Abstract
There is considerable interest in transplanting stem cells or progenitors into the injured nervous system and enhancing their differentiation into mature, integrated, functional neurons. Little is known, however, about what intrinsic or extrinsic signals control the integration of differentiated neurons, either during development or in the adult. Here we ask whether purified, postmitotic, differentiated retinal ganglion cells (RGCs) directly isolated from rat retina or derived from in vitro-differentiated retinal progenitor cells can survive, migrate, extend neurites, and form morphologic synapses in a host retina, in vivo and ex vivo. We found that acutely purified primary and in vitro-differentiated RGCs survive transplantation and migrate into deeper retinal layers, including into their normal environment, the ganglion cell layer (GCL). Transplanted RGCs from a wide range of developmental ages, but not from adults, were capable of extending lengthy neurites in the normal and injured adult rat retina ex vivo and to a lesser degree after transplantation in vivo. We have also demonstrated that RGCs may be differentiated and purified from retinal precursor cultures and that they share many of the same cell biological properties as primary RGCs. We have established that progenitor-derived RGCs have similar capacity for integration as developing primary RGCs but appear to form a lower number of presynaptic punctae. This work provides insight for further understanding of the integration of developing RGCs into their normal environment and following injury.
Introduction
How does a developing neuron integrate into its environment? In the retina, for example, retinal ganglion cells (RGCs) are among the first neurons to arise from a common pool of multipotent progenitors during retinal development, from embryonic day 11.5 (E11.5) to E18 in the rodent (1). After exiting the cell cycle, RGCs migrate across the retina, forming the ganglion cell layer (GCL) (22,23). In the GCL, RGCs extend axons toward the optic nerve head, ultimately forming the optic nerve connecting the eye to the brain. RGCs proceed to form synaptic connections with presynaptic amacrine and bipolar interneurons (25). Little is known about what signals regulate many of the steps involved in this complex process or to what degree each facet—survival, neurite growth, migration, and synaptic integration—depends on the preceding one.
These questions are particularly important because there is significant interest in transplanting stem cells and other multipotent progenitors into the retina as well as various other regions of the central nervous system (CNS). Under the right conditions, these cells may differentiate into the proper neuronal subtype and functionally integrate with the host's preexisting circuitry. Transplanted embryonic stem cells, embryonic and adult neural progenitors, and other proliferating cells have demonstrated only limited success at integrating into various regions of the central nervous system (5,31,37). Several factors, including injury reactivity, immune response, and a lack of crucial developmental factors in the adult microenvironment are likely to play a role in the failure of these cells to properly differentiate into appropriate cell types and integrate with surrounding cells (34,36).
Retinal progenitors from various donor ages transplanted to the retina have thus far not yielded significant differentiation and integration of RGCs. Indeed, it is not yet known whether actual RGCs are capable of integration into preexisting retinal circuitry after transplantation to the adult retina (13,24). Here we ask whether RGCs purified from developing or mature retinas or purified from differentiating retinal progenitor cultures, survive, extend neurites in the nerve fiber layer, migrate into the GCL, and form morphologic synapses after transplantation to the adult retina in vivo, ex vivo, and after optic nerve injury.
Materials and Methods
Animals
All use of animals conformed to the Association for Research in Vision and Ophthalmology (ARVO) Statement for the Use of Animals in Research, and was approved by the Institutional Animal Care and Use Committee (IACUC) and the Institutional Biosafety Committee of the University of Miami.
Sprague–Dawley rats of varying ages and equally male and female were obtained from Harlan Laboratories (Indianapolis, IN, USA). Equally male and female mice were bred from the following strains: C57BL/6-Tg(CAG-EGFP)10sb/J [enhanced green fluorescent protein (EGFP) under the control of the chicken β-actin promoter; Stock # 003291, Jackson Laboratories, Bar Harbor, ME, USA].
RGC Purification and Culture
RGCs from embryonic day 17 (E17) through postnatal (P) and adult Sprague–Dawley (SD) rats or postnatal transgenic β-actin-GFP mice were acutely purified by sequential immunopanning with the T11D7 antibody (generated in lab from hybridoma cultures) for rat or the cluster of differentiation 90 (CD90) (thymocyte antigen 1; Thy1.2; AbD Serotec, Düsseldorf, Germany) antibody for mouse, yielding 99.5% pure RGCs (3,11,20). Purified rat primary RGCs were labeled in vitro with chloromethylbenzamido-1,1′-dioctadecyl-3,3,3′3′-tetramethylindocarbocyanine perchlorate (CM-DiI; red; Invitrogen, Carlsbad, CA, USA) or with Oregon Green Cell Trace (green; Invitrogen), and then cultured for 24 h or 1 week on adult retinal explants in serum-free defined medium as described containing brain-derived neurotrophic factor (BDNF; 50 ng/ml; Peprotech; Rocky Hill, NJ, USA), ciliary neurotrophic factor (CNTF; 10 ng/ ml; Peprotech), insulin (5 μg/ml; Invitrogen), and forskolin (5 μM; Sigma, St. Louis, MO, USA) (3,20). Purified RGCs from β-actin–GFP mice did not undergo cell trace or CM-DiI incubation but were further labeled with anti-GFP antibody immunofluorescence (see below). A subset of RGCs from each immunopanning preparation were cultured on poly-d-lysine (PDL) (70 kDa, 10 μg/ml; Sigma) and laminin (2 mg/ml; Telios/Invitrogen) in the same media used for explants (see above).
