Abstract
Cryopreservation of bone marrow (BM), mobilized peripheral blood (mPB), and cord blood (CB) hematopoietic stem/progenitor cells (HSPCs) is a routine procedure before transplantation. The most commonly used cryoprotectant for HSPCs is dimethyl sulfoxide (DMSO). The objective of this study was to evaluate the influence of DMSO on surface receptor expression and chemotactic activities of HSPCs. We found that 10 min of incubation of human mononuclear cells (MNCs) with 10% DMSO significantly increases the percentage of CXCR4+, CD38+, and CD34+ cells, resulting in an increase of CD34+, CD34+CXCR4+, and CD34+CXCR4+CD38– subpopulations. Furthermore, DMSO significantly increased chemotactic responsiveness of MNCs and CXCR4+ human hematopoietic Jurkat cell line to a stromal cell-derived factor-1 (SDF-1) gradient. Furthermore, we demonstrated enhanced chemotaxis of human clonogenic progenitor cells to an SDF-1 gradient, which suggests that DMSO directly enhances the chemotactic responsiveness of early human progenitors. DMSO preincubation also caused lower internalization of the CXCR4 receptor. In parallel experiments, we found that approximately 30% more of DMSO-preincubated human CD45+ and CD45+CD34+ cells homed to the mouse BM 24 h after transplantation in comparison to control cells. Finally, we demonstrated considerably higher (25 days) survival of mice transplanted with DMSO-exposed MNCs than those transplanted with the control cells. We show in this study an unexpected beneficial influence of DMSO on HSPC homing and suggest that a short priming with DMSO before transplantation could be considered a new strategy to enhance cell homing and engraftment.
Keywords
Introduction
Intensive radio- and/or chemotherapy followed by hematopoietic stem/progenitor cell (HSPC) transplantation is used for the treatment of certain hemato-oncologic and nonproliferative diseases. The role of transplanted HSPCs is to provide a rapid and sustained hematopoietic reconstitution after myeloablative and nonmyeloablative conditioning regimens (23). The importance of a rapid reconstitution arises from the risk of bacterial, viral, and mycotic infections and a need for repetitive erythrocyte and platelet infusions. For patients for whom obtaining a high CD34+ cell number is impossible, it would be highly beneficial if the HSPC homing potential could be increased.
The process in which HSPCs home to the bone marrow (BM) is a multistep process, regulated by the chemokine stromal cell-derived factor-1 (SDF-1) and mediated through its receptor CXCR4 (12,14). CXCR4 is a seven-transmembrane receptor coupled to G proteins, and it is expressed on a variety of hematopoietic cells (3). Different strategies of increasing homing and engraftment of HSPCs have been proposed. Peled et al. showed that CD34+ cells had increased CXCR4 expression after exposure to interleukin-6 (IL-6) and stem cell factor (SCF), which has been associated with increased homing and repopulation in vivo, and suggested that an upregulation of this receptor expression is a way to improve engraftment of repopulating stem cells in a clinical transplantation setting (24). Similarly, Kahn et al. showed that CXCR4 overexpression on CD34+ progenitors increased cell proliferation and migration in nonobese diabetic/severe combined immunodeficient (NOD/SCID) mice (13). In vitro studies demonstrated that complement C3a increases chemotaxis of BM hematopoietic cells to an SDF-1 gradient (11). Xia et al. demonstrated that active fucosylation of human cord blood (CB) mononuclear cells (MNCs) increases the engraftment of these cells within the BM of immunodeficient mice (28). It was discovered that BM HSPCs covered with platelet-derived microparticles (PMPs) engraft significantly faster than those not covered (12).
Stem cells collected before a transplantation procedure (for autologous and, rarely, for allogeneic transplantation) are typically stored for prolonged periods of time. The cells should be frozen in the presence of a cryoprotectant and stored in a vapor phase of liquid nitrogen to enable cells to be stored for extended periods of time without loss of stem/progenitor cell viability and functionality,
The most clinically relevant cryoprotectant is dimethyl sulfoxide (DMSO) (4,25), a colligative cryoprotectant that reduces the formation of water crystals at any given temperature and decreases the degree of cellular dehydration and the level of osmotic pressure (6,27). To date, there has been only one report describing an ex vivo effect of DMSO exposure on hematopoietic stem cells and BM stromal cells. Rowley and Anderson tested the effect of DMSO on hematopoietic cells without cryopreservation in vitro and showed that this cryoprotectant, when used in the concentrations commonly used for HSPC freezing, does not decrease clonogenicity of the cells (26).
