Abstract
Transplantation of adipose-derived stem cells (ADSCs) is an emerging therapeutic option for addressing intractable diseases such as critical limb ischemia (CLI). Evidence suggests that therapeutic effects of ADSCs are primarily mediated through paracrine mechanisms rather than transdifferentiation. These secreted factors can be captured in conditioned medium (CM) and concentrated to prepare a therapeutic factor concentrate (TFC) composed of a cocktail of beneficial growth factors and cytokines that individually and in combination demonstrate disease-modifying effects. The ability of a TFC to promote reperfusion in a rabbit model of CLI was evaluated. A total of 27 adult female rabbits underwent surgery to induce ischemia in the left hindlimb. An additional five rabbits served as sham controls. One week after surgery, the ischemic limbs received intramuscular injections of either (1) placebo (control medium), (2) a low dose of TFC, or (3) a high dose of TFC. Limb perfusion was serially assessed with a Doppler probe. Blood samples were analyzed for growth factors and cytokines. Tissue was harvested postmortem on day 35 and assessed for capillary density by immunohistochemistry. At 1 month after treatment, tissue perfusion in ischemic limbs treated with a high dose of TFC was almost double (p < 0.05) that of the placebo group [58.8 ± 23 relative perfusion units (RPU) vs. 30.7 ± 13.6 RPU; mean ± SD]. This effect was correlated with greater capillary density in the affected tissues and with transiently higher serum levels of the angiogenic and prosurvival factors vascular endothelial growth factor (VEGF) and hepatocyte growth factor (HGF). The conclusions from this study are that a single bolus administration of TFC demonstrated robust effects for promoting tissue reperfusion in a rabbit model of CLI and that a possible mechanism of revascularization was promotion of angiogenesis by TFC. Results of this study demonstrate that TFC represents a potent therapeutic cocktail for patients with CLI, many of whom are at risk for amputation of the affected limb.
Keywords
Introduction
The nonadipocyte stromal vascular fraction (SVF) of human adipose tissues contains a population of mesenchymal stem cells (MSCs) intimately associated with blood vessels 1 . These adipose-derived stem cells (ADSCs) possess many phenotypic and functional similarities to bone marrow-derived MSCs (BM-MSCs). Unlike BM-MSCs, though, multipotent ADSCs can be harvested at relatively high numbers (0.5 × 106/g fat with a typical harvest of 500 g of tissue) using minimally invasive techniques of lipoaspiration or lipectomy of subcutaneous adipose tissue 2 . ADSCs rapidly proliferate in culture, allowing fast expansion to obtain cell quantities sufficient to treat large volume lesions. Transplantation of ADSCs is an emerging therapeutic option for addressing many intractable diseases, including cardiovascular diseases such as peripheral artery disease (PAD)3–8.
Evidence suggests that the therapeutic effects of MSCs, including ADSCs, are primarily mediated through paracrine mechanisms rather than cellular transdifferentiation9–12. The three predominant paracrine actions of stem cell-secreted factors are (1) trophic support of survival and repair of cells in diseased or injured tissues, (2) modulation of the immune system, and (3) recruitment of endogenous stem and progenitor cells 11 . Trophic factors produced by ADSCs include the potent paracrine prosurvival and proangiogenic proteins hepatocyte growth factor (HGF) and vascular endothelial growth factor (VEGF) that reduce cell death and promote reperfusion of ischemic tissues. The immunomodulatory factors secreted by ADSCs, such as prostaglandin E2 (PGE2), tumor necrosis factor-inducible gene 6 protein (TSG-6), and interleukin-10 (IL-10), support reduction of inflammation and suppression of T-cell pathology. This, in turn, suppresses atherothrombotic circulatory defects in the lower extremities in diabetic patients with diabetic foot ulcers and the most severe form of PAD—critical limb ischemia (CLI) 13 . Chemotactic factors, such as stromal cell-derived factor-1α (SDF-1α) 14 , attract circulating stem and progenitor cells to the injury site where they support regeneration of damaged tissues 15 . Moreover, these factors have the potential to work in concert to affect physiological improvements in diseased and injured tissues. The contribution of secreted factors to ADSC potency was established by knocking down HGF expression using a stably integrated small interfering RNA construct, which severely attenuated ADSC-mediated promotion of ischemic limb revascularization in a mouse model of CLI 16 .
