Abstract
Falciform ligaments in the liver are surrounded by adipose tissue. We investigated the capability of adipose-derived stem cells from human liver falciform ligaments (hLF-ADSCs) to differentiate into hepatic-type cells and confirmed the functional capacity of the cells. Mesenchymal stem cells (MSCs) were isolated from the liver falciform ligament and abdominal subcutaneous adipose tissue in patients undergoing partial hepatectomy for liver disease. Cells were cultivated in MSC culture medium. Properties of MSCs were confirmed by flow cytometry, RT-PCR analysis, immunocytochemistry assays, and multilineage differentiation. Hepatic induction was performed using a three-step differentiation protocol with various growth factors. Morphology, capacity for expansion, and characteristics were similar between hLF-ADSCs and adipose-derived stem cells from human abdominal subcutaneous adipose tissue (hAS-ADSCs). However, hematopoietic– and mesenchymal–epithelial transition (MET)-related surface markers (CD133, CD34, CD45, and E-cadherin) had a higher expression in hLF-ADSCs. The hepatic induction marker genes had a higher expression in hLF-ADSCs on days 7 and 10 after the hepatic induction. Albumin secretion was similar between hLF-ADSCs and hAS-ADSCs at 20 days after the hepatic induction. The hLF-ADSCs had a different pattern of surface marker expression relative to hAS-ADSCs. However, proliferation, multilineage capacity, and hepatic induction were similar between the cell types. Accordingly, it may be a useful source of MSCs for patients with liver disease.
Introduction
Adipose tissue-derived stem cells (ADSCs) have different characteristics depending on their location in the body. For example, eyelid-derived ADSCs have a greater expression of genes related to neural function, compared to subcutaneous-derived cells, and are more easily differentiated into insulin-secreting cells than other ADSCs 1 . These differences in the properties of ADSCs likely help determine the differentiation yield in target cells. In vitro hepatic induction using ADSCs has been shown to yield a high expression of hepatic tissue via various methods, indicating that they may be a good source for induction of hepatic cells in vivo2-4.
The liver falciform ligaments, derived from the fetal umbilical vein and embryonic ventral mesentery5-7, are strings of tissue that attach the liver to the anterior body wall in vertebrates 8 . These ligaments are composed of a crescent-shaped fold of peritoneum that extends from the diaphragm and anterior abdominal wall to the surface of the liver, and are continuous with the surrounding adipose tissue. The liver falciform ligaments are removed during partial hepatectomy in patients with liver disease and then usually discarded. However, adipose tissues from liver falciform ligaments have mesenchymal lineage cells, which have characteristics similar to mesenchymal stem cells (MSCs) derived from subcutaneous adipose tissue in the abdomen. Isolation of mesenchymal cells from liver falciform ligaments, which have MSC-like characteristics with efficient hepatic differentiation, may be a novel source for stem cell therapy in patients with liver disease. Furthermore, isolation of MSCs from falciform ligaments from canine models has already been utilized in previous literature9,10.
The aim of this study was to isolate, characterize, and confirm the effectiveness of in vitro hepatic differentiation through a three-step differentiation method described in our previous study 11 for a new type of ADSC present in liver falciform tissues. As MSCs for therapeutic use must be isolated with minimal harm to the patient, available in large quantities, proliferate in vitro, and able to differentiate into a broad spectrum of lineages, ADSCs may be an ideal type of MSCs as specified by the International Society for Cellular Therapy 12 . By using adipose tissue from liver falciform ligament, ADSCs can be obtained without harm. This may help to identify an effective source for liver therapy and the feasibility of using discarded tissue for therapeutic purposes.
Materials and Methods
Primary Isolation and Cultivation of Human Abdominal Subcutaneous ADSCs and Human Liver Falciform ADSCs
Harvest of tissues was performed under anesthesia according to procedures approved by the institutional review board (IRB; 4-2013-0757) of Yonsei University Health System. Informed consent was obtained from all patients after explanation of the research protocol. Tissues were obtained from a 57-year-old female and a 48-year-old male.
Abdominal Subcutaneous Adipose-Derived Stem Cells (hAS-ADSCs)
Abdominal subcutaneous adipose tissues were harvested from patients via lipectomy. To isolate ADSCs, adipose tissues (2 g) were washed with phosphate-buffered saline (PBS), minced into 0.5-mm squares using surgical scissors, and digested at 37°C for 30 min using 0.2% collagenase type I (Roche, Penzberg, Germany).