In the current study, we evaluated the influence of preincubation of HSPCs with 10% DMSO on the expression of CD34, CD38, and CXCR4 antigens, as well as the viability, clonogenicity, chemotactic activity, homing, and survival of the cells.
Materials and Methods
Isolation of Human CB-MNCs and DMSO Preincubation
Umbilical CB samples were obtained according to the procedures approved by the Institutional Bioethics Committee of Jagiellonian University (Cracow, Poland). Light-density MNCs were separated on a lymphocyte separation medium (PAA Laboratories GmbH, Pasching, Austria) by density gradient centrifugation at 400 × g for 30 min. For most studies performed, 10 min of preincubation with 10% DMSO (Merck, Darmstadt, Germany) was used. For viability and clonogenicity experiments, 5, 10, and 30 min of preincubation were used. In chemotactic experiments performed on Jurkat cells, 1%, 5%, 10%, or 20% concentrations of DMSO were used.
Cell Lines
The Jurkat cell line was purchased from the American Type Culture Collection (ATCC; Manassas, VA, USA) and maintained in RPMI-1640 medium supplemented with 10% fetal bovine serum (FBS; PAA Laboratories GmbH), 50 U/ml penicillin (Gibco, Life Technologies, Grand Island, NY, USA), 50 μg/ml streptomycin (Gibco, Life Technologies), 2 mM L-glutamine (Lonza, CELLLAB, Warsaw, Poland) at 37°C in a fully humidified atmosphere supplemented with 5% CO2.
Flow Cytometry
Human CB-derived MNCs (CB-MNCs; 1 × 106) were stained with 20 μl of anti-CD34, anti-CD38, or anti-CXCR4 monoclonal antibodies (1:4) (all from BD Immunocytometry Systems, San Jose, CA, USA) or additionally with 20 μl of 7-AAD (BD), incubated in the dark, in a final volume of 100 μl, for 30 min at 4°C, and then washed. Finally, samples were collected using a FACSCanto cytometer and analyzed with FACSDiva software (BD Biosciences, San Jose, CA, USA). For some experiments, cells preincubated with DMSO were kept at 4°C for 1, 2, or 24 h and then stained to analyze the duration of DMSO-mediated effects.
Intracellular Staining
A pellet of 1 × 106 phosphate-buffered saline (PBS)-washed cells was resuspended in 250 μl of BD Cytofix/Cytoperm solution (BD Pharmingen, Franklin Lakes, NJ, USA), incubated for 20 min at 4°C, and then washed twice with BD Perm/Wash™ buffer (BD Pharmingen). Fixed and permeabilized cells were stained with anti-CD34, anti-CD38, or anti-CXCR4 monoclonal antibodies. Samples were then collected using the FACSCanto cytometer and analyzed with the FACSDiva software.
Viability
Cell viability was determined with the trypan blue (TB) exclusion method (Sigma-Aldrich, Seelze, Germany) and 7-AAD staining. For the TB method, cells treated with DMSO were mixed with TB (4:1) and immediately live and dead cells were counted under a light microscope. For 7-AAD staining, PBS-washed cells were resuspended in 100 μl of PBS with 2% FBS, and 5 μl of 7-AAD was added. Cells were incubated for 10 min at 4°C. After washing unbound antibody, cells were collected with the use of a FACSCanto cytometer and analyzed with the FACSDiva software.
Clonogenic Assay
For evaluation of the DMSO effect on clonogenicity of CB-MNCs, 1 × 104 cells from each experimental variant were seeded in methylcellulose into wells of a 24-well plate in duplicate, and colony forming unit granulocyte/macrophage (CFU-GM) and burst-forming unit-erythroid (BFU-E) colonies were counted on day 14. In another experiment, all MNCs exposed to DMSO that were collected after the chemotaxis assay were seeded in 2 ml of complete commercial MethoCult medium (STEMCELL Technologies, Vancouver, Canada) containing human growth factors [SCF, granulocyte/macrophage colony-stimulating factor (GM-CSF), IL-3, and erythropoietin (EPO)] that support growth of BFU-E and CFU-GM colonies. Cells were incubated at 37°C in a fully humidified atmosphere, supplemented with 5% CO2. CFU-GM and BFU-E colonies were identified and counted using an inverted microscope after 14 days of culture, based on the typical morphological appearance reported elsewhere (7,20).