Paracrine-mediated effects explain the frequently reported functional benefits of MSC treatment in the absence of stable cell engraftment on the scale required to replace significant portions of injured tissues. Given that each cell secretes a number of different factors that function to modulate a spectrum of molecular pathways required for preserving tissues and correcting disease, paracrine effects have the benefit over direct tissue replacement of amplifying the effects of cell treatment. Each cell produces an abundance of these factors, and even brief exposure to target tissues preceding clearance is sufficient to produce therapeutic effects. Thus, a paracrine mechanism of action overcomes limitations in numbers of recoverable and deliverable therapeutic cells through amplification of the effect produced by each cell; conversely, direct tissue replacement by multipotent stem cells requires a sufficient number of cells to stably engraft and replace each cell type lost due to the insult.
Recognition that the functional component of adult stem cells may be attributable to secreted factors led our group and others to explore the potential therapeutic benefit of delivering these factors in the absence of cells10,17–20. ADSC-derived paracrine factors can be captured by briefly (up to 72 h) incubating freshly isolated or culture-expanded ADSCs in minimal medium lacking added protein supplements 20 . Incubation conditions are not permissive for cell growth; however, ADSCs remain viable and “condition the medium” by secreting paracrine factors. The resulting therapeutic factor concentrate (TFC), composed of a cocktail of beneficial growth factors and cytokines, is filtered to remove all cells and then concentrated before storage by freezing.
The potential for modifying disease status through treatment with the array of paracrine factors present in TFC may offer advantages over cellular therapies. First, there is reduced concern regarding maintaining cell viability during preparation of cryopreserved cells for injection. Additionally, because of their size, cells possess a potential risk of embolization. Conversely, stem cells, such as ADSCs, have the potential to home to target tissues and produce a greater local concentration of paracrine factors, although targeted effects will be limited in duration due to rapid clearance of most cells within the first 24 h following administration21,22.
The purpose of the present study was to test the efficacy of a single intramuscular administration of human TFC (hTFC) in the experimental setting of the rabbit subacute CLI model. A positive result could open up the possibility of subsequent clinical studies designed to evaluate TFC as a novel therapy for CLI.
Materials and Methods
Human and Animal Research Approval
Collection of adipose tissues and blood from human volunteers was approved by the Ethics Committee of FN Ostrava (Approval No. 654/2014). All procedures involving animals were approved by the Local Bioethics Commission of the Polish Academy of Science, Kraków, Poland (Approval No. 1103/2014).
ADSC Isolation and Culture
ADSCs were isolated from adipose tissue by Cytori Celution CRS 800 (Cytori Therapeutics Inc., San Diego, CA, USA). Adipose tissue was obtained with written informed consent from a single male patient [40 years of age and body mass index (BMI) of 54] undergoing elective lipoplasty at the University Hospital Ostrava. Erythrocytes in the obtained cell suspension were lysed with red blood cell (RBC) lysis buffer (eBioscience, San Diego, CA, USA). The number and viability of cells were determined by 0.4% trypan blue solution staining (Sigma-Aldrich, St. Louis, MO, USA) and Burker's chamber counting method. Cells were seeded in cell culture flasks (Nunc, Rochester, NY, USA) at density of 1 × 104 cells/cm2 in endothelial cell growth medium (EGM)-2MV (Lonza, Valais, Switzerland) supplemented with 5% (v/v) fetal bovine serum (FBS; Hyclone, Logan, UT, USA). Cell cultures were maintained at 37°C in a humidified atmosphere containing 5% CO2. This initial passage of the primary cell culture was designated passage 0 (P0). After 24 h, the medium was changed to remove nonadherent cells. When 70% confluence was reached, cells were detached using CTS TrypLE Select Enzyme (Gibco, Paisley, Scotland, UK) and replated at a density of 5 × 103 cells/cm2 (P1). Cells were expanded by passaging repeatedly until P5. Viable nucleated cell counts were obtained using a Luna-STEM™ automated cell counter (Logos Biosystems, Annandale, VA, USA) according to the manufacturer's instructions.