Liver Falciform Adipose-Derived Stem Cells (hLF-ADSCs)
Liver falciform tissues were obtained from patients who underwent partial hepatectomy. Liver falciform ligaments were removed using surgical scissors. Adipose tissues (approximately 2 g) surrounding the ligaments were minced into 0.5-mm squares using surgical scissors and digested at 37°C for 30 min with 0.2% collagenase type I.
For both types of ADSCs, enzyme activity was neutralized with low glucose containing Dulbecco's modified Eagle's medium (DMEM; Gibco BRL, Grand Island, NY, USA), 10% fetal bovine serum (FBS; WelGENE, Daegu, South Korea), and 1% antibiotic/antimycotic solution (Gibco BRL). Samples were centrifuged at 1,200 × g for 10 min to obtain a pellet. After removing the supernatant, the pellet was resuspended in the medium, filtered through a cell strainer (100-μm pore size; BD Biosciences, Seoul, South Korea) to remove cellular debris, and incubated overnight at 37°C/5% CO2 in low-glucose DMEM supplemented with 10% FBS and 1% antibiotic/antimycotic solution (Gibco BRL). After 24 h of incubation, the plates were washed with PBS to remove residual nonadherent red blood cells, and the remaining cells were incubated in control medium at 37°C/5% CO2.
To expand the ADSCs, cells were incubated in the conditioning medium containing 60% low-glucose DMEM, 40% MCDB-201 (Sigma-Aldrich, St. Louis, MO, USA), 0.1 mM ascorbic acid 2-phosphate (Sigma-Aldrich), 1% (w/v) antibiotic/antimycotic solution, and 10% FBS. Medium was changed two times per week, and cells were maintained at 37°C and 5% CO2.
Flow Cytometry
Cells subcultivated from the second passage were washed with PBS and stained using a human pluripotency stem cell transcription kit (BD Biosciences) according to the manufacturer's protocol. Flow cytometry was performed using FACScan argon laser cytometer (Beckman Coulter Inc., Fullerton, CA, USA).
Reverse Transcriptase Polymerase Chain Reaction (RT-PCR) Analysis
Total RNA was extracted from cells by the TRIzol isolation method (Invitrogen, Seoul, South Korea). TRIzol solution (1 ml) was added into a tube containing isolated cells, and vortexing was performed for 5 min. Then chloroform (Sigma-Aldrich) was added into the tube, mixed for 15 s, and centrifuged at 12,000 × g for 15 min at 4°C. The aqueous phase was separated into a new tube, and isopropanol (Sigma-Aldrich) was added. The tube was then incubated at room temperature for 15 min, and centrifugation was performed at 12,000 × g for 15 min at 4°C. After discarding the supernatant, the isolated RNA pellet was rinsed with 75% ethanol in diethyl pyrocarbonate (DEPC)-treated water (Thermo Fisher Scientific, Wilmington, DE, USA), and centrifugation was performed at 7,500 × g for 10 min at 4°C. After discarding the supernatant, the isolated RNA pellet was dried and released with DEPC-treated water. RT-PCR was performed using reverse transcriptase (Promega, Madison, WI, USA) according to the manufacturer's protocol. Complementary DNA (cDNA) was amplified manually and quantified using a NanoDrop™ Lite Spectrophotometer (Thermo Fisher Scientific). PCR analysis was performed with various primers (Bioneer Corporation, Daejeon, South Korea) on cells (passage 2). Primer sequences and conditions are listed in Table 1. The amplification of cDNA was performed by PTC-100 (Bio-Rad, Irvine, CA, USA) with primers. The RT-PCR cycle has been described by standard method: (1) initial denaturation on 95°C for 2 min; (2) denaturation on 95°C for 30 s, annealing on 55°C for 30 s, polymerization on 72°C for 30 s; (3) final extension on 72°C for 5 min. Cycles were performed for 35 cycles. Then cDNA was migrated by agarose gel electrophoresis (Takara Bio Inc., Shiga, Japan).
