Abstract
Psoriasis is a dermatologic disease of immune origins with no definitive cure. We report the Makati Medical Center experience of utilizing autologous mesenchymal stromal cells (MSCs) for one patient with psoriasis vulgaris (PV) and another with psoriatic arthritis (PA). Patients were educated and gave informed consent, according to the principles of the Declaration of Helsinki. The protocol was approved by the Cellular Transplantation Ethics Committee of the Makati Medical Center. Autologous MSCs were cultured from lipoaspirate and expanded in a clean room class 100 facility (Cellular Therapeutics Center, Makati Medical Center). MSCs were infused intravenously at a dose of 0.5–3.1 million cells/kg after complying with quality control parameters. Psoriasis area and severity index (PASI) evaluations were conducted by third-party dermatologists. The PA patient, who was previously unresponsive to standard treatment modalities, demonstrated a decrease in PASI (from 21.6 to 9.0, mild state after two infusions). No improvements were noted in joint pain until further treatment with etanercept and infliximab. The PV patient, who was previously dependent on methotrexate, showed a decrease in PASI from 24.0 to 8.3 after three infusions; this clinical improvement was sustained for 292 days (9.7 months) without methotrexate. The PV patient illustrated a marginal reduction in serum tumor necrosis factor-α (TNF-α), while significant (3.5- to 5-fold) decreases in reactive oxygen species (ROS) activity were noted. The ROS levels correlated with the clinical improvement of the PV patient. No serious adverse events were noted for either patient as a result of MSC infusions. This report demonstrates safe and tolerable transplantation of autologous MSCs for the treatment of psoriasis and warrants large clinical studies to investigate the long-term safety and efficacy of this approach.
Introduction
Background on Psoriasis
Psoriasis is a chronic inflammatory dermatologic disease exhibiting a wide spectrum of clinical signs. Its cutaneous manifestation is characterized by hyperproliferation of basal keratinocytes, thickened and scaly epidermis, and recruitment of inflammatory cells to the skin (12). Psoriasis was primarily considered a proliferative disorder with secondary inflammation, but some observations have led to the reexamination of this idea: T-cell infiltrates precede epidermal changes (lesions) (14), and bone marrow transplantation from psoriatic donors has led to T-cell infiltrates preceding epidermal changes (lesions) and development of psoriasis (15). Immune system dysregulation is now considered a root cause of the disease, which leads to the inflammatory and hyperproliferative manifestations. Genetics, environmental triggers, and infections also influence its occurrence and severity (8).
The basic characteristics of psoriatic lesions (redness, thickness, and scaliness) provide a means of assessing the severity of psoriasis. The current gold standard for the assessment of extensive psoriasis is the psoriasis area and severity index (PASI) (6). The PASI is a measure of the average redness, thickness, and scaliness of lesions, accounting for skin area.
Psoriasis patients may also develop arthritis, ranging from mild and nondestructive symptoms, to severe and rapidly degrading arthropathy. Psoriasis is correlated with other conditions such as uveitis and, possibly, reduced life expectancy due to adverse events associated with the drugs used in treatment (12).
Standards of Care for Psoriasis
Treatment strategies for psoriasis include combinations of emollients, topical corticosteroids, vitamin D analogs, cyclosporine, tar preparations, psoralen with ultraviolet A, and oral and topical retinoids. At present, there is no cure for psoriasis; current treatments are meant to manage symptoms. Therapeutic approaches such as biologics and cell transplantation are currently being developed and explored in search for disease-modifying cures.
Cellular Therapy for Psoriasis and Autoimmune Inflammatory Diseases
A few studies have demonstrated the clinical potential of hematopoietic stem cell transplantation (HSCT) following immunoablation for psoriasis patients. It may be considered that the immunosuppressive drugs are responsible for this effect, as it may deplete the autoreactive T cells, while HSCT is necessary to reconstitute the immune system and prevent infection. Among the longest spanning cases reported was clinical benefit lasting 20 years for a male with disabling psoriatic arthropathy who received allogeneic HSCT (16,17). The psoriasis remitted for 1 year, the arthritis remitted for 5 years, and the patient's overall quality of life improved for 15 years.
