Abstract
Hemophilia B (HB) is an inherited deficiency in coagulation factor IX (FIX) that leads to prolonged bleeding after injury. Although hepatocyte transplantation has been demonstrated to be an effective therapeutic strategy for HB, the quality and sources of hepatocytes still limit their application. Recently, stem cells were proposed as an alternative source of donor cells for cell-based therapy. Much research has been devoted to the properties of stem cells that can be differentiated into functional hepatocytes, thereby providing a new cell source for cell-based therapy. Induced pluripotent stem cells (iPSCs) represent a renewable source of hepatocytes for cell-based therapy; these cells exhibit pluripotency and differentiation ability and can be derived from somatic cells. These iPSCs are highly similar to embryonic stem cells (ESCs). We hypothesized that hepatocyte-like cells derived from iPSCs would have therapeutic efficiency in a mouse model of HB. To test this hypothesis, we differentiated iPSCs toward hepatocytes by stepwise protocol and then transplanted these cells into HB mice. We found that these cells shared many characteristics with hepatocytes, such as albumin synthesis, metabolic capacity, glycogen storage, and ureagenesis. Moreover, iPSC-derived hepatocyte transplantation led to increased coagulation factor IX activity, improved thrombus generation, and better hemostasis parameters, and the transferred cells were localized in the liver in recipient HB mice. In conclusion, our results clearly demonstrate that hepatocyte-like cells derived from iPSCs represent a potential cell source for cell-based therapy in the treatment of HB.
Introduction
Hemophilia B (HB) is a bleeding disorder caused by lack of functional coagulation factor IX (FIX). This disorder is X-linked and affects 1 in 30,000 individuals (3). A FIX activity level of 1% results in severe disease and possibly death, while improvement to a level of approximately 5% is sufficient to prevent spontaneous and life-threatening bleeding events (4). Current therapies include FIX replacement and gene therapy. However, FIX replacement is not completely efficient, and some issues remain unresolved, such as its high cost, high risk of anaphylaxis, and life-threatening hemorrhagic complications. Gene therapy is also limited by host immune tolerance and the long-term expression of therapeutic gene products (2,14). Hepatocyte transplantation offers promise for the treatment of many metabolic liver diseases. It was first confirmed by Tatsumi et al. that efficacy of hepatocyte transplantation on HB (19) was consistent with our previously published study, in which the bioengineering of FIX for HB treatment was successful (22); however, the next issue to be considered is cell sourcing. Although natural hepatocytes are an excellent cell resource for cell-based therapy for liver failure, obstacles remain in sourcing and maintaining viable hepatocytes. Over the past few years, a considerable number of studies have investigated the production of hepatocyte-like cells from stem cells, and these cells provide exciting new opportunities for cell-based therapy (8,20). For instance, one recent study demonstrated that endothelial cells derived from induced pluripotent stem cells (iPSCs) showed therapeutic potential in a hemophilia A mouse disease model (23). Thus, we hypothesized that iPSC-derived hepatocytes could also be used to treat HB.
A growing body of literature has demonstrated that somatic cells can be reprogrammed into pluripotent stem cells through the introduction of several transcription factors [octamer-binding transcription factor 4 (Oct4), sex-determining region Y box 2 (Sox2), c-Myc, and Krüpple-like factor 4 (Klf4)] (18). These iPSCs are highly similar to embryonic stem cells (ESCs) with respect to their morphology, gene expression, differentiation potential, and teratoma formation (1,11), thus bypassing the ethical and immunological concerns regarding ESCs. Modern research has shed significant light on cell-based therapy, disease modeling, personalized medicine, and drug toxicity screening (7,15). In particular, there is considerable evidence supporting the therapeutic potential of iPSCs in regenerative medicine, and numerous publications have been devoted to the study of iPSC technology. Because of their wide differentiation potential, iPSCs have been used to generate somatic cells to treat several diseases (9,10), such as Parkinson's disease (PD), Alzheimer's disease (AD), sickle cell anemia, and heart disease. In hepatology, numerous studies have investigated iPSC differentiation into hepatocytes; however, little is known about the therapeutic potential of iPSC-derived hepatocytes in genetic diseases.