Retinal Explants
Retinal explant culture was performed as described previously (33). Briefly, adult SD rat eyes were enucleated and transferred to cold Dulbecco's phosphate-buffered saline (DPBS; Invitrogen). Eyes were rinsed repeatedly and dissected under sterile conditions. The cornea and lens were removed, creating an eye cup preparation. The neural retina was teased off the retinal pigment epithelium with special care taken not to disturb the retinal ganglion cell side of the retina. The retinas were adsorbed to Millicell chamber filters (Millipore, Billerica, MA, USA; diameter 30 mm, pore size 0.4 um) with the ganglion cell layer upward. The chamber was transferred to a six-well culture plate (Falcon, VWR, Radnor, PA, USA) containing RGC medium as above. After 24 h or 7 days, retinas were fixed, counterstained with 4′,6-diamidino-2-phenylindole (DAPI; Vector Labs, Burlingame, CA, USA) to highlight nuclei, and imaged using confocal microscopy (Leica, Buffalo Grove, IL, USA). Confocal microscopy was used because neurite outgrowth occurred in three dimensions, and tissue sections would only contain a limited view of the neurite outgrowth of each cell. Thus, whole mount retinas were imaged to allow a broader, clearer image in the xy-plane while retaining z-plane capabilities. Transplanted RGC survival was quantified by nuclear morphology 24 h after transplantation; condensed pyknotic nuclei marked dead cells. Migration was quantified with 3D Z-stack projections using confocal fluorescence microscopy (Leica).
In Vitro Differentiation of RGCs
Retinas from five litters of E14 timed pregnant rats were dissected, dissociated enzymatically, and cultured in proliferation-promoting media containing epidermal growth factor (EGF) and basic fibroblast growth factor (bFGF) (20 ng/ml each; Sigma) for 7 days at clonal density (10 cells/μl) on nonadherent Petri dishes (Falcon, VWR). After 7 days, progenitor-enriched neurospheres were cultured on PDL (70 kDa, 10 μg/ml; Sigma) and laminin (2 mg/ml; Telios/Invitrogen) in RGC growth-promoting, serum-free medium as above without EGF or bFGF. After 4 days, the cells were released from the plates with trypsin (Sigma), and Thy1+ in vitro-differentiated RGCs were purified by immunopanning as above.
Optic Nerve Axotomy
Optic nerve axotomy was performed as described previously (15,16). RGCs from the host retina were first labeled with Fluoro-Gold™ (Sigma) by prior injection into the superior colliculus (15). In some cases, gel foam soaked in Alexa-488-conjugated cholera toxin β-subunit (Invitrogen) was placed on the optic nerve stump following transection in order to retrogradely label host retina ganglion cells. The contralateral eye was used as a noninjured control.
Ex Vivo Transplantation
RGCs purified from acutely dissected retinas or derived from neurospheres were labeled in vitro with the lipophilic dye CM-DiI or Cell Trace Oregon Green. RGCs were diluted in DPBS to 2,500 μl and a total volume of 20 μl (50,000 cells per retina) were transplanted directly over the center of the retina and allowed to float down to the retina by gravity.
In Vivo Transplantation
RGCs were purified by immunopanning from both rat and β-actin–GFP mice. The rat RGCs were labeled in vitro with the lipophilic dye CM-DiI (red). Using a pulled glass pipette, 50,000 labeled RGCs in 2 μl of PBS or 2 μl PBS alone for controls were injected intravitreally into one eye of an anesthetized adult rat as previously described (15). Three rat eyes were injected per RGC donor age tested, and all sections of each eye were examined. Remaining RGCs were plated on PDL/laminin-coated plates in growth media for 3 h, after which these extra cells were fixed and immunostained for RGC and neural-specific markers [brain-specific homeobox 3 (Brn3) and βIII-tubulin, respectively] to verify RGC purity (data not shown), consistent with published data on the RGC immunopanning procedure (11,20).
Immunostaining
Flat mount retinas were fixed in 4% paraformaldehyde (VWR) for 30 min and either placed on glass slides or further processed for cryosectioning. The samples isolated for cryosectioning were incubated in 30% sucrose (Sigma) overnight and then frozen in optimal cutting temperature solution (OCT; TissueTek; Sakura Finetek, Alphen aan den Rijn, Netherlands) using liquid nitrogen. Both flat mount samples, as well as retinal sections, were simultaneously blocked and permeabilized with 20% donkey serum (Sigma) and 0.4% Triton X-100 (Sigma), respectively, for 30 min. Flat mount retinas and retinal sections were then incubated in primary antibodies, including anti-Brn3 (1:200; SantaCruz; Dallas, TX, USA; goat polyclonal), anti-synaptophysin (1:200; SantaCruz; rabbit polyclonal), anti-βIII-tubulin (1:200; Covance, Princeton, NJ, USA; mouse monoclonal), and anti-nestin (1:50; Developmental Studies Hybridoma Bank, Iowa City, IA, USA; mouse monoclonal), and purified RGCs from β-actin–GFP mice with anti-GFP (1:200; Invitrogen; rabbit polyclonal) overnight at 4°C. Retinal samples were rinsed three times with PBS and incubated with the matching secondary antibody overnight at 4°C. Fluorophore-conjugated secondary antibodies (1:500; Invitrogen) were used to label primary antibodies. The retinal samples were then sealed with coverslips on slides with Vecta-Shield (Vector Labs, Burlingame, CA, USA) solution containing DAPI nuclear stain.
Cell Counting
Explanted retina and retinal section were stained with βIII-tubulin and the nuclear stain DAPI to highlight the different layers of the retina. For retinal sections, lines defining each layer were superimposed over the micrograph image. For explant confocal micrographs, Z-stack projection movies were constructed, and βIII-tubulin and DAPI were used to demarcate each retina layer. The location of the nuclei of the transplanted cell in or on the retina was used to define each cell body's localization.
RGC Neurite Direction and Orientation Assay
The orientation of neurites of RGCs on scaffolds was measured using the ImageJ NIH, Bethesda, MD, USA) plugin OrientationJ, written by Daniel Sage and available at http://bigwww.epfl.ch/demo/orientation/. The ellipses were made using the “measure” function of the plugin with the Laplacian of Gaussian set to zero. The eccentricity of the ellipses demonstrates calculated coherence of the direction of neurites. The narrower the ellipse, the higher the coherence, and the direction of the ellipse also indicates the direction of lines in the region of interest measured. A perfect circle would denote a completely random configuration. The frequency of neurites extending toward the optic nerve head was measured by the distribution function of the OrientationJ plugin. The Gaussian gradient was used with a V value of 1, a minimum coherency of 0, and a minimum energy of 0. Values from the output were normalized and then wrapped around 90° for each region to indicate the direction toward the 90° vertical. Data were partitioned into three bins of 30° each.