Complete remission of psoriasis has also been observed after autologous HSCT. A psoriatic patient with a 15-year history of psoriasis vulgaris (PV) and psoriatic arthritis (PA) developed multiple myeloma and was given an immunoablative regimen followed by autologous HSCT (2). After treatment, the psoriasis completely remitted for 15 months. In a review of 19 HSCT cases of hematologic patients with psoriasis, allogeneic HSCT performed on 10/13 resolved the psoriasis (mean of 49 months), but was associated with higher transplantation-related mortality. In contrast, autologous HSCT performed on 5/6 patients was demonstrably safer than allogeneic transplantation, but relapsing disease was noted within 2 years posttransplantation (9). It was speculated that retained T-cell receptor diversity after autologous transplantation limited the efficacy of this approach.
We investigated the use of autologous mesenchymal stromal cells (MSCs) instead of HSCs to avoid the safety problems associated with immunoablation and allogeneic transplantation. We also assessed whether MSCs could provide clinical benefit due to their well-documented immunomodulatory properties (11). While the mechanisms of MSC-mediated immunosuppression are still uncertain, some functions of MSCs have been established: they inhibit T-cell proliferation in vitro, inhibit natural killer (NK) cell activity in vitro, disrupt the differentiation of HSCs and monocytes in vitro, and decrease the antigen-presenting capacity of dendritic cells. Furthermore, they have pleiotropic effects on B-cell proliferation and possibly act through myriad signaling pathways. One study established the synergistic roles of interferon-γ (IFN-γ) and tumor necrosis factor-α (TNF-α) in the ability of MSCs to induce a regulatory/immunosuppressive phenotype in Th17 cells (a subset of T cells significant in psoriasis), downregulate IL-17 production, and increase IL-10 production (7).
MSCs are currently being evaluated in several clinical trials on immune disorders such as Crohn's disease, multiple sclerosis, type 1 diabetes, autoimmune hepatitis, and graft-versus-host disease. One trial is registered at the NIH ClinicalTrials.gov database utilizing umbilical cord-derived MSCs for psoriasis (3). We explored the utility of adipose-derived MSCs (ADSCs) for the following reasons: relative ease of sample acquisition, abundance of MSCs in fat, and the observation that dermal MSCs in psoriasis patients have atypical properties (higher growth rates, varying gene expression patterns, and lower free radical-scavenging activity) (13).
The main objectives were to measure safety and note any indicators of potential clinical utility for these conditions.
Case Presentation
Patient A, Case CTC P 0021 00 13AT01
A 58-year-old Filipino male weighing 52 kg was diagnosed with psoriasis since 1987 and arthritis since 2008. The patient suffered from generalized erythematous scaly patches and thin plaques sparing the central face and soles of both feet; nail pitting and onycholysis on all fingernails and toenails; scales distributed on the upper extremities, lower extremities, chest, abdomen, and bulk; and difficulty ambulating, standing, and bathing.
Over the course of the patient's disease, he underwent the following treatment modalities: clobetasol ointment, methotrexate, prednisone, etanercept, celecoxib, and various herbal remedies. He did not experience durable remission of symptoms at the time of consultation for MSC therapy.
Patient B, Case CTC P 0030 00 14AT01
A 28-year-old Filipino female weighing 45 kg was periodically receiving methotrexate weekly for a period of 5 years (from the age of 16 to 21) for PV. She was later diagnosed with hyperthyroidism at the age of 25. An attempt to wean off the steroid was instigated at age 22, but pulse doses were still required every month when symptoms flared. The patient also reported a variable therapeutic outcome using methotrexate. MSC therapy was considered with the goal of reducing the patient's methotrexate dependence for her condition.
Materials and Methods
In accordance with the Declaration of Helsinki, patients were educated regarding the protocol (including the potential for no benefit) and given ample time to give their informed consent. Afterward, patients were screened for basic eligibility according to the standards set by the Center: complete blood chemistry, complete blood count, clotting parameters, human immunodeficiency virus and hepatitis virus profiles, pregnancy test, X-rays, and cardiopulmonary clearance. The protocol that follows was approved by the Cell Transplantation Ethics Committee, duly recognized by the Philippine Department of Health.
Cell Harvest
Patients underwent miniliposuction procedures under tumescent anesthesia (lidocaine). About 200 ml of subcutaneous abdominal fat was aspirated and transported to the Cellular Therapeutics Center. Patients were discharged after no evidence of adverse reactions was observed.