In this study, we assessed the therapeutic potential of iPSC-derived hepatocytes in an HB mouse model. Our results demonstrated that iPSCs could be differentiated into hepatocytes that expressed FIX both in vivo and in vitro. Moreover, these iPSC-derived hepatocytes were successfully engrafted within the hepatic parenchyma, where they produced FIX. These findings indicate that iPSC-derived hepatocytes represent another potential cell source for the clinical treatment of hemophilia and other genetic disorders.
Materials and Methods
Animal Model
All mouse experiments were approved by the institutional animal care and use committee at National Taiwan University. The HB mice (male, 6–8 weeks old) used in this study were obtained from Professor S. W. Lin. HB mice deficient in FIX were used as recipient mice in this study. C57BL/6 mice were used to isolate normal hepatocytes as the control group and were purchased from National Laboratory Animal Center, Taipei, Taiwan. A preconditioned animal model was generated according to previous experiments (21). Rifampicin (Sigma-Aldrich, St. Louis, MO, USA) was dissolved in 20 M of sodium hydroxide (NaOH; Sigma-Aldrich), and monocrotaline (MCT; Sigma-Aldrich) was dissolved in normal saline at a pH of 7.4. HB mice received 75 mg/kg rifampicin intraperitoneally daily for 2 days. MCT was administered intraperitoneally as a single 200 mg/kg dose on the third day. Cells were transplanted on the fourth day. Each mouse was injected intrasplenically with 5 × 105 iPSC-derived hepatocytes. Thirty minutes before surgery, HB mice were infused intravenously with 100 IU/kg recombinant FIX protein (Bene FIX®; Wyeth Pharmaceuticals, Philadelphia, PA, USA) (Fig. 1). In the cell tracing experiment, the differentiated cells or primary hepatocytes were stained with PKH26 fluorescent dye (Sigma-Aldrich) according to the manufacturer's instructions. After fluorescent staining, the cells were transplanted into HB mice. At 1 and 4 weeks after transplantation, the recipient mice were sacrificed, and the liver tissue was embedded in TissueTek OCT (Sakura Fintek, Torrance, CA, USA) for immunocytochemistry. For quantitation assay, the number of PKH positive cells was examined under 200× magnification and counted by 20 continuous field per liver lobule for three individual mice.
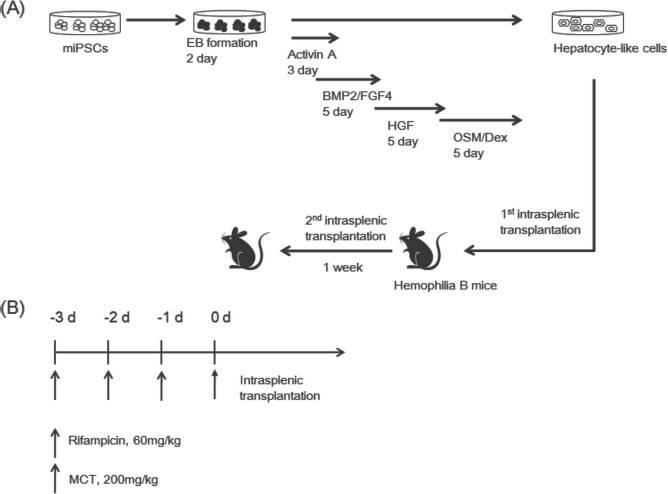
(A) Strategy for differentiating iPSCs into hepatocyte-like cells and subsequent transplantation. (B) Strategy for preconditioning. EB, embryonic body; Dex, dexamethasone.
Cell Culture and Hepatocyte Differentiation
Mouse iPSCs were obtained from S. Yamanaka. The iPSCs were cultured and expanded from mouse embryonic fibroblasts with mytomycin C (Sigma-Aldrich) treatment in Dulbecco's modified Eagle medium (DMEM; Gibco, Grand Island, NY, USA) supplemented with 10% fetal bovine serum (FBS; Hyclone Laboratories, Logan, UT, USA), 0.3% leukemia inhibitory factor (LIF; Millipore, Bedford, MA, USA), 1 mM of L-glutamine, 1% nonessential amino acid, and 0.1 mM of β-mercaptoethanol (all were purchased from Gibco).
