Abstract
The mechanism by which stem cell-based therapy improves heart function is still unknown, but paracrine mechanisms seem to be involved. Adipose-derived stem cells (ADSCs) secrete several factors, including insulin-like growth factor-1 (IGF-1), which may contribute to myocardial regeneration. Our aim was to investigate whether the overexpression of IGF-1 in ADSCs (IGF-1-ADSCs) improves treatment of chronically infarcted rat hearts. ADSCs were transduced with a lentiviral vector to induce IGF-1 overexpression. IGF-1-ADSCs transcribe100- to 200-fold more IGF-1 mRNA levels compared to nontransduced ADSCs. IGF-1 transduction did not alter ADSC immunophenotypic characteristics even under hypoxic conditions. However, IGF-1-ADSCs proliferate at higher rates and release greater amounts of growth factors such as IGF-1, vascular endothelial growth factor (VEGF), and hepatocyte growth factor (HGF) under normoxic and hypoxic conditions. Importantly, IGF-1 secreted by IGF-1-ADSCs is functional given that Akt-1 phosphorylation was remarkably induced in neonatal cardiomyocytes cocultured with IGF-1-ADSCs, and this increase was prevented with phosphatidylinositol 3-kinase (PI3K) inhibitor treatment. Next, we tested IGF-1-ADSCs in a rat myocardial infarction (MI) model. MI was performed by coronary ligation, and 4 weeks after MI, animals received intramyocardial injections of either ADSCs (n = 7), IGF-1-ADSCs (n = 7), or vehicle (n = 7) into the infarcted border zone. Left ventricular function was evaluated by echocardiography before and after 6 weeks of treatment, and left ventricular hemodynamics were assessed 7 weeks after cell injection. Notably, IGF-1-ADSCs improved left ventricular ejection fraction and cardiac contractility index, but did not reduce scar size when compared to the ADSC-treated group. In summary, transplantation of ADSCs transduced with IGF-1 is a superior therapeutic approach to treat MI compared to nontransduced ADSCs, suggesting that gene and cell therapy may bring additional benefits to the treatment of MI.
Keywords
Introduction
The repair of an injured tissue/organ through stem cell therapy has obvious appeal in regenerative medicine. However, the source of stem cells and, most importantly, the mechanism by which stem cell therapy repairs or regenerates the host tissue are still in debate1,2. Among various adult tissues where stem cells reside, adipose tissue contains a great amount of stem cells capable of differentiating into osteogenic, chondrogenic, adipogenic, and myogenic cell lines when treated with specific factors 3 . Moreover, adipose-derived stem cells (ADSCs) can be easily obtained from patients through a simple and minimally invasive procedure in the subcutaneous tissue. Because of these properties, ADSCs have become good candidates for cellular therapy, especially in chronic degenerative diseases3–7.
Recently, we demonstrated that intramyocardial ADSC injection reduced left ventricle akinetic area and slightly increased the ejection fraction (EF) in healed infarcted rat hearts when compared to the group receiving vehicle, which progressively deteriorated its cardiac function 8 . ADSCs also increased cardiac contractility and decreased the perimeter of the infarct area 8 . Our data corroborate other basic9,10 and preclinical studies11,12, although the mechanism of improvement has not been clarified yet.
Two of the most important characteristics of ADSCs are their angiogenic potential and antiapoptotic potential. ADSCs secrete growth factors such as vascular endothelial growth factor (VEGF) and hepatocyte growth factor (HGF), and when challenged by hypoxic conditions in vitro, the rate of VEGF secretion increases considerably 13 . Moreover, ADSCs protect primary cardiomyocytes from apoptosis induced by hypoxia and serum deprivation 14 , an effect that was not observed after silencing the insulin-like growth factor-1 (IGF-1) gene in the ADSCs 14 .
IGF-1 activates pathways that regulate several important physiological functions, including cell growth, proliferation, and survival15,16, and both IGF-1 and its receptor (IGF-1R) are expressed by cardiomyocytes17,18. IGF-1 activation of the phosphatidylinositol 3-kinase (PI3K)-Akt pathway induces physiological cardiac hypertrophy, which leads to an increase in heart contractile function18–21. Indeed, IGF-1-deficient mice have increased apoptosis after myocardial infarction (MI) 22 , whereas overexpression of IGF-1 specifically in cardiac cells protected against apoptosis and prevented ventricular dilatation after MI23,24. Furthermore, IGF-1 overexpression also protected against cellular death and improved cardiac function in aged animals24,25. In the context of cell therapy, treatment of bone marrow-derived mesenchymal stem cells (BM-MSCs) with IGF-1 (10 mg/ml) for 15 min activates PI3K-Akt signaling and thus increases cell survival under hypoxic conditions in vitro while also improving cardiac function in vivo, whereas untreated cells only stabilized cardiac functional parameters 26 .
Sustained release of growth factors by stem cells is a promising tool to increase the rate of success after cardiac transplant with the aim of intensifying the beneficial paracrine effects of the transplanted cells27–29. BM-MSCs genetically modified to overexpress IGF-1 improved the indices of left ventricular function in an acute rat model of MI 27 . Recently, it was reported that isolated human cardiac stem cells transduced with IGF-1 exhibited enhanced proliferation and stimulation of prosurvival signaling 29 . Infarcted mice treated with these cells showed improved cardiac function. However, IGF-1 overexpression targeting the easily obtainable ADSCs had not been tested. Furthermore, in our work, we tested the ability of the combined cell/gene therapy to improve cardiac function in a healed MI model, since the IGF-1-transduced cells were injected 4 weeks after MI.
