Abstract
There is growing interest in the use of mesenchymal stem cells (MSCs) to improve the outcome of organ transplantation. The immunogenicity of MSCs is, however, unclear and is important for the efficacy of MSC therapy and for potential sensitization against donor antigens. We investigated the susceptibility of autologous and allogeneic MSCs for lysis by CD8+ T-lymphocytes and NK cells in a kidney transplant setting. MSCs were derived from adipose tissue of human kidney donors and were CD90+, CD105+, CD166+, and HLA class I+. They showed differentiation ability and immunosuppressive capacity. Lysis of MSCs by peripheral blood mononuclear cells (PBMCs), FACS-sorted CD8+ T cells, and NK cells was measured by europium release assay. Allogeneic MSCs were susceptible for lysis by cytotoxic CD8+ T cells and NK cells, while autologous MSCs were lysed by NK cells only. NK cell-mediated lysis was inversely correlated with the expression of HLA class I on MSCs. Lysis of autologous MSCs was not dependent on culturing of MSCs in FBS, and MSCs in suspension as well as adherent to plastic were lysed by NK cells. Pretransplant recipient PBMCs did not lyse donor MSCs, but PBMCs isolated 3, 6, and 12 months after transplantation showed increasing lysing ability. After 12 months, CD8+ T-cell-mediated lysis of donor MSCs persisted, indicating there was no evidence for desensitization against donor MSCs. Lysis of MSCs is important to take into account when MSCs are considered for clinical application. Our results suggest that the HLA background of MSCs and timing of MSC administration are important for the efficacy of MSC therapy.
Introduction
Mesenchymal stem cells (MSCs) have beneficial regenerative and immunomodulatory properties that make them promising for use in the treatment and prevention of a variety of disorders, including graft-versus-host disease and multiple sclerosis (19,20,28). Recently, interest has risen in the use of MSCs to improve the outcome of organ transplantation (14). Animal models have demonstrated the ability of MSCs to prolong the survival of skin (5), heart (9,37), and islets allografts (15) and to facilitate the recovery of kidney function after acute and chronic injury (30,41). It is therefore anticipated that MSC therapy in transplant patients may allow a reduction in the dosing of immunosuppressive drugs and thereby limit their side effects, and to aid the repair of ischemia-reperfusion injury of the transplant (25).
The immunomodulatory functions of MSCs are largely mediated via soluble factors. Via these factors MSCs inhibit T-cell proliferation (1,15), maturation of dendritic cells, and differentiation of B lymphocytes (3,11,45). Moreover, MSCs inhibit the formation of cytotoxic CD8+ T cells (2) and proliferation and cytotoxic activity of natural killer (NK) cells (38,42,43). Reversely, it is less clear how immune cells affect MSCs. It is reported that MSCs have a low immunophenotype as they express low levels of human leukocyte antigen (HLA) class I, no HLA class II, and costimulatory molecules like CD80 and CD86, and that they therefore do not provoke immune responses (6,39). This would make MSCs transplantable across HLA barriers (29). In contrast, there is evidence that MSCs are immunogenic and can induce memory T-cell responses (4,32). Furthermore, it has been reported that MSCs are susceptible for lysis by NK cells (2,34,42) as MSCs express the activating NK cell receptor ligands NKG2D and UL16 (35). Interesting in this respect is the fact that intravenously administered MSCs have repeatedly been demonstrated to disappear within days after infusion in immunocompetent mice (37). It is not unlikely that lysis by cytotoxic cells is responsible for the disappearance of MSCs (4, 17). The observation of tumor formation after administration of autologous MSCs in immunodeficient mice further strengthens this possibility (18).
It has been demonstrated that exposure of MSCs to inflammatory conditions has an important impact on the immunomodulatory function of MSCs (27,36). In concert with the increased immunosuppressive properties of MSCs, these conditions also change the immunophenotype of MSCs by upregulating the expression of HLA molecules, costimulatory molecules, and adhesion molecules (8). This would make allogeneic MSCs potentially more susceptible for recognition by CD8+ T cells. The immunogenicity and survival of MSCs after administration is important for the efficacy of MSC therapy. Furthermore, in an organ transplant setting potential priming by donor MSCs is of great relevance as it may induce rejection of the donor graft or perhaps desensitize against donor antigens. Therefore, in the present study we investigated the susceptibility of autologous and allogeneic adipose tissue-derived MSCs for lysis by NK cells and CD8+ T cells. In addition, it was examined whether the susceptibility of donor-derived MSCs for lysis by recipient cytotoxic cells changed in time after kidney transplantation. The results of this study are important for the choice of MSCs and the timing of this therapy in organ transplantation.