ADSC Conditioned Medium (ADSC-CM) and TFC
ADSCs (P6) were seeded into 225-cm2 culture flasks (Corning, Inc., Corning, NY, USA) at a density of 1 × 104 cells/cm2 in cultivation media. When reaching 80% confluence, cells were rinsed three times with Dulbecco's phosphate-buffered saline (DPBS; Gibco), and Dulbecco's modified Eagle's medium (DMEM; Lonza) was applied to the cells for 48-h conditioning at 37°C in a humidified atmosphere containing 10% CO2. At the end of incubation, the conditioned medium was harvested, centrifuged at 1,000 × g to remove debris and detached cells, concentrated 50-fold using Vivacell 70 (10 kDa; Sartorius, Goettingen, Germany), and stored at −80°C. All studies were performed with concentrated TFC.
Flow Cytometry
The cell suspension product from the Celution isolation device was analyzed by flow cytometry for cell surface marker expression. Cells (2 × 105) were added to tubes containing complete medium (100 μl) on ice. Fluorescently conjugated monoclonal antibodies to cell surface markers or isotype controls (1 μl) were added to the tubes and incubated for 20–30 min on ice. Cells were subsequently washed with PBS, fixed in paraformaldehyde (PFA; Tousimis Research Corporation, Rockville, MD, USA), and analyzed using a Cytomics FC500 (Beckman Coulter, Brea, CA, USA). The following antibody conjugates (Beckman Coulter, Brea, CA, USA) were used: CD34-phycoerythrin (PE) (catalog #A0776); CD34-fluroscein isothiocyanate (FITC) (catalog #IM1870); CD34-PE/Cy5 (PC5) (catalog #A07777); CD10-FITC (catalog #IM0471U); CD29-PE (catalog #6604159); CD31-PE (catalog #IM2409); CD45-FITC (catalog #A07782); CD49-FITC (catalog #IM1425); CD73-PE (catalog #344003); CD90-FITC (catalog #IM1893U); CD105-PE (catalog #A07414); CD13-PE/CD14-FITC (catalog #A07722); and IgG1 isotype control-PE/FITC (catalog #A07794).
Characterization of TFC
The concentrated TFC from ADSCs was analyzed using Quantibody multiplex enzyme-linked immunosorbent assays (ELISAs) (RayBiotech Inc., Norcross, GA, USA) for the presence of multiple growth factors, including acidic fibroblast growth factor (aFGF), insulin-like growth factor 1 (IGF-1), platelet-derived growth factor AA (PDGF-AA), transforming growth factor-β1 (TGF-β1), VEGF, interferon-γ (IFN-γ), IL-1β, IL-2, IL-4, IL-6, IL-7, IL-8, IL-10, IL-12 p70, IL-13, brain-derived neurotrophic factor (BDNF), matrix metalloproteinase-9 (MMP-9), and tumor necrosis factor-α (TNF-α). For comparison, human serum obtained from a healthy volunteer unrelated to the adipose tissue donor was also analyzed. Fluorescence signals from the membranes (arrays) were collected using a SureScan Microarray Scanner (Agilent Technologies, Santa Clara, CA, USA) and evaluated using software from RayBiotech Inc.
Animals
Animal studies were approved by the Second Local Ethical Committee on Animal Testing at the Institute of Pharmacology of the Polish Academy of Science, Cracow, Poland (Resolution No. 1103/2014).
A total of 32 female New Zealand white rabbits (3.5 months, 2.7 ± 0.2 kg) were purchased from Zakład Doświadczalny Instytutu Zootechniki PIB (Chorzelów, Poland). The environmental controls on temperature and humidity were set according to the Center for Cardiovascular Research and Development (Kostkowice, Poland) standards for animal care. Animals were acclimated for 28 days prior to beginning the study. A sufficient amount of feed was offered for maintenance, and water was supplied ad libitum. Each rabbit had a unique ID number. Body weights were recorded upon receipt and weekly thereafter until the end of the study. The most recent body weight was used for calculation of all perioperative medications. Body temperature was measured using a rectal probe thermometer.