Culture of Mesenchymal Stromal Cells
Lipoaspirate was washed three times with phosphate-buffered saline (PBS; pH 7.2) to separate the bloody fraction from the lipid fraction. Afterward, the lipid fractions were pooled and digested using collagenase I, collagenase II, and thermolysin (Roche, Mannheim, Germany) in a shaking water bath (37°C) for 1.5 h. The cell fraction was isolated via centrifugation and plated in complete culture media: Dulbecco's modified Eagle's medium (Life Technologies, Grand Island, NY, USA), 10% fetal bovine serum (filter sterilized using a 0.20-μm microfilter; Life Technologies), 2 mM L-glutamine (Life Technologies), 100 U/ml penicillin (Life Technologies), and 100 μg/ml streptomycin (Life Technologies). Cells were fed every 4–5 days and passaged as necessary (up to passage 3) via enzymatic digestion with trypsin/ethylenediaminetetraacetic acid (EDTA) (Life Technologies). All culture activities were performed under aseptic conditions in an ISO 14644-1 cleanroom class V facility with stringent quality control standards, and all reagents were tested for sterility beforehand.
Quality Testing
In accordance with the position statement of the International Society for Cellular Therapy, MSCs at minimum are characterized by the following: plastic adherence, a CD73+/90+/105+/11b-/19-/34-/45-/HLA-DR- surface phenotype, and the ability to differentiate into adipocytes, chondrocytes, and osteocytes in vitro (4). Flow cytometry (BD FACSCalibur; BD Biosciences, San Jose, CA, USA) was used to determine the cell surface phenotype of cultured MSCs (BD Stemflow Kit; BD Biosciences), while standard growth factor cocktails to induce trilineage differentiation were utilized to test for differentiation potential (Differentiation Kits; Life Technologies).
Sterility of cultures was determined via testing for mycoplasma DNA, endotoxins, and bacterial/fungal growth. Mycoplasma DNA was measured by polymerase chain reaction (PCR; iNtRON Biotechnology, Kyungki-do, South Korea), endotoxins by the Limulus amoebocyte lysate gel clot assay (Lonza, Walkersville, MD, USA), and microbial growth by culture for 2–5 days in an independent facility (Department of Pathology and Laboratories, Makati Medical Center).
For cell quantification and viability, trypan blue (Life Technologies) exclusion was used.
Intravenous Cell Administration
On the days of infusion, cells were collected from the culture vessels via trypsin/EDTA and repeatedly washed with PBS (pH 7.2) to remove any residual media. Afterward, cells were suspended in a final solution of 0.9% parenteral normal saline solution (NSS).
For each infusion, an initial assessment (including vital signs) was performed on the patient, NSS was run intravenously, and the cellular product was infused as a concurrent side drip. Patients were discharged after additional monitoring for adverse events for 2 h. Follow-up over the course of 12 months included documentation of adverse events, PASI scoring, as well as clinical photography.
Results
Culture and Infusion
Patient A received two infusions of MSCs (days 0 and 40), and patient B received three infusions (days 0, 30, and 71). The timing of infusions was based on our unpublished study set by the attending physician. Patient B received more infusions than patient A in order to maximize any potential clinical benefit. Cultures showed no signs of contamination, and cell identity parameters corresponded well with MSC characteristics (summarized in Table 1). Both transplantation procedures were well tolerated, with no adverse reactions being recorded throughout the first year of follow-up.
Summary of Quality Parameters for Infused Cells

Sterility data not shown: all cultures were negative for mycoplasma DNA, endotoxins, and microbial growth. MSC, mesenchymal stromal cell.
Patient A
On the day of the first infusion, the PASI score of patient A was 21.6 (summarized in Fig. 1), with massive scaling, notable right knee swelling and pain, shoulder pain, finger pain, and toe pain. Forty days later, his PASI was 8.9, with notable decreases in erythema, induration, scaling, and lesions. Joint pain, condition of nails, and knee swelling were unchanged, although itching decreased significantly. The reduced PASI was sustained up to day 157, with noted reduction in onycholysis.

Summary of psoriasis area and severity index (PASI) scoring for both patients pre- and posttransplantation of MSCs. The PASI score of each patient decreased during follow-up visits postinfusion of MSCs. Scoring was performed by third-party dermatologists. Clinical improvement was durable and sustained beyond the 150th day for patient A and beyond the 200th day for patient B.