Primers for Characterization and Hepatic Differentiation of Human Abdominal Subcutaneous Adipose-Derived Stem Cells (hAS-ADSCs) and Human Liver Falciform Adipose-Derived Mesenchymal Stem Cells (hLF-ADSCs)

OCT4, octamer-binding transcription factor 4; SOX-2, sex-determining region Y box 2; CXCR4, C-X-C chemokine receptor type 4; N-cadherin, neural-cadherin; E-cadherin, epithelial-cadherin; GATA-4, guanine-adenine-thymine-adenine-binding protein 4; FOXA2, forkhead box A2; CK19, cytokeratin 19; CK7, cytokeratin 7; AFP, α fetoprotein; HNF-4α, hepatocyte nuclear factor 4 α; albumin, ALB; CYP3A4, cytochrome P450 3A4; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Cell Proliferation Assay Using Cell Counting Kit-8
Cells were seeded into a 96-well plate and incubated in a humidified incubator at 37°C and 5% CO2. Cell counting kit-8 (CCK-8; Dojindo Molecular Technologies Inc., Rockville, MD, USA) solution was added to each well and incubated for 3 h in the incubator. Proliferation capacity was measured at 450 nm using a microplate reader (Molecular Devices, Sunnyvale, CA, USA). Proliferation assays were performed in two passages of cells at days 1, 4, 7, and 10 of cultivation per passage. Both samples of hLF-ADSCs were used for proliferation assays.
Immunocytochemistry Assay for Surface Markers
Cells were fixed with 10% formaldehyde solution (Sigma-Aldrich) at 20°C for 10 min and permeabilized with 100% ice-cold methanol (Sigma-Aldrich) for 5 min. The solution used for blocking and dilution of the primary antibody consisted of PBS and 5% bovine serum albumin (BSA; Amresco, Solon, OH, USA). Cells were blocked for 45 min and incubated sequentially overnight at 4°C with primary antibodies. Positive markers included rabbit polyclonal anti-human CD105 (Cat. No. sc-20632; 1:250; Santa Cruz Biotechnology, Dallas, TX, USA), CD90 (Cat. No. sc-9163; 1:250; Santa Cruz Biotechnology), and CD73 (Cat. No. sc-25603; 1:250; Santa Cruz Biotechnology), and goat polyclonal anti-human vimentin (Cat. No. sc-7870; 1:250; Santa Cruz Biotechnology). Negative markers included goat polyclonal anti-human CD34 (Cat. No. sc-7045; 1:250; Santa Cruz Biotechnology), CD45 (Cat. No. sc-1123; 1:250; Santa Cruz Biotechnology), and CD133 (Cat. No. sc-32596; 1:250; Santa Cruz Biotechnology); mouse monoclonal anti-human CD13 (Cat. No. ab7417; 1:500; Abcam, Cambridge, MA, USA); and rabbit polyclonal anti-human E-cadherin (Cat. No. sc-7870; 1:250; Santa Cruz Biotechnology). As secondary antibodies, coupled products were used: goat anti-rabbit immunoglobulin G (IgG) conjugated with Texas Red (TR) (Cat. No. sc-2780; 1:500; Santa Cruz Biotechnology), donkey anti-goat IgG conjugated with fluorescein isothiocyanate (FITC; Cat. No. sc-2024; 1:500; Santa Cruz Biotechnology), and goat anti-mouse IgG conjugated with FITC (Cat. No. ab6785; 1:1,000; Abcam). The quantification of fluorescence images was performed using the ImageJ software [1.46 ver, National Institutes of Health (NIH), Bethesda, MD, USA].
Multilineage Differentiation Into Adipogenic and Osteogenic Lineage
For adipogenic and osteogenic differentiation, cultivated cells were replated on 24-well culture plates at a density of 1.0 × 104 and 5 × 103 cells/cm2 in α-MEM basal medium (Gibco BRL) supplemented with 10% FBS and 1% antibiotic/antimycotic. At a confluence of more than 90%, cells were treated with adipogenic or osteogenic agents using human mesenchymal stem cell functional identification kit (R&D Systems, Inc., Minneapolis, MN, USA) according to manufacturer's protocol.
Adipogenic induction was performed for 2 weeks. Induced cells were fixed with a 10% formaldehyde solution, rinsed using distilled water (DW), and stained using Oil red O staining agent (red; R&D Systems Inc.) for 10 min at room temperature.
Osteogenic induction was performed for 3 weeks. Induced cells were fixed with a 10% formaldehyde solution, rinsed with DW, and stained using Alizarin red S staining agent (bright orange red; R&D Systems, Inc.) for 10 min at room temperature.
Rapid Endoderm and Hepatic Induction on Collagen Type I-Coated Plates
Collagen type I (Col I; BD Biosciences, Bedford, England) was coated onto six-well culture plates at a concentration of 5 μg/cm2. Endoderm and primitive hepatic induction was conducted using a three-step differentiation protocol published from a previous study 11 (Fig. 1).
