Abstract
Allogeneic and xenogeneic transplantation are suitable alternatives for treating patients with stem cell defects and autoimmune diseases. The purpose of this study was to compare the effects of long-term serial transplantation of adipose tissue-derived mesenchymal stem cells (ASCs) from (NZB × NZW) F1 mice (syngeneic), BALB/c mice (allogeneic), or humans (xenogeneic) on systemic lupus erythematosus (SLE). The effects of transplanting human ASCs overproducing CTLA4Ig (CTLA4Ig-hASC) were also compared. Animals were divided into five experimental groups, according to the transplanted cell type. Approximately 500,000 ASCs were administered intravenously every 2 weeks from 6 to 60 weeks of age to all mice except for the control mice, which received saline. The human ASC groups (hASC and CTLA4Ig-hASC) showed a 13-week increase in average life spans and increased survival rates and decreased blood urea nitrogen, proteinuria, and glomerular IgG deposition. The allogeneic group also showed higher survival rates compared to those of the control, up to 40, 41, 42, 43, 44, 45, 52, and 53 weeks of age. Syngeneic ASC transplantation did not accelerate the mortality of the mice. The mean life span of both the syngeneic and allogeneic groups was prolonged for 6–7 weeks. Both human ASC groups displayed increased serum interleukin-10 and interleukin-4 levels, whereas both mouse ASC groups displayed significantly increased GM-CSF and interferon-γ levels in the serum. The strongest humoral immune response was induced by xenogeneic transplantation, followed by allogeneic, CTLA4Ig-xenogeneic, and syngeneic transplantations. Long-term serial transplantation of the ASCs from various sources displayed different patterns of cytokine expression and humoral responses, but all of them increased life spans in an SLE mouse model.
Keywords
Introduction
Systemic lupus erythematosus (SLE) is a multisystem autoimmune disease characterized by the presence of serum autoantibodies directed against nucleic acids including anti-dsDNA antibodies, deposition of immune complexes in various organs such as the kidney, inflammation, and tissue damage (3,7,28,32).
Common mouse models of spontaneous SLE include the F1 hybrids of New Zealand Black and New Zealand White mice [(NZB × NZW) F1], MRL/lpr mice, and BxSB/Yaa mice (26,30,35). Among these models, (NZB × NZW) F1 mice are considered to be most representative of SLE in humans (8,21,38). Female (NZB × NZW) F1 mice are characterized by the production of antinuclear antibodies, including anti-dsDNA antibodies, and the development of severe immune complex-mediated glomerulonephritis (17,42). According to data from the Jackson laboratory, these female mice have an average life span of only 245 days (35 weeks) owing to the development of renal failure.
Recently, it has been reported that mesenchymal stem cells (MSCs) are immunoprevileged, have paracrine effects, and have immunomodulatory properties (2,15,16,22,39–41). In previous studies, single or short-term allogeneic bone marrow-derived MSC (BM-MSC) transplantation in murine SLE [(NZB × NZW) F1] mice at an advanced disease stage did not have favorable effects on the level of anti-dsDNA antibodies, proteinuria, or survival; in a study by Schena et al., (NZB × NZW) F1 mice were treated intravenously with allogeneic C57BL/6-derived BM-MSCs (1.25 × 106) at 27, 28, and 29 weeks of age. Proteinuria was evaluated weekly at 25–32 weeks of age, and histopathological assessment of kidneys was performed at 33 weeks of age. According to their results, MSC treatment significantly improved the histological characteristics of kidneys but did not improve the production of autoantibodies, the level of proteinuria, or the mortality rates (33). In a study by Youd et al., (NZB × NZW) F1 mice were treated with intraperitoneal injection of allogeneic BALB/c-derived BM-MSCs (1 × 106) biweekly starting at 21 or 32 weeks of age. In their study, systemic administration of MSCs worsened the disease and increased anti-dsDNA autoantibody production (44).
Furthermore, a single injection of umbilical cord blood-derived MSCs (1 × 106) at 8 weeks of age showed just a short-term minor effect on anti-dsDNA antibody production and proteinuria; none of the mice showed severe proteinuria at 6 months, but 50% of mice showed severe proteinuria at 8 months of age. There was no significant difference in serum levels of anti-dsDNA antibody between the control group and the treatment group at 8 months of age (5). In our preliminary study, a single injection of adipose tissue-derived mesenchymal stem cells (ASCs) before disease onset (2 × 106 at 6 weeks of age) also did not significantly affect the levels of blood urea nitrogen (BUN), the incidences of proteinuria, or the rates of survival (unpublished data).
However, data from our previous study (13) showed that long-term serial human ASC (hASC) transplantation in a murine SLE [(NZB × NZW) F1] model at an early disease stage (from age 6 weeks until age 60 weeks, biweekly) ameliorated histological and serological abnormalities and enhanced immunological functions. The survival rate was significantly higher, and the proteinuria rate was significantly lower in the hASC group than in the control group. ASCs are regarded as an ideal source for practical regenerative medicine using stem cell-based therapy because adipose tissues are abundant and easily obtainable in large quantities (4,10). Furthermore, it was reported recently that the proliferation and differentiation of ASCs were less affected by age and multiple passages than were the proliferation and differentiation of BM-MSCs (6).
The purpose of this study was to compare the effects of long-term serial syngeneic, allogeneic, xenogeneic, and CTLA4Ig-overproducing xenogeneic ASC transplantations in an SLE mouse model. In human patients with stem cell defects, it is important to understand the differences between autologous and allogeneic stem cell transplantation for further clinical applications, especially for autoimmune diseases (14). Therefore, we compared the characteristics of ASCs derived from adipose tissue from two different mouse strains and analyzed differences in the efficacy and humoral response between transplanted syngeneic ASCs acquired from animals with spontaneously induced autoimmune diseases and transplanted allogeneic ASCs acquired from animals without diseases, in a murine model that develops a spontaneous lupus-like disease.