The patient's self-reported quality of life did not improve significantly, and a third-party rheumatologist was consulted for persistent joint pain. According to follow-up by telephone, after receiving an etanercept regimen 10 months posttransplantation, significant improvement was noted in lesions, scaling of the trunk and extremities, and joint pain, allowing him to ambulate and perform physical exercise. Building on this observation, the assessment for patients after patient A included serum TNF-α and ROS in blood. In a final follow-up with the patient in October 2015, it was found that upon receiving infliximab 1 month after the aforementioned etanercept regimen, further clinical improvement was noted, with only about 10% of body surface area containing erythematous patches. However, upon contracting pulmonary tuberculosis 2 years after the MSC transplantation, his psoriatic lesions and arthritis relapsed.
Patient B
Photographic documentation for this patient is shown in Figure 2. The PASI of patient B during the time of consultation was 9.9, while receiving methotrexate, clobetasol, loratadine, and hydroxyzine. In order to prevent any untoward incident related to MSC–drug interactions, the patient was weaned off methotrexate for 23 days before the day of the first infusion. (To date, no human in vivo study demonstrated the safety of coadministering MSCs with methotrexate in psoariasis.) On the day of the first infusion, her PASI was 24 in the absence of medication such as clobetasol, anthralin, and hydroxyzine.

Photographic documentation (patient B). (A) Baseline (prior to cell transplantation); (B) 30 days after the first infusion; (C) 121 days after; and (D) 231 days after. Significant improvements were noted in the reduction of lesions in the back. Reductions in scaling cannot be appreciated in the photographs provided. All photographs were taken with the patient's consent, and no digital enhancements were made.
On the day of the second infusion (30 days after the first), her PASI was 18.3 (summarized in Fig. 1); onycholysis on fingernails and toenails was noted. At this point, the patient was receiving topical clobetasol propionate, aquaphor, and betamethasone valerate.
During the third infusion, her PASI reduced further to 9.4 with no indications of onycholysis or pitting. At this point, the patient was receiving topical clobetasol propionate, aquaphor, betamethasone valerate, and oral hydroxyzine medications. Forty-five days after, her PASI score was maintained at 9.6 (similar to levels with methotrexate use), with recurrence of onycholysis and pitting.
By the 231st day, her PASI was 8.3 while receiving calcipotriol, halobetasol–aquaphor, carbimazole, and antihistamines as needed. Onycholysis and pitting were sustained. The patient required methotrexate for the treatment of a recurrent flare after 292 days after MSC therapy.
Tumor Necrosis Factor-α and Oxidative Stress
While an overall decrease in serum TNF-α was seen by the 197th day (Fig 3), values did not deviate significantly from the range of healthy individuals, 11.2 ± 7.3 pg/ml (1). In contrast to TNF-α levels, the levels of ROS in whole blood decreased 3.5-fold (from 1,286 to 355 fluorescence units) on the 94th day, reaching a fivefold reduction (255 fluorescence units) on the 221st day posttransplantation (Fig 4). Additionally, the proportion of sampled cells in whole-blood fluorescing beyond an arbitrary threshold (1,000 fluorescence units) decreased from 56% to 1.2% over a 100-day period and was sustained at day 221 (Fig. 4, inset).
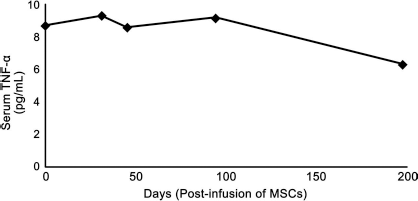
Serum TNF-α measurements by ELISA (patient B). By day 197, the concentration of TNF-α decreased to 6.3 pg/ml from a baseline measurement of 8.7 pg/ml. However, all values were still within the ranges of healthy subjects.