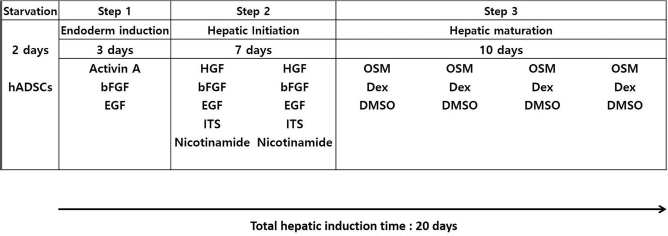
Detailed schedule for the three-step differentiation protocol.
Treatment with Activin A
In step 1 (rapid early endoderm induction), cells were cultivated in serum-free expansion medium supplemented with 10 ng/ml basic fibroblast growth factor (bFGF; R&D Systems, Inc.), 10 ng/ml epidermal growth factor (EFG; R&D Systems, Inc.), and 100 ng/ml activin A (R&D Systems Inc.) at a density of 1.5 × 104 cells/cm2 for 3 days.
Hepatic Differentiation with Various Growth Factors
Cells activated by activin A were cultured in serum-free conditioning medium supplemented with 50 ng/ml hepatocyte growth factor (HGF; R&D Systems, Inc.), 30 ng/ml bFGF, 20 ng/ml EFG, 1x insulin–transferrin–selenium (ITS; Gibco BRL), and 0.6 mg/ml nicotinamide (Sigma-Aldrich). The maturation medium was administered for 7 days in step 2. For step 3, the same medium supplemented with 20 ng/ml oncostatin M (OSM; R&D Systems, Inc.), 4.9 mmol dexamethasone (Sigma-Aldrich), and 0.1% dimethyl sulfoxide (DMSO; Sigma-Aldrich) was administered for 10 days. Medium change was performed two times per week.
Immunocytochemistry Assay for Confirmation of Hepatic Induction
When the cells were at 7 days into hepatic differentiation step 3, they were fixed with 10% formaldehyde solution at 20°C for 10 min and permeabilized with 100% ice-cold methanol for 5 min. Solutions for blocking and dilution of the primary antibody consisted of PBS and 5% BSA. Cells were blocked for 45 min and incubated sequentially overnight at 4°C with rabbit polyclonal anti-human cytokeratin 19 (CK19) (Cat. No. sc-25724; 1:250; Santa Cruz Biotechnology) and goat polyclonal anti-human albumin (ALB; Cat. No. sc-46293; 1:250; Santa Cruz Biotechnology). Goat anti-rabbit IgG conjugated with TR (Cat. No. sc-2780; 1:500; Santa Cruz Biotechnology) and donkey anti-goat IgG conjugated with FITC (Cat. No. sc-2024; 1:500; Santa Cruz Biotechnology) were used according to the species of the primary antibody. Nuclei staining was performed using 4′,6-diamidino-2-phenylindole (DAPI; Invitrogen, Carlsbad, CA, USA).
RT-PCR Analysis for Confirmation of Endoderm and Hepatic Induction
Cells were trypsinized on the third passage. Total RNA was extracted from the cells using the TRIzol isolation method on day 7 of induction in passage 2 and days 7 and 10 of induction in passage 3. RT-PCR was performed using reverse transcriptase according to the manufacturer's protocol. The cDNA was amplified manually and quantified using NanoDrop™ Lite Spectrophotometer. PCR analysis was performed with various primers. Primer sequences and conditions are listed in Table 1. Density of genes relative to the glyceraldehyde 3-phosphate dehydrogenase (GAPDH) gene was determined by ImageJ software.
Periodic Acid-Schiff Staining for Glycogen Storage
Cells were fixed with a 10% formaldehyde solution for 10 min after 10 days on the third passage and rinsed with DW. Cells were stained with periodic acid–Schiff solution (PAS; Sigma-Aldrich) for 5 min, rinsed with DW, and immersed with Schiff's reagent for 15 min. Hematoxylin (Sigma-Aldrich) was provided for cell counterstaining.
ELISA for Albumin Secretion
At days 7 and 10 of hepatic induction on the third passage, the medium from the cultivated cells was extracted and added to a 96-well plate coated with anti-human serum albumin (HSA) antibody (Cell Biolabs Inc., San Diego, CA, USA). After the incubation for 2 h at 37°C, the next procedure was performed according to the Human Albumin ELISA kit (Cell Biolabs Inc.) protocol.