Materials and Methods
Isolation and Culture of Perirenal Adipose Tissue-Derived MSCs
During the kidney donation procedure, perirenal fat tissue was surgically removed after written informed consent as approved by the Medical Ethical Committee of the Erasmus Medical Center Rotterdam (protocol No. MEC-2006-190) and collected in α-MEM medium (Gibco BRL, Paisley, UK) with 100 IU/ml penicillin and 100 μg/ml streptomycin (p/s, Gibco BRL). Adipose tissue-derived mesenchymal stem cells (ASCs) were subsequently isolated as previously described (12,24). Cultures were kept at 37°C, 5% CO2, and 95% humidity and refreshed twice weekly with MSC culture medium, consisting of α-MEM (Gibco BRL) with p/s and 15% fetal bovine serum (FBS, Biowithaker, Verviers, Belgium). At 90% confluence, adherent cells were removed from culture flasks by incubation in 0.05% trypsin-EDTA at 37°C and cells were used for experiments described below or frozen at −150°C until further use. MSCs were used for experiments between passages 2 and 5. When indicated, MSCs were cultured with 50 ng/ ml interferon-γ (IFN-γ; U-CyTech biosciences, Utrecht, The Netherlands) for 7 days.
Silencing of HLA Class I Expression
HLA class I expression by MSCs was modulated by small interfering RNA (siRNA). Seven μl of siRNA for HLA class I (Ambion, Austin, TX, USA) was added to 50 μl Opti-MEM (Gibco BRL). Twelve μl of si-Port (Ambion) was added to 50 μl Opti-MEM. siRNA and si-Port were mixed and incubated for 10 min at room temperature and added to a six-well plate. Cell suspension in α-MEM with 15% FBS (0.5 ml) was added and incubated for 3 days. Controls were treated with noncoding siRNA.
Flow Cytometric Characterization of MSCs
MSCs were trypsinized, washed twice with FACS Flow (BD Biosciences, San Jose, CA, USA), and incubated with antibodies against CD90-APC (allophycocyanin), CD105-FITC (fluorescein isothiocyanate; all R&D Systems, Abingdon, UK), CD166-PE (phycoerythrin; BD Biosciences), HLA class I-ABC-PE, CD86-PE (Serotec), and HLA class II-DR-FITC (BD Biosciences) for 30 min. After incubation, cells were washed twice with FACSFlow and flow cytometric analysis performed using an eight-color FACSCANTO-II with FACSDIVA Software (BD Biosciences).
Differentiation of MSCs
Osteogenic differentiation was induced by culturing confluent MSC cultures in α-MEM supplemented with 1% p/s, 15% heat-inactivated FBS, 5 mM β-glyceropho-sphate (Sigma-Aldrich, Munich, Germany), 50 μg/ml L-ascorbic acid phosphate (Sigma-Aldrich), and 10 nM dexamethasone (Sigma-Aldrich) for 21 days. Cells were then washed with PBS and fixed in cold 4% paraformaldehyde for 5 min at room temperature. After two washes with H2O, cells were incubated in 1% silver nitrate in H2O at room temperature on a light box until blackening occurred. The cells were then washed three times with H2O, incubated in 2.5% sodium thiosulfate in H2O for 5 min at room temperature, washed twice with H2O, and photographed.
Adipogenic differentiation was induced by culturing confluent ASC cultures in α-MEM supplemented with 1% p/s, 15% heat-inactivated FBS, 50 μg/ml L-ascorbic acid phosphate (Sigma-Aldrich), 500 μM 3-isobutyl-1-methylxanthine (IBMX, Fluka, Buchs, Switzerland), 60 μM indomethacin (Fluka), and 10 nM dexamethasone (Sigma-Aldrich) for 21 days. Cells were then fixed in 60% isopropanol for 1 min, and incubated in filtered 0.3% oil red O (Sigma-Aldrich) solution in 60% isopropanol for 10 min to stain lipid droplets. After several washes with PBS the cells were photographed.
Inhibition of Mixed-Lymphocyte Reactions by MSCs
The immunosuppressive capacity of MSCs was tested in mixed-lymphocyte reactions (MLR). Responder peripheral blood mononuclear cells (PBMCs; 5 × 104) were stimulated by 5 × 104 γ-irradiated (40 Gy) allogeneic PBMCs in RPMI + 10% HI-FBS in round-bottom 96-well plates (Nunc, Roskilde, Denmark). MSCs were added at various ratios to responder cells. On day 7, proliferation was measured by incorporation of [3H] thymidine (0.5 μCi/well) during a 16-h incubation using a β-plate reader.