Derivation of hepatocytes from iPSCs was performed according to the protocols described and modified by Song et al. (18). The iPSCs were cultured on low-attachment dishes (Corning Life Science, Corning, NY, USA) to form embryonic bodies, which were cultured in Iscove's modified Dulbecco's medium (IMDM; Gibco) supplemented with 1% FBS and 100 ng/ml of activin A (ProSpec, Rehovot, Israel) for 1 day. On the following days, 0.1% insulin–transferrin–selenium (ITS; Sigma-Aldrich) was added to this medium, and 1% ITS was added 1 day later. After endoderm induction, the cells were cultured in IMDM supplemented with 10% FBS, 30 ng/ml fibroblast growth factor 4 (FGF4), and 20 ng/ml bone morphogenetic protein 2 (BMP2) for 5 days (all were purchased from ProSpec). Next, the cells were incubated in IMDM supplemented with 10% FBS, 10 ng/ml of hepatocyte growth factor (HGF; ProSpec), 10 ng/ml of oncostatin M (ProSpec), and 0.1 μM of dexamethasone (Sigma-Aldrich) for 5 days (Fig. 1A).
Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
Total RNA was extracted using RNeasy mini kits (Qiagen Ltd, Crawley, UK), and RT-PCR was performed using a Fast-Run™ Hotstart RT-PCR kit (Protech Technology, Taipei, Taiwan) according to the manufacturer's instructions. The PCR program consisted of 35 cycles of 94°C for 30 s, 72°C for 1 min, and extension at 72°C for 5 min. All PCR primers are shown in Table 1.
Primer Sequences

CK18, cytokeratin 18; ALB, albumin; G6P, glucose-6-phosphate; TAT, tyrosine aminotransferase; AFP, α-fetoprotein; Tdo2, tryptophan 2,3-dioxygenase; HNF4, hepatic nuclear factor 4α.
Functional Assay for iPSC-Derived Hepatocytes
Periodic acid–Schiff (PAS) staining was performed to identify glycogen storage. Cells were fixed in 4% paraformaldehyde (Sigma-Aldrich) for 20 min and then stained with PAS (Sigma-Aldrich). Cellular uptake and indocyanine green (ICG; Sigma-Aldrich) release were measured. ICG (1 mg/ml) was added to the culture medium for 30 min at 37°C, and ICG release was measured after 6 h. During cell differentiation, albumin and uric acid were secreted into the culture medium. The albumin concentration was quantified using a mouse albumin enzyme-linked immunosorbent assay (ELISA) kit (Bethyl Laboratories, Montgomery, TX, USA). The total protein content was determined using a BCA protein assay kit (Pierce, Rockford, IL, USA). The albumin content was normalized to the total cellular protein level. The uric acid levels were measured using a uric acid assay kit (BioVision, Milpitas, CA, USA) according to the manufacturer's protocol.
Immunofluorescence
Cells were fixed in 4% paraformaldehyde for 20 min, permeabilized in 0.1% Triton X-100 (Sigma-Aldrich) for 20 min, and blocked in 3% bovine serum albumin (BSA; Sigma-Aldrich) for 1 h. The cells were then incubated with the primary antibody at 4°C overnight. The mouse anti-α-fetoprotein (Cell Signaling Technology, Beverly, MA, USA), rabbit anti-albumin (Abcam, Cambridge, UK), rabbit anti-cytokeratin 7 (Abcam), anti-cytokeratin 18 (Abcam), and sheep anti-FIX (Molecular Innovations, Novi, MI, USA) primary antibodies were used at a dilution of 1:100, and the rabbit anti-cytokeratin 19 (Abcam) primary antibody was used at a dilution of 1:1,000. After washing with PBS–0.02% Tween-20 (Sigma-Aldrich), cells were incubated with FITC- or PE-conjugated secondary antibody (Invitrogen, Carlsbad, CA, USA) diluted to 1:200 for 2 h and then stained with 4′,6-diamidino-2-phenylindole (DAPI; Biogenex, San Ramon, CA) to stain the cell nuclei.
FIX Clotting Activity Assay
FIX clotting activity was determined using the APTT reagent (Actin® FSL; Dade Behring, Newark, DE, USA) as the activator in a semiautomated blood coagulation analyzer according to the manufacturer's instructions (CA-50; Sysmex, Kobe, Japan). Pooled mouse plasma was obtained from inbred C57BL/6 male mice (n = 18) to generate standard curves for the quantification of test samples. FIX clotting activity in the standard was assumed to be 100%. The test samples were diluted 1:5 in imidazole buffer prior to analysis. A one-stage clotting assay was performed by incubating 50 μl of the test sample with 50 μl of human FIX-deficient plasma (American Diagnostica, Stamford, CT, USA) for 1 min at 37°C, after which 50 μl of activator was added for 3 min at 37°C. Next, 50 μl of 25 mM calcium chloride (CaCl2; Sigma-Aldrich) was added, and the time required for clotting was measured. Each value was reported after subtracting the mean baseline level in HB mice.