Aggarwal and Pittenger investigated the immunomodulatory functions of human MSCs by coculturing MSCs with purified subpopulations of immune cells. In their study, MSCs caused mature dendritic cell type 1 (DC1) cells to secrete decreased levels of tumor necrosis factor-α (TNF-α) and mature DC2 cells to secrete increased levels of interleukin (IL)-10. MSCs also led to decreased interferon-γ (IFN-γ) secretion and increased IL-4 secretion by Th1 and Th2 cells, respectively. Furthermore, an increase in the proportion of regulatory T cells and decreased IFN-γ secretion from natural killer (NK) cells were also observed (1). The results from that study were useful in understanding the immunomodulatory actions of MSCs on immune cell subpopulations in culture. However, the interplay and interactions between the immune cells, the pathophysiology of disease, and the microenvironment of the host are all much more complex in a living organism. Thus, the in vivo study of the immunomodulatory functions of stem cells under a specific disease condition by using animal models is essential to understand disease-specific mechanisms comprehensively. Therefore, in this study, we investigated the immunomodulatory effects of syngeneic, allogeneic, and xenogeneic ASC transplantations in a murine SLE model by comparing their cytokine secretion profiles. The lack of a costimulatory signal after the coupling of T-cell receptors to antigens further induces an antigen-specific unresponsive state, also known as T-cell anergy. Thus, blocking the B7-CD28 costimulatory pathway by CTLA4Ig is considered a useful strategy for achieving immunosuppression or tolerance (23,27,45).
Recombinant proteins have long faced practical problems, as they are short acting and expensive, whereas ASCs can induce more specific targeting and synergic effects owing to their homing effects and immunomodulatory properties (9,10). We, therefore, also evaluated whether CTLA4Ig-overproducing xenogeneic ASCs might show synergistic immunomodulatory effects.
Materials and Methods
Isolation and Culture of hASCs
hASCs were prepared under good manufacturing practice conditions (K-Stemcell, Seoul, Korea) as described previously (12); hASCs were obtained by simple liposuction from abdominal subcutaneous fat after the donor had provided informed consent. Adipose tissues were digested with 4 ml RTase (K-Stemcell) per 1 g fat under gentle agitation for 60 min at 37°C and were filtered through a 100-μm nylon sieve, followed by centrifugation at 470 × g for 5 min. The pellet was resuspended in RCME (hASC attachment medium; K-Stemcell) containing 10% fetal bovine serum (FBS; Gibco BRL, Rockville, MD, USA) and centrifuged again at 470 × g for 5 min. The supernatant was discarded, and the cell pellet was collected. The cell fraction was cultured overnight at 37°C at 5% CO2 in RCME containing 10% FBS. After 24 h, nonadherent cells were removed by washing with Dulbecco's phosphate-buffered saline (DPBS; Welgene, Daegu, Korea). The cell medium was then changed to RKCM (hASC culture medium; K-Stemcell) containing 5% FBS. The cells were maintained for 4 or 5 days until confluent (passage 0). When the cells reached 90% confluency, they were detached using TryPLE express (Gibco BRL) and subculture expanded in RKCM until passage 3. Anonymized hASCs from one donor were used in this study under an institutional review board (IRB)-approved exemption (Samsung Medical Center IRB, SMC 2014-05-018); according to the enforced regulation of the National Bio-Ethics Committee, the research can exempt the deliberation if the researcher cannot realize the personal identification information of the human body origin donor, and the research does not have the relationship with the genetic characteristic of the donor person.
Preparation and Confirmation of CTLA4Ig-Overproducing hASCs (CTLA4Ig-hASCs)
The construction of the therapeutic mouse CTLA4Ig gene and the preparation of CTLA4Ig-hASCs were performed as described in our previous study (12); we constructed a therapeutic gene comprising the extracellular domain of mouse CTLA-4 (GenBank accession number NM_009843, nt 258–629) and the CH2–CH3 domains of the human immunoglobulin γ 1 constant (IGHGC) region (J00228, nt503, 892-936, 1055-1384, 1481-1803). The human oncostatin M (NM_020530.3) signal sequence (nt 53–127) was cloned along with the therapeutic gene to enable protein secretion into body fluid. The construct was transduced into hASCs using a ViraPower™ Lentiviral Expression System (Invitrogen, Carlsbad, CA, USA). The reverse transcription-polymerase chain reaction (RT-PCR) was used to detect transcription of therapeutic gene (primers: hOnco-F 5′-CACCATGGGGGTACTGCTCA-3′ and hIgG1-R 5′-TCATTTACCCGGAGACAGGGA-3′), and enzyme-linked immunosorbent assays [ELISAs, CTLA4 Duoset ELISA Development kit (DY476; R&D systems, Minneapolis, MN, USA)] were used to quantify the concentration of the mouse CLTA4 protein in stem cell culture supernatants, as described in our previous study (12).