Materials and Methods
Ethics Statement and Animals
All procedures were performed in accordance with the Revised Guide for the Care and Use of Laboratory Animals [National Institutes of Health (NIH), Bethesda, MD, USA] and were approved by the Committee on the Ethics of Animal Use of the Federal University of Rio de Janeiro under No. IBCCF 026. Syngeneic female and male Wistar rats (from the animal facility of the Cellular and Molecular Cardiology Laboratory at University of Rio de Janeiro, Rio de Janeiro, Brazil) were exposed to a 12-h light/dark cycle (lights on at 7:00 a.m. to 7:00 p.m.) and had access to both water and standard rat chow ad libitum. For cell isolation, five male Wistar rats (12–16 weeks old) weighing 300 to 450 g were used. For in vivo procedures, 39 female Wistar rats (8–10 weeks old) weighing 200 to 250 g were used.
Cell Culture
ADSCs were isolated by a method based on density and differential adhesion, as previously described 8 . Inguinal subcutaneous adipose tissues of five male syngeneic Wistar rats (12–16 weeks old; 300–450 g) were washed with sterile phosphate-buffered saline (PBS; Cultilab, Campinas, SP, Brazil) to remove contaminating debris and red blood cells. Washed tissue was minced, and cells were isolated by enzymatic digestion (0.1% type II collagenase; Worthington Biochemical, Lakewood, NJ, USA) in PBS for 45 min at 37°C with gentle agitation. Enzyme activity was neutralized by Dulbecco's modified Eagle's medium (DMEM; Cultilab) supplemented with 20% fetal bovine serum (FBS; Cultilab), and cells were centrifuged for 10 min at 200 × g at room temperature. The supernatant, containing mature adipocytes, was discarded. Pellets were filtered through 80-μm filters; cells were washed in PBS three times, counted in a Neubauer chamber, and checked for viability using 0.4% trypan blue (Sigma-Aldrich, St. Louis, MO, USA). Cells were then plated at a density of 5 × 104/cm2 in 150-cm2 cell culture flasks. Culture medium used was DMEM-high glucose, containing 20% FBS (Cultilab), 2 mM L-glutamine (Sigma-Aldrich), 100 U/ml penicillin (Gibco, Saranac, NY, USA), and 100 mg/ml streptomycin (Gibco). Following 24 h of incubation, nonadherent cells were removed by washing with PBS, and the cultures were maintained at 37°C under normoxic conditions (95% air, 5% CO2). Medium was changed twice a week, and cells were trypsinized [tripsin-ethylenediaminetetraacetic acid (EDTA) 0.25%; Sigma-Aldrich] at 80% confluence. Cells were used at passage 3 (P3) in this study.
IGF-1 Vector Construction
The pEF1.IGF-1.T2A.puro lentiviral vector was constructed by cloning the coding regions of the rat IGF-1 gene in a multiple cloning site of the commercial vector CDH.EF1.MSC.T2A.puro (Thermo Fisher Scientific, Carlsbad, CA, USA) (Fig. 1). The lentiviral particles containing the IGF-1 gene and the puromycin N-acetyl transferase gene (which confers resistance to the antibiotic puromycin) were produced by cotransfection of HEK293T packaging cells with vectors pEF1.IGF-1.T2A. puro and the vectors and accessories pΔ8.9 pMD.G. After incubation for 16 h, the culture medium was replaced with fresh medium (DMEM 10% FBS). Forty-eight and 72 h after transfection, culture media containing the lentiviral particles were collected, filtered through 0.45-μm filter (Sigma-Aldrich), and the lentiviral particles were concentrated by centrifugation at 49,000 × g for 90 min at 4°C. Viral pellets were used immediately or frozen at −70°C for future use.

Lentiviral vector construct. The pEF1.IGF-1.T2A.puro lentiviral vector was constructed by cloning the coding regions of the insulin-like growth factor-1 (IGF-1) gene in a rat multiple cloning site of the commercial vector CDH.EF1.MSC.T2A.puro.
Transduction and Selection of ADSCs
ADSCs in culture dishes (100 mm2; Corning Inc. Corning, NY, USA) at third passage and 50% confluence were transduced with vector pEF1.IGF-1. T2A.puro in a total volume of 8 ml in the presence of 8 μg/ml polybrene (Sigma-Aldrich). The medium was replaced with fresh medium 16 h after transduction, and 48 h after transduction we initiated the selection of positively transduced cells by the addition of puromycin (Sigma-Aldrich) at a concentration of 2.5 mg/ml. At the end of selection, a portion of the cells was frozen, and the remainder was expanded for subsequent experiments.
Total RNA Extraction and Quantitative RT-PCR
IGF-1 mRNA expression in transduced and nontransduced cells, IGF-1-ADSCs and ADSCs, respectively, was analyzed by polymerase chain reaction (PCR) and quantified by real-time PCR (qRT-PCR). Total RNA was extracted and treated with DNase using the kit RNeasy Mini Kit (Qiagen, Hilden, Germany) following the manufacturer's instructions. Total RNA (1 μg) was transcribed into cDNA with random primers using a high-capacity reverse transcription kit (Thermo Fisher Scientific) following the manufacturer's instructions. Amplifications were performed in an ABI Prism Model 7500 thermocycler (Applied Biosystems, Carlsbad, CA, USA) using a Power SYBR Green Master Mix (Applied Biosystems) and sense and antisense primers. The amplification program was: 55°C for 2 min, 95°C for 10 min, followed by 40 cycles of 95°C for 30 s and 58°C for 1 min.
The efficiency of amplification was evaluated by serial dilutions of the cDNA template. Each cDNA was amplified in triplicate, and a corresponding sample without the enzyme reverse transcriptase (RT sample) was included as a negative control. Furthermore, the expression of the target gene (IGF-1) was normalized by the expression of glyceraldehyde-3-phosphate dehydrogenase (GAPDH), which was used as an endogenous control.
The variation in the expression of the target gene in response to treatments was calculated as follows: mean ± SD of 2 – (ΔCt) for each group (transduced and nontransduced) and followed by determining the relationship between the transduced/nontransduced.