Statistical Analysis
Student t tests and analysis of variance (ANOVA) with post hoc Dunnett's statistical tests were conducted to identify significant differences among control and variable groups in all experiments [Graphpad (San Diego, CA, USA) and Excel (Microsoft, Redmond, WA, USA)].
Results
Transplanted RGCs Survive and Migrate Following In Vivo Transplantation
We first asked if primary RGCs survive and migrate into the adult retina following intravitreal transplantation. Embryonic and postnatal RGCs were purified by immunopanning, acutely labeled in vitro with CellTrace or CM-DiI and transplanted intravitreally using a pulled glass capillary. Transplanted RGCs displayed robust survival, as measured by nuclear morphology (Fig. 1). Using DAPI nuclear staining to define the retinal lamina, we observed that RGCs migrated away from the initial site of injection into the ganglion cell layer as well as, less commonly, deeper layers of the retina (Fig. 1). When we compared this capacity between various ages of donor cells, we observed that donor cells across a broad developmental time window had a similar capacity. Furthermore, we observed RGCs from both embryonic and postnatal RGCs surviving following 7 days postinjection, although the survival was significantly reduced compared with 24 h (Fig. 1e). Adult RGCs, as donor cells, were not tested in these assays due to relatively lower yields, survival, and neurite outgrowth following immunopanning (see below). Cross-sections of the retina revealed that the transplanted RGCs predominately resided on the vitreous side of the nerve fiber layer (NFL) with a significantly smaller proportion of cells migrating into the ganglion cell layer (GCL) or remaining in the vitreous (Fig. 1f, g). There was no correlation between RGC migration into deeper retinal layers and the needle injection site. Potential issues and concerns with using a viable Cell Trace dye were verified by comparing these results with transplantation experiments using RGCs derived from β-actin–EGFP mice as the donor cells. GFP– RGCs had similar survival and migration capacity to Cell Trace-dyed RGCs following transplantation (Fig. 1d). In order to ensure our criteria for determining in which layer the donor cell resides, we stained a subset of retinas with βIII-tubulin to label the nerve fiber layer (NFL) (Fig. 1d, bottom). Using this marker, as well as DAPI nuclear labeling, as lines of demarcation to define each layer, we found similar migration results to DAPI nuclear stain-based criteria alone. Taken together, our results demonstrate that transplanted RGCs can survive and occasionally migrate into the GCL in vivo, opening new avenues for studying the integration capacity of neurons transplanted directly into their appropriate region.

Developing retinal ganglion cells survive and migrate into the adult retina after transplantation in vivo. Embryonic and postnatal retinal ganglion cells (RGCs) were purified by immunopanning, labeled in vitro with CellTrace (green in panels a and b), or derived from transgenic β-actin–green fluorescent protein (GFP; green in panels c and d) mice and transplanted intravitreally into adult rat eyes. Nuclear staining (4′,6-diamidino-2-phenylindole; DAPI, blue) defined the retinal lamina. Purified RGCs survived and extended neurites 24 h (a–d, f) and 7 days (e, g) following intravitreal transplant. Scale bar: 200 μm for (a, b); 100 μm for (c, d). P4 RGCs (green) were found along the nerve fiber layer (arrow in b) and in the ganglion cell layer (arrowheads in a, b, d), in the vitreous (arrows in a, b), and in the inner plexiform layer (c, d) 24 h after transplantation. Lower panel in d shows βIII-tubulin colocalization with GFP-labeled RGCs. (e) Approximately 1,600 and 600 cells of 50,000 injected cells survived 24 h and 7 days after injection, respectively, demonstrating a significant decrease in survival (*p < 0.001, paired t test; mean ± SEM, N = 3). (f, g) Donor cells were most frequently encountered in the nerve fiber layer both after 24 h (f) and 7 days (g) after transplantation. NFL, nerve fiber layer; GCL, ganglion cell layer; IPL, inner plexiform layer; INL, inner nuclear layer.
RGC Survival Following Ex Vivo Transplantation
In order to observe the capacity of RGCs to integrate into a host retina, we developed an ex vivo model system taking advantage of our capability to isolate, purify, and label retinal ganglion cells, combined with a well-characterized organotypic retinal explant culture system (Fig. 2) (17). We first investigated RGC viability following transplantation onto a normal adult retinal explant in serum-free medium that strongly supports RGC survival in vitro and in vivo (20,28). RGCs purified from a broad developmental time window readily survived in the ex vivo model (Fig. 2); the even distribution and survival of RGCs across the retina was apparent at lower magnifications as well (Fig. 3a). The survival of transplanted adult RGCs was significantly lower than all the other developmental ages tested (Fig. 2c), consistent with our observations that adult RGCs are less likely to survive following purification and culture in vitro (data not shown). Thus, our results demonstrate that the host retinal explant readily supports RGC survival, providing a system to investigate neurite growth, migration, and synapse formation.

Acutely purified RGCs survive following ex vivo transplantation. (a) RGCs were acutely purified by immunopanning and labeled in vitro with CM-DiI (red, a) or Cell Trace (green, not shown), or derived from transgenic β-actin–GFP mice and cultured for 24 h on adult retinal explants in serum-free growth medium containing brain-derived neurotrophic factor (BDNF), ciliary neurotrophic factor (CNTF), insulin, and forskolin. After 24 h, retinas were fixed, counterstained with DAPI (blue) to highlight nuclei, and imaged using confocal microscopy. (b) Nuclear staining (DAPI, blue) was used to identify living (arrow) and dead (condensed, arrowhead) nuclear morphologies of transplanted RGCs (red) 24 h after transplantation. Scale bar: 100 μm for (a); 20 μm for (b). (c) Quantification of survival based on nuclear morphology demonstrated similar survival of transplanted developing RGCs and reduced survival of transplanted adult RGCs (N = 3, *p < 0.001, ANOVA with post hoc Dunnett's test; mean ± SEM).