Isolation and Culture of Mouse ASCs (mASCs)
Mouse adipose tissues were obtained from the axillary and inguinal subcutaneous fat from different donors [syngeneic: (NZB × NZW) F1 mice (female, n = 30; Jackson Laboratory, Bar Harbor, ME, USA); allogeneic: BALB/c mice (male, n = 30; Orient Bio, Sungnam, Korea)] at 16–22 weeks of age under deep anesthesia with intraperitoneal injection of a ketamine (80 mg/kg, Ketamine 50®; Yuhan Co., Seoul, Korea)/xylazine (10 mg/kg, Rompun®, Bayer Korea, Seoul, Korea) combination. mASCs were prepared as described previously (14). Briefly, the stromal-vascular fraction from adipose tissues was cultured at 37°C in 5% CO2 and 3% O2 in the STEMXvivo MSC expansion medium (R&D Systems). After 24 h, nonadherent cells were removed by medium replacement, and adherent cells (mASCs) were maintained until 80–90% confluent. mASCs were detached using TryPLE express (Gibco BRL) and expanded until passage 1 or 2 with medium changes every 2–3 days.
Immunophenotype and Differentiation Capacity of mASCs
mASC cultures from passage 2 were immunophenotyped by flow cytometric analysis. Immunophenotyping was conducted as we described previously (14); fluorescein isothiocyanate (FITC)-conjugated hamster anti-mouse CD29 (clone: Ha2/5, 555005; BD Biosciences, San Jose, CA, USA), phycoerythrin (PE)-conjugated rat anti-mouse CD31 (clone: MEC13.3, 553373; BD Biosciences), FITC-conjugated rat anti-mouse CD34 (clone: MEC14.7, MCA1825F; BD Biosciences), FITC-conjugated rat anti-mouse CD44 (clone: IM7, 553133; BD Biosciences), FITC-conjugated rat anti-mouse CD45 (clone: 30-F11, 553079; BD Biosciences), PE-conjugated rat anti-mouse CD73 (clone: TY/23, 550741; BD Biosciences), PE-conjugated rat anti-mouse CD 90.2 (clone: 53-2.1, 553006; BD Biosciences), PE-conjugated rat anti-mouse CD105/endoglin (clone: 209701, FAB1320P; R&D Systems), PE-conjugated hamster anti-mouse CD95 (clone: Jo2, #554258; BD Biosciences), FITC-conjugated mouse anti-mouse H-2Kd (MHC class I, clone: SF1-1.1, #553565; BD Biosciences), and FITC-conjugated mouse anti-mouse I-Ad (MHC class II, clone: AMS-32.1, #553547; BD Biosciences) were used to detect cell surface antigens. Corresponding isotypic antibodies served as controls. Differentiation of ASCs was conducted using the MSC Adipogenesis Kit (Millipore, Darmstadt, Germany) and NH OsteoDiff Medium (Miltenyi Biotec GmbH, Bergisch Gladbach, Germany), according to the manufacturers' instructions. Adipogenesis and osteogenesis was assessed quantitatively using the Adipogenesis Assay Kit (Millipore) based on Oil red O staining dye and the Osteogenesis Assay Kit (Millipore) based on Alizarin red staining dye, respectively. Quantitative assessment was conducted in two independent experiments repeated three times.
Experimental Animals
(NZB × NZW) F1 female mice (n = 75) were purchased from the Jackson Laboratory. This study was reviewed and approved by the Institutional Animal Care and Use Committee of Samsung Biomedical Research Institute. Samsung Biomedical Research Institute is accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International and abides by the guidelines of the Institute of Laboratory Animal Resources.
Experimental Groups
For the in vivo study, the experimental groups consisted of a control group (n = 15), the hASC group (xenogeneic, hASCs, n = 15), the CTLA4Ig-hASC group (CTLA4Ig-overproducing hASCs, n = 15), the syngeneic group [(NZB × NZW) F1 ASCs, n = 15], and the allogeneic group (BALB/c ASCs, n = 15). Each mouse in the ASC groups was intravenously administered 5 × 105 ASCs/150 μl of saline every 2 weeks from the age of 6 weeks until the age of 60 weeks. The control mice were administered 150 μl of saline and were subjected to the same schedule. The survival rate and incidence of severe proteinuria (percent of individual mice showing ≥ 300 mg/dl of urine protein) were calculated. Survival was monitored until 62 weeks of age.
Determination of Proteinuria
Coomassie brilliant blue (CBB) reagent was made by Lott et al.'s method (24); 50 ml of 95% ethanol (DaeJung Chemicals & Metals, Shiheung-city, Korea) was added to about 100 mg of CBB G-250 (B1131; Sigma-Aldrich, St. Louis, MO, USA) and mixed to dissolve. The mixture was warmed to 50°C for about 10 min. One hundred milliliters of phosphoric acid (Fluka 79606; Sigma-Aldrich) was added to 500 ml of distilled water, and the diluted phosphoric acid was added to the CBB in ethanol. Distilled water was added to bring total solution to 1 L. Eight to 10 grams of siliceous earth (392545; Sigma-Aldrich) was added and stirred for 6 h. The CBB solution was stored at 4°C for 24 h and then filtered twice through Whatman No. 1 filter paper (GE Healthcare, Piscataway, NJ, USA). CBB reagent was centrifuged at 800 × g for 10 min before using.
During the experiments, urine protein levels were measured every 2 weeks. Fresh urine was collected by performing abdominal massages. Urine proteins were measured as described in the methods of our previous study; the protein concentration of pooled fresh murine sera was standardized using the biuret method and used as a standard. The protein standard was diluted with a solution of 154 mmol/L NaCl to obtain working standards of 25, 50, 100, and 150 mg serum protein/dl. A 10-μl specimen (standard or urine) was added to 1 ml of CBB reagent, incubated for 39 min, and the absorbance values of samples and water blanks were measured at 465 nm and 595 nm (11,24). The ratio of the absorbance (A595/A465) versus the concentration of the standards was plotted on linear graph paper, and a standard curve was constructed for every experiment. The specimens with protein concentrations exceeding 1,500 mg/L were diluted with 154 mmol/L NaCl and reanalyzed.
