Abstract
Pericapsular fibrotic overgrowth (PFO) is a problem that thwarts full implementation of cellular replacement therapies involving encapsulation in an immunoprotective material, such as for the treatment of diabetes. Mesenchymal stem cells (MSCs) have inherent anti-inflammatory properties. We postulated that coencapsulation of MSCs with the target cells would reduce PFO. A hepatoinsulinoma cell line (HUH7) was used to model human target cells and was coencapsulated with either human or mouse MSCs at different ratios in alginate microcapsules. Viability of encapsulated cells was assessed in vitro and xenografted either intraperitoneally or subcutaneously into C57BL/6 mice. Graft retrieval was performed at 3 weeks posttransplantation and assessed for PFO. Coencapsulation of human MSCs (hMSCs) or mouse MSCs (mMSCs) with HUH7 at different ratios did not alter cell viability in vitro. In vivo data from intraperitoneal infusions showed that PFO for HUH7 cells coencapsulated with hMSCs and mMSCs in a ratio of 1:1 was significantly reduced by ~30% and ~35%, respectively, compared to HUH7 encapsulated alone. PFO for HUH7 cells was reduced by ~51% when the ratio of mMSC/HUH7 was increased to 2:1. Implanting the microcapsules subcutaneously rather than intraperitoneally substantially reduced PFO in all treatment groups, which was most significant in the mMSC/HUH7 2:1 group with a ~53% reduction in PFO compared with HUH7 alone. Despite the reduced PFO reaction to the individual microcapsules implanted subcutaneously, all microcapsule treatment groups were contained in a vascularized fibrotic pouch at 3 weeks. The presence of MSCs in microcapsules retrieved from these fibrotic pouches improved graft survival with significantly higher cell viabilities of 83.1 ± 0.6% and 79.1 ± 0.8% seen with microcapsules containing mMSC/HUH7 at 2:1 and 1:1 ratios, respectively, compared to HUH7 alone (51.5 ± 0.7%) transplanted subcutaneously. This study showed that coencapsulation of MSCs with target cells has a dose-dependent effect on reducing PFO and improving graft survival when implanted either intraperitoneally or subcutaneously in a stringent xenotransplantation setting.
Introduction
Cellular replacement is driving treatments for metabolic disease states using living cells to produce specific therapeutic molecules. One example is the use of insulin-producing cells in the treatment of type 1 diabetes. The currently available cell treatment for type 1 diabetes is the surgical transplantation of donor pancreatic islets, but this is associated with serious problems from toxic immunosuppressive drugs coadministered to prevent graft rejection (37). To avoid the need for such drugs, cells can be placed in an immune isolation device to protect them from the host's immune system. One such strategy is the use of alginate to create a selectively permeable, protective microcapsule to contain the target therapeutic cells. Pancreatic islets encapsulated in alginate microcapsules have been shown to survive well and normalize blood sugar levels for prolonged periods in varied diabetic animal models of a syngeneic, allogeneic, and xenogeneic transplantation setting (12,36,40). However, graft survival was limited and varied significantly, ranging from months to a few years. A major factor responsible for the poor graft survival and which is limiting any successful clinical outcome with encapsulation technologies is the growth of fibrotic tissue around the implanted microcapsules, referred to as pericapsular fibrotic overgrowth (PFO).
PFO is a problem reported in humans (20,44), the baboon (47), and in rodents (15,50) and is thought to be either a foreign body reaction against the alginate microcapsules and/or an inflammatory response to the release of small molecular weight antigens shed by the grafted encapsulated cells (8,9,18). A recent study has reported PFO in response to empty alginate microcapsules that was associated with the purity of the commercially sourced alginate due to the presence of pathogen-associated molecular patterns, despite it being labeled ultrapure (32). Clearly, the species of animal model used in studies is an important determinant of the outcome (35), and indeed we have previously demonstrated that the PFO to transplanted alginate microcapsules is species specific despite using the same batch of commercially sourced ultrapure alginate (47). Especially in mouse models, it has been shown that severity of PFO is strain specific with C57BL/6 provoking a significantly higher fibrotic response to alginate microcapsules and encapsulated cells compared to BALB/c (23). Ultimately, PFO will interfere with the effective transport of metabolites and therapeutic molecules to the cells contained in the microcapsules, causing the encapsulated cells to fail and die. This raises a crucial need for the careful selection of a suitable animal model (species and strain) to test the biological tolerance to alginate microcapsules and to various antifibrotic strategies aimed at overcoming PFO.