Chemotaxis Assay
CB-MNCs and Jurkat cells were exposed to DMSO, and a chemotaxis assay was performed as described previously (15). Briefly, 650 μl/point of prewarmed 0.5% bovine serum albumin (BSA) containing RPMI-1640 medium, with 100 ng/ml SDF-1β (PeproTech Inc., Rocky Hill, NJ, USA) for CB-MNCs or various concentrations of SDF-1β (1 to 2,000 ng/ml) for Jurkat cells, was added to the lower chamber of the Costar Transwell 24-well plate (Corning Inc., Corning, NY, USA). Cell suspensions were distributed to the upper chambers and incubated at 37°C, in 95% humidity with 5% CO2 for 3 h. Cells from the lower chambers were collected, and cell number was scored by FACSCalibur (BD Immunocytometry Systems). Results are demonstrated as a percentage of the cells in reference to the number of the cells in the input wells. In some experiments, cells from the lower chambers were seeded in methylcellulose and stimulated to grow CFU-GM and BFU-E colonies.
Internalization of the CXCR4 Receptor
For the determination of chemokine-induced receptor internalization, control cells and cells exposed to DMSO were incubated at 37°C in culture medium for 30 min in the presence or absence of SDF-1β (500 ng/ml) (8). Before analysis, cell-bound chemokine was removed by an ice-cold acidic glycine wash [50 mM glycine (BioShop Canada Inc., Burlington, Ontario, Canada)/HCl, pH 3, 100 mM NaCl (POCH SA, Gliwice, Poland)] for 90 s (2). Cells were labeled with allophycocyanin (APC)–anti-CXCR4 monoclonal antibody (1:4) (clone No. 12.G5; BD Pharmingen). The expression of CXCR4 was measured by flow cytometry. The receptor internalization was evaluated by comparison of the mean channel fluorescence values.
BM Transplantation Into Sublethally Irradiated NOD/SCID Mice
Mice were obtained from Jackson Laboratory (Bar Harbor, ME, USA). For the transplantation experiments, two groups of 8-week-old male mice were sublethally irradiated with a double dose of 150 cGy γ radiation at 4-h intervals (total dose of 300 cGy). After 24 h, the irradiated mice were transplanted by retro-orbital plexus injection with 0.75 × 106 –1 × 106 CB-MNCs containing 4.5 × 104–8.4 × 104 CD34+ cells. The first group was used to evaluate short-term engraftment and the second group to evaluate survival.
Twenty-Four-Hour Homing Evaluation
For the homing evaluation, transplanted mice were sacrificed 24 h after the transplantation, and BM was aspirated. The number of human CD45+ and CD45+CD34+ cells within 1 × 106 of murine BM cells was assessed via cytofluorimetric evaluation (n = 6 per group).
Mouse Survival
The transplanted mice were left alive and assessed for survival over a 25-day period after transplantation (n = 8 per group).
Statistical Analysis
Statistical analysis was performed with Microsoft Office Excel 2003 (Microsoft, Redmond, WA, USA), with log-rank (Mantel–Cox) chi-square tests and Gehan–Breslow–Wilcoxon chi-square tests. Variables were analyzed with a one-tail Student's t-test for unpaired samples. Results were deemed significant if a value of p ≤ 0.05 was attained. In most experiments, mean ± standard deviation (SD) is shown. N indicates the number of replicates for all in vitro experiments, and n is the number of animals in both in vivo experiments.