Body Weight, Spleen Weight, and Spleen Weight/Body Weight Ratios
The spleens of mice that died of natural causes were harvested and weighed immediately upon death or at the end of the study (at 62 weeks of age). Body weights were also measured at these times.
Determination of BUN, Serum Creatinine, and dsDNA Antibody Levels
Blood samples were collected under isoflurane anesthesia every 2 months, and sera were stored at −70°C. BUN and serum creatinine levels were determined with a DRI-CHEM 3000 Colorimetric analyzer (Fujifilm, Tokyo, Japan). Anti-dsDNA antibody concentrations were measured using a mouse anti-dsDNA ELISA kit (Shibayagi Co. Ltd., Ishihara, Shibukawa, Japan).
ELISAs Analyzing Multiple Cytokine Levels in Spleen Cell Culture Supernatants and Sera
Serum samples (n≥8, each group) were assayed using a Milliplex® MAP Kits (Millipore) for granulocyte macrophage-colony stimulating factor (GM-CSF), TNF-α, IFN-γ, IL-1α, IL-1β, IL-2, IL-4, IL-6, IL-10, IL-15, IL-17, MCP-1, and RANTES.
Hematoxylin and Eosin (H&E), Periodic Acid/Schiff (PAS) Reagent, and Masson Trichrome Staining
Kidneys were harvested from mice that died of natural causes immediately upon death or at the end of the study (at 62 weeks of age). For light microscopy, the tissues were fixed in 10% neutral buffered formalin, embedded in paraffin wax, sectioned at a thickness of 5 μm, and stained with H&E (BBC Biochemical, Mount Vernon, WA, USA) by using routine histological techniques. Periodic acid/Schiff (PAS stain kit; BBC Biochemical) staining and Masson trichrome (BBC Biochemical) staining were used for the detection of proteinuria and fibrosis, respectively.
Kidney Immunofluorescence Experiments
The fluorescence intensity of IgG or C3 deposits in freshly harvested kidneys acquired from the final survivors was evaluated [n = 4–5 per group; ages of mice from which tissue was taken: control group: 50 weeks of age (n = 2), 52 weeks of age (n = 1), 62 weeks of age (n = 1); hASC group: 62 weeks of age (n = 4), CTLA4Ig-hASC group: 62 weeks of age (n = 4), syngeneic group: 47 weeks of age (n = 1), 48 weeks of age (n = 1), 53 weeks of age (n = 1), 62 weeks of age (n = 2); allogeneic group: 48 weeks of age (n = 1), 51 weeks of age (n = 1), 53 weeks of age (n = 2)]. Immunofluorescence staining procedures were performed as described in our previous study (13); frozen sections (4 μm) of kidney tissues were fixed in ice-cold acetone for 5 min and washed two times in ice-cold phosphate-buffered saline (PBS; Sigma-Aldrich). Nonspecific binding was blocked with blocking solution (1% bovine serum albumin and 0.05% Tween 20 in PBS; Koma Biotech, Seoul, Korea) for 30 min. The slides were drained, wiped with tissue paper, and incubated at room temperature with FITC-conjugated goat anti-mouse IgG (1:200; AP 308F; Millipore) or FITC-tconjugated goat anti-mouse C3 (1:100; MP Biomedicals, West Chester, PA, USA) for 1 h. The slides were washed three times in PBS and mounted with mounting medium containing 4′,6-diamidino-2-phenylindole (DAPI; Vector Laboratories, Southfield, MI, USA) and examined with an LSM 700 laser-scanning confocal microscope (Carl Zeiss, Jena, Germany). The fluorescence staining intensities of IgG or C3 deposits were graded as 0 (none), 1+ (mild), 2+ (moderate), 3+ (moderate to strong), and 4+ (strong).
Confocal Microscopy Experiments with CM-DiI-Labeled ASCs
Three mice from each group were administered fluorescently labeled ASCs by using red fluorochrome–conjugated Cell Tracker CM-DiI (Life Technologies, Carlsbad, CA, USA) for histopathological analysis. The tissue samples of naturally deceased mice were harvested immediately after death or at the end of the study [mouse ages of tissue taken: control group: 41 weeks of age (n = 1), 47 weeks of age (n = 1), 50 weeks of age (n = 1); hASC group: 48 week of age (n = 1), 56 weeks of age (n = 1), 58 weeks of age (n = 1); CTLA4Ig-hASC group: 54 weeks of age (n = 1), 62 weeks of age (n = 2); syngeneic group: 36 weeks of age (n = 1), 48 weeks of age (n = 1), 53 weeks of age (n = 1); allogeneic group: 46 weeks of age (n = 1), 53 weeks of age (n = 1), 54 weeks of age (n = 1)]. The presence of CM-DiI-labeled ASCs was examined in various tissues, such as the spleen, kidney, liver, lung, and heart by counterstaining with mounting medium containing DAPI and observing the sections under a Radiance 2100 laser-scanning confocal microscope (Bio-Rad Laboratories, Hercules, CA, USA).