Various strategies have been employed to try and reduce PFO, thereby allowing the encapsulated cells to survive and function, including the use of highly purified alginate or altering alginate chemistry (8,26), surface modification of alginate microcapsules (17,48,52), and delivering encapsulated cells to different transplant sites (28,43). A newer strategy of trying to reduce PFO is to coencapsulate the target cells with mesenchymal stem cells (MSCs). MSCs are hypoimmunogeneic and are currently being used in clinical trials as immunomodulators in bone marrow transplants (4). Nonencapsulated MSCs also have been cotransplanted with pancreatic islets into rodents and monkeys to reduce/prevent the occurrence of tissue rejection (2). The rationale for testing coencapsulated MSCs to prevent/reduce PFO is that once activated, they are known to produce a variety of factors, such as indoleamine 2,3-dioxygenase, prostaglandin E2, and nitric oxide (13), all of which are molecules that are small enough to exit the permeable microcapsules and modulate the immune response. The concept of coencapsulation with MSCs has been tested previously for the treatment of both acute liver failure (39) and diabetes (22), but only in syngeneic transplantation models. In both studies, mouse islets and rat hepatocytes functioned more efficiently when coencapsulated with MSCs. While it is unclear whether PFO occurred in the acute liver failure study with encapsulated rat hepatocytes (39), PFO did occur when encapsulated syngeneic islets were transplanted into C57BL/6 mice with no benefit offered from coencapsulation with MSCs on PFO reduction (22). These studies suggest the effect of MSC coencapsulation on PFO occurrence cannot be studied effectively in syngeneic transplantation models as the extent of PFO formation was minimal, with the majority of microcapsules having little or no fibrotic overgrowth compared to xenogeneic models where PFO was maximal (24,46). Furthermore, it has been reported previously that the barium alginate microcapsules provide better immunoprotection for encapsulated syngeneic or allogeneic, but not xenogeneic, tissue (6,18).
Presently, we can find no published studies that examine the direct effects of transplanting an aggressive mouse model, at different anatomical sites, with coencapsulated MSCs in barium alginate microcapsules on the occurrence of PFO. To address this question, we examined the effects of coencapsulated MSCs on PFO occurrence in an aggressive xenotransplantation model. To this end, the experiments described in this article focus on 1) testing the biotolerability of alginate microcapsules in different mouse strains to establish the best murine model for subsequent work; 2) testing the ability of MSCs to reduce PFO when coencapsulated with human cells and transplanted into the selected mouse model; 3) determining if the source of the MSCs made a difference to the outcome by testing MSCs from both human and mouse sources; and 4) exploring the potential benefit of two different delivery sites for encapsulated cells, namely, intraperitoneal (IP) infusion and subcutaneous injection.
Materials and Methods
Ethical Approval
All animal experiments were conducted in accordance with the Australian Code of Practice for the Care and Use of Animals for Scientific Purposes (National Health and Medical Research Council), and all experimental procedures were approved by the Animal Care and Ethics Committee of Commonwealth Scientific and Industrial Research Organization (CSIRO), North Ryde, Australia (AEC No: 2011-03). The human placenta-derived MSCs used in this study were isolated by Prof. Kerry Atkinson from two human placentas obtained from healthy mothers as described previously (3), with full informed consent who fulfilled the donor selection guidelines of both the Australian Red Cross and Auscord (The Australian Cord Blood Registry). All procedures relating to isolating human placental-derived MSCs were approved by the Mater Health Services Human Research Ethics Committee and The Ethics Committee of Westmead Hospital, Sydney. In addition, all experiments were carried out in compliance with the Helsinki Declaration.
Tissue Culture
All media used for cell culture were manufactured by Gibco and purchased from Life Technologies (Melbourne, Australia). All fetal bovine serum (FBS) was purchased from Bovogen Biologicals Pty. Ltd. (Melbourne, Australia). All cell cultures were incubated in a humidified atmosphere of 5% CO2 in air at 37°C. The human hepatoinsulinoma cell line (HUH7) was generated and cultured in DMEM high-glucose media (Life Technologies) with 10% FBS as described previously (45) and was used as highly metabolic model target effector cells. Mouse MSCs (mMSCs) isolated from C57BL/6 bone marrow were purchased from Invitrogen (Melbourne, Australia) and expanded in DMEM/F-12 (Life Technologies) with 10% FBS. Human MSCs (hMSCs) isolated from placenta (kindly provided by Prof. Kerry Atkinson) were grown in low-glucose DMEM with 10% FBS as described previously (3).