Results
Influence of DMSO Preincubation on CB-MNC Surface Marker Expression
Fluorescence-activated cell sorting (FACS) analysis showed that preincubation of MNCs with 10% DMSO resulted in an increase in CXCR4+ cells from 50% to 81% (Fig. 1A). Similarly, the percentage of CD34+ cells nearly doubled from 0.43% to 0.88%, and the percentage of CD38+ cells also increased (from 38% to 90%). The observed increase was statistically significant for all tested receptors with p < 0.05. Figure 1B shows the percentage of overall receptor expression (both surface and intracellular) in tested cells. Interestingly, the level of the increases after DMSO preincubation was only as high as the overall (both surface and intracellular) percentages of CXCR4+, CD34+, and CD38+ cells. Figure 1C demonstrates changes in a dot plot image of control and DMSO-exposed cells. It was shown that contamination by remaining granulocytes disappeared, and the amount of debris increased after DMSO pretreatment. Furthermore, Figure 1D reveals that DMSO causes a statistically significant (199%) increase of median fluorescence intensity (MFI) of the entire CB-MNC population stained with anti-CXCR4 antibody, whereas the effect was not seen in the isotype-stained cells. Additionally, Figure 1F presents the overlay of untreated isotype- and CXCR4-stained, and DMSO-preincubated isotype- and CXCR4-stained CB-MNCs, which shows no or negligible DMSO influence on isotype binding. It also shows some increase in MFI of CXCR4+ population besides the increase of percentage of positive cells shown in Figure 1A. The duration of the observed effect was checked for the CXCR4 receptor only, as the most important for cell homing, and shown to last at least 20 h (Fig. 1E).

Influence of DMSO preincubation on the expression of CXCR4, CD34, and CD38 receptors on CB-MNCs. CB-MNCs (5 × 106) were incubated in cold culture medium alone (RPMI-1640 medium supplemented with 10% FBS) or cold culture medium containing 10% DMSO for 10 min, washed out, and split into three aliquots. The first aliquot of both the control and the DMSO-treated cells was stained with monoclonal anti-CXCR4, anti-CD34, and anti-CD38 antibodies to check surface expression of these receptors. Isotype- and CXCR4-stained control and treated cells were overlayed on one histogram. A second aliquot of the cells was first fixed, permeabilized, and then stained with the same antibodies as the first group to check overall (both surface and intracellular) surface expression of these receptors. The last aliquot of the cells was kept in RPMI-1640 with 10% FBS at 4°C after DMSO incubation and subsequently washed for 1, 2, and 24 h; it was then stained in the same manner as the other cells. Finally, cells were submitted to the cytofluorimetric analysis. Values are means ± SD. Number of analyzed samples for each experiment is stated above. (A) Surface expression level of CXCR4, CD34, and CD38 receptors (n = 6); *p < 0.05, significantly different from the control, by Student's t-test. (B) Overall expression level of CXCR4, CD34, and CD38 receptors (n = 2–4). (C) A dot plot view of untreated and DMSO-preincubated CB-MNCs; representative results out of n = 6 are shown. (D) Analysis of DMSO influence on MFI of the CXCR4 receptor (n = 6); *p < 0.05, significantly different from the control by Student's t-test. (E) Duration of the increased percentage of CXCR4+ cells in the 24-h time period (n = 3); *p < 0.05, significantly different from the control by Student's t-test. (F) Histogram overlay analysis of CXCR4 expression prior and after DMSO preincubation; representative results out of n = 6 are shown.
Influence of the DMSO Incubation on Viability and Clonogenicity of CB-MNCs
In this experiment, the evaluated cells were divided into four groups: untreated and treated with 10% DMSO for 5, 10, and 30 min. As shown in Figure 2A, incubation with 10% DMSO for 5, 10, or 30 min caused an approximately 10% considerable decrease in viability of the cells, as determined by a TB exclusion assay. Figure 2B presents the evaluation of viability of the cells using 7-AAD staining. An increase in the level of 7-AAD+ cells after exposure to DMSO correlated with the results obtained from the TB assay.

Influence of DMSO preincubation on viability and clonogenicity of CB-MNCs. CB-MNSs (1 × 106, cooled at 4°C) were incubated in cold culture medium alone (RPMI-1640 medium supplemented with 10% FBS) or cold culture medium containing 10% DMSO for 5, 10, and 30 min, washed by adding 10× volume of culture medium, centrifuged, and split into two aliquots. The first aliquot was used to test viability of the cells in TB exclusion assay, and the second aliquot was used to check clonogenicity of the cells. Additionally, we assessed viability of the control and preincubated cells at 4°C with 10% DMSO for 10 min, using 7-AAD staining and cytofluorimetric analysis. Values are means ± SD. The number of analyzed samples in each experiment is stated above. (A) Viability of DMSO-preincubated CB-MNCs assessed with TB exclusion assay (n = 3); *p < 0.05, significantly different from the control by Student's t-test. (B) Viability of preincubated CB-MNCs assessed with 7-AAD staining; representative results out of n = 4 are shown. (C) Clonogenicity of preincubated CB-MNCs (n = 5).