Hindlimb Ischemia Surgery
At day 0, rabbits were anesthetized with an intramuscular injection of 100 mg/kg ketamine and 20 mg/kg xylazine (Vetoquinol, Gorzów Wielkopolski, Poland) and subcutaneous injection of 0.5 mg/kg atropine (Warszawie Zakłady Farmaceutyczne Polfa, Warszawa, Poland). Surgical and ligation/excision were conducted to establish ischemia in the left hindlimb. The femoral artery and its branches as well as femoral vein were ligated, beginning from the inguinal ligament to the bifurcation of saphenous and popliteal arteries and excised. The incision was closed with 3-0 silk sutures. A sham group underwent the same surgery without ligation or excision of the artery and vein.
Study Groups and Treatments
Rabbits with induced limb ischemia were randomly assigned to one of three treatment groups (nine/group): (1) placebo control (unconditioned DMEM), (2) low-dose TFC, and (3) high-dose TFC. Treatments were provided to animals on day 7 after surgery by intramuscular injection using a 1-ml luer-lock syringe with a 0.45 × 13-mm needle. The doses of placebo or TFC were divided into five injection sites: two into each bundle of the m. gastrocnemius (four injections) and one into the m. tibialis anterior. The volume of injection per site was 0.4 ml for placebo and low-dose TFC and 0.8 ml for high-dose TFC.
Laser Doppler Perfusion
Perfusion status of the ischemic (left) and intact (right) limbs was assessed with a Laser Doppler Perfilux 5000 device (Perimed AB, Järfälla, Sweden) on days 0 (before and after the surgery), 14, 28, and 35. Perfusion in ischemic hindlimbs immediately following surgery was approximately 20% of presurgery levels.
Rabbit Serum Analysis for Proteins
Venous blood (4–5 ml) was collected from the auricular vein; serum was obtained and frozen for later use. Serum samples were analyzed for multiple rabbit growth factors by ELISA: HGF, granulocyte macrophage colony-stimulating factor (GM-CSF), VEGF, basic fibroblast growth factor (bFGF), glial cell-derived neurotrophic factor (GDNF), SDF-1α, and TGF-β (all kits were purchased from BlueGene Biotech, Shanghai, China).
Necropsy and Histological Examination
After the final Doppler assessment on day 35, the rabbits, while under general anesthesia, were euthanized by intravenous injection of pentobarbital (Biowet Puławy, Puławy, Poland) euthanasia solution. After death, the tibialis anterior and gastrocnemius muscles were harvested for evaluation. The tibialis anterior muscle was immediately frozen by placing on dry ice. Gastrocnemius muscles were marked for orientation and fixed in 10% formaldehyde (Bio-Optica Milano, Milan, Italy). Both muscles were photographed, and their size was measured. The fixed muscles were grossly (0.3–0.5 cm thick) dissected from the broadest part of the proximal muscle region. Sections were divided into four pieces and enclosed into bioptic sectors in the direction from medial to lateral side.
Tissue segments were embedded in paraffin, and paraffin blocks were sectioned at 2- to 4-μm thickness and stained with hematoxylin and eosin (H&E; Merck, Kenilworth, NJ, USA). After examination of H&E-stained sections to confirm tissue integrity, additional sections were processed for immunohistochemical analyses. Antibodies used were CD31 (1:20; DakoCytomation, Glostrup, Denmark) and α-smooth muscle actin (αSMA; 1:100; DakoCytomation). The numbers of positively staining vessels in a 10-mm2 area of each block were counted. Capillary density measurements were normalized by subtracting the values from the nonischemic limb from the ischemic limb. Thus, the values range from both positive to negative.
Data Representation and Statistical Analyses
Data were analyzed with open source R (R-project, The R Foundation for Statistical Computing, Vienna, Austria) and Matlab (MathWorks, Natic, MA, USA) institutional license software. Reported summary statistics are arithmetic mean with 1 standard deviation (SD), denoted as mean ± SD. Most of the measurements are shown as box and whisker plots (box plot) to account for strong non-Gaussianity of much of the data. On each box, the central mark is the median; the top and bottom edges of the box are the 75th and 25th percentiles, respectively; and the whiskers extend to the most extreme data points not considering outliers, which are marked with the black dots. To test the key statistical hypotheses about the treatment groups for all the endpoints, Kruskal–Wallis test, Mann–Whitney U test, and Welch's t-test were calculated, reported, and selected according to their suitability for the particular statistical properties (probability density moments) of the distributions of the measurements being compared. In addition to classical statistics, an alternative approach of gnostics23,24 was used here to analyze and validate our findings.
