and image analysis sections of this study by Orla Hanrahan, Ph.D., and Gavin McManus, Ph.D., at the Microscopy and Imaging Facility at the School of Biochemistry and Immunology, Trinity College Dublin, Ireland. The authors also appreciate the assistance provided with statistical analysis by Timothy Grant, C.Stat., C.Sci., and Ricardo Segurado, Ph.D., at the Centre for Support and Training in Analysis and Research, University College Dublin, Ireland. The authors declare no conflicts of interest.