The sequences of the primers were as follows: endogenous IGF-1 (NM_001082477)—3′-untranslated region (3′-UTR) of the IGF-1 gene in rat: 5′-AAAGTCAGCTCGTTCCATCC-3′ (sense) and 5′-TGTTTCCTGCACTTCCTCTAC-3′ (antisense); total IGF-1 (NM_001082477)—gene coding region of rat IGF-1: 5′-GTCGTCTTCACATCTCTTCTACC-3′ (sense) and 5′-ACAGTACATCTCCAGCCTCC-3′ (antisense); and GAPDH (NM_017008)—constitutive gene in all cells: 5′-GACATGCCGCCTGGAGAAAC-3′ (sense) and 5′-AGCCCAGGATGCCCTTTAGT-3′ (antisense).
Population Doubling Time (PDT) Study
All characterizations of ADSCs and IGF-1-ADSCs were done at the third passage. ADSCs (transduced and nontransduced) were submitted to population doubling time (PDT) assays. The cells were grown in a culture plate grid (Thermo Fisher Scientific) where each square has a 4-mm2 area. Cells (104) were plated per dish on day 0. The number of cells in four quadrants chosen at random was estimated daily by manual counting under phase-contrast microscopy, until the culture reached a stage of confluent monolayer. With these values, we constructed a graph of the average number of cells per unit area (y-axis) for each day of the experiment (x-axis). The y-axis was transformed to a base 2 logarithm scale, and we performed linear regression. The inverse of the angular coefficient was used to calculate PDT. Three independent experiments were made in duplicate for each experimental condition.
ADSC and IGF-1-ADSC Resistance to Oxidative Stress
When cells were 70% confluent, they were exposed to different concentrations of H2O2 (50, 70, and 100 μM). Cells were incubated with MTT [0.5 mg/ml of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide, a yellow tetrazole; Sigma-Aldrich] for 0.5 h in the dark at 37°C. Subsequently, after 5 min of incubation with dimethyl sulfoxide (DMSO; Sigma-Aldrich), optical density (OD) was measured at 570 nm in a VictorTMX4 spectrophotometer (PerkinElmer, Waltham, MA, USA). Four independent experiments were done for each experimental condition.
Hypoxic and Normoxic Assays
ADSC and IGF-1-ADSC cultures were subjected to hypoxic conditions in an incubator with a humidified atmosphere with the conditions of 1% O2 and 5% CO2 for a period of 48 h. Cells were also maintained at normoxic conditions (control). In these experiments, we subjected all cultures to serum deprivation. Subsequently, cells and conditioned media (CM) were collected and analyzed by flow cytometry and enzyme-linked immunosorbent assay (ELISA).
Collection of ADSC and IGF-1-ADSC Conditioned Medium
After 48 h of conditioning (hypoxic or normoxic), the CM obtained were centrifuged (speed of 300 × g) and filtered using 0.22-μM filters. After collecting the medium, 200-μl aliquots were prepared and used in an ELISA assay. Approximately 1 ml was obtained by conditioning of 105 cells.
Flow Cytometry and Differentiation Protocols
ADSCs and IGF-1-ADSCs were subjected to immunophenotypic characterization. Cultured ADSCs were harvested with 0.25% trypsin and suspended as single cells at a concentration of 5 × 105 cells in 500 μl PBS (10 mM, pH 7.4). To verify that cultured cells were MSCs, the cells were analyzed by flow cytometry (FACSCanto; BD Biosciences, Franklin Lakes, NJ, USA) after labeling with CD45-PercP-CY5 (BD Pharmingen™, San Diego, CA, USA), CD34-PE (Santa Cruz Biotechnology, Dallas, TX, USA), CD11b-FITC (Caltag Laboratories, Burlingame, CA, USA), CD29-FITC (BD Pharmingen), and CD90-FITC (Caltag Laboratories). Isotype IgG1 immunoglobulin served as a control. In addition, these cells were cultured in the presence of osteogenic medium [10 nM dexamethasone, 10 mM β-glycerophosphate, 50 μg/ml L-ascorbate 2-phospate, and 10 nM 1α, 25-dihydroxyvitamin D3 (all from BIOMOL Research Laboratories Inc., Plymouth Meeting, PA, USA)] and adipogenic medium [DMEM-low glucose supplemented with 15% fetal calf serum (FCS), 50 U/ml penicillin, 50 mg/ml streptomycin, 5 × 10−4 M indomethacin, 10 μg/ml human insulin, and 10−7 M dexamethasone (all from Sigma-Aldrich)].
Measurement of Growth Factor Secretion by ELISA (VEGF, IGF-1, and HGF)
CM from ADSCs and IGF-1-ADSCs were collected, and concentrations of angiogenic and antiapoptotic growth factors (IGF-1, VEGF, and HGF) secreted by them were measured quantitatively using ELISA kits [IGF-I DuoSet (No. DY791), VEGF DuoSet (No. DY564), and HGF DuoSet (No. DY2207) (all from R&D Systems, Minneapolis, MN, USA)]. IGF-1 (SRP3069; 100 μg; Sigma-Aldrich) was added to serum-free medium and used as a positive control at a dose of 0.1 mg/μl. The optical density of each well was determined immediately using a microplate reader (ELISA plate spectrophotometer; Molecular Devices, Sunnyvale, CA, USA). The secreted cytokines were standardized to the quantity of DNA as picograms of cytokine per 1 g of DNA. Four independent experiments were done in duplicate for each experimental condition.