Results
Preparation and Characterization of TFC
Approximately 3.62 × 106 total nucleated cells/ml were obtained from 70 ml of lipoaspirate (Table 1). Freshly harvested cells had a typical morphology and surface marker profiles of SVF cells (Fig. 1 and Table 2) and ADSCs. These cells were expanded to P5 and were similar to ones that were previously reported 2 (Table 2 and Fig. 2). Levels of selected angiogenic and inflammatory cytokines and growth factors in 50-fold concentrated TFC were compared to those of human serum (Table 3). While the concentrations of the majority of proteins were similar between TFC and human serum, the levels of VEGF, IL-6, and IL-8 were >10-fold higher in TFC. Conversely, levels of MMP-9, IL-7, and PDGF-AA were much lower in TFC compared to serum.
Characteristics of Stromal Vascular Fraction (SVF) and ADSC Suspensions Obtained With the Luna-STEM™ Automated Cell Counter


Morphology of stromal vascular fraction (SVF) cells. Image from Luna-STEM™ automated cell counter showing morphology of cells obtained from SVF. The green marking corresponds to live cells, and the red coloring highlights dead cells. Counted cells are encircled with red and green lines for dead and live cells, respectively. Scale bar: 50 μm.
Characterization of Cell Composition of Freshly Obtained SVF and Cultured ADSCs at Passage 3 (P3) and P5 by Flow Cytometric Technique


Outgrowth of adipose-derived stem cells (ADSCs) from SVF. Morphology of cultured ADSCs. Photomicroscopic image of cultured ADSCs at passage 3. Scale bar: 50 μm.
Analysis of Selected Proteins Levels in 50-Fold Concentrated Therapeutic Factor Concentrate (TFC) and in Human Serum From a Healthy Donor

All values are expressed as pg/ml. <: below detection limit.
Characterization of the Hindlimb Ischemia Model
A total of 32 animals were originally enrolled in the study. A total of eight rabbits died before the end of the study; five were found dead or had to be euthanized as a result of complications due to the surgery before administration of the therapeutic agent, and two died after administration of TFC or placebo. Early deaths were in sham (one rabbit), placebo (two rabbits before administration and one after), and low-dose TFC (three rabbits before administration) and high-dose TFC (one rabbit after administration) groups. In an attempt to rebalance the group numbers, one rabbit from the control group and one rabbit from the high-dose TFC group were reassigned to the middle dose group before administration. The number of rabbits (in parentheses) in each group surviving to the end of the study were as follows: sham group (four), placebo (five), low-dose TFC (eight), and high-dose TFC (seven). There were no differences between groups in body temperatures or weights (data not shown).
TFC Augments Reperfusion in Part by Stimulating Angiogenesis
Blood perfusion in the left ischemic and right intact limbs was determined immediately pre- and postsurgery and at 2, 4, and 5 weeks after surgery. Sham animals displayed very little change in blood perfusion during this period (Fig. 3). Trends toward greater perfusion in the ischemic limbs of low- or high-dose TFC group animals compared to the placebo control group were evident as early as 1 week following treatment (day 14 of the study). The effect was significant in the group receiving the highest dose of TFC [58.5 ± 24.3 relative perfusion units (RPU)] versus the placebo control group (30.5 ± 15.1 RPU) at day 35 following surgery, which was 1 month after treatment. The level of perfusion at day 35 in the treated limb of the high-dose group was not different compared to that in the sham group (54.3 ± 15.4 RPU) (Fig. 4).

(A) Validation of the rabbit critical limb ischemia model used in this study. (A) Surgical procedure of induction of acute hindlimb ischemia (HLI) in rabbits. The superficial femoral artery and vein were ligated and excised. (A) Common femoral artery, (B) dissected superficial femoral artery, (C) femoral nerve, and (D) branching popliteal artery. (B) Box and whisker plot comparing serial laser Doppler perfusion of the sham (white) and placebo control (gray) groups. The median of relative perfusion units (RPU) for each day of measurement is represented by a horizontal line in the box; the lower and upper boundaries of the box are the 25th and 75th percentiles, respectively. The whiskers extend to the most extreme data points not considering outliers, which are plotted as the filled circles.