Statistical Analysis
Quantitative data are expressed as means±SD (standard deviation). Analysis was repeated more than five times on the results of two samples from each patient. Significance was determined using paired t-tests and two-way analysis of variance (ANOVA) with Turkey's HSD (honest significant difference) tests for multiple comparisons post hoc using GraphPad Prism 5 for Windows (GraphPad Software, Inc., La Jolla, CA, USA). Values of p < 0.05 were considered statistically significant.
Results
Characteristics of hLF-ADSCs Versus hAS-ADSCs
Both the hLF-ADSCs and the hAS-ADSCs had a similar fibroblast-like shape after 24 h (Fig. 2A). The hLF-ADSCs had a higher rate of proliferation than the hAS-ADSCs (Fig. 2B). The hLF-ADSCs expressed all embryonic stem cell markers such as octamer-binding transcription factor 4 (OCT4), NANOG, sex-determining region Y box 2 (SOX2), and chemokine receptor type 4 (CXCR4), as well as mesenchymal lineage markers such as VIMENTIN, TWIST, SNAIL1, and N-cadherin (Fig. 2C). However, unlike hAS-ADSCs, hLF-ADSCs showed a weak expression of E-cadherin, a marker of mesenchymal–epithelial transition (MET). Cytokeratin 7 (CK7), a hepatic endodermal marker13,14, was more highly expressed in cultured hLF-ADSCs. On flow cytometry datum, OCT4, NANOG, and SOX2 had similar expressions between hAS-ADSCs and hLF-ADSCs (Fig. 2D).
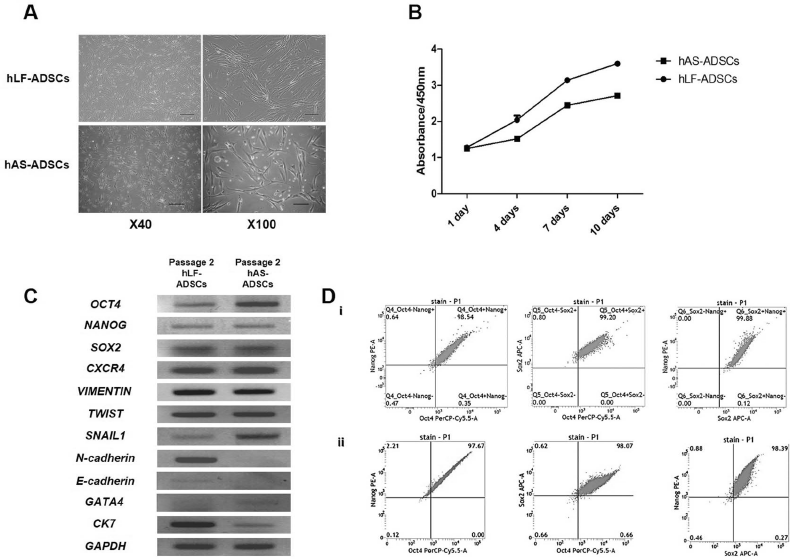
Cultivation, proliferation, and characterization of human liver falciform adipose-derived stem cells (hLF-ADSCs) harvested from human liver falciform ligament tissue. (A) hLF-ADSCs and human abdominal subcutaneous (hAS)-ADSCs transformed similarly 24 h after plating. (B) Both samples of hLF-ADSC were used for proliferation assays on passage 2. After 10 days of proliferation, hLF-ADSCs had greater cell proliferation than hAS-ADSCs. (C) In reverse transcriptase polymerase chain reaction (RT-PCR) analysis, similar to hAS-ADSCs, hLF-ADSCs on passage 2 had a high expression of the embryonic stem cell markers octamer-binding transcription factor 4 (OCT4), NANOG, sex-determining region Y box 2 (SOX2), and C-X-C chemokine receptor type 4 (CXCR4) genes, and the mesenchymal lineage markers VIMENTIN, TWIST, and SNAIL1. Unlike hAS-ADSCs, hLF-ADSCs had a higher expression of the mesenchymal–epithelial transition (MET) markers N-cadherin and E-cadherin, as well as the early hepatic endoderm marker cytokeratin 7 (CK7). (D) OCT4, NANOG, and SOX2 were highly expressed on hAS-ADSCs (i) and hLF-ADSCs (ii) on flow cytometry datum. Scale bars: x40 = 500 μm, x100 = 200 μm.