Humoral Responses to Infused Cells
Humoral responses to infused cells were evaluated by detecting antibodies against the transplanted ASCs, as described in our previous study (14); for the detection of antibodies against administered ASCs, flow cytometric cross-matching for circulating anti-(NZB × NZW) F1 ASC antibodies or anti-BALB/c ASC antibodies was conducted using sera collected from each recipient and from control mice. Circulating anti-hASC antibodies or anti-CTLA4Ig-hASC antibodies were also evaluated in recipient and control mouse sera. Specifically, recipient mouse sera (1:25 dilution in DPBS) and control mouse sera (non-ASCs infused, negative control) were incubated with donor ASCs for 30 min at 37°C. After washing, ASCs were incubated with FITC-conjugated anti-mouse IgG antibody (1:1,000 per 106 ASCs; Millipore) for 1 h at 4°C, then washed in DPBS (Welgene) and assayed in a flow cytometer (FACSAria, BD Biosciences). Fluorescence signals from 10,000 cells were counted and the percentage of FITC-positive cells recorded. Sera acquired at 4 and 10 months of age were used for analysis, each of which was performed in duplicate experiments.
Statistical Analysis
All results except for proteinuria and survival rate data were expressed as the mean ± SEM. Data obtained from groups were compared using one-way analysis of variance (ANOVA), followed by either post hoc Tukey's multiple comparison tests or Dunnett's test. Proteinuria and survival rate data were analyzed using Kaplan–Meier curves and the log-rank test. Two-group comparisons were performed with the Student's t-test. A value of p < 0.05 was considered to be statistically significant. All statistical analyses were conducted using SPSS version 17.0 (IBM, Armonk, NY, USA).
Results
Immunophenotyping and Differentiation Capacity of ASC
Immunophenotyping results, obtained using flow cytometry, demonstrated a high expression of CD29, CD44, CD73, CD90, and CD105 and the absence of CD31, CD34, CD45, and HLA-DR surface markers in hASCs and CTLA4Ig-hASCs (data not shown), which is in agreement with results of our previous study (12). Like ASCs from BALB/c mice in a previous study [(14); data not shown], ASCs from (NZB × NZW) F1 mice were positive for CD29, CD44, and CD90.2 expression, while the expression of CD31, CD34, and CD45 was absent, and the surface markers CD73 and CD105 were expressed at moderate levels. ASCs from (NZB × NZW) F1 did not express MHC class II (I-Ad), as shown in Figure 1 (five independent experiments per group). ASCs from BALB/c mice were positive for MHC Class I (H-2Kd) expression in a previous study [(14); data not shown], while those from (NZB × NZW) F1 mice exhibited moderate expression of MHC Class I (H-2Kd). In addition, we found that ASCs from both strains possessed the ability to differentiate; ASCs from (NZB × NZW) F1 mice showed similar adipogenic but decreased osteogenic differentiation abilities [two independent experiments per each group (mouse ages of ASCs taken: 40 weeks of age and 50 weeks of age), triplicates, Student's t-test, p = 0.013] compared to those of ASCs from BALB/c mice. The features of differentiation of ASCs from both strains into adipocytes and osteocytes are presented in Figure 1.

Differentiation of ASCs from two strains, BALB/c and (NZB × NZW) F1 mice, into adipocytes and osteocytes, and ASC immunophenotypes from (NZB × NZW) F1 mice. (A) ASCs that differentiated into adipocytes contained Oil red O-positive lipid droplets. BALB/c (upper left), (NZB × NZW) F1 (upper right), and ASCs differentiated into osteocytes, which were stained with Alizarin red S; BALB/c (lower left), (NZB × NZW) F1 (lower right). (B) Immunophenotype of ASCs from (NZB × NZW) F1 at passage 2. Data are expressed as mean ± SEM (unit: %, except for cell size), SEM: the standard error of the mean.
Survival Rate, Average Survival Time, and Incidence of Proteinuria
Upon comparison up to the end of the experiment, survival rates were significantly higher and the incidences of proteinuria were significantly lower in the hASC and CTLA4Ig-hASC groups compared to the control group, as assessed using Kaplan–Meier curves and a log-rank test (Fig. 2A,B). No significant differences were observed in the survival rates and proteinuria incidences between the control, syngeneic, and allogeneic groups (Fig. 2A,B). However, upon comparison, up to 40, 41, 42, 43, 44, 45, 52, and 53 weeks of age, the allogeneic group showed significantly higher survival rates compared to those of the control group. The allogeneic group showed significantly lower incidence of proteinuria compared to those of the control group upon comparison up to 36 and 38 weeks of age.
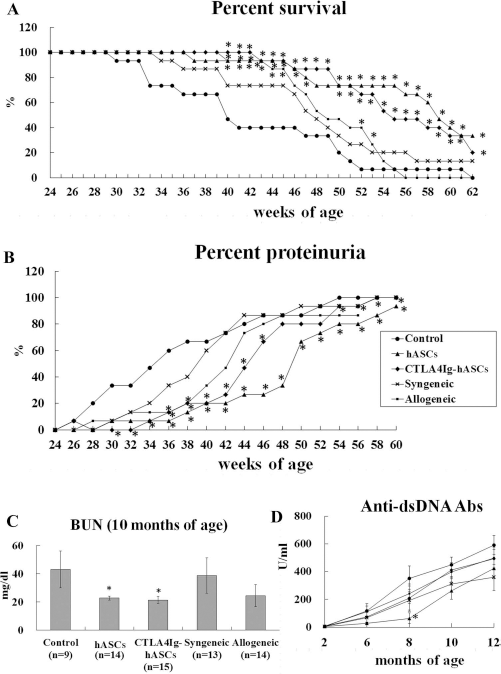
Survival and progression of severe proteinuria and serology in (NZB × NZW) F1 mice after transplantation of ASCs from various sources. Experimental groups were divided into the control group (n = 15), the hASC group (xenogeneic, hASC; n = 15), the CTLA4Ig-hASC group (CTLA4Ig overexpressing hASC; n = 15), the syngeneic group [(NZB × NZW) F1 ASC; n = 15], and the allogeneic group (BALB/c ASC; n = 15). Each mouse in the ASC groups was intravenously administered 5 × 105 ASCs/150 μl of saline every 2 weeks from 6 to 60 weeks of age. The control mice were administered 150 μl of saline and subjected to the same schedule. (A) Survival rate. (B) Incidence of severe proteinuria (percent of individuals showing ≥ 300 mg/dl of urine protein). (C) BUN levels at 10 months of age. (D) Anti-dsDNA Ab production. Data from (A) and (B) were analyzed using Kaplan–Meier survival curves and a log-rank test. The data in (C) and (D) were analyzed using the data obtained from the indicated groups, which were compared using one-way analysis of variance and a post hoc multiple comparison; *p < 0.05 relative to the control.