Figure 2C demonstrates no influence of DMSO pretreatment on BFU-E colony number and a slight, but not statistically significant, decrease in CFU-GM colony number.
Upregulation of SDF-1-Induced Cell Motility After DMSO Preincubation
Figure 3A illustrates that CB-MNCs preincubated for 10 min with 10% DMSO had four times higher chemotactic activity to SDF-1 gradient than control cells (p < 0.05). Specificity of the DMSO influence on CB-MNCs was confirmed with the Jurkat cell line, using different SDF-1 and DMSO concentrations. We observed that the level of the increase in chemotactic activity of Jurkat cells was dependent on the DMSO concentration (Fig. 3B). The experiment also revealed that DMSO increased chemotactic responsiveness of Jurkat cells to an SDF-1 gradient in a wide range of concentrations (100–2,000 ng/ml), and the level of the increase was approximately the same throughout the whole range of concentrations (Fig. 3C).
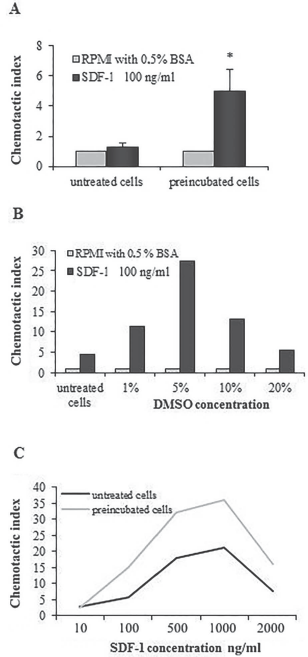
Influence of DMSO preincubation on the migratory abilities of CB-MNCs and Jurkat cells. Control and DMSO-preincubated CB-MNCs were tested for chemotactic activity. To test specificity of the observed effect, two additional experiments were performed. First, control and 1%, 5%, 10%, and 20% DMSO-preincubated Jurkat cells were tested for chemotactic activity. In the second experiment, control and 10% DMSO-preincubated Jurkat cells were submitted to chemotaxis assay to 10, 100, 500, 1,000 and 2,000 ng/ml of SDF-1β. Finally, cells from the lower chambers were collected, and cell number was scored by FACSCalibur. (A) DMSO influence on the migratory abilities of CB-MNCs. Values are means ± SD of n = 4 samples; *p < 0.05, significantly different from the control. (B) Influence of DMSO concentration (1%, 5%, 10%, and 20%) on the chemotactic activity of Jurkat cells. (C) Chemoattractant gradient (10–2,000 ng/ml) influence on the chemotactic activity of Jurkat cells.
Moreover, DMSO preincubation nearly doubled the percentage of live CD34+CXCR4+ progenitors and their subpopulations: CD34+CXCR4+CD38– and CD34+ CXCR4+CD38+ (Fig. 4A). In the next part of our study, we showed that a higher number of BFU-E and CFU-GM progenitors migrated to an SDF-1 gradient after preincubation with DMSO, in comparison to untreated cells (Fig. 4B).

Influence of DMSO preincubation on CXCR4 expression in CD34+ subsets and migratory abilities of clonogenic progenitors. Control and DMSO-preincubated CB-MNCs were divided into two aliquots. The first aliquot of the cells was submitted to multicolor cytofluorimetric analysis against CXCR4, CD34, and CD38 receptors and 7-AAD. Gating system from P1 (to exclude debris) through P8 (CD34+CD38–) or P10 (CD34+CD38+) to final dot plot, showing alive CXCR4+ cells in the lower right square, was applied to check the number of alive CD34+CXCR4+ cells and their CD38+ and CD38– subpopulations. The second aliquot of the cells was submitted to chemotactic assay to check number of migrated BFU-E and CFU-GM progenitors. Values are means ± SD. (A) Upper panel presents number of CD34+CXCR4+, CD34+CD38+CXCR4+, and CD34+CD38-CXCR4+ cells among 1 × 105 CB-MNCs (n = 5); *p < 0.05, significantly different from the control by Student's t-test; ▴ indicates a trend (p value between 0.05 and 0.1) by Student's t-test. Lower panel presents a representative example of gating strategy. (B) Migratory abilities of BFU-E and CFU-GM control and DMSO-preincubated progenitors (n = 2).