Differentiation Potential of MSCs
The potential of mMSCs to differentiate into defined cells types was tested by culturing them in six-well plates (In Vitro Technologies, Melbourne, Australia) in either osteogenic or adipogenic differentiation media or in standard mMSC culture conditions as a control. MSCs maintained under osteogenic conditions were cultured in α-MEM (Life Technologies) with 10% FBS containing dexamethasone (1 μM) (Sigma-Aldrich, St. Louis, MO, USA), ascorbic acid phosphate magnesium salt (70 μM) (Sigma-Aldrich), and β-glycerophosphate (7 mM) (Sigma-Aldrich) for 28 days and then routinely stained with Alizarin red (Sigma-Aldrich) and von Kossa stain using silver nitrate (Sigma-Aldrich) to identify mineralized deposits, which occur when cells differentiate into osteocytes. mMSCs maintained under adipogenic conditions were cultured in DMEM/F-12 with 10% FBS containing insulin (1 μM) (Sigma-Aldrich) and dexamethasone (1 μM) (Sigma-Aldrich) for 28 days then routinely stained with Oil red O (Sigma-Aldrich) to identify neutral fat produced by differentiated adipocytes. The control set of mMSCs was maintained under the usual mMSC culture regime (DMEM/F-12 with 10% FBS) for 28 days, and then separate wells were routinely stained with von Kossa or Oil red O.
Encapsulation
Cells were microencapsulated using an air-driven droplet generator as described previously (48). Briefly, the cells were trypsinized (Life Technologies), mixed with 2.2% sodium alginate (Batch No. BP-0908-01; UPMVG Pronova, FMC Biopolymer, Sandvika, Norway) in a ratio of 1:6, and injected through the droplet generator (with air being introduced at 5 L/min and 80 kPa). The microcapsules were gelated in 20 mM barium chloride (Sigma-Aldrich) for 2 min and washed three times with phosphate-buffered saline (PBS) (Life Technologies). Empty capsules were used as controls in all experiments, and these were made in exactly the same way, but no cells were added. The diameter of a representative sample of 100 microcapsules taken from the control and encapsulated cell groups was measured using an Olympus microscope (Olympus Australia Pty. Ltd., Notting Hill, Australia) fitted with an eyepiece graticule in a 10× objective lens. The average size of microcapsules was 750.8 ± 8.6 μm for capsules containing cells and 756.8 ± 6.7 μm (mean ± SEM) for empty capsules. Microencapsulated cells were equilibrated overnight in respective media and held in an incubator at 37°C in a humidified atmosphere of 5% CO2 in air and then washed in PBS prior to transplantation into mice.
Viability
Viability of encapsulated cells was assessed on the day of transplant using routine live/dead staining based on the fluorescent dyes 6-carboxyfluorescein diacetate (6-CFDA; Sigma-Aldrich) and propidium iodide (PI; Sigma-Aldrich) as described previously (17). The percentage of green (live cells) to red (dead cells) was estimated using a fluorescent microscope (Nikon Eclipse Iii, Nikon, Rhodes, Australia).
Transplantation
All mice (NOD/SCID, BALB/c, C57BL/6) were female aged ~8 weeks and obtained from the Australian Resources Centre (Canning Vale, Australia). Isoflurane (Ethical Agents Ltd., Manukau, New Zealand) was used for anesthesia in mice and was induced using 3% isoflurane in air and maintained on 1–2% isoflurane in air. Microcapsules were delivered IP through a 14-gauge catheter (Terumo, Macquarie Park, Australia), as described previously (48). For subcutaneous transplants, microcapsules were injected using a similar 14-gauge catheter inserted through a small incision that was expanded to create a pocket in the dermis on the left flank of the mouse and was closed with stainless steel staples.