Isolation of PBMCs
Peripheral blood samples were collected from living kidney donors and the corresponding kidney transplant recipients. Heparinized blood of kidney transplant recipients was drawn before transplantation, and 3, 6, and 12 months after kidney transplantation. PBMCs were isolated by density gradient centrifugation using Ficoll Isopaque (δ=1.077, Amersham, Uppsala, Sweden) and frozen at −150°C until use.
Living Kidney Transplant Recipient and Donor Pairs
To investigate the cytotoxicity-mediated lysis of MSCs in a clinical kidney transplantation setting, non-HLA-identical living kidney donor–recipient pairs were included. Recipient PBMCs isolated before transplantation and 3, 6, 12, 16, 18, and 24 months after transplantation were used as effector cells, while PBMCs and MSCs of the corresponding donor were used as targets in the cell-mediated lysis assays. Immunosuppressive therapy of kidney transplant recipients consisted of tacrolimus, mycophenolate mofetil, and low-dose steroids.
Cytotoxicity-Mediated Lysis by Europium Release Assay
Cytotoxicity-mediated lysis of target cells was determined by europium release assays as described previously (7,47). In brief, for effector cells PBMCs derived from recipients and healthy kidney donors were stimulated with allogeneic γ-irradiated PBMCs and 200 IU/ml IL-2 (proleukin; Chiron BV, Amsterdam, The Netherlands) in round-bottom 96-well plates for 7 days at 37°C. Phytohemagglutinin (PHA) blasts and MSCs were used as target cells. To obtain PHA blasts, PBMCs were stimulated with 200 IU/ml recombinant IL-2 and 2 μg/mL PHA in 24-well plates (Greiner Bio-one, Alphen a/d Rijn, The Netherlands) for 7 days at 37°C. At day 3 and 6, cell cultures were split in two and refreshed with culture medium containing 200 IU/ml recombinant IL-2. After 7 days, target cells were washed and living cells counted using an automatic cell counter system Casy (Schärfe System, Reutlingen, Germany). In addition, 100,000 target cells were used for flow cytometric analysis of HLA class I-ABC. The remaining target cells were labeled with europium-diethylenetriaminepentaacetate (DTPA) (Sigma, St. Louis, MO, USA). Autologous PHA blasts were used as negative control; allogeneic PHA blasts were used as positive control. MSCs were cultured under control conditions or with 50 ng/ml IFN-γ for 7 days.
At day 7, cytotoxicity-mediated lysis assays were performed by incubating 5 × 103 63Eu-labeled targets with various numbers of effector cells in 96-well plates for 4 h. Effector/target (ET) ratios ranged from 40:1 to 1.25:1. After incubation, the plates were centrifuged and 20 μl of the supernatant was transferred to 96-well plates with low background fluorescence (Fluoroimmunoplate, Nunc, Roskilde, Denmark). Subsequently, 100 μl enhancement solution (PerkinElmer, Groningen, The Netherlands) was added to each well. Released europium was measured in a time-resolved fluorometer (Victor 1420 Multilabel Counter, LKB-Wallac, Turku, Finland). Fluorescence was expressed in counts per second (cps).
Maximum release of europium by target cells was measured by incubation of 5 × 103 labeled target cells with 1% triton (Sigma-Aldrich, Zwijndrecht, The Netherlands) for 4 h. Spontaneous release of europium by target cells was measured by incubation of labeled target cells without effector cells. Percentage leakage was then calculated as: (spontaneous release/maximum release) x 100%. Mean percentage of leakage was 29 ± 9% for PHA blasts, 25 ± 6% for MSCs, and 26 ± 7% for IFN-γ pretreated MSCs. Finally, the percentage of cytotoxicity-mediated lysis was calculated as: % lysis = [(measured lysis – spontaneous release)/(maximum release –spontaneous release)] x 100%.