Surface Markers of hLF-ADSCs Versus hAS-ADSCs
The expression of surface markers on cultured hAS-ADSCs and hLF-ADSCs (third passage) was compared (Fig. 3A). The hAS-ADSCs had a high expression of positive surface markers such as CD105, CD73, and CD90, and no or weak expression of negative markers such as CD133, CD34, CD45, and CD13. Vimentin was highly expressed in hAS-ADSCs, but E-cadherin was barely expressed. Conversely, hLF-ADSCs showed expression of all MSCs and hematopoietic markers and were aggregated more during cultivation than hAS-ADSCs.

Expression of surface markers on human abdominal subcutaneous adipose-derived stem cells (hAS-ADSCs) and human liver falciform adipose-derived stem cells (hLF-ADSCs), and multilineage differentiation. (A) Human hAS-ADSCs on passage 3 expressed positive surface markers (CD105, CD90, and CD73) indicating presence of mesenchymal stem cells (MSCs). However, hLF-ADSCs expressed positive and negative markers (CD133, CD34, CD45, and CD13). Mesenchymal–epithelial transition (MET) markers [vimentin and E-cadherin (E-cad)] were also expressed on hLF-ADSCs. The hLF-ADSCs showed more cell aggregation during cultivation than hAS-ADSCs. (B) Quantified intensity of surface markers in (A) is shown. (C) The hLF-ADSCs were induced to differentiate into adipogenic and osteogenic lineages to confirm these cells had mesenchymal stem cell differentiation properties. In adipogenic-differentiated hLF-ADSCs, cells formed vacuoles and showed extensive staining with Oil red O for lipid (arrowhead). In osteogenic-differentiated cells, hLF-ADSCs were transformed to osteocyte-like cells, as indicated by extensive staining with Alizarin red S for calcium deposit (arrowhead). Scale bars: 200 μm. LM, light microscope.
Quantified intensity of surface markers in Figure 3A is shown in the graph in Figure 3B. Isolated and cultivated hLF-ADSCs differed in expression of markers for MSC characteristics (Fig. 3C). To confirm the substantial multipotency, hLF-ADSCs were differentiated into adipogenic and osteogenic lineages. After 2 weeks of adipogenic differentiation, cells formed various vacuoles, which were clearly identified with Oil red O staining (arrowhead). For osteogenic differentiation, cells were transformed to osteocyte-like cells after 3 weeks and broadly stained with Alizarin red S solution to confirm calcium deposit (arrowhead).
These results have shown that hLF-ADSCs and hAS-ADSCs have slightly different characteristics, but the potential for multilineage differentiation was present in both. Therefore, hLF-ADSCs were also considered to be a sufficient source of cells for differentiation into hepatic lineage.
Hepatic Induction of hLF-ADSCs
Cultured hLF-ADSCs and hAS-ADSCs (third passage) were differentiated into hepatic lineage using a 20-day three-step differentiation protocol.
In step 1 of differentiation, the cells were treated with a high concentration of activin A, which promotes rapid and efficient hepatic endoderm induction that is characterized by cells that have a small cytoplasm and large nucleus 15 . After the second step involving HGF treatment, cells return to a fibroblast-like shape. The third step, using OSM, transformed the cells into a polygonal shape 16 . Both cell types transformed into a polygonal shape by 17 days after differentiation. As differentiation progressed, the shape of these cells resembled that of hepatocytes and had a hexagonal shape. The changes in morphology were similar between hAS-ADSCs and hLF-ADSCs (Fig. 4A). PAS staining was performed to confirm the glycogen storage in hepatic-induced hAS-ADSCs and hLF-ADSCs. Hepatic-induced hAS-ADSCs and hLF-ADSCs both showed glycogen storage (arrowhead) on day 20 after the differentiation. Nondifferentiated hAS-ADSCs and hLF-ADSCs remained unstained (Fig. 4B).

Morphological changes, periodic acid–Schiff (PAS) staining, and gene expressions during the three-step hepatic differentiation of human abdominal subcutaneous adipose-derived stem cells (hAS-ADSCs) and human liver falciform adipose-derived stem cells (hLF-ADSCs) on plates coated with collagen I. (A) Both hAS-ADSCs and hLF-ADSCs adopted a polygonal shape on day 7 into step 3 (S3-7D), and a hexagonal shape (similar to hepatocytes) on day 10 into step 3 (S3-10D) of hepatic induction. (B) Hepatic differentiation of hAS-ADSCs and hLF-ADSCs showed similar staining with PAS agent, which confirmed glycogen storage in both cells (arrowhead). (C) Polymerase chain reaction (PCR) analysis was performed with cDNA extracted on passage 3 of differentiating cells after step 2 of induction (S2-7D) and on days 7 and 10 of step 3. PCR analysis of undifferentiated cells (Undiff.) after passage 2 was performed for guanine–adenine–thymine–adenine-binding protein 4 (GATA4) and cytokeratin 7 (CK7). With the exception of GATA4, all hepatic endoderm markers had a higher expression in hLF-ADSCs after step 2 of induction (S2-7D). In particular, the hepatic progenitor markers cytokeratin 19 (CK19) and albumin (ALB) increased throughout differentiation in the hLF-ADSCs. FOXA2, forkhead box A2; HNF4α, hepatocyte nuclear factor 4 α. Scale bars: 200 μm.