Cocultivation of Cardiomyocytes with ADSCs/IGF-1-ADSCs
In vitro effects of ADSCs and IGF-1-ADSCs co-cultivated with cardiomyocytes isolated from the hearts of neonatal Wistar rats (1–2 days after birth) were tested. Briefly, hearts from neonatal rats were removed, minced, and serially digested by dissociation solution (NaCl, 136.7 mM; KCl, 2.68 mM; Na2HPO4, 0.352 mM; NaHCO3, 11.9 mM; dextrose, 11 mM) containing pancreatin (1.25 mg/ml; Sigma-Aldrich) and bovine serum albumin (BSA; 3 mg/ml; Sigma-Aldrich). Each successive digestion lasted for 5 min at 37°C with gentle stirring, and the cells obtained were suspended in growth medium containing 15% FBS for inhibition of proteolytic enzymes. Elimination of nonmuscle cells was achieved by preplating twice for 1 h each. After that, the cells were counted and seeded in 24-well plates at a density of 104 cells/well and cultured in DMEM-high glucose containing 10% FBS at 37°C and 5% CO2. Experimental in vitro conditions were established 24 h after plating.
ADSCs and IGF-1-ADSCs were plated on Transwell membranes of 0.4 μM (Millipore, Billerica, MA, USA). After adhesion of neonatal rat cardiomyocytes (NRCs) on the culture plate, the membranes containing ADSCs or IGF-1-ADSCs were placed in the wells for cocultivation. The coculture was maintained for 15 min at 37°C in a humid atmosphere in the presence of 5% CO2 with 1.3 ml of DMEM culture medium without FBS. After 15 min of cocultivation, the NRCs were processed for immunofluorescence assay. In some wells, LY294002 (a PI3K pathway inhibitor; #9901; Cell Signaling Technology, Danvers, MA, USA) was added 1 h before and throughout the coculture at a concentration of 50 μM. The inhibition was done in order to check the biological effect of IGF-1 secreted by ADSCs (transduced and nontransduced with the IGF-1 gene) in the cardiomyocytes. As a positive control, we added IGF-1 (SRP3069; Sigma-Aldrich) in two wells at a concentration of 10 mM, and as negative control, we used NRCs and ADSC cultures alone. Three independent experiments were done in duplicate for each experimental condition.
Immunofluorescence
The cells were fixed in a solution containing 0.1 M phosphate buffer and 4% paraformaldehyde (PFA; Sigma-Aldrich) at room temperature and maintained at 37°C for 15 min. After this time, all of the PFA was removed, and the fixed cells were washed with PBS and stored in PBS at 4°C until immunofluorescence was performed. Negative controls were used to test the specificity of the secondary antibodies. The primary antibodies used were Akt (polyclonal #9272; Cell Signaling Technology) at 1:200 dilution; phospho-Akt (Ser473) (monoclonal #4058; Cell Signaling Technology) at 1:200 dilution; and cardiac troponin T (monoclonal #ab8295; Abcam, Cambridge, UK) at 1:200 dilution. The secondary antibodies used were anti-rabbit conjugated to Cy3 (No. 711165152; Jackson ImmunoResearch, West Grove, PA, USA) at 1:1,000 dilution and anti-mouse conjugated to Alexa 488 (A-11029; Thermo Fisher Scientific) at a dilution of 1:400. Cells were incubated with the primary antibodies overnight at 4°C, and with the secondary antibodies for 2 h at 37° C. The coverslips were then incubated for 10 min with TO-PRO-3 iodide (No. 3605; diluted 1:1,000; Thermo Fisher Scientific), and the slides were assembled. The slides were analyzed in an inverted fluorescence microscope Axiovert 130 (Zeiss, Oberkochen, Germany) coupled to a digital photomicrograph capture system (Axiovision 4.7; Zeiss) where the images obtained were processed (n = 3).
MI and Cell Transplantation
A total of 39 female Wistar rats (8–10 weeks) weighing 200 to 250 g were used for in vivo procedures. MI was induced by left coronary artery (LCA) ligation as previously described 30 in 34 female Wistar rats. Sham-operated (SHAM, n = 5) animals underwent the same procedure, but the coronary ligature was left untied. Twenty-nine percent of the animals (10 out of 34) died during MI induction or in the 24 h after surgery. Four weeks after MI induction, 3 out of 24 infarcted animals that survived had left ventricular EF (LVEF) above 50% and were excluded. The large initial number of animals was chosen to provide at least five to six rats per group for each of the measurements, as a high mortality rate is expected for rats subjected to MI (30–40%). Four weeks post-MI, rats were subjected to another thoracotomy and received an intramyocardial injection of 100 μl of 2 × 106 IGF-1-ADSCs in Matrigel(BD Biosciences) (n = 7), 100 μl of 2 × 106 ADSCs in Matrigel (BD Biosciences) (n = 7), or 100 μl of vehicle (PBS/Matrigel solution) (n = 7). Six weeks after treatment, the animals were subjected to functional evaluation and then sacrificed by cervical dislocation under anesthesia. The hearts were excised and processed for histology (see below) and to track the injected cells by PCR detection of the Y chromosome.
Echocardiographic Assessment (ECHO)
Echocardiographic examination was performed blinded by the same operator, following the American Society of Echocardiography (ASE) recommendations. We used an echocardiographic color system (Bio Sound Esaote, Indianapolis, IN, USA) equipped with a 10-MHz electronic-phased array transducer. Short-axis two-dimensional views of the left ventricle (LV) were taken at the level of the papillary muscles to obtain the M-mode recordings. EF was measured as EF% = (EDV – ESV)/EDV, where the end-diastolic (EDV) and systolic volumes (ESV) were calculated by the method of Teichholz et al. 31 , using the ultrasound software. Parameters were evaluated 28 days after infarction (1 h before treatment) to compare the baseline values in all groups and 6 weeks after treatment.