DMSO Inhibits Internalization of CXCR4
The chemotactic response to chemokines is dependent on the level of the receptor availability. Because DMSO influences cell membranes, we investigated the possible influence of DMSO on CXCR4 internalization. Surprisingly, the level of the CXCR4 receptor internalization after SDF-1 stimulation was lower in the cells exposed to DMSO than in the control cells (Fig. 5).

Influence of DMSO preincubation on the internalization of CXCR4 receptor on CB-MNCs. Control and DMSO-preincubated CB-MNCs were divided into two aliquots. The first aliquot was stained with anti-CXCR4 antibody to check the initial level of the receptor of the control and preincubated cells. The second aliquot was incubated at 37°C in culture medium for 30 min in the presence of SDF-1β (500 ng/ml) and level of the receptor was evaluated. The experiment was repeated three times (n = 3), and representative results are shown.
Influence of DMSO Preincubation on Short-Term Engraftment and Mouse Survival
Figure 6A demonstrates approximately 30% more CD45+ and CD45+CD34+ human cells per 1 × 106 of mouse BM cells in animals transplanted with DMSO-preincubated CB-MNCs than animals transplanted with control cells. Figure 6B illustrates a higher (25 days) survival rate in a group of mice transplanted with DMSO-preincubated CB-MNCs than in animals transplanted with control cells.

Influence of DMSO preincubation on CB-MNC homing abilities and mouse survival. Male mice, sublethally irradiated with a double dose of 200 cGy γ irradiation, were transplanted the next day with control and DMSO-preincubated 0.75 × 106–1 × 106 CB-MNCs, containing 4.5 × 104–8.4 × 104 CD34+ cells. Six mice from the control and treatment groups were sacrificed 24 h later, and the number of human CD45+ and CD45+CD34+ cells/1 × 106 of mice BM cells was checked. Eight other mice in each group were left alive, and their survival was evaluated. (A) Homing of the control and DMSO-preincubated CB-MNCs CD45+ subsets (n = 6); *p < 0.05, significantly different from control by Student's t-test. (B) Survival of the mice transplanted with the control and DMSO-preincubated CB-MNCs (n = 8 in each group).
Discussion
DMSO is a widely used cryoprotectant, but its influence on cell biology has not been fully elucidated. In this study, we tested the influence of DMSO pretreatment on the expression of chosen surface antigens on CB-MNCs.
We observed a considerable increase in the percentage of CXCR4+, CD38+, and CD34+ CB cells (65–110%) as a result of 10 min of exposure to 10% DMSO. We also demonstrated that CB-MNCs contain a considerable percentage of cells carrying a cytoplasmic pole of the receptors. The percentage after DMSO exposure reflected the overall, both surface and cytoplasmic, number of cells expressing the studied receptors. The fact that the level of the increase reached, but did not exceed, the level of the overall expression suggests that DMSO can increase availability of the cytoplasmic pool of the receptor to the antibody. The increased percentage of CXCR4+, CD38+, and CD34+ cells was not due to cell death, as the gating strategy used during analysis excluded dead cells. The combination of 7-AAD and dot plot analysis showed that an approximately 10% increase in dead cells after exposure to DMSO comes mostly from remaining granulocytic contamination. The main population of CB-MNCs, including lymphocytes and monocytes, remains of the same size and granularity. The increase was also not due to increased binding of the exposed cells to antibodies. Additionally, a greater increase in MFI of CXCR4+ DMSO-preincubated cells than of untreated CXCR4+ cells might suggest an increase in surface receptor availability. On the basis of this result, we speculate that DMSO could mobilize the cytoplasmic, submembrane reservoir of the receptors to the surface of the cells and/or, alternatively, may unmask receptors, which were not accessible for the antibody.
A 10% DMSO exposure caused a significant (approximately 10%) viability decrease of CB-MNCs in both applied tests. Dot plot analysis suggests that most of the dead cells came from the remaining granulocytic contamination. The DMSO exposure had no influence on BFU-E and reduced the CFU-GM colony number; the reduction was not significant. This confirms Rowley and Anderson's observation that DMSO was not toxic to myeloid and erythroid progenitors (26).
The CXCR4 receptor has been shown to be expressed on human lymphocytes, BM, CB, and mobilized peripheral blood (mPB) CD34+ progenitors (20,29). It was demonstrated that 20% to 25% of CB CD34+ cells and no more than 40% of CD34+ cells, regardless of their source, migrated toward an SDF-1 gradient in vitro (1,5,24,29). Moreover, the percentage of migrating cells correlated with the percentage of CXCR4+ cells (1). As the CXCR4–SDF-1 axis is involved in the homing of transplanted cells, increasing the CXCR4 expression on hematopoietic progenitors may be a way to accelerate this process.