Isolation of CD8+ T Cells and CD16+56+ NK Cells by FACS
PBMCs cultured with allogeneic γ-irradiated PBMCs and 200 IU/ml IL-2 for 7 days were washed twice with PBS with 1% heat-inactivated FBS and stained with antibodies against CD3-PE, CD4-FITC, CD8-PE-Cy7, CD56-APC, CD19-Amcyan, 7AAD-viaprobe (all BD Biosciences), and CD16-APC (Serotec) at room temperature and protected from light for 30 min. Subsequently, cells were sorted using a FACS ARIA-II with FACS DIVA Software (BD Biosciences). The lymphocytes were gated for CD3. The CD3+ cells were then gated for CD8 to obtain CD8+ T cells. The CD3- cells were gated for CD16/56 to obtain NK cells. Purity of sorted CD8+ T cells and CD16+56+ NK cells was ≥ 98%. PBMCs of healthy kidney donors used as effector cells consisted of 23 ± 10% (mean ± SD) CD8+ T cells and 22 ± 11% NK cells. Effector cells derived from PBMCs of kidney recipients (12 months after transplantation) contained 22 ± 9% CD8+ T cells and 16 ± 8% NK cells.
Microarray Analysis
The expression of activating NK cell receptor ligands on MSCs was examined by microarray analysis, according to the manufacturer's description. In brief, 100 ng of total RNA of four MSC cultures was used to prepare sense-strand biotinylated DNA according to the manufacturer's “Whole transcript sense target labeling” protocol (Affymetrix, Santa Clara, CA, USA). Hybridization to Affymetrix Human Gene 1.0 ST Arrays (764,885 probe sets, representing 28,869 annotated genes), staining, washing, and scanning (Scanner 3000) procedures were performed as described by Affymetrix (Affymetrix) and performed by Erasmus MC Center for Biomics. Probe set summarization, Array QC, and annotations of the probesets were performed using Affymetrix “Gene Expression Consolle” (Affymetrix).
Statistical Analysis
Distribution of the data was tested with Kolmogorov-Smirnov test for normality. Parametric data were analyzed using two-way ANOVA for repeated measures with Bonferroni posttests, while nonparametric data were analyzed by Kruskal-Wallis. Parametric data are expressed as mean ± SD, while nonparametric data are expressed as median (25th–75th percentile, interquartile range). Statistical significance was defined as p < 0.05 (two-tailed).
Results
Characterization of MSCs
The cells isolated from perirenal adipose tissue showed an immunophenotype typical for MSCs with positive expression of CD90, CD105, CD166, HLA class I-ABC, and very low expression of CD86 and HLA class II-DR (Fig. 1A), as described earlier (12). They were adherent with a spindle-shaped morphology (Fig. 1B) and showed the capacity to differentiate into the osteogenic lineage, as demonstrated by von Kossa staining for calcified nodules (Fig. 1C) and the adipogenic lineage, as demonstrated by Oil red O staining for lipid vesicles (Fig. 1D). Typically for MSCs, they showed the capacity to dose-dependently inhibit the proliferation of allo-activated PBMCs (Fig. 1E).

Characterization of adipose tissue-derived mesenchymal stem cells (MSCs). (A) Immunophenotype of MSCs determined by flow cytometry. Gray histograms depict unstained MSCs, open histograms depict MSCs stained for the indicated marker. (B) Bright-field image of cultured MSCs. (C) Von Kossa staining of osteogenic differentiated MSCs. Black staining indicates calcified nodules. (D) Oil red O staining of adipogenic differentiated MSCs. Red staining indicates lipid-filled vesicles. (E) Effect of MSCs on allo-activated peripheral blood mononuclear cell (PBMC) proliferation. MSCs show dose-dependent inhibitory effect on PBMC proliferation.
Allogeneic MSCs Are Susceptible for Cytotoxicity-Mediated Lysis by CD8+ T Cells and NK Cells
To investigate the susceptibility of MSCs for lysis by cytotoxic immune cells, lysis of MSCs was compared to lysis of PHA blasts. Over 95% of MSCs and PHA blasts generated from PBMCs of healthy kidney donors expressed HLA class I-ABC. Cytotoxic effector cells were generated from PBMCs of healthy kidney donors by culture in the presence of γ-irradiated allogeneic PBMCs and 200 IU/ml IL-2 for 1 week. Effector cells lysed allogeneic PHA blasts in a dose-dependent manner (Fig. 2A). Allogeneic MSCs were lysed in a comparable fashion. To investigate whether lysis of allogeneic PHA blasts and MSCs was mediated by CD8+ T cells or NK cells, effector cells were sorted in CD3+/CD8+ T cells and CD3-/CD16+56+ NK cells. PHA blasts were lysed by CD8+ T cells, but not by NK cells (Fig. 2B). MSCs, in contrast, were lysed by both CD8+ T cells and NK cells (Fig. 2B). Lysis of MSCs by allogeneic NK cells was most efficient (58% of MSCs at an ET ratio of 1: 40 vs. 41% of MSCs by allogeneic CD8+ T cells at the same ET ratio). The lysis of MSCs by NK cells may be related to the expression of activating NK cell receptor ligands by MSCs (Fig. 2C).