The average survival times of the control, hASC, CTLA4Ig-hASC, syngeneic, and allogeneic groups were 42.5 ± 2.4, > 56.1 ± 2.0, > 55.4 ± 1.6, > 48.1 ± 2.1, and 49.6 ± 1.2 weeks, respectively. Compared to the control group, the hASC and CTLA4Ig-hASC groups showed significantly longer average survival times. For statistical analyses, mice surviving after 62 weeks of age were considered as surviving up to 62 weeks of age, and groups containing these surviving mice are distinguished with “>” sign. Statistical differences were determined by oneway ANOVA testing, followed by post hoc Tukey's multiple comparison tests (p < 0.001).
Body Weights, Spleen Weights, and Spleen Weight/Body Weight Ratios
To investigate whether host immune responses occur after ASC transplantation, we measured body and spleen weights, from which we calculated spleen weight/body weight ratios. During the experiments, body weights were not significantly different among the groups (control group: 26.6 ± 1.2 g; hASC: 28.8 ± 2.5 g; CTLA4Ig-hASC: 29.1 ± 2.0 g; syngeneic: 28.5 ± 2.1 g; allogeneic: 27.1 ± 2.3 g; ANOVA, p = 0.901). Spleen weights were also similar (control group: 0.12 ± 0.02 g; hASC: 0.13 ± 0.02 g; CTLA4Ig-hASC: 0.16 ± 0.03 g; syngeneic: 0.13 ± 0.03 g; allogeneic: 0.11 ± 0.02 g; ANOVA, p = 0.818), as were the spleen weight/body weight ratios (control group: 0.00440 ± 0.00073; hASC: 0.00402 ± 0.00064; CTLA4Ig-hASC: 0.00497 ± 0.00075; syngeneic: 0.00420 ± 0.00060; allogeneic: 0.00384 ± 0.00069; ANOVA, p = 0.814, data not shown).
The data from our other study indicated that the spleen weight/body weight ratios of the control, hASC, CTLA4Ig-hASC, syngeneic, and allogeneic groups (n = 4–5 per group; 0.00468 ± 0.00098, 0.00252 ± 0.00016, 0.00265 ± 0.00012, 0.00413 ± 0.00075, and 0.00301 ± 0.00042, respectively) were not significantly different at 35 weeks of age (ANOVA, p = 0.066, data not shown).
Determination of BUN, Serum Creatinine, and dsDNA Antibody Levels
To investigate kidney function and the level of autoantibody production, we measured the production of BUN, serum creatinine, and dsDNA antibodies. Anti-dsDNA levels at 8 months were significantly lower in the hASC group than in the control group. BUN levels at 10 months were significantly lower in the hASC and CTLA4Ig-hASC groups than in the control group (Fig. 2C,D). Serum creatinine levels at 10 months did not differ significantly among the groups; serum creatinine levels of the control, hASC, CTLA4Ig-hASC, syngeneic, and allogeneic groups were 0.2 ± 0.1, 0.1 ± 0.0, 0.1 ± 0.0, 0.2 ± 0.1, and 0.1 ± 0.0 mg/dl, respectively (data not shown).
Multiple Cytokines in Sera
We detected the presence of cytokines in the serum after ASC transplantation to identify cytokines that exhibited differing patterns of expression. The xenogeneic transplantation (hASC and CTLA4Ig-hASC groups) showed significantly increased IL-10 and IL-4 levels in the serum at 8 months of age (Table 1) and showed increased expression of IL-10 and IL-4 at 2, 4, 6, and 10 months of age, although these differences did not reach statistical significance. However, syngeneic and allogeneic transplantation significantly increased GM-CSF production in the serum at 4, 6, and 10 months of age, IFN-γ production in the serum at 6 and 10 months of age (Table 1) and was associated with increased IL-10 levels at all observed time points and with increased GM-CSF and IFN-γ levels at 8 months of age.
Long-Term Monitoring of Serum Cytokine Levels (pg/ml) After Adipose Tissue-Derived Stem Cell (ASC) Transplantation

Data obtained from groups were compared using one-way analysis of variance and a post hoc multiple comparison.
Significant (p < 0.05) differences relative to control mice.
Findings of Kidney Histopathology and Immunofluorescence Staining
In this study, we observed a high correlation between histopathological findings (H&E, PAS, MT) and clinicopathological findings (urine protein), as shown in Figure 3A. The fluorescence intensities of IgG deposits were significantly lower in the hASC and CTLA4-hASC groups than in the control group (Fig. 3B,C).