Hemostatic Function Assay
Whole blood mixed with a 1/10 volume of 3.2% sodium citrate (Sigma-Aldrich) was assayed by thromboelastography (TEG® analyzer; Haemonetics, Braintree, MA, USA) using the citrated kaolin mode. Analyses for the variables were conducted according to the manufacturer's instructions.
Statistical Analysis
Data are shown as the mean ± standard error of the mean (SEM) and were compared using two-tailed unpaired Student's t-tests or one-way analysis of variance (ANOVA) and post hoc test followed by Tukey's multiple comparisons. Statistical significance was set at a value of p < 0.05.
Results
Generation and Characterization of Hepatocytes From Mouse iPSCs
We previously demonstrated that hepatocyte transplantation improved liver function in an HB mouse model (22). In this study, we examined the therapeutic potential of hepatocyte-like cells derived from iPSCs to treat genetic liver diseases (Fig. 1). We utilized HB mice as a proof-of-principle preclinical model to test the therapeutic potential of iPSC-derived hepatocytes, and we used a previously developed preconditioned animal model to investigate cell engraftment (21).
Using a hepatic differentiation protocol, iPSCs (Fig. 2A) were induced and differentiated to the hepatoblast stage by BMP2 and FGF4 through endoderm induction by activin A (at day 7, Fig. 2B), and finally differentiation to hepatocyte-like cell by HGF/OSM/Dex (at day 20, Fig. 2C). During differentiation, the cell morphology changed from colonical to polygonal. We checked the mRNA expression at day 18 in hepatocytes. The results of RT-PCR demonstrated that hepatocyte markers, including α-fetoprotein (AFP), albumin (ALB), hepatocyte nuclear factor 4 (HNF4), glucose 6-phosphate (G6P), tyrosine amino transferase (TAT), and cytokeratin 18 (CK18) could be detected by protein expression or immunofluorescent staining. AFP was expressed in immature hepatocytes and was detected at day 10, ALB and cytokeratin 7 (CK7) at day 15, and CK18 and cytokeratin 19 (CK19) at day 20 when the cells were further differentiated (Fig. 3). Importantly, the RT-PCR and immunofluorescence results confirmed that the iPSC-derived hepatocytes expressed FIX mRNA (Fig. 2D) and protein (first detected at day 10) (Fig. 3). Taken together, these data show that hepatocytes can be derived from iPSCs using our differentiation procedure.

Generation of hepatocytes from mouse iPSCs. Mouse iPSCs (A) were differentiated into hepatocyte-like cells for 7 (B) and 20 days (C). At day 18, the differentiated cells were expressed hepatocyte-related gene by RT-PCR (D). These cells showed functional characteristics of hepatocytes, including glycogen storage, as determined by PAS staining (E), ICG uptake (F), ALB secretion (G), and uric acid production (H) as measured by ELISA. Data are expressed as the mean ± SEM. Magnification: 100×. Scale bar: 100 μm.

Immunostaining analysis. Expression of AFP and FIX in day 10 induced differentiated cells. ALB and CK7 were expressed in day 15 induced differentiated cells, and CK218 and CK19 were expressed in day 20 induced differentiated cells. Magnification: 100×, scale bar: 100 μm (AFP, CK7, and CK18) or magnification: 200×, scale bar: 5 μm (ALB, CK19, and FIX).
We further examined the functionality of the iPSC-derived hepatocytes. PAS staining demonstrated the ability of the derived cells to store glycogen (Fig. 2D), and ICG analysis indicated their capacity to accumulate and excrete compounds (Fig. 2E). Protein synthesis is another characteristic of functional hepatocytes, which can be monitored by the secretion of proteins into the culture medium. Thus, we monitored the concentration of albumin during the period of differentiation. Albumin production was first detected after 13 days of differentiation, and reached the maximum level at 21 days of differentiation (1.51 ± 0.65 ng/mg protein/day) (Fig. 2F). Moreover, after 18 days of differentiation, the uric acid production level was 4.45 ± 0.48 nmol/ml/day (Fig. 2G). These results revealed that the generated iPSC-derived hepatocytes exhibited hepatic functionality.