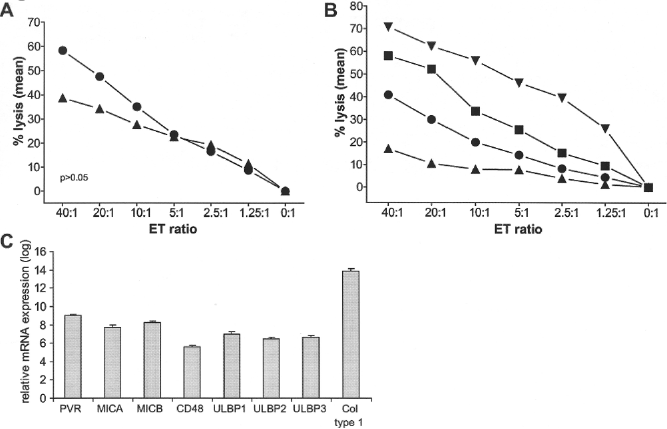
Cytotoxicity-mediated lysis of PHA blasts and MSCs. (A) Cytotoxicity-mediated lysis of phytohemagglutinin (PHA) blasts (▴) (n = 15) and MSC (?)(n = 5) by allogeneic effector cells of healthy kidney donors. MSCs are lysed by allogeneic PBMCs with comparable or even greater efficacy than PHA blasts. (B) Cytotoxicity-mediated lysis of MSCs by purified allogeneic CD8+ T cells (•) and natural killer (NK) cells (▪) of healthy kidney donors (n = 4) and lysis of PHA blasts by purified allogeneic CD8+ T cells (▾) and NK cells (▴) of healthy kidney donors (n = 3). Purity of CD8+ T cell and CD16+56+ NK cell fractions was ≥ 98%. (C) mRNA expression of seven activating NK cell receptor ligands by MSCs measured by microarray analyses. The last column shows the expression of collagen type I to demonstrate the relative expression of the ligands. The average expression levels of four MSC cultures with SD are shown. PVR, poliovirus receptor; MICA, MHC I polypeptide-related sequence A; MICB, MHC I polypeptide-related sequence B; ULBP1, UL16 binding protein 1; ULBP2, UL16 binding protein 2; ULBP3, UL16 binding protein 3; Col type I, collagen type 1. ET ratio: effector/target ratio.
IFN-γ Enhances CD8+ T-Cell-Mediated Lysis, but Reduces NK Cell-Mediated Lysis of Allogeneic MSCs
To investigate whether the susceptibility of MSCs for cytotoxicity-mediated lysis was affected by inflammatory conditions, MSCs were cultured for 7 days with IFN-γ. Culturing of MSCs with IFN-γ increased the mean fluorescence intensity (MFI) of HLA-ABC 4.7-fold (Fig. 3A). The susceptibility of allogeneic MSCs for NK cell-mediated lysis was decreased by IFN-γ treatment (Fig. 3B), while lysis of MSCs by CD8+ T cells was increased (Fig. 3C).

Effect of IFN-γ on cytotoxicity-mediated lysis of MSCs. (A) Flow cytometric detection of human leukocyte antigen (HLA) class I-ABC expression on MSCs after 7 days of culturing under control conditions or with interferon-γ (IFN-γ) (50 ng/ml). Representative example shown of n = 16. (B) Cytotoxicity-mediated lysis of allogeneic MSCs cultured under control conditions (□) or with IFN-γ (▪) by purified NK cells of healthy kidney donors (n = 4). (C) Cytotoxicity-mediated lysis of allogeneic MSCs cultured w (○) or with IFN-γ (•) by purified CD8+ T cells of healthy kidney donors (n = 4). ET ratio: effector/target ratio.
Autologous MSCs Are Susceptible for Cytotoxicity-Mediated Lysis by NK Cells
After demonstrating that MSCs were effectively lysed by allogeneic CD8+ T cells and NK cells, we questioned whether MSCs were susceptible for lysis by autologous cytotoxic cells. Cytotoxic effector cells, isolated from kidney donors, were not capable of lysing autologous PHA blasts, isolated from the same kidney donors, at any ET ratio, but lysed 27 ± 20% of autologous MSCs at an ET ratio of 40:1 (Fig. 4A). FACS purification of CD8+ T cells and NK cells showed that lysis of autologous MSCs was the result of NK cell cytotoxic activity (Fig. 4B).