Left Ventricular Pressure Measurements in Anesthetized Rats
One week after the last echocardiogram measurement, animals were anesthetized with ketamine (Dopalen; Vetbrands, Paulínia, SP, Brazil)/xylazine (Anasedan; Vetbrands) (50 and 5 mg/kg, respectively, by intraperitoneal administration), and the right carotid artery was cannulated with a PE10 catheter. One extremity of the catheter was inserted into the left ventricle, and the opposite end was connected to a pressure transducer (MLT0380/D; ADInstruments, Colorado Springs CO, USA) coupled to a Power Lab400 acquisition system (ADInstruments), and LV pressure acquisition started. The left ventricular parameters evaluated were as follows: left ventricular end-diastolic pressure (LVEDP), left ventricular end-systolic pressure (LVESP), left ventricular developed pressure (LVDP = LVESP – LVEDP), cardiac index of left ventricular contractility (dP/dt+: rate of pressure rise), and index of left ventricular relaxation (dP/dt–: rate of pressure decay). All values were measured using Chart 4 software (PowerLab; ADInstruments).
Histological Study
After all cardiac functional analyses were performed, the animals were sacrificed and had their hearts excised for histological analysis. After being arrested in diastole by KCl (30 mM) perfusion, the hearts were fixed by immersion in 4% PFA in phosphate buffer. Ventricles were sectioned into four slices from the apex to the base (A, B, C, and D, respectively). Infarct size determination was performed by computer-assisted planimetry (ImageJ version 1.27z; NIH) as the mean percentage of the circumference occupied with the scar tissue on each section, after staining with picrosirius (Sirius red; Sigma-Aldrich) for collagen scar evaluation.
PCR for Identifying Y Chromosome
The PCR amplification was used to identify the Y chromosome to detect male transplanted cells. LV tissue from the injection site was isolated and stored in liquid nitrogen. Genomic DNA templates (150 to 300 ng/μl) from different groups were used in PCR reactions (20 μl) with rat sex determining region Y (Sry) primers according to the protocols published by Dvorianchikov et al. 32 . The primer sequence was as follows: 5′-CGTGAAGTTGCCTCAACAAA-3′ (sense) and 5′-TCTGGTTCTTGGAGGACTGG-3′ (antisense) (GenBank: NM_012772). Four independent PCR assays were done for each heart sample. Control reactions were performed using GAPDH primers with the following sequences: 5′-GGTGAAGGTCGGTGTGAAC-3′ (sense) and 5′-CGTTGATGGCAACAATGTC-3′ (antisense) (Gen Bank: NM_017008.3). PCR products were separated by electrophoresis on a 1.5% agarose gel with 1× Trisaminomethane Tris-acetate-ethylenediaminetetraacetic acid buffer (Tris buffer; Bio-Rad, Hercules, CA, USA), stained with EtBr (UltraPure Ethidium Bromide; Thermo Fisher Scientific), and photographed under ultraviolet light.
Statistical Analysis
All data are expressed as mean ± standard error of the mean (SEM). Unpaired or paired Student's t-tests were performed to compare two groups. One-way analysis of variance (ANOVA) was used to compare three or more groups followed by Bonferroni's post hoc test. When more than one time point was included, two-way ANOVA was applied followed by Bonferroni's post hoc test. Values were considered significant when p < 0.05. All analyses were performed with GraphPad Prism 6.0 software (GraphPad Software Inc., La Jolla, CA, USA).
Results
Characterization of IGF1-Transduced ADSCs (IGF-1-ADSCs)
Adherent ADSCs at third passage were transduced with pEF1.IGF-1.T2A.puro lentiviral vector (Fig. 1) and selected by puromycin treatment. IGF-1 transduction successfully induced overexpression of IGF-1 in ADSCs (Fig. 2A). We transduced two independent cultured ADSCs, and both total and endogenous IGF-1 mRNA levels increased more than 100-fold (Fig. 2A). The two transduced ADSCs were then combined and used for further characterization.
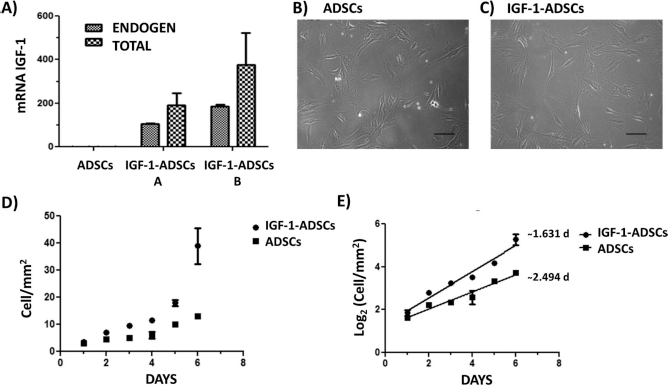
Characterization of IGF-1-transduced ADSCs (IGF-1-ADSCs). (A) IGF-1 mRNA in nontransduced cell cultures and two different cell cultures transduced with the lentiviral vector containing the IGF-1 gene. In both cases, transduction resulted in significant increases in both total and endogenous IGF-1. (B) Cultured ADSCs, after three passages, exhibit a spindle-shaped fibroblastic morphology (scale bar: 30 μm). (C) Cultured IGF-1-ADSCs, after three passages, exhibit the same spindle-shaped fibroblastic morphology (scale bar: 30 μm). (D) Number of cells (cells/mm2) in culture after plating 104 cells at day 0 and following cell number for 6 days of the experiment population doubling time (PDT). (E) Linear regression of logarithmic cell growth (cells/mm2).
IGF-1-ADSCswere adherent to culture flasks, showed fibroblast-like morphology, and were easily expanded in culture similarly to nontransduced ADSCs (Fig. 2B and C). However, IGF-1 overexpression enhanced the PDT from 2.5 (wild-type ADSCs) to 1.6 days (IGF-1-ADSCs) after plating at a density of 104 cells and expansion for 7 consecutive days (Fig. 2D and E).