Histological staining and immunofluorescence staining of kidney samples. (NZB × NZW) F1 mice were administered ASCs originating from (NZB × NZW) F1 mice (syngeneic, S), BALB/c mice (allogeneic, A), humans (hASC, xenogeneic, H), or CTLA4Ig overexpressing human ASCs (CTLA4Ig-hASCs, CT), or saline (Control, C). (A) H&E, PAS, and Masson trichrome staining of kidneys from mice without proteinuria (< 30 mg, upper) or with severe proteinuria (> 300 mg, lower). The kidneys from mice with severe proteinuria showed extensive mesangial matrix deposition and crescent formation. PAS staining showed pink material in kidney tubule sections (arrow), and Masson trichrome staining showed a blue color in the kidney sections (arrow), which indicated fibrosis. Original magnification, 400×. (B) Immunofluorescence staining of kidney sections from the experimental groups was conducted using FITC-conjugated anti-IgG or FITC-conjugated anti-C3 antibodies to compare the effects of long-term serial transplantation of ASCs. Original magnification, 200×. (C) The scores of IgG and C3 deposition in glomeruli are presented.
Examination of CM-DiI-Labeled ASCs by Confocal Microscopy
hASCs (hASC and CTLA4Ig-hASC groups) and mASCs (syngeneic and allogeneic groups) labeled with the red fluorescent CM-DiI tracker dye were predominantly found in the spleen, and many cells were evident in the kidney and liver (Fig. 4). However, there was little evidence of fluorescent-labeled cells in the lung and heart.
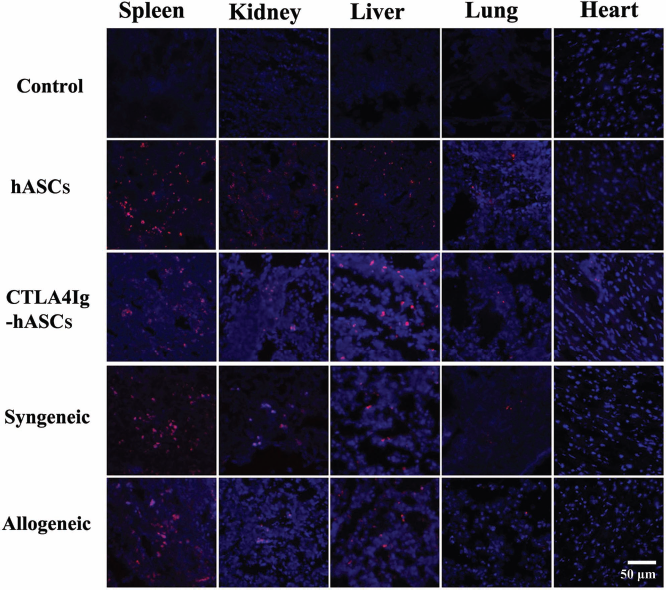
Biodistribution of transplanted ASCs from various sources. Three mice from each of the ASC groups were administered ASCs that were fluorescently labeled using conjugated red fluorochrome Cell Tracker CM-DiI and used for identification purposes in histopathological sections. The presence of CM-DiI-labeled cells was examined by laser-scanning confocal microscopy in various tissues, such as the spleen, kidney, liver, lung, and heart by counterstaining with mounting medium containing DAPI. Original magnification, 400×.
Humoral Responses to Infused Cells
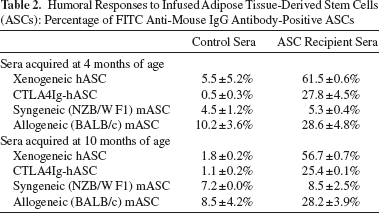
The level of IgGs against syngeneic-transplanted cells [(NZB × NZW) F1 ASCs] in sera from syngeneic ASC-transplanted mice was similar to that in the control sera from non-ASC-transplanted mice. An increased level of IgG against the allogeneic-transplanted cells (BALB/c ASCs) and a higher percentage of IgG-positive cells was observed in the allogeneic-transplanted cells when compared to the syngeneic-transplanted cells (Table 2). The level of IgGs against hASCs was higher than that against allogeneic-transplanted cells. However, the level of IgGs against CTLA4Ig-overproducing xenogeneic transplanted cells (CTLA4Ig-ASCs) was found to be the same as that observed against allogeneic-transplanted cells (Table 2).
Humoral Responses to Infused Adipose Tissue-Derived Stem Cells (ASCs): Percentage of FITC Anti-Mouse IgG Antibody-Positive ASCs
Discussion
In this study, in vivo treatment with human ASCs (hASCs or CTLA4Ig-hASCs) reduced BUN levels and proteinuria incidence, increased the survival rate, and significantly prolonged the average life span by ~13 weeks. Considering that the mean life span of (NZB × NZW) F1 mice is 35–39 weeks, an increase of 13 weeks is significant and represents a beneficial improvement.
The CTLA4Ig-ASC group showed the highest survival rate (100%) at 42 weeks of age (control group, 40%), but monitoring during the total experimental period of up to 62 weeks of age did not reveal any synergistic effects in the CTLA4Ig-ASC group compared to the ASC group. Long-term CTLA4Ig secretion might not have any additional benefits in these mice because aging is associated with a progressive decline in T-cell-mediated immune responses (29).
The analysis of all these parameters (survival rates: comparison up to 62 weeks of age; proteinuria incidence: comparison up to 60 weeks of age; BUN levels of 10 months of age; and average life span) in syngeneic and allogeneic mouse ASCs did not show any statistical differences compared to the control group. This discrepancy may relate to the different sources of ASCs used in the treatments: syngeneic and allogeneic (murine) versus xenogeneic (human). Nevertheless, when the mice were compared up to 40, 41, 42, 43, 44, 45, 52, and 53 weeks of age, the allogeneic group also showed significantly higher survival rates compared to those observed in the control group, and the mean life span of both the syngeneic and allogeneic groups was prolonged by ~6–7 weeks. In a previous study on hematopoietic stem cell (HSC) transplantation, syngeneic HSC transplantation accelerated mortality in (NZB × NZW) F1 mice (36). However, syngeneic ASC transplantation did not accelerate mortality in this study.