Cytotoxicity-mediated lysis of autologous MSCs. (A) Cytotoxicity-mediated lysis of autologous MSC (▴; n = 12), allogeneic MSC (Δ; n = 5) and of autologous PHA blasts (•; n = 15) by effector cells of healthy kidney donors. (B) Cytotoxicity-mediated lysis of autologous MSCs by purified CD8+ T cells (▪) or NK cells (•) by effector cells of healthy kidney donors (n = 3). (C) Cytotoxicity-mediated lysis of autologous MSCs isolated and cultured in fetal bovine serum (FBS) (Δ) or in human serum (HuS) (•) by effector cells of healthy kidney donors (n = 4). (D) Flow cytometric detection of HLA class I-ABC expression on MSCs cultured under control conditions, MSCs cultured with siRNA for HLA class I-ABC for 3 days, or MSCs cultured with IFN-γ (50 ng/ml) for 7 days. Representative example shown of n = 3. (E) Cytotoxicity-mediated lysis of IFN-γ-treated MSCs by purified autologous NK cells (Δ) or CD8+ T cells (∇) of healthy kidney donors and of control MSCs by autologous purified NK cells (○) or CD8+ T cells (□) (n = 3). (F) Cytotoxicity-mediated lysis of control MSCs (○) and of MSCs treated with siRNA for HLA class I (∇) by purified autologous NK cells of healthy kidney donors (n = 2). (G) Cytotoxicity-mediated lysis of adherent autologous MSCs (○; n = 4) or IFN-γ-pretreated MSCs (□; n = 3) by NK cells of healthy kidney donors. ET ratio: effector/target ratio.
To exclude the possibility that the observed lysis of autologous MSCs was the result of recognition of bovine proteins by NK cells due to culture of MSCs in FBS, MSCs were isolated from adipose tissue and cultures established and maintained in either medium containing 15% FBS or 15% human serum (HuS). Autologous effector cells lysed MSCs cultured in FBS as efficiently as MSCs cultured in HuS (52 ± 25% vs. 49 ± 20% lysis at an ET ratio of 40:1) (Fig. 4C). These results indicate that culturing of MSCs in FBS is not the cause of their susceptibility to cytotoxicity-mediated lysis.
To examine the role of HLA class I on NK cell lysis of autologous MSCs, HLA expression was increased by treatment of MSCs with IFN-γ for 7 days or decreased by culturing MSCs with siRNA for HLA class I for 3 days. Culturing MSCs with siRNA for HLA class I strongly reduced HLA class I-ABC expression on MSCs and only 13% of MSCs showed positive expression, while the MFI reduced 2.8-fold (Fig. 4D).
In agreement with the findings with allogeneic MSCs, the lysis of autologous MSCs by NK cells was decreased after IFN-γ treatment of MSCs to 50 ± 2% (ET ratio of 40:1) (Fig. 4E). MSCs cultured with siRNA for HLA class I showed significantly increased susceptibility of MSCs for autologous NK cell-mediated lysis (Fig. 4F).
The fate of MSCs after infusion is currently unclear. One possibility is that MSCs home to sites of injury where they adhere to local stroma or epithelial cell layers. To determine whether adherent MSCs would remain susceptible for lysis by autologous NK cells, MSCs were seeded in six-well plates and purified NK cells added. Autologous NK cells showed potent capacity to lyse adherent MSCs (92% lysis at an ET ratio) (Fig. 4G). IFN-γ treatment strongly reduced the susceptibility of adherent MSCs for lysis by autologous NK cells to 52% lysis.
Kidney Donor-Derived MSCs Are Lysed by Recipient Cytotoxic Immune Cells
The immunogenicity and survival of MSCs is an important issue for their application in organ transplantation. As we found that MSCs are susceptible for CD8+ T-cell- and NK cell-mediated lysis, we wanted to examine this in a kidney transplant setting using donor MSCs and kidney recipient immune cells isolated at various time points after kidney transplantation. Cytotoxic effector cells of recipients isolated before transplantation showed poor capacity to lyse donor MSCs (5 ± 5% lysis at an ET ratio of 40:1). Recipient cytotoxic cells isolated 3, 6, and 12 months after transplantation showed increasing capacity to lyse MSCs in time after transplantation (Fig. 5A). Lysis of donor MSCs 12 months after transplantation was for 33% (9–57%) dependent on CD8+ T-cell-mediated lysis while the CD8+ T-cell-depleted cell fraction was responsible for 55% (46–68%) of MSC lysis (Fig. 5B). This indicates that the recovered lysis of donor MSCs after transplantation is for a major part dependent on NK cell cytotoxicity.