ADSCs are capable of differentiating into mesodermal cell lineages 8 . This cell population is characterized by the expression of high levels of CD90 and CD29 and low levels of hematopoietic protein markers such as CD34 and CD45 on its cell membrane3,6,8. Thus, we analyzed the expression of these proteins by flow cytometry on IGF-1-ADSCs under normoxic or hypoxic conditions. Transduction of IGF-1 into ADSCs did not change the phenotypic characteristics of ADSCs under standard culture conditions nor under hypoxia (Table 1).
Cell Surface Markers Assessed by Flow Cytometry of Adipose-Derived Stem Cells (ADSCs) and Insulin-Like Growth Factor (IGF-1) in ADSCs

Cells were positive for CD29 and CD90 under normoxic and hypoxic conditions.
IGF-1-ADSCs Secrete Great Amounts of IGF, VEGF, and HGF
We next measured IGF-1 protein levels in the CM of ADSCs and IGF-1-ADSCs to address whether the increased IGF-1 mRNA expression induced increased secretion of IGF-1. Nontransduced ADSC medium conditioned for 48 h contained 0.007 ± 0.002 pg/ml of IGF-1 (Fig. 3A). Remarkably, overexpression of IGF-1 increased IGF-1 protein levels by 177-fold (Fig. 3A). To mimic the hypoxic environment of an infarcted myocardium, we challenged the cells under hypoxic conditions for 48 h and collected the medium. Interestingly, under low levels of oxygen (1%), IGF-1-ADSCs secreted even more IGF-1 (Fig. 3A). Furthermore, the combination of IGF-1 overexpression and hypoxia boosted the expression of HGF and VEGF (Fig. 3B and C).
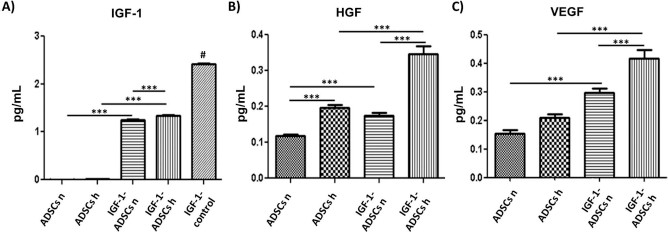
ADSC and IGF-1-ADSCsecretion of growth factors. (A) IGF-1 protein levels in the conditioned media of ADSCs and IGF-1-ADSCs under normoxia (ADSCs n and IGF-1-ADSCs n, respectively) and hypoxia (ADSCs h and IGF-1-ADSCs h, respectively). Medium with no serum but supplemented with IGF-1 was used as positive control. (B) Same as in (A) except that HGF protein levels were measured. (C) VEGF protein levels measured in the same conditions as in (B). IGF-1, insulin-like growth factor-1; HFG, hepatocyte growth factor; VEGF, vascular endothelial growth factor. One-way analysis of variance (ANOVA) was used to compare all groups followed by Bonferroni's post hoc test; ***p < 0.001; ns, nonsignificant. #p < 0.005 IGF-1 control versus ADSCs h, IGF-1-ADSCs h, ADSCs n, and IGF-1-ADSCs n.
IGF-1-ADSC-Secreted Soluble Factors Activate PI3K-Akt Signaling in Cardiomyocytes
So far, our results confirm that IGF-1 gene overexpression in ADSCs also increased the release of IGF-1 protein. However, one critical question is whether the experimentally induced IGF-1 produced by ADSCs is biologically active and able to activate the PI3K-Akt pathway. Thus, we cocultured ADSCs and IGF-1-ADSCs with NRCs using a setup where the cocultured cells shared the culture medium without physical contact and analyzed Akt phosphorylation (P-Akt) in the cardiomyocytes (Fig. 4). When cocultured with nontransduced ADSCs, troponin+ (TNPT), cardiomyocytes stained for P-Akt (Fig. 4A). Pretreatment of NRCs with the PI3K inhibitor LY294002 decreased Akt phosphorylation (Fig. 4B), confirming that ADSCs release soluble factors that activate the PI3K-Akt signaling pathway. Remarkably, Akt phosphorylation of NRCs was much higher when cocultured with IGF-1-ADSCs (Fig. 4C) compared to wild-type ADSCs (Fig. 4A). Also, inhibition of PI3K decreased Akt activation induced by IGF-1-ADSCs (Fig. 4D). As a positive control, we treated NRCs with recombinant IGF-1 and observed P-Akt expression in two different cultures of cardiomyocytes (Fig. 4E and F). Importantly, when cultured alone, neither NRCs nor ADSCs stained for P-Akt (Fig. 4G and H), and as expected, ADSCs were negative for TPNT (Fig. 4G).

Immunofluorescence for Akt and phospho-Akt (P-Akt) in neonatal rat cardiomyocytes (NRCs) when cocultured with ADSCs or IGF-1-ADSCs. In all images shown, NRCs are stained green for cardiac troponin T, red for P-Akt, and blue for cells nuclei (TOPRO). (A) NRCs cocultured with ADSCs. (B) NRCs pretreated with PI3K inhibitor LY294002 and cocultured with ADSCs. (C) NRCs cocultured with IGF-1-ADSCs. (D) NRCs pretreated with PI3K inhibitor LY294002 and cocultured with IGF-1-ADSCs. (E, F) NRCs treated with IGF-1. (G) ADSCs alone. (H) NRCs alone. All photos were taken with 20× objective and 10× viewing lenses (scale bars: 200 μm).