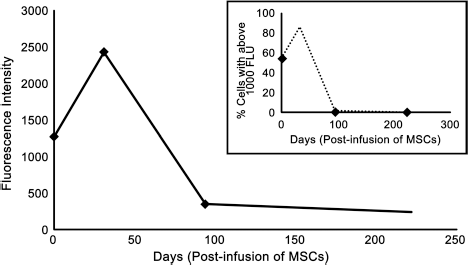
ROS measurement by flow cytometry (patient B). The mononuclear cell fraction in whole blood was assayed using a fluorescein derivative and measured using flow cytometry. By day 31, levels of ROS increased 1.9-fold. By day 94, ROS decreased 3.5-fold from baseline. The observation was sustained by day 221, with a 5.0-fold decrease from baseline. Inset: In order to substantiate the median fluorescence values obtained, an arbitrary threshold was chosen at 1,000 fluorescence units (FLU) to stipulate “high” and “low” levels of ROS activity. Proportions of cells above and below this threshold were then measured. Cell expressing levels of ROS above this threshold were present in 55.61% of cells at baseline, 86.36% of cells at day 31, 1.15% of cells at day 94, and only 0.54% of cells at day 221, reflecting the observations in median fluorescence.
Discussion
This study demonstrates the safety of autologous lipoaspirate-derived MSCs in psoriasis patients. The finding for patient A that etanercept provided clinical benefit after, but not prior to, MSC transplantation is of interest in light of in vivo immunomodulation. Our study was not designed to capture objectively the possibility of MSC-mediated immune “resetting,” which is considered as a plausible rationale for the treatment of autoimmune diseases by MSCs. We thus expanded the assessment for patients after patient A to include serum TNF-α and ROS in blood.
This finding also supports the hypothesis that MSC transplantation may have effects on the inflammatory microenvironment. On the basis of objective assessments of patient B, PASI scoring suggested a durable response (292 days after MSC therapy) comparable to the use of methotrexate with the aforementioned concomitant medications. This demonstrates the potential utility of MSC transplantation together with other standard medications aside from methotrexate, which has an established adverse effects profile upon extended use.
The timing and the number of infusions in cell-based therapy are two different parameters that are hard to establish. Unlike conventional drugs, infused cells do not follow classical pharmacokinetic behavior. On the basis of our preliminary unpublished study, we noted decreases in ROS after single infusion of autologous MSCs from 30 to 100 days. Thus, we designed the study to administer two to three MSC infusions covering the period between 30 and 100 days.
Many cytokines are implicated in psoriasis, but TNF-α plays a key role according to preclinical and clinical studies (5,10). TNF-α levels in skin blister fluids of psoriasis patients are higher than in uninvolved skin or in healthy controls, and serum levels were shown to correlate with PASI score pre- and posttherapy. Thus, serum TNF-α was measured for patient B. However, results did not indicate significant differences in serum TNF-α prior to and after cell transplantation (Fig 3). The measured TNF-α concentrations were maintained in the healthy range throughout.
Following this, we hypothesized that ROS may serve as a potential alternative biomarker for the clinical course of psoriasis. To this end, our Center validated an assay for measuring ROS-induced fluorescence via dihydrodichlorofluorescein diacetate in blood. We have previously noted decreased circulating ROS activity in healthy subjects after receiving autologous MSCs (unpublished data).
In contrast to serum TNF-α, measured ROS levels for patient B significantly decreased and were consistent with the observed PASI score. This finding suggests the potential utility of blood ROS measurement as a biomarker for the clinical course of psoriasis. Our preliminary findings on ROS levels suggest that oxidative stress may be involved, whether directly or indirectly, to observations made after transplantation of MSCs.
Conclusion
This report illustrates the safety and potential clinical utility of ADSCs for the treatment of autoimmune conditions such as PV and PA. Our experience with patient A suggests that response to standards of care may be modulated by MSCs. Patient B, on the other hand, demonstrated a durable response to MSC transplantation, resulting in lesser dependence on the potent immunosuppressant drug methotrexate. The current observational study can serve as preliminary evidence to support clinical investigations into the use of MSCs for these conditions.
Footnotes
Acknowledgments
We would like to thank Dr. Diorella Anne Capco for her management of patient A. Furthermore, we express our gratitude to Dr. Ma. Purita G. Paz-Lao for her insights and inputs. We also greatly appreciate the helpful discussions and contributions of Dr. Baltazar D. Aguda to the manuscript. Last, we would like to thank the Philippine Department of Science and Technology (DOST) for financial assistance for the publication of this manuscript. Francisco S. Chung, Ph.D., is a DOST balik scientist program recipient. The authors declare no conflicts of interest.