Cytotoxicity-mediated lysis of MSC in a kidney transplantation setting. (A) Cytotoxicity-mediated lysis of kidney donor-derived MSCs by recipient immune cells collected before transplantation (•; n = 5), and 3 months (▪; n = 5), 6 months (▴; n = 8), and 12 months (⋄; n = 9) after transplantation. (B) Cytotoxicity-mediated lysis of kidney donor-derived MSCs by purified CD8+ T cells (○) and CD8+ T-cell-depleted (•) recipient cells isolated 12 months after transplantation (n = 6). (C) Cytotoxicity-mediated lysis of kidney donor-derived MSCs by purified CD8+ T cells (▴) and CD8+ T-cell-depleted (•) recipient cells isolated 16 months (n = 1), 18 months (n = 1), and 24 months (n = 1) after transplantation (n = 6). ET ratio: effector/target ratio. Tx: transplantation.
Finally, we examined whether there was evidence for the development of hypo- or hyperresponsiveness (sensitization) of kidney transplant recipients against donor MSC after transplantation by looking at the capacity of recipient CD8+ T cells to lyse donor MSCs. As we found that non-CD8+ T-cell-mediated lysis of donor MSCs was recovered 12 months after transplantation, we examined whether the balance between CD8+ T-cell-mediated and non-CD8+ T-cell-mediated lysis of donor MSC was shifted more than 12 months after transplantation compared to nontransplant healthy kidney donors. CD8+ T-cell-mediated lysis of donor MSCs remained well below the lysis of donor MSCs by CD8+ T-cell-depleted cells in three recipients that were 16, 18, and 24 months after transplantation (Fig. 5C). These results suggest that there is no evidence for hypo- or hyperresponsiveness against donor MSCs long after kidney transplantation.
Discussion
In an increasing number of clinical trials the use of MSCs for immune therapy is studied. Trials investigating the use of MSCs in organ transplantation have recently initiated (25). Although the use of autologous MSCs may be preferable to avoid recognition of MSCs by the immune system, the use of “off-the-shelf” allogeneic MSCs is more practical. The immunogenicity and survival of autologous and allogeneic MSCs are, however, unknown and are important for the efficacy of clinical trials. In particular, they may have implications in an organ transplant setting where cross-reactivity between MSCs and the organ transplant may induce allograft rejection. In the present study we investigated the susceptibility of autologous and allogeneic MSCs cultured under control and inflammatory conditions for lysis by cytotoxic CD8+ T cells and NK cells. Subsequently, the susceptibility of donor-derived MSCs for lysis by kidney transplant recipient cytotoxic cells isolated before and after kidney transplantation was examined.
Several studies reported that MSCs can inhibit the formation of cytotoxic cells (2,38,40,48). Data on the susceptibility of MSCs for lysis by cytotoxic cells are, however, contradictory. It has been suggested that MSCs are immune privileged and that they are not susceptible for lysis by cytotoxic immune cells (38,39). Others reported that MSCs are susceptible for cytotoxicity-mediated lysis, especially by NK cells (44,49). In our hands, allogeneic adipose tissue-derived MSCs were highly susceptible for cytotoxicity-mediated lysis, at a rate comparable to allogeneic PHA blasts. Whereas allogeneic PHA blasts were lysed by CD8+ T cells only, allogeneic MSCs were lysed by CD8+ T cells and NK cells. This difference may lie in expression of activating NK cell receptor ligands on MSCs and PHA blasts of which several are found on MSCs (35,44). Autologous MSC were not lysed by CD8+ T cells, but were susceptible for lysis by NK cells. Expression levels of HLA class I molecules modulated the susceptibility of MSCs for NK cell lysis, as downregulation of HLA class I increased lysis of MSCs by NK cells, whereas upregulation of HLA class I by IFN-γ was associated with protection of MSCs. We ruled out the possibility that lysis of MSCs was the result of recognition of bovine antigens due to culture of MSCs in FBS by establishing and maintaining MSC cultures in medium with human serum. Culturing in human serum had no effect on lysis of MSCs.