IGF-1-ADSCs Improve Cardiac Function in Chronically Infarcted Rat Hearts
ADSCs improved the heart function of rats with chronic infarction and skeletal muscle function of injured rats8,33. We tested whether infarcted hearts transplanted with ADSCs overexpressing IGF-1 would benefit from the combination of the new genetic and cellular approaches in comparison to the cellular treatment alone. ADSCs, IGF-1-ADSCs, or vehicle was injected directly into the myocardium in the border zone between infarcted and viable cardiomyocytes 4 weeks after coronary occlusion. Large chronic infarcts were confirmed by the significant fall in EF (~40–50% in infarcted rats compared to 80–90% of EF in sham-operated rats) (Fig. 5A) and the presence of Q-wave in the L1 ECG derivation. Importantly, there was no difference in EF between infarcted groups before treatment, indicating similar degree of cardiac injury.
Remarkably, IGF-1-ADSC-treated hearts showed improved EF (by ~25% when compared to baseline), while the ADSC-only group showed stabilized cardiac function 6 weeks after transplantation (Fig. 5B). On the other hand, vehicle-treated hearts continued to show deteriorating cardiac function (by ~20% when compared to baseline) over the course of the follow-up (Fig. 5B). We also measured left intraventricular pressures 7 weeks after cell injection (Table 2). Hemodynamic analysis revealed that intramyocardial injection of IGF-1-ADSCs improved cardiac contractility as measured by the intraventricular contractility index (dP/dt+) when compared to ADSC and vehicle administration (Table 2).
Hemodynamic Values for Experimental Groups 7 Weeks After Therapy

Functional parameters were obtained from the left ventricular pressure record. LVEDP, end-diastolic pressure; LVSP, systolic pressure; LVDP, pressure developed by the left ventricle; HR, heart rate; dP/dt+, index of cardiac contractility; dP/dt–, index cardiac relaxation. One-way ANOVA was used to compare all groups followed by Bonferroni's post hoc test.
p < 0.05 versus Sham group.
p = 0.0002, IGF-1-ADSCs versus infarcted groups: Vehicle and ADSCs.
IGF-1-ADSCs Reduce Myocardial Infarct Size in Chronically Infarcted Rat Hearts
After the ischemic acute phase, characterized by intense inflammatory responses and cell death, collagen deposition leads to the substitution of dead cardiomyocytes by scar tissue 34 . We measured MI size of the hearts in order to determine the role of IGF-1-ADSCs in scar remodeling. MI size as a percentage of the total left ventricle perimeter was significantly smaller in the IGF-1-ADSC and ADSC groups versus the vehicle group (vehicle, 48.3 ± 1.9%; ADSC, 37.1 ± 1.7%; and IGF-1-ADSC, 30.4 ± 3.3%) (Fig. 5C). However, there was no significant difference in MI size between the two cell-treated groups (Fig. 5C).

IGF-1-ADSC effect in infarcted rat hearts. (A) Ejection fraction (EF) determined by echocardiography 4 weeks after myocardial infarction (MI) and before cell treatment (baseline) of SHAM (n = 5), VEHICLE (n = 7), ADSC (n = 7), and IGF-1-ADSC (n = 7) groups. (B) EF of the infarcted groups before and 6 weeks after cell injection. *p < 0.05 and ***p < 0.001. Data are normalized by baseline values. (C) Morphometric analysis of MI perimeter (see Materials and Methods) of the infarcted groups (n = 5). *p < 0.05; ***p < 0.001 compared to vehicle. One-way ANOVA was used to compare all groups followed by Bonferroni's post hoc test. When more than one time point was included, two-way ANOVA was applied followed by Bonferroni's post hoc test. (D) Sry gene expression in cardiac tissue analyzed by RT-PCR. Animals 1 and 2 belong to the VEHICLE group; animals 3 and 4 belong to the ADSC group; and animals 5 and 6 belong to the IGF-1-ADSC group. After the sixth column: positive control using male rat heart (+), negative control using female rat heart (–), and ladder (L). (E) GAPDH control samples.
Cell Tracking
The transplanted cells were tracked by RT-PCR in left ventricular tissue samples from the three experimental groups (n = 2) by using the sex-determining region of the Y chromosome (Sry gene). All tissues from animals of the ADSC group were positive for Sry (Fig. 5D), confirming the presence of the transplanted cells even after 7 weeks posttreatment. Unexpectedly, all tissues of the IGF-1-ADSC were negative for Sry at the same time point (Fig. 5D). Tissues from the vehicle group, as expected, were also negative (Fig. 5D). These results suggest that although IGF-1-ADSCs induce better cardiac function outcomes, they do not survive as long as the nontransduced ADSCs. This unexpected result led us to test the resistance to oxidative stress by exposing the cells to different concentrations of H2O2 (50–100 μM). Exposure to 50 μM H2O2 did not alter either ADSC or IGF-1-ADSC proliferation (Fig. 6). Increasing H2O2 concentration to 70 and 100 μM impaired MTT incorporation in both cell types (Fig. 6). While there was no difference between cells at 70 μM, IGF-1-ADSCs were more sensitive to ROS when exposed to 100 μM H2O2 compared to ADSCs (Fig. 6).

Resistance to oxidative stress analysis. ADSCs and IGF-1-ADSCs exposed to increasing concentrations of H2O2 and MTT were measured. Two-way ANOVA was applied followed by Bonferroni's post hoc test; *p < 0.05, **p < 0.01, and ***p < 0.0001 compared to respective cells not exposed to H2O2; #p < 0.05 ADSCs versus IGF-1-ADSCs.
Discussion
IGF-1 activates pathways that regulate several important physiological functions, including growth, cell proliferation, and survival15,16. It is also a protein produced by the heart, and its receptor (IGF-1R) is expressed in cardiomyocytes17,18. IGF-1 leads to PI3K pathway activation in the heart, which is responsible for physiological hypertrophy 35 . Since the 1990s, several groups have studied the role of the IGF-1/PI3-K pathway in mouse hearts to determine its role in cardiomyocyte growth19,23,36. The beneficial effects of IGF-1 therapy in mouse and rat models of acute MI were well established20,23, but in a rat model of chronic ischemic heart failure, IGF-1 and growth hormone (GH) therapy was unable to increase EF or left ventricular dP/dtmax 37 .