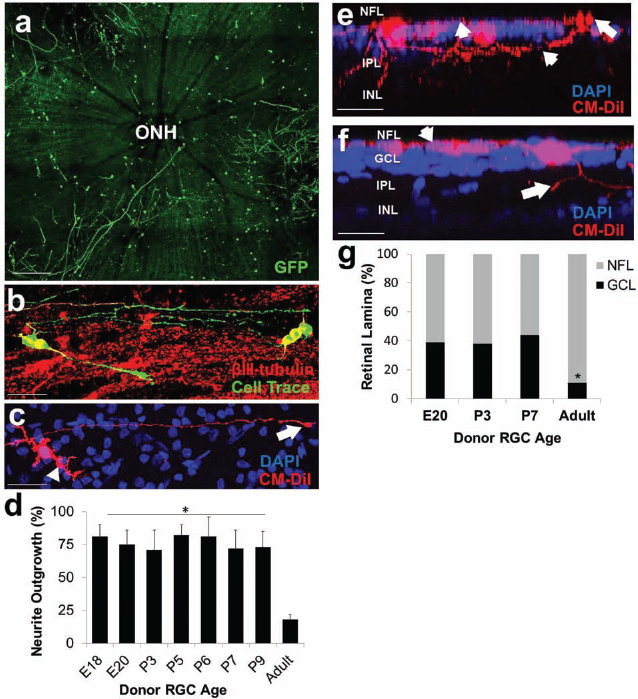
Acutely purified RGCs extend lengthy neurites and migrate into the ganglion cell layer following transplantation. (a) Postnatal RGCs (red) transplanted onto retinal explants (blue) under low magnification displaying robust survival and neurite outgrowth throughout the retinal surface. (b) Postnatal RGCs (green) extended lengthy neurites along host βIII-tubulin-positive (red) neurite tracks. (c) Postnatal RGC displaying dendritic-like arborization (arrowhead) and one long axon with a growth cone (arrow). Scale bar: 100 μm for (a, b); 50 μm for (c). (d) Quantification of neurite outgrowth 24 h after transplantation across the various ages tested demonstrated little difference among developing RGCs' ability to extend neurites, and significantly reduced neurite outgrowth was observed among surviving adult RGCs (N = 3, *p < 0.001, ANOVA with post hoc Dunnett's test; mean ± SEM). (e, f) RGC somas (red) of embryonic (e) and postnatal (f) were observed in the nerve fiber layer (NFL; arrowhead in e) and ganglion cell layer (GCL; arrow in e) with processes projecting both deeper into the inner plexiform layer (IPL, arrow in f) as well as along the NFL (arrowhead in f). Scale bar: 50 μm for (e, f). (g) Migration was quantified with 3D confocal Z-stack projections using DAPI nuclear morphology as a guide for defining the distinct retina lamina. Approximately 40% of RGCs migrated into the ganglion cell layer, independent of the age of the immature donor RGCs, and significantly reduced migration was observed in adult RGCs (N = 3, *p < 0.01, ANOVA with post hoc Dunnett's test; mean ± SEM). ONH, optic nerve head.
RGC Neurite Growth Following Ex Vivo Transplantation
We next investigated the ability of transplanted neurons to extend neurites in the host adult retina. RGCs purified from all of the developmental ages tested were readily capable of extending robust neurites, often greater than 500 μm in length (Fig. 3a, b). Furthermore, RGCs generally displayed dendritic-like arborization and one long axon containing a growth cone (Fig. 3c). Of the surviving cells, we quantified how many (%) of these cells were capable of extending at least one 50 μm neurite. Quantification of neurite outgrowth 24 h after transplantation across the various ages tested demonstrated little difference among developing RGCs' ability to extend neurites, although surviving transplanted adult RGCs had significantly less neurite outgrowth compared to any of the developmental ages tested (Fig. 3d). Using Z-stack confocal microscopy as well as cryosections, we found transplanted neurons mostly extended neurites along the NFL but were also capable of sending neurites deeper into the inner plexiform layer (IPL) (Fig. 3e, f). Thus, purified primary RGCs from a range of developmental ages can survive and extend neurites after transplantation.
RGC Migration Into the Ganglion Cell Layer
The transplanted RGCs are initially transplanted on top of the inner limiting membrane (ILM) and the NFL. Using DAPI nuclear staining to define the retinal laminae, we next asked how many of the transplanted RGCs migrated into deeper layers of the retina, including the GCL, where RGC cell bodies normally reside. Within the first 24 h, approximately 40% of the transplanted developing RGC somas were found in the GCL; on a percentage basis, adult RGCs were significantly less able to migrate into deeper layers after transplantation (Fig. 3e–g). As noted above, RGCs in the GCL extended neurites along the NFL as well as in the deeper IPL (Fig. 3e, f). Even though the cells were transplanted at clonal density (500 cells/mm2), some cells were found clustered together. We do not know whether or not this reflected RGCs migrating toward neighboring cells transplanted nearby. Taken together, these data demonstrate that embryonic and postnatal RGCs have a similar capacity to migrate deeper in the retina following transplantation.
Prolonged Transplant Culture Results in Donor RGCs with Robust Survival and Neurite Outgrowth and Increased Ganglion Cell Layer Localization
We next investigated whether transplanted RGCs would continue to survive, extend lengthy neurites, and migrate deeper into the retina 1 week in culture (Fig. 4). We found robust survival of transplanted RGCs following 1 week of retinal explant coculture. After 24 h, around 45% of the observed labeled cells were viable, while following 1 week of culture, around 90% of the observed labeled cells were viable (Fig. 4b). We hypothesize that, similarly to RGCs in culture, a proportion of the cells do not survive past the first 24 h following immunopanning, but that the RGCs that do survive past this time point retain robust survival even in long-term cultures.