The fate of MSCs after intravenous infusion is not yet clear. One possibility is that MSCs home to specific tissue sites or to sites of injury, and adhere to the stroma or epithelial cell layers at these sites. To find out whether homing would protect MSCs from lysis by NK cells, we added autologous NK cells to adherent MSC monolayers. Under these conditions, MSCs remained susceptible for lysis by NK cells, suggesting that homed MSCs can still be cleared by NK cells. More research is needed to investigate the distribution and integration of the engrafted cells into host tissues. Labeling MSCs with micron-sized iron oxide particles (MPIOs) is a promising tool for in vivo cell tracking and evaluation of cell-based therapies, as reported previously (33).
Our finding that both autologous and allogeneic MSCs are susceptible for lysis by cytotoxic cells may explain why MSCs seem to disappear shortly after infusion (14,17,46). Nevertheless, lysis of autologous MSCs remains remarkable. We previously demonstrated that MSCs of donor origin can be isolated from heart biopsies up to 6 years after heart transplantation in human (23). These heart transplant patients were obviously under immunosuppression, suggesting that the lysis of MSCs may be preventable with immunosuppressive drugs.
It has been reported previously that exposure of MSCs to inflammatory conditions affects immunophenotype and enhances the immunosuppressive function of MSCs (13,22,27,31). This is particularly relevant when MSCs are used as clinical immune therapy. Proinflammatory cytokines, such as IFN-γ, increase the expression of HLA class I and II subtypes on MSCs. Pretreatment of autologous and allogeneic MSCs with IFN-γ made them less susceptible for NK-mediated lysis, whereas it increased CD8+ T-cell-mediated lysis of allogeneic MSCs as a result of elevated HLA class I expression. In line with the results obtained from healthy individuals, we found that kidney transplant recipient-derived cytotoxic CD8+ T cells and NK cells were capable of lysing donor-derived MSCs. Recipient cytotoxic cells isolated with increasing time after transplantation were increasingly efficient in lysing MSCs compared to recipient cells isolated before transplantation. The lack of lysis of MSCs by recipient immune cells isolated prior to transplantation could reflect the poor renal function and general health of the patients, while the increasingly efficient lysis of MSCs after transplantation may be the result of recovery of the renal function or improvement of the recipient's immune system and tapering of immunosuppressive drugs after transplantation. We also found that CD8+ T-cell-mediated lysis of donor MSC more than a year after transplantation remained well below the lysing efficiency of NK cells, similar to that found in healthy kidney donors, indicating that hyperresponsiveness against donor MSCs may not occur.
The susceptibility of MSCs for lysis by cells of the innate and adaptive immune system may be of important relevance for the efficacy, but also for the safety of MSC therapy. If MSCs are removed rapidly after infusion, this may impair the time span of the effects of the MSCs. This could explain why MSCs appear to have a transient effect on the inflammatory milieu in graft-versus-host disease (10). On the other hand, evidence indicates that MSCs can have long-lasting effects by passing on some of their effect to other cell types, such as regulatory T cells (21), suggesting that even though MSCs may be lysed shortly after infusion, this does not prevent a long-term effect of MSCs to occur. While the removal of MSCs after infusion may raise debate on the long-term efficacy of MSC therapy, it clearly diminishes worries about the safety of MSC therapy as it takes some concerns about tumorgenicity away. Taken together, our results do not impair the validity of MSC therapies, but they rather help in understanding the potential mechanisms involved and aid in optimizing this therapy.
In conclusion, our results suggest that controlling MSC lysis by cytotoxic immune cells may be important for successful MSC therapy in organ transplantation. Recent data show that blocking of CD16 on NK cells inhibits lysis of MSCs (26). Other approaches to improve MSC survival may include in vitro modulation of MSCs to reduce their immunogenicity. Preclinical models should be used to find out whether such approaches affect the balance between the efficacy and safety of MSC therapy.
Footnotes
Acknowledgments
The authors thank the Department of Surgery of the Erasmus University Medical Center for collecting the adipose tissue of the living kidney donors. M.J.C. participated in research design, performance of the research, data analysis and writing of the paper. S.S.K. participated in the performance of the research and data analysis. R.de K. participated in the performance of the research. N.M.van B. participated in research design. J.N.M.I. participated in research design. C.C.B. participated in research design. W.W. participated in research design and writing of the paper. M.J.H. participated in research design and writing of the paper. The authors declare no conflicts of interest.