Transplantation of iPSC-Derived Hepatocytes
Next, we assessed the therapeutic efficacy of iPSC-derived hepatocytes in the HB mouse model. The HB mice were divided into two groups (one that received normal hepatocyte transplantation and the other iPSC-derived hepatocyte transplantation), and the circulating FIX activity was monitored after 4 weeks of transplantation. Our data showed that iPSC-derived hepatocytes enhanced FIX clotting activity by 2–3%, and the circulating FIX activity was stable for up to 1 month (Fig. 4A). These results reflected a substantial improvement in HB mice.

Hepatocyte transplantation in the HB mouse model. (A) The FIX clotting activity after transplantation with iPSC-derived hepatocytes was similar with those transplanted with normal hepatocytes (WT-Hep) at 1, 2, 3, and 4 weeks after transplantation. Data are expressed as the mean ± SEM. (B) The iPSC-Hep induced greater thrombus generation. Cell localization 1 week after WT-Hep (C) and iPSC-Hep (D) transplantation. (E) Preconditioned model. The arrows in (C)–(E) denote transplanted cells. (F) Quantification of iPSC-Hep engraftment in the HB mice. Red, PKH 26; blue, DAPI. Magnification: 200×, scale bar: 50 mm.
Because the clotting test could not fully reflect the in vivo hemostatic response, we utilized the TEG assay to further examine the in vivo hemostatic state. Moreover, we used transplantation to improve the therapeutic efficacy. For this purpose, HB mice underwent a second intrasplenic transplantation (separated by 1 week) of iPSC-derived hepatocytes or normal hepatocytes, and we assessed hemostasis in whole blood 7 days after the second transplantation. The peak level of thrombin generation progressively decreased, and the time of thrombus generation was 43 min in HB mice. However, repeated normal hepatocyte transplantation improved the peak thrombin level and reduced the time of thrombus generation to 14 min. In addition, the effects of repeated iPSC-derived hepatocyte transplantation were similar to the effects of repeated normal hepatocyte transplantation (Fig. 4B). Moreover, repeated iPSC-derived hepatocyte transplantation improved both the reaction time and strength of thrombus generation and the coagulation index (CI) value, which is another hemostatic parameter that reflects the overall ability of blood to coagulate. The CI value of mice after repeated iPSC-derived hepatocyte transplantation improved to –6.4 (the value before transplantation was –32.3) (Table 2). Taken together, these results indicate that iPSC-derived hepatocyte transplantation markedly improved hemostasis, suggesting that iPSC-derived hepatocytes could be used to treat HB.
Effects of Transplantation on Whole Blood Hemostasis in HB Recipient Mice

B6, inbred C57BL/6 mice; HB, hemophilia B mice; R (reaction time), the time to initial fibrin formation; K, the speed to reach a specific level of clot strength; Angle, the rapidity of fibrin build-up and cross-linking; MA (maximum amplitude), the reflection of clot strength contributed by platelets and fibrin; CI (coagulation index), overall assessment of coagulation ability; MRTG, maximum rate of thrombus generation; TTG, total thrombus generation. Data are expressed as the mean ± SEM, n ≥ 3. Compared with the HB group,
p < 0.05,
p < 0.01,
p < 0.001.