Transplanted GFP+ RGCs demonstrate robust survival and extensive neurite outgrowth with a majority of cells localized to the GCL following long-term coculture on retinal explants. GFP+ RGCs were acutely purified by immunopanning and cultured on adult retinal explants in serum-free growth medium containing BDNF, CNTF, insulin, and forskolin. After 24 h or 7 days, retinas were fixed, immunostained for GFP (green), βIII-tubulin (red), and counterstained with DAPI (blue) to highlight nuclei, and imaged using confocal microscopy. Nuclear staining was used to identify living and dead (condensed) nuclear morphologies of transplanted RGCs (green). (b) Quantification of survival based on nuclear morphology demonstrated significantly (p < 0.02) higher survival following 7 days in culture compared to 24 h in culture. We found extensive neurite outgrowth toward the ONH (a, top, arrowheads) with neurites displaying prototypical axonal growth cone tips (a, bottom, arrowheads). (c) Quantification of the percent of live cells extending lengthy neurites demonstrated that following 1 week in culture, most (90%) of the cells extended extensive and elaborate neurites. (d) Z-stack images of the retinal explants were acquired from (d,1) above the NFL to the (d,5) INL in order to localize the cell soma (green) to a specific layer. Cell somas were localized (e) on or in the NFL or (f) in the GCL. (g) After 24 h, most of the cells (~80%) were either on or embedded in the NFL with only 20% of the cells localized to the GFL. After 1 week in culture, we observed significantly (p < 0.02) higher proportion of the live cells in the GCL (65%) and less cells in the NFL (35%). Scale bar: 300 μm for (a), top; 50 μm for (a), bottom; 200 μm for (d); 50 μm for (e, f). *p < 0.02, paired t test; mean ± SEM, N = 3.
When next asked whether these surviving cells retain their lengthy and elaborate neurite outgrowth over 1 week in culture compared to 24 h. We found that, indeed, the cells that survived 1 week had extensive and lengthy (over 1 mm longer) neurites (Fig. 4a). In certain instances, we observed lengthy RGC projections directed and even entering the optic nerve head (ONH) (Fig. 4a, top and bottom). Although the RGCs projected neurites in all directions, it should be noted that many of the projections that were directed toward the ONH had thicker axon-like projections with classic growth cone tips (Fig. 4a, bottom, arrowhead).
We examined the location of the transplanted cells using confocal microscopy by generating tiled Z-stack projection images of the retinal explants (Fig. 4d) beginning above the NFL (slide 1) and ending at the INL (slide 5). Although we found neurites in all the layers, we only found cell somas on or in the NFL or among the host soma in the GCL (Fig. 4e, f). After 24 h, most of the cells (~80%) were either on or embedded in the NFL with only 20% of the cells localized to the GFL. After 1 week in culture, we observed a significantly higher proportion of the live cells in the GCL (65%) and less cells in the NFL (35%) (Fig. 4g). This suggests that, over time, either more cells migrate to the GCL or that cells that migrate to the GCL are somehow more neuroprotected and survive more robustly. Taken together, we have shown here that over 1 week, transplanted RGCs survive and extend elaborate growth cone-producing neurites that in some instances project over long distances toward the ONH. Furthermore, over time, we observe more cells in the GCL, which demonstrates the capacity of transplanted RGCs to migrate into appropriate neural layers.
In Vitro Differentiation and Purification of Retinal Ganglion Cells From Retinal Progenitors
It is not known whether RGCs differentiated in vitro from retinal progenitor cells would be similar in their properties to RGCs purified from the richer context of the developing retina. We proliferated retinal progenitors from the E14 retina into neurospheres (Fig. 5a), which were subsequently induced, through mitogen withdrawal, to spontaneously differentiate into retinal neurons in culture media that promotes RGC survival (see Materials and Methods) (Fig. 5b) (20). After 4 days, cells were trypsinized, and presumptive RGCs were isolated by Thy1 immunopanning. Immunostaining of a subset of cells revealed that after a 4-day differentiation period, 3% of the total cells were positive for the retinal ganglion cell marker Brn3 (Fig. 5c), and 15% were positive for the neural cell marker βIII-tubulin (Fig. 5b). From a total of 180 million cells, 300,000 RGCs (0.16%) were purified by Thy1 immunopanning. The cells were either transplanted into our organotypic explant model or plated on PDL-and laminin-coated dishes. The in vitro-differentiated RGCs demonstrated similar morphology to purified primary neurons including the ability to form lengthy axon-and dendrite-like structures (Fig. 5d). Furthermore, all of the purified cells stained positive for the neuronal marker βIII-tubulin and the RGC-specific marker Brn3, and none of the cells stained positive for the neural progenitor marker nestin demonstrating the efficiency of immunopanning in isolating progenitor-derived RGCs away from progenitor cells or other derived progeny (Fig. 5e–h). Thus, large numbers of RGCs may be differentiated and purified from retinal progenitor cell cultures.

Isolation and purification of in vitro-differentiated RGCs. (a) E14 rat retinal neurospheres were expanded in culture in the presence of mitogens. (b) Mitogen-withdrawal in culture conditions initiated retinal differentiation from nestin-expressing progenitors (green) to βIII-tubulin-expressing neurons (red; nuclei counterstained with DAPI in blue throughout figure). (c) Approximately 3% of cells in in vitro-differentiated cultures stained positive for brain-specific homeobox 3 (Brn3; green), an RGC-specific marker. (d) In vitro-differentiated RGCs after purification by thymocyte antigen 1 (Thy1) immunopanning, labeled with CellTrace (green), and cultured on poly-d-lysine/laminin-coated dishes. Isolated Thy1+ cells demonstrated RGC morphology, and almost all cells stained positive for βIII-tubulin (e, g) and Brn3 (f, h). Scale bar: 50 μm for (a-c); 30 μm for (d-f). *p < 0.001, paired t test; mean ± SEM, N = 3.