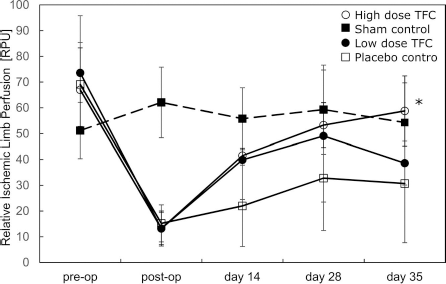
TFC treatment enhances ischemic limb perfusion. A laser Doppler probe was used to serially measure perfusion (expressed as relative perfusion units) at the medial aspect of the ischemic limb gastrocnemius muscle. Each symbol represents the gnostic median value for a group on the indicated days. Vertical lines represent standard deviations. Values for “preop” and “postop” were obtained on the first day of the experiment immediately before and after, respectively, the surgery to induce unilateral hindlimb ischemia. Filled box, sham control; open box, placebo control; filled circle, low-dose TFC; open circle, high-dose TFC. *p < 0.05.
Enhanced reperfusion in the high-dose TFC group correlated with greater capillary density in the treated muscles. Histological analysis of CD31+ endothelial cell density in the gastrocnemius muscle of all animals in the study was performed (Fig. 5A). The ischemic (index) limbs of animals treated with high-dose TFC (23.91 ± 12.82) possessed significantly higher capillary density (p < 0.01) compared to the nonischemic control limb (18.41 ± 11.36) (Fig. 5B). Capillary density in the index limbs of the high-dose TFC group (8.41 ± 12.82) was also greater (p < 0.05) than in the placebo group (3.32 ± 12.19), respectively (Fig. 5C).
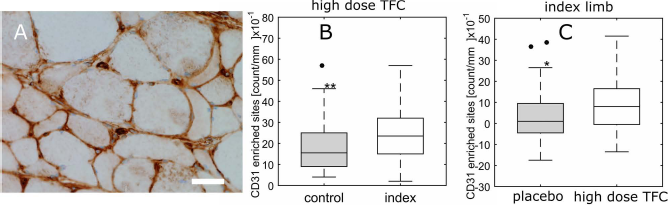
TFC induces increased capillary density in the ischemic leg. Capillary densities in the gastrocnemius muscles at 5 weeks following surgically induced ischemia. (A) Representative image of a cross section of the gastrocnemius muscle in the ischemic hindlimb that had been immunohistochemically stained for CD31 antigen (marker of endothelial cells). Scale bar: 20 μm. (B) A box and whisker plot of capillary density (counts of CD31+ cells) in the high-dose TFC group for untreated, nonischemic limb (filled box) versus ischemic limb treated with high-dose TFC (open box). (C) A box and whisker plot of capillary density (CD31+ cell count per 10 mm2) in the ischemic limb of the placebo group (filled box) versus ischemic limb of the high-dose TFC group (unfilled box). Median of control limbs was subtracted from ischemic limb values to equalize baselines between the two groups. Description of box plots provided in Figure 3. *p < 0.05; **p < 0.01.
The increased capillary density leading to enhanced muscle perfusion in the high-dose-treated group correlated with increased levels of angiogenic factors in the circulation. Analysis of blood samples collected at different time points revealed that rabbits treated with either high- and low-dose TFC had transiently higher blood levels of rabbit VEGF (p < 0.05) compared to levels in blood of the placebo group at 1 week following treatment (Fig. 6). Rabbit serum levels of VEGF at 1 week following treatment (day 14 of the study) were 60.9 ± 33.4 pg/ml (placebo control), 141 ± 70.7 pg/ml (high-dose TFC), and 142.5 ± 96 pg/ml (low-dose TFC). Circulating levels of HGF and bFGF were also higher following treatment with TFC compared to the placebo control group, although the effect was not significant at all time points.