To confirm the functional capacity of hepatic differentiation, PCR analysis was performed with cDNA extracted from differentiating cells (passage 3). For comparison, analysis was carried out after step 2 of induction and on days 7 and 10 after step 3 of induction. Differentiating hAS-ADSCs showed greater expression of early endoderm marker guanine–adenine–thymine–adenine-binding protein 4 (GATA4) than hLF-ADSCs. However, all hepatic endoderm markers, with the exception of GATA4, had a higher expression in hLF-ADSCs than in hAS-ADSCs after step 2 of induction. In particular, the hepatic progenitor markers CK19, AFP, and ALB were notably increased as differentiation progressed (Fig. 4C). Furthermore, the cytochrome family 3A4 (CYP3A4), a functional hepatic induction marker 18 , had a higher expression in hLF-ADSCs than in hAS-ADSCs.
Our immunohistochemistry results showed that hLF-ADSCs and hAS-ADSCs had similar expressions of CK19 and ALB (Fig. 5A). Secretion of albumin in hepatic-induced hLF-ADSCs tended to be higher than in hAS-ADSCs, but the difference was not significant (Fig. 5B).
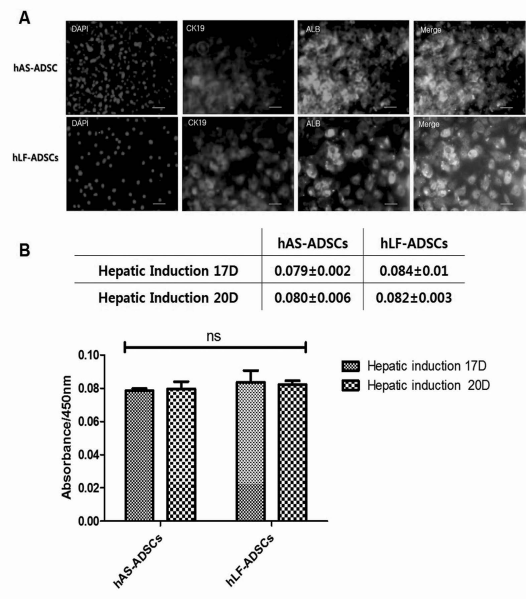
Cytokeratin 19 (CK19) and albumin (ALB) expression and albumin secretion in hepatic-induced human abdominal subcutaneous adipose-derived stem cells (hAS-ADSCs) and human liver falciform adipose-derived stem cells (hLF-ADSCs). (A) Immunocytochemistry assays showed that hLF-ADSCs and hAS-ADSCs had similar expression of hepatic induction markers such as CK19 and ALB. (B) ELISAs performed on days 17 and 20 of hepatic induction showed that hLF-ADSCs tended to have higher expression of secreted albumin protein than hAS-ADSCs, although this was not statistically significant. DAPI, 4′,6-diamidino-2-pheylindole. Scale bars: 200 μm.
Discussion
As stem cells develop, their characteristics and differentiation processes are determined by the origin of their tissue 19 . Moreover, different characteristics are possible even in the case of similar types of MSCs 20 . Among them, ADSCs have been known for easier isolation than MSCs derived from the bone marrow, and for reducing patient's burden on pain 21 . For these reasons, ADSCs are getting more attention as a source of MSCs for therapy22,23.
Differentiation studies for liver therapy utilizing ADSCs have been reported by many researchers2,3,21,24-26. Even though adipose tissues have similar characteristics, they have subtle genetic differences depending on their location.