In Vitro-Differentiated RGC Survival, Neurite Outgrowth, and Migration Following Transplantation Onto Retinal Explants
We next asked how these in vitro-derived RGCs compare to primary RGCs in their ability to survive, elaborate neurites, and form synapses after transplantation. Immediately after isolation, in vitro-differentiated, purified RGCs were acutely labeled with CellTrace Oregon Green (pseudocolored red) and transplanted using the ex vivo model as described above. Compared to RGCs purified directly from developing retinas, in vitro-differentiated RGCs demonstrated reduced survival in retinal explants (Fig. 6a–e); similarly reduced survival was also observed when plated in vitro on plastic dishes (data not shown). In contrast, quantification of neurite outgrowth of surviving in vitro-derived RGCs demonstrated little difference compared to that of developing primary RGCs (Fig. 6a–d, f). Approximately 40% of in vitro-differentiated RGCs migrated into the GCL, similar to the migration observed with primary RGCs and considerably more than the significant reduction in migration observed with adult RGCs (data not shown). Thus, other than reduced survival, in vitro-differentiated RGCs exhibit similar capacity for neurite growth and migration in the adult retina compared to primary RGCs.

Acutely purified and in vitro-differentiated RGC survival, neurite outgrowth, and migration into the ganglion cell layer improves in retinal explants 28 days after optic nerve axotomy. (a, b, c, d) RGCs labeled with CellTrace (pseudocolored red; arrowheads) were transplanted onto ex vivo retinas explanted after sham surgery (a, c, c′) or 7, 14, or 28 days after optic nerve axotomy (b, d, d′). Host retinas were subsequently labeled with βIII-tubulin (pseudocolored blue); note the relative absence of host RGC bodies at 28 days after axotomy (d′) compared to the control retina (arrows in c′). Scale bar: 30 μm for (a, b). (e, f) Acutely purified and in vitro-differentiated RGC survival and neurite outgrowth was increased in retinal explants at 28 days after optic nerve axotomy (N = 3, *p < 0.001, ANOVA with post hoc Dunnett's test; mean ± SEM; IV-RGCs were only tested in the sham and 28-day conditions).
RGC Transplantation Into Injured Adult Retina
An injured host environment may influence the integration capacity of transplanted cells (9). The specific degeneration of RGCs following optic nerve axotomy follows a well-characterized degeneration rate in which ~40% of RGCs have died after 7 days, 95% have died after 14 days, and 98% of RGCs have died 28 days after axotomy (4). We first asked whether the injured adult retina would provide a more or less supportive environment for survival, neurite growth, or migration of transplanted RGCs. Adult rat RGCs were retrogradely labeled with Fluoro-Gold™ prior to optic nerve axotomy. Retinas were then explanted at 7, 14, and 28 days after axotomy for the ex vivo RGC transplantation paradigm as above. We found that retinas from 7 and 14 days postaxotomy supported transplanted primary RGCs' survival as well as control, uninjured retinas (Fig. 6a, c, c′, e). Similar levels of survival were observed for both embryonic (E20) and postnatal (P3) transplanted RGCs as well as in vitro-differentiated RGCs. However, we found that E20, P3, and in vitro-differentiated donor RGC survival was significantly (p < 0.001) greater when transplanted onto retinas explanted 28 days postaxotomy (Fig. 6b, d, d′, e). Primary and in vitro-differentiated RGCs transplanted into the 28-day postaxotomy host retina also displayed enhanced neurite outgrowth (Fig. 6f). Taken together, these results suggest that the retina becomes more supportive of transplanted RGC integration only after significant host RGC degeneration, irrespective of whether the donor RGCs are primary or in vitro derived.
Transplanted RGC Neurites Orient Along Host Retinal Neurites
After observing that transplanted neurons mostly extended their neurites along the NFL, we investigated in what directions the transplanted neurons were sending neurites in relation to host axons by immunostaining with βIII-tubulin (Fig. 7a, b) to highlight all neurites. By labeling both host and donor neurites, we found that some of the transplanted RGCs appeared to orient their neurites with host axons directed toward the optic nerve head. When we quantified how many of the surviving transplanted RGCs oriented their neurites with host neuronal projections, we found that roughly 40% of E20, P3 RGCs, as well as in vitro-differentiated RGCs had neurites that oriented along the nerve fiber path toward the optic nerve head (Fig. 7c). Interestingly, there was a significant (p < 0.01) increase (~58%) in neurite orientation observed among all donor sources when transplanted into explants of 28-day postaxotomized retinas (Fig. 7c). Thus, transplanted RGCs tend to elongate their axons along the host RGC axon bundles, and this effect is more pronounced after host RGC/NFL degeneration.
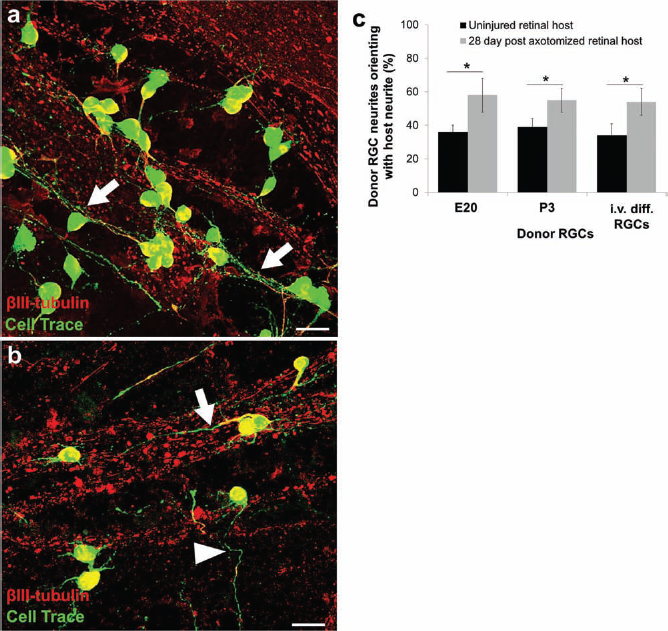
Purified primary and in vitro-differentiated RGCs orient along host neurites. (a, b) RGCs (green) transplanted onto retinal explants oriented with (arrows) or across (arrowheads) host retinal neurites (stained with βIII-tubulin, red). Scale bar: 50 μm for (a, b). (c) Quantification demonstrated that donor RGCs oriented with host neurites independent of donor source, but RGCs transplanted into 28-day postaxotomized retinas demonstrated significantly increased neurite orientation (*p < 0.01, paired t test; mean ± SEM, N = 3).