The use of MSCs derived from bone marrow or adipose tissue has been extensively tested in small and large animal models of acute and chronic ischemic heart disease, and the positive results in the preclinical models led to clinical trials in patients (for a review see Karantalis and Hare 38 ). Of note, the PRECISE trial, using ADSCs, was not able to show a decrease in left ventricular infarcted mass in chronic ischemic patients 39 , which was in contrast to the APOLLO trial, wherein the same cell type promoted a significant decrease in left ventricular infarcted mass in acute MI patients 40 . The need to explore combination therapies for chronic ischemia is thus warranted, and we chose a combination of ADSCs and IGF-1.
Associating cell therapy with supplemental growth factors 26 and combining gene therapy with cell therapy is not a novel approach41–46, but to our knowledge, the present study was the first to use rat isogenic ADSCs transduced with the IGF-1 gene for therapy in a healed MI model. The studies performed herein show that IGF-1-mediated lentivirus transduction of ADSCs (IGF-1-ADSCs) successfully increased IGF-1 expression at both the mRNA and protein levels without altering MSC membrane surface markers. In contrast, as expected, IGF-1-ADSCs exhibited a higher proliferation rate. IGF-1 secreted by transduced ADSCs was biologically active, as shown by coculture experiments, where PI3K-Akt was more substantially expressed in cardiomyocytes cocultured in the presence of IGF-1-ADSCs compared to wild-type nontransduced ADSCs. Importantly, the transduced cells secrete not only higher levels of IFG-1 but also had significantly increased secretion of HGF and VEGF under normoxia or hypoxia.
The in vivo studies revealed that IGF-1-ADSCs improved cardiac function when injected into healed infarcted rats by increasing EF and dP/dtmax in relation to placebo or to nontransduced ADSCs, indicating that the cell and growth factor effects are additive. In contrast to the functional results, IGF-1ADSCs were not able to significantly decrease left ventricle scar formation when compared to nontransduced ADSC transplanted hearts. However, both cell types reduced MI size compared to vehicle-treated animals, and there was a clear trend of the transduced cells to have small MI perimeter, although they did not reach significance.
It has been shown that the grafted stem cells have the potential to secrete cytokines and growth factors through paracrine effects, protecting the remaining cells at the lesion site13,47–51. However, it is also known that the grafted cells do not survive for long in the site 52 . Therefore, several authors have tried to transduce the cells with genes like HGF, VEGF, Akt, and stromal cell-derived factor 1 (SDF-1), in an attempt to improve and prolong cell grafting41–43,45,46. Surprisingly, at the end of follow-up (7 weeks post-cell injection), PCR for the Sry gene showed that IGF-1-ADSCs were not present in the host heart, while the Sry gene was still present in the ADSC transplanted hearts. The faster disappearance of the transduced cells may be explained by the reduced resistance to oxidative stress displayed by IGF-1-ADSCs when compared to nontransduced ADSCs. At any rate, this finding has important implications, suggesting that the functional effects of cell therapy are not linearly related to cell survival in the heart.
Our findings are in agreement with the results reported by Enoki et al. 26 , where 1 month after the induction of MI in mice, the group of animals that received intramyocardial BM-MSCs previously treated with 10 μg/ml IGF-1 showed a significant improvement in left ventricular function, compared to all the other groups: treated with cells; treated with cells previously supplemented with IGF-1 and blocked with PI3-K inhibitor; which received only the growth factor IGF-1; and which received only vehicle 26 . In addition, we showed that intramyocardial injection of IGF-1-ADSCs in the healed infarct promoted a significant improvement in systolic function (cardiac contractility index) of this group compared to the ADSC and vehicle groups, which showed stabilization and progressive worsening, respectively. These findings are similar to those found by Blumenthal et al., who reported hemodynamic improvement in the group of infarcted rats treated with skeletal myoblasts overexpressing SDF-1 (with or without a biomaterial transplant) 45 .
Conclusion
In conclusion, the present study suggests that cell therapy combined with gene therapy has greater potential in regenerative medicine. In particular, IGF-1 overexpression in MSCs leads to greater cardiac improvement than MSC therapy alone in a chronic model of MI.
Footnotes
Acknowledgment
The present work was supported by CNPq, CAPES, Decit/MS, FAPERJ, and the National Institute of Structural Biology and Bioimaging. Author contributions: Concept and design (L. L. Bagno, J. P. Werneck-de-Castro, and A. C. Campos-de-Carvalho), cell culture (L. L. Bagno and D. Cipitelli), lentiviral vector construction (D. Carvalho), ASC transduction (L. L. Bagno and D. Carvalho), PCR assay (L. L. Bagno and D. Carvalho), ELISA assay (L. L. Bagno), flow cytometry (T. H. Kasai-Brunswick and L. L. Bagno), MTT assay (B. Andrade and D. Cipitelli), coculture and immunofluorescence assay (F. Mesquita), myocardial infarction induction (J. P. Werneck-de-Castro and G. Suhet), cell transplantation (L. L. Bagno, J. P. Werneck-de-Castro, and G. Suhet), ECG acquisition and analysis (L. L. Bagno), echocardiography acquisition and analysis (R. A. Louzada and L. L. Bagno), hemodynamic procedures and evaluation (R. A. Louzada and L. L. Bagno), PCR for identifying Y chromosome (V. M. Lago), histology (L. L. Bagno), data analysis and interpretation (L. L. Bagno, J. P. Werneck-de-Castro, and A. C. Campos-de-Carvalho), manuscript writing (L. L. Bagno, J. P. Werneck-de-Castro, and A. C. Campos de Carvalho). The authors declare no conflicts of interest.