Aparnathi et al. 27 have successfully isolated canine ADSCs from falciform and subcutaneous adipose tissue. The multipotency of canine ADSCs has been demonstrated from adipogenic and osteogenic differentiation. Differentiation potential of canine ADSCs was similar for both falciform and subcutaneous adipose tissue. Our study utilized liver falciform ligament as a source of autologous tissue for liver therapy. The beneficial effect of MSCs in liver cirrhosis and acute liver failure has been extensively demonstrated in both animal and clinical studies 28 . MSCs derived from adipose tissue of liver falciform ligaments, which maintain and support the anatomical position of the liver 5 , are formed during liver development. They are hypothesized to differentiate more efficiently into hepatic endoderm lineage than ADSCs from abdominal subcutaneous adipose tissue.
Isolation and cultivation of hLF-ADSCs have shown that its morphology, cell proliferation, and adipogenic and osteogenic differentiation were similar to hAS-ADSCs, which are commonly used by many researchers. The hLF-ADSCs also expressed ESCs and mesenchymal lineage markers such as OCT4, NANOG, SOX2, CXCR4, VIMENTIN, TWIST, and SNAIL1 genes, while the expression of E-cadherin, a marker of MET, was higher than in hAS-ADSCs. Therefore, hLF-ADSCs were confirmed to be mesenchymal lineage cells that had genetic characteristics for differentiation.
The expression of surface proteins on hLF-ADSCs was slightly different than on hAS-ADSCs. The hLF-ADSCs expressed both positive markers (CD105, CD73, and CD90) that are expressed in all MSCs, as well as negative markers (CD133, CD34, CD45, and CD13). The hLF-ADSCs contain numerous capillaries due to their origination from the liver falciform ligaments 6 . Additionally, unlike hAS-ADSCs, the hLF-ADSCs had marked aggregation during cell proliferation. Lang et al. 29 have reported that aggregates of CD133+ cells derived from the prostate cell line showed active cell division and multipotency. This morphological phenotype could have resulted from aggregation of self-assembled CD133+ cells. However, we were not able to evaluate negative control (e.g., IgG-treated cells) to confirm this finding, and limitation exists on the explanation of the results.
Hepatic induction of hLF-ADSCs was performed to confirm the effect of tissue origin on properties of cell differentiation. Differentiation was more efficient with hepatic induction of hLF-ADSCs than with hAS-ADSCs. GATA4 30 , an early endoderm marker in cell differentiation, had a higher expression in hAS-ADSCs during hepatic induction. However, the expression of late endoderm and hepatic endoderm markers, such as FOXA2, CK19, CK7, AFP, HNF4α, and ALB13,11, increased more rapidly in hLF-ADSCs during the second step of hepatic induction. Also, CYP3A4, a gene related to detoxification in the liver 18 , was highly expressed in hLF-ADSCs. These gene expression findings indicated that hLF-ADSCs were more efficient at undergoing hepatic differentiation into functional hepatic-like cells than hAS-ADSCs. This finding may be explained by the fact that the hLF-ADSCs were derived from neighboring liver tissue.
Furthermore, our immunocytochemistry results have shown that albumin, an indicator of a functional protein within the cell, had a higher expression in hLF-ADSCs than in hAS-ADSCs after 17 days of hepatic induction. Although the difference lacked statistical significance, taken together with the data on gene and protein expressions this may indicate that the differentiation of the hLF-ADSCs into hepatic-like cells was more efficient than hAS-ADSCs. These results support our hypothesis and suggest that the hLF-ADSCs may be a better candidate in the investigation of cell therapies for patients with liver disease. However, the degree of secreted ALB showed no significant increase in the ELISA assay at 20 days after hepatic induction. It could be said that hAS-ADSCs and hLF-ADSCs may have a similar functional property, and it could be suggested that the hLF-ADSCs may be another candidate in the investigation of cell therapies for patients with liver disease.
Conclusion
In this study, the authors examined the degree of differentiation into hepatocyte-like cells utilizing two types of ADSCs. Utilized human ADSCs were extracted from abdominal subcutaneous adipose tissue and the liver falciform ligaments. Abdominal subcutaneous tissue is widely used for the development of general ADSCs. Liver falciform ligament tissue is typically discarded in patients undergoing partial hepatectomy for liver disease. Using this tissue as a source of ADSCs may be a more effective therapeutic approach for patients with liver disease. As evidenced by its rapid induction into hepatic-like cells, ADSCs from the liver falciform ligament may be a source of autologous adult stem cells for therapeutic purposes related to liver disease while minimizing side effects related to autoimmunity.
Footnotes
Acknowledgment
This study was supported by a faculty research grant of Yonsei University College of Medicine for 2013 (6-2013-0152). The authors declare no conflicts of interest.