Engraftment of iPSC-Derived Hepatocytes
Finally, to investigate whether the iPSC-derived hepatocytes were retained in the liver parenchyma, we utilized immunofluorescence to trace the grafted cells in recipient mice. Before transplantation, the differentiated cells were stained with PKH26 fluorescent dye, and liver sections were examined at 1 week after transplantation. The immunofluorescence staining showed that PKH26-positive cells were localized in the livers of recipient mice, thus indicating that the transplanted cells were engrafted (Fig. 4C and D). To improve the engraftment efficiency, we adopted a preconditioned model that included immunosuppression (MCT and rifampicin). For iPSC-derived hepatocyte engraftment in the livers of preconditioned animals 1 week after transplantation, the number of engrafted cells was similar to the value observed in nonpreconditioned mice (Fig. 4F), whereas more PKH26-positive cells were detected at 4 weeks after transplantation (Fig. 4E). These results indicated that the transplantation of iPSC-derived hepatocytes into the livers of preconditioned mice could enhance the efficiency of engraftment. Additionally, we assessed hemostasis in the preconditioned animal model with iPSC-derived hepatocyte transplantation. The TEG assay revealed that the time of thrombin generation for iPSC-derived hepatocyte transplantation was similar to the result for normal hepatocyte transplantation, with a shorter time of thrombus generation. iPSC-derived hepatocyte transplantation resulted in improved CI values and hemostasis parameters in the preconditioned model, compared with the normal hepatocyte transplantation group. Therefore, iPSC-derived hepatocytes possess therapeutic potential in HB mice.
Discussion
Our previous study demonstrated the therapeutic efficacy of hepatocyte transplantation in the HB mouse model, although the shortage of hepatocyte sources limits their therapeutic application. Therefore, another hepatocyte source is urgently needed for cell-based therapy. The use of iPSCs has revolutionized the field of regenerative medicine, as these cells have been shown to represent a novel source of hepatocytes for drug development and cell-based therapy (6). In this study, we utilized the ability of iPSCs to differentiate into functional hepatocytes in vitro and then transplanted these cells into HB mice to assess their therapeutic potential. Our results showed that iPSCs differentiated into functional hepatocytes in our optimized culture conditions and that iPSC-derived hepatocyte transplantation resulted in enhanced FIX clotting activity and improved hemostasis.
Recently, numerous reports have indicated that iPSC-based therapy could improve liver function in disease models. Specifically, Xu et al. reported that the transplantation of iPSC-derived endothelial cells into a hemophilia A mouse model resulted in factor VIII expression, providing a proof-of-principle for genetic disorder treatment (23). Moreover, iPSC-derived hepatocytes transplanted into mice with lethal acute hepatic failure induced by thioacetamide, a hepatotoxin that causes the production of reactive oxygen species (ROS), alleviated acute hepatitis. These results indicate that iPSC-derived hepatocytes can improve liver function and prevent oxidative stress-induced damage (13). In another liver failure model, iPSC-derived hepatocyte transplantation improved survival and protected against liver injury induced by CCL4 (5). We demonstrated that iPSC-derived hepatocyte transplantation in HB mice resulted in FIX clotting activity and improved hemostasis. Similar effects of transplantation have been reported for other diseases. Furthermore, the combination of iPSC-based therapy and gene therapy has been reported for therapeutic applications in sickle cell anemia (17). It is precisely on such grounds that we believe that gene therapy combined with iPSC-based therapy holds promise for treating HB in the future (12).
The repopulation capacity of grafted cells may limit the efficiency of cell-based therapy; however, 2–5% replacement has been shown to sufficiently improve liver function (16). To examine this aspect, we adopted a preconditioned model (21) and repeated transplantation to improve the engraftment efficiency. Our data reveal that repeated transplantation can improve hemostasis in HB mice with results similar to normal hepatocyte transplantation. Further experiments showed that the engraftment efficiency of transplanted iPSC-derived hepatocytes was further improved in the preconditioned model, and the transplanted cells were engrafted in the liver parenchyma. Although the time of thrombus generation was shortened, the strength of thrombus was insufficient, partly because the differentiated cells did not fully acquire hepatic function. This problem will hinder clinical application, as undifferentiated cells can induce tumorigenicity. Currently, none of the strategies for fully separating differentiated cells from heterogeneous cell populations eliminates the risk of teratoma formation. Thus, providing a promising strategy for separating cell populations would help to solve this problem.
In this study, we acquired functional hepatocytes from iPSCs using a stepwise differentiation protocol and compared the therapeutic efficacy of iPSC-derived hepatocytes and freshly isolated hepatocytes in HB mice. Although iPSC-derived hepatocytes can be used as an alternative cell source to treat HB, there are still some obstacles that must be overcome before clinical application of this type of cell therapy. It is important to eliminate the undifferentiated cells before transplantation and to assess the risk of tumorigenesis with long-term follow-up.
Footnotes
Acknowledgments
This study was supported by a grant from the National Science Council NSC 101-2314-B-002-016 and NSC 2314-B-002-038. The authors declare no conflicts of interest.