TFC treatment induces transient increases in circulating growth factors. Concentrations of selected factors in sera of placebo and low- and high-dose TFC groups were measured by enzyme-linked immunosorbent assay (ELISA). Data are represented by box plots comparing serum levels of (A, D) VEGF (pg/ml), (B, E) HGF (ng/ml), and (C, F) bFGF (pg/ml) at 1 and 4 weeks following treatment with placebo (filled boxes) or TFC at the high (A–C) or low (D–F) doses (open boxes). Description of box plots provided in Figure 3. *p < 0.05.
Discussion
This study demonstrated that factors secreted by ADSCs in culture and delivered as a single intramuscular bolus to the affected limb promoted revascularization of ischemic skeletal tissue in a rabbit model of subacute PAD. The rationale for using TFC is based on an increasing recognition that the salutary effects of adult stem/progenitor cells are a function of paracrine factors as opposed to direct tissue regeneration through transdifferentiation 9 . These factors have the potential to stimulate endogenous repair, modulate inflammation, and promote survival of at-risk tissues 11 . TFC contains factors involved in each of these processes. For instance, the prosurvival and potent angiogenic factor VEGF was present in TFC at levels that were 46-fold higher than detected in human serum. Importantly, for skeletal muscle protection and recovery from ischemia, TFC contained high levels of the anti-inflammatory myocyte cytokine IL-6 25 . Furthermore, the exposure of ischemic tissues to the factors contained in TFC produced a systemic increase in levels of the prosurvival angiogenic factor VEGF. The angiogenic and prosurvival factors HGF and bFGF were also elevated following treatment. Thus, a single administration was sufficient to induce recovery over the duration of the study.
It has been postulated that therapeutic cells differentially home to the injury site and respond to the host environmental cues; thus, the cells produce a concentrated combination of factors that are optimized to address a particular disease state 11 . However, there is scant in vivo experimental evidence to support this hypothesis, and at least one study demonstrated that paracrine factors acting at a distal from the lesion could be responsible for the majority of the beneficial effects following treatment with BM-MSCs 26 . This study found that the vast majority of intravenously administered BM-MSCs were trapped in the lung, where they persisted with a half-life of 24 h and released factors that promoted protection from myocardial infarction. Even with direct injection to the injury site, the frequency of cellular retention is quite low and the retention time of all but the rare population of cells that engraft long term is extremely short (with half-lives of a few hours)21,22. Thus, the period of exposure to the environment would appear to be too brief to allow for effective modulation of the paracrine factor profile secreted by those few cells that homed to the injury site, although the exact kinetics of clearance would depend on the route of delivery.
There are several advantages of an ex vivo-derived cocktail of the factors produced by therapeutic cells over administering cells themselves. As opposed to treatment with cells, direct injection of TFC provides a bolus of “ready-made” paracrine factors that are immediately available to alter the pathophysiological process. We and others have found that TFC produced using standard cell culture conditions possesses robust activity in ischemic models17–20. By varying the physiochemical environment used in culture, it is possible to modify the factors expressed by the cells, which allows for producing disease-specific combinations of factors. Furthermore, the composition of TFC can be carefully characterized and tested in order to ascertain the levels of each factor contained within. TFC can also be tested for safety and efficacy before administration. Conversely, the response of a cell to the complex host environment after local or systemic delivery is difficult to predict with respect to composition of secreted factors, as well as those released through cell lysis. Finally, it is becoming apparent that, although they possess some degree of immunoprivilege, allogeneic MSCs do evoke an immune response that may limit repeated administration27,28. Conversely, the proteins in CM are less likely to stimulate an immune response, which would allow repeated administration in order to enhance the response.
On the basis of the results of this preclinical study, we believe that TFC could be potentially efficacious in CLI patients. We envision an off-the-shelf product that would be immediately available for administration with little preparation. To this end, we are planning a clinical trial with patients who are not candidates for conventional revascularization. If successful, TFC could increase the quality of lives and reduce morbidity, such as amputation, for a significant patient population that is not currently eligible for or helped by conventional therapy.
Footnotes
Acknowledgment
This work was supported by the Ministry of Education, Youth and Sports, Czech Republic, GESHER/MOST Program, Project No. LJ14003 (DiaCellix—new regenerative medicine product for the treatment of secondary effects of T2DM). The authors declare no conflicts of interest.