Transplanted RGCs' Neurites Are Slightly Oriented to Intraretinal Axons
Using the OrientationJ plugin (see Materials and Methods for details) for ImageJ, we next asked if all the combined processes of the transplanted cells were completely random in direction or whether they orient toward the ONH (Fig. 8a, b). We found that in the central, middle, and peripheral areas of the eye, there was a significantly (p < 0.02) higher proportion of neurites oriented 0°–30° away from the ONH compared with 30°–60° or 60°–90° (Fig. 8c). These data suggest that all three regions of the retina support a similar level of neurite orientation toward the ONH.

Total transplanted RGC neurites orient to intraretinal axon tracts of the host retina. (a, b) GFP+ RGC neurites were imaged and assigned net orientations (ellipses; see Materials and Methods). Furthermore, the retinal explant was divided into central, middle, and peripheral regions of interest. (c) The frequency of neurites extending in each direction, partitioned into three bins of 30°. There was a significant difference comparing 0°–30° and 30°–60° as well as comparing 0°–30° and 60°–90°, indicating some degree of alignment of transplanted RGC neurite growth with the host retina. Scale bar: 300 μm for (a), 200 μm for (b), left and 50 μm for (b), right.*p < 0.02, paired t test; mean ± SEM, N = 3.
Synapse Formation Onto Primary and In Vitro-Differentiated RGCs Following Transplantation Onto Adult Retinal Explants
Finally, we asked whether we could detect morphologic presynaptic puncta following RGC transplantation. Purified primary and in vitro-differentiated RGCs were transplanted onto ex vivo adult rat retinas as above. The presynaptic marker synaptophysin was found immediately adjacent to the neurites and cell bodies of transplanted RGCs (Fig. 9). Interestingly, primary postnatal donor RGCs were surrounded by significantly more (p < 0.001) synaptophysin-positive puncta compared with the in vitro-differentiated donor RGCs (Fig. 9). These data suggest that purified primary neurons may more readily synaptically integrate into the host retinal environment compared with neurons that were purified from retinal progenitor cells differentiated in vitro or that the in vitro-differentiated RGCs are less mature and require more time to receive as many presynaptic connections.
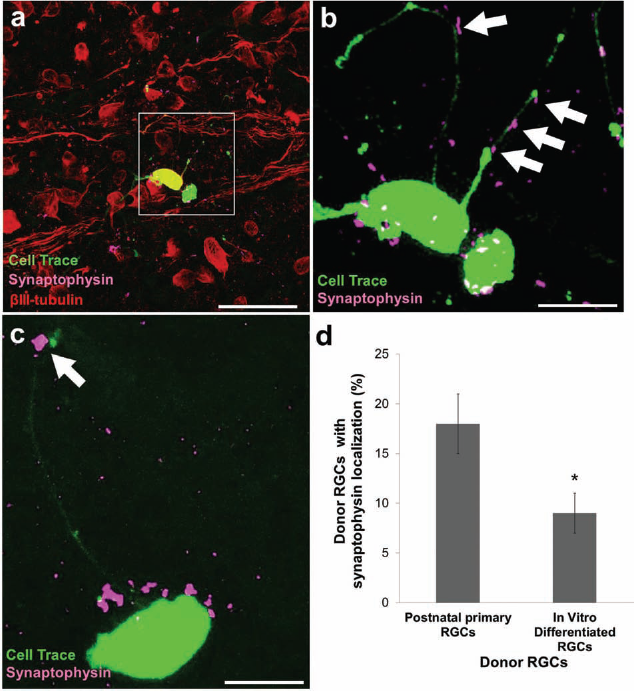
Acutely purified and in vitro-differentiated transplanted RGCs form morphologic presynaptic punctic in retina. (a, b, c, d) Purified primary (a, b) and in vitro-differentiated (c) RGC neurites and somas (green) localized with the presynaptic marker synaptophysin (purple; colocalization shown with white arrows). Scale bar: 15 μm for (a, b). (d) Transplanted primary postnatal RGCs showed more presynaptic marker colocalization then in vitro-differentiated RGCs (*p < 0.01, paired t test; mean ± SEM, N = 3).
Discussion
These data reflect three main findings. First, we have created an explant model of retinal transplantation that allows detailed examination of RGC-retinal morphologic integration. Second, we have demonstrated that RGCs may be purified from retinal precursor cultures and that they share many of the same cell biological properties as primary RGCs. Third, we have established that progenitor-derived RGCs have similar capacity for integration as developing primary RGCs but appear to form a lower number of morphologic synapses.
Transplantation of Primary Neurons
Here we examined the short-term survival, migration, neurite growth, and morphological synapse formation of retinal ganglion cells after transplantation into the adult retina in an ex vivo explant model and in vivo. An advantage to this explant model is that it allowed us to provide an optimal media for survival and growth and to carefully control for factors like cell density. This model system also allows us to transplant RGCs in close proximity to the GCL by culturing the donor cells on top of and directly adjacent to the GCL and avoiding the need to forcibly inject the cells into tissue, which may also decrease initial survival. Longer term studies and further application to in vivo work may benefit from advances made in such ex vivo models.
To our knowledge, this is the first report studying the transplantation of a purified, identified neuron to its host tissue and also the first report to purify a specific neuron type from in vitro-differentiated progenitor cells. With both primary RGCs purified from a range of developmental ages and RGCs immunopanned from differentiating retinal progenitor neurosphere cultures, we found that RGCs survive transplantation and migrate into the GCL in similar fashion. The transplanted RGCs were capable of extending lengthy neurites in the normal and injured adult rat retina ex vivo. Overall, we found that transplanted RGCs mainly resided on the NFL, although this may have been due to relatively shorter culture times or to the strongly trophic media provided on the outside of the retina. Recent findings demonstrate how the extracellular matrix and other environmental signals limit the integration of transplanted cells, and the degradation of some of these molecules with enzymes such as MMP2 has been shown to support integration and retinal repopulation following transplantation (32). Even so, limited migration may not necessarily limit integration with the host retina. Indeed, our explant model demonstrates that transplanted RGCs that reside on the NFL project long neurites, which fasciculate with and form morphologic presynapses with host intraretinal axon tracts.