In our studies, the increased percentage of CXCR4+ cells appears to correlate with an increased number of CB-MNCs migrating to an SDF-1 gradient. Specificity of the observed increased migration was confirmed with Jurkat cells, demonstrating a typical pattern of SDF-1 dose and chemotactic response dependency described for hematopoietic cells (1).
To check if the observed effect also pertains to CB-HSPCs, we performed multicolor cytofluorimetric analysis, which revealed an increase in the number of CXCR4+ CB-HSPCs: CD34+CXCR4+ cells and both their CD34+CXCR4+CD38– and CD34+CXCR4+CD38+ subpopulations, responsible for long- and short-term hematopoietic recovery (10). Clonogenic CB cells exposed to DMSO migrated to an SDF-1 gradient, similar to CB-MNCs, in considerably increased numbers. Although the increase was much lower for clonogenic progenitors than for CB-MNCs, the range of the increase reflected the increase in the number of CD34+CXCR4+CD38+ cells.
We showed that most of the CB-MNCs harbor the intracellular poles of the CXCR4 receptors. Kollet et al. demonstrated that the CD34+CXCR4– subpopulation harbors low intracellular CXCR4 and that its surface expression appears 24 h after cytokine stimulation and allows homing to BM (16). Our observations were in agreement with this report and expand upon the findings therein, indicating that some CD34+CD38+ and CD34+CD38– cells probably also harbor intracellular CXCR4, as multicolor analysis showed an increase in the number of living CD34+CXCR4+, CD34+CXCR4+38+, and CD34+CXCR4+CD38– cells after DMSO exposure. Lower CXCR4 internalization in addition to an increased percentage of CXCR4+ CB-MNCs may work together to increase chemotactic activity of cells exposed to DMSO. Additionally, we speculate that the effect might also lead to an increase in survival of the cells, as CXCR4–SDF-1 interactions, in synergy with other early acting cytokines, enhance proliferation/survival of human CD34+ cells (19).
Different strategies have been suggested to enhance homing and engraftment of transplanted HSPCs (11,12, 20,28), and two studies aimed to increase CXCR4 expression (13,24). Kahn et al. suggested lentiviral CXCR4 transduction (13). Peled et al. demonstrated a considerable increase in CXCR4 expression after 48 h of exposure of CD34+ mPB cells to SCF (24). The 48 h needed for the increase suggests stimulation of de novo synthesis of the receptor. Conditions used in our experiments were too short for inducing de novo synthesis; thus, other mechanisms had to be involved. It seems more probable that the observed increase could be explained by the well-documented influence of DMSO on the cell membrane, such as membrane thinning and/or increased fluidity of the membrane's hydrophobic core with transient water pores (9,22). DMSO was already demonstrated to increase membrane fluidity, which influenced the expression of the membrane receptors on Friend virus- and Moloney virus-infected hematopoietic and lymphoid cells (18). The latter mechanism would assume some cytoplasmic level/submembrane reservoir of the receptor, which we (CXCR4, CD34, and CD38) and others (CXCR4) have shown (17).
The described effect persists for at least 24 h, which should allow the cells to home to BM in increased numbers, as intravenously infused human HSPCs have been demonstrated to find their way to BM niches within a few hours (16). In fact, we demonstrated that DMSO preincubation can considerably increase homing of CB-HSPCs present in MNC populations and survival of transplanted animals.
In conclusion, it seems that DMSO acting through different unspecific changes, regarding receptor expression and inhibition of CXCR4 internalization, increases the chemotactic response of hematopoietic cells to an SDF-1 gradient in vitro and increases homing and survival of CB-HSPCs present in MNC populations in a more complicated animal model. We therefore conclude that a short incubation with 10% DMSO could be considered a way of increasing homing and engraftment efficacy of transplanted cells, and may be a viable alternative to already suggested strategies (11–13,20,21,24,28). In addition, we believe that our results may have wider implications, as it is probable that DMSO influences expression of many antigens and many types of cells.
Footnotes
Acknowledgments
This study was supported by a research grant from Jagiellonian University Medical College. The authors declare no conflicts of interest.