BM-MSCs derived from MRL/lpr mice were previously shown to be functionally impaired compared to those derived from control mice (37). In that report, BM-MSCs from MRL/lpr mice (fas mutation) demonstrated impairment of osteogenic and adipogenic differentiation, and we found that ASCs from these mice showed similar defects (unpublished data). Compared to ASCs from BALB/c mice, ASCs from (NZB × NZW) F1 mice showed similar adipogenic and decreased osteogenic differentiation abilities in this study. Thus, the integrity and the extent of defects in ASCs from SLE mouse models or SLE patients might be determined by individual pathogenic risk factors such as critical genetic defects. The therapeutic effects of hASC and CTLA4Ig-hASC transplantation were not significantly different, but different humoral responses were observed. The strongest humoral immune response was induced by xenogeneic (human) ASC transplantations followed by xenogeneic, allogeneic, CTLA4Ig-overexpressing xenogeneic, and syngeneic administrations. This result may reveal that CTLA4Ig decreased humoral responses in in vivo stem cell therapy. Human ASCs showed a better therapeutic effect on murine SLE than did mouse ASCs, indicating that these antibodies induced by the humoral response may be binding antibodies and not neutralizing antibodies.
Compared to xenogeneic transplantations, the syngeneic and allogeneic transplantations evoked different cytokine patterns in the SLE mouse model. This finding indicates that syngeneic, allogeneic, and xenogeneic ASCs proved to be efficient in this model through independent cytokine mechanisms. Xenogeneic transplantation significantly increased IL-10 and IL-4 production in serum, and IL-10 plays an important role in xenogeneic immunosuppression. However, syngeneic and allogeneic ASCs induced the secretion of IFN-γ and GM-CSF. IFN-γ and the concomitant presence of TNF-α and IL-1β can induce immunosuppressive functions of MSCs (34). Ren et al. reported that immunosuppression by MSCs through the concerted action of chemokines and nitric oxide requires proinflammatory cytokines (31). IFN-γ-dependent inhibition of B-cell activation by allogeneic BM-MSCs was also reported by Schena et al. (33). According to their study, the inhibitory effects of BM-MSCs on antigen-dependent proliferation and differentiation of follicular and marginal zone B cells to plasma cells was dependent on IFN-γ and was mediated by the programmed death 1 (PD-1)/PD ligand pathway. IFN-γ also stimulates the expression of indoleamine 2,3-dioxygenase in BM-MSCs, which inhibits the proliferation of activated T or NK cells (20).
The role of IL-10 in the pathogenesis of lupus remains controversial. Continuous administration of IL-10 to SLE mice has been shown to accelerate the development of autoimmunity (19). However, in another study, SLE mice deficient in IL-10 showed disease exacerbation, and SLE mice administered IL-10 at 6 weeks of age showed decreased Th1-type autoantibodies and amelioration of symptoms (43). Taken together, these findings indicate that IL-10 may serve more than one purpose in disease pathogenesis. However, in general, the administration of IL-10 can be beneficial in treating excessive inflammatory conditions of either type Th1 or Th2 and can prevent tissue destruction resulting from Th1 polarization in various autoimmune diseases (25).
According to results from a study by Gangi et al. on autoimmune thyroiditis, GM-CSF facilitates the expansion of IL-10-producing regulatory T cells and thus induces a shift from Th1-type to Th2-type immune response against autoantigens (18). In this study, IL-10 levels in the sera from both mouse ASC groups showed patterns of increased expression, although the levels were not statistically significant.
In our previous studies on murine experimental autoimmune thyroiditis, all of the syngeneic, allogeneic, and xenogeneic (hASC or CTLA4Ig-hASC) transplantations showed the same cytokine expression patterns, including the reduction of inflammatory immune responses and an improvement in the Th1/Th2 balance (12,14). These findings suggest that during in vivo ASC treatment, the host's cytokine expression pattern varies depending on complex interactions relating to the source of the ASCs, the immunopathophysiology of the disease, and the microenvironment of the host.
In conclusion, the data presented in this study showed that long-term serial transplantation of hASCs (or CTLA4Ig-hASCs) has beneficial effects for SLE treatment, without the development of adverse effects. The therapeutic effects of hASC and CTLA4Ig-hASC transplantation were similar, although different humoral immune responses were observed. Long-term multiple transplantations of CTLA4Ig-hASCs did not show any synergistic therapeutic effects, although they were associated with decreased humoral immune responses to infused xenogeneic ASCs, which was significant when compared to that observed in hASC transplant mice. Human (xenogeneic) and mouse ASCs (syngeneic and allogeneic) have a similar but not identical immunophenotype and display differential patterns of cytokine expression in the (NZB × NZW) F1 mouse model of SLE.
Footnotes
Acknowledgments
This research was supported by a grant to E. W. Choi from the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education, Science and Technology (basic research, NRF-2009-0077165). Further support was provided by a grant from the Samsung Biomedical Research Institute (SMX1131211/SMX1140141) to E. W. Choi. The authors thank the staff of the Laboratory Animal Research Center at the Samsung Biomedical Research Institute for providing technical support and assistance. The authors declare no conflicts of interest.