There were three experimental manipulations in which we could identify more optimal “integration”: [1] developing primary RGCs and in vitro-differentiated RGCs consistently demonstrated higher levels of survival and migration than adult primary RGCs, [2] primary RGCs formed more morphologic synapses than in vitro-differentiated RGCs, and [3] the degeneration of host retinal RGCs after optic nerve injury only enhanced donor RGC survival after 28 days but not at 7 or 14 days when RGC degeneration was still ongoing.
Developing RGCs Integrate Better Than Adult RGCs
We found that postmitotic RGCs isolated across a broad developmental window display similar capacities to survive, migrate deeper into the retina, and extend lengthy neurites in the adult retina in vivo and ex vivo. Donor RGCs from a wide range of developmental ages equally extended neurites along the nerve fiber layer with some neurites growing deeper into the inner plexiform layer, where RGC dendrites normally reside. These findings differ slightly from prior photoreceptor and cortical transplantation experiments that found that donor cells isolated at a developmental period close to the peak birthdate in vivo had the greatest capacity for integration, and this capacity fell off very rapidly, even while still in early development (14,18). MacLaren and colleagues transplanted dissociated retinal cells from various donor ages subretinally in mouse and found that postmitotic rod precursors were capable of reintegrating synaptically in the photoreceptor layer better than multipotent progenitors and that donor cells from rods' peak retinal birthdate had the most highly structured morphological and synaptic integration. The transplantation of these immature rod cells was capable of improving visually evoked potentials in genetic models of mouse photoreceptor degeneration, either by synaptically reintegrating into the preexisting adult circuitry or by attenuating the ongoing degeneration and enhancing the function of the residual photoreceptors (18,32). It is not yet known whether similar developmental differences in integrative capacity will be found among other long projection neurons in the central nervous system.
The equal capacity for neurite extension among all the developmental ages tested matches previous work in RGCs, where both E20 and P8 RGCs can elaborate axons in this optimized growth medium (12). The prior finding that intrinsic ability to rapidly extend axons is down-regulated across development was not addressed in these studies, as we did not measure neurite lengths after transplantation, but we did observe that donor primary RGCs from postnatal day 2 (P2) had more neurite fasciculation with host axons compared with P8 donor RGCs. The long fasciculating neurites projecting from the donor RGCs will have to be further analyzed to determine whether these are indeed axonal or dendritic. An interest in donor axons growing along host fascicles toward the optic nerve head might argue for the use of younger primary RGCs, based on these data.
In Vitro-Differentiated RGCs Demonstrate Decreased Synapse Formation
Immature neurons predifferentiated in culture from stem cells may potentially be the best candidates for neuronal reintegration; however, the prospect of successful neural transplantation will require an improved understanding of the intrinsic properties of the donor cells, as well as how these properties change during increasingly restricted neuronal differentiation. Interestingly, we observed that primary RGCs from any developmental age consistently aligned with more synaptophysin-positive puncta than did in vitro-differentiated RGCs. Perhaps in vitro-differentiated RGCs are more immature and are less competent to receive synapses, or the environment does not support synapse formation due to a lack of necessary signals, for example, from mature glial cells (21), or synapse formation is merely delayed (2). Furthermore, it is unknown how these differentiation conditions affect the communication of the donor cells with extrinsic signals in the host environment, especially in an injured or degenerating host environment. Future work including electrophysiological recording and identification of the host cells involved should help elucidate the correlation between the marker staining seen here and the function of these synapses.
The Degenerating Retina Enhances RGC Transplant Survival
Targeted apoptotic neurodegeneration has been used to produce highly controlled spatially and temporally specific cell death of selected types of projection neurons within defined regions of the cortex. Photoactivated induction of cell death in the neocortex has been shown to affect migration and differentiation of transplanted neurons as well as transplanted neural precursors (19,26,27,29,30). In these experiments, later-stage and region-specific immature neurons integrated when transplanted back into the injured adult cortex where they usually are located more efficiently than after transplantation to ectopic regions of injured cortex. However, at postnatal stages of development, limits in the survival of the donor, immature cortical neurons offset this improved efficiency (10). Experiments transplanting RGCs into other regions of the CNS or perhaps transplanting cortical neurons into the retina may better elucidate whether neurons are more “universal” or more “specific” in their capacity to integrate into adult neural circuits.
Molecular Signals Critical to Integration of Transplanted Neurons
Are there sufficient signals in the adult retina or perhaps increased in the injured adult retina that can facilitate survival, migration, neurite outgrowth and guidance, and synapse formation of transplanted RGCs? Many developmentally expressed molecular signals persist in the adult retina, including netrin, an RGC axon chemoattractant, and its receptor DCC on RGC neurites (8), N-CAM (7), and laminin along Muller glial end feet in the nerve fiber layer (35). Other signals for RGC synapse formation such as thrombospondin (6) may be downregulated during normal development but could be reexpressed after transplant or in an injured tissue. As CNS neurons largely fail to regenerate from endogenous precursors after injury, stem cell-based cell replacement therapies are being actively investigated. The decision of whether and to what extent neurons are differentiated before transplantation will be influenced by a better understanding of the cellular and molecular biology of neuronal integration.
Footnotes
Acknowledgments
This work was supported by the Department of Defense, the Hope for Vision Foundation, the Pew Foundation, the National Eye Institute (P30 EY022589 to UCSD; P30 EY014081 to Miami), an unrestricted grant from Research to Prevent Blindness, and the Walter G. Ross distinguished chair in ophthalmic research. J.H. was supported in part by NIH T32-NS07492 and by a Lois Pope Life Fellowship. We thank Gabe Gaidosh and Eleut Hernandez for excellent assistance with confocal microscopy and animal husbandry, respectively. The authors declare no conflicts of interest.