Graft Retrieval
At 3 weeks posttransplantation, mice were sacrificed with an excess of sodium pentobarbitone (Virbac, Milperra, Australia) injected into the peritoneal cavity, and microcapsules were retrieved from there or the subcutaneous tissue space immediately postmortem. A time point of 3 weeks was chosen based on our earlier study in which PFO formation was maximal when encapsulated fetal pig islets were xenografted into C57BL/6 mice (46). Retrieval of microcapsules from the peritoneal cavity of mice was carried out by a peritoneal lavage with PBS. Retrieval of microcapsules from the subcutaneous tissue space required excision of a pouch that had formed around the microcapsules in the dermal space. Cell viability of retrieved grafts was measured using live/dead staining (as described above), and capsules were also assessed for evidence of breakage (percentage) and PFO (as described below).
Assessment of PFO
A random aliquot of retrieved microcapsules was taken from each mouse, and the degree of fibrotic overgrowth assessed in a blinded fashion using a scoring system was described previously (48). The microcapsules were scored as follows: 0 = no overgrowth, 1 = <25% of capsule overgrowth, 2 = 25–50% of capsule overgrowth, 3 = 50–75% of capsule overgrowth, and 4 = >75% of capsule overgrowth. The fibrotic score index was calculated for each mouse using the formula (0 × % score 0) + (2 × % score 1) + (4 × % score 2) + (8 × % score 3) + (16 × % score 4), giving a minimum possible fibrotic score of 0 and a maximum possible fibrotic score of 16.
DNA Content Measurement
To quantify PFO on empty microcapsules retrieved from different mouse strains, 30 microcapsules were randomly picked for each mouse and placed in 200 μl of distilled nuclease-free water in a sterile Eppendorf tube. The microcapsules were then sonicated for 5 min to produce a homogeneous solution, and DNA measurements were carried out on homogenized samples using a Nanodrop 2000 Spectrophotometer (Thermo Fisher Scientific, Melbourne, Australia).
Histology
The subcutaneous pouches containing microcapsules from different treatment groups excised at 3 weeks posttransplantation were fixed in 4% neutral-buffered formalin (Ajax Finechem, Sydney, Australia) and routinely embedded in paraffin (Paraplast; Sigma-Aldrich) blocks that were sectioned at 5-μm thickness, routinely stained using hematoxylin and eosin (H&E) (Sigma-Aldrich), and examined using a light microscope.
Experimental Design
Several transplant experiments were performed as part of this study, which examined the beneficial effects of MSCs on the fibrotic response to implanted microcapsules as follows (and summarized in Table 1).
Experimental Series Performed With Details of Different Mice Strains, Different Treatment Groups, and Different Transplant Sites

IP, intraperitoneal; SC, subcutaneous; hMSCs, human mesenchymal stem cells; mMSCs, mouse mesenchymal stem cells; HUH7, human hepatoinsulinoma cell line.
A preliminary experiment was conducted to select the most discriminating mouse strain for use in subsequent experiments. This was performed using empty alginate microcapsules infused into the peritoneal cavity of C57BL/6, BALB/c, and NOD/SCID and retrieved after 3 weeks and analyzed for PFO (as described above).
An experiment to test the effect of coencapsulation with human MSCs was conducted in C57BL/6 mice with treatment delivered IP; viability and PFO were assessed after 3 weeks (as described above).
An experiment to test the effect of coencapsulation with mouse MSCs was conducted in C57BL/6 mice with cells delivered either IP or subcutaneously. Viability and PFO were assessed after 3 weeks (as described above).
Statistical Analysis
All data were expressed as mean ± SEM. Student's t-test was used to compare data between two groups, and one-way analysis of variance (ANOVA) with post hoc Duncan's multiple comparison test was used to compare data among groups, and the results were considered significant with a value of p < 0.05. All statistical analyses were performed using the statistical software NCSS97 (NCSS LLC, Kaysville, UT, USA).
Results
Selection of Aggressive Mouse Model
A preliminary experiment was conducted to compare the reaction to empty alginate microcapsules in two immunocompetent mouse strains (C57BL/6 and BALB/c) compared to an immunocompromised strain (NOD/SCID). Outcomes are presented in Figure 1. The greatest fibrotic reaction to empty alginate microcapsules occurred in the C57BL/6 mice with ~41% of retrieved microcapsules having a score of ≥2+ compared to BALB/c and NOD/SCID, where the percentage of microcapsules with a score of ≥2+ was ~16% and ~0.5%, respectively (Fig. 1A, B). Accordingly, the extent of PFO, as measured by fibrotic score index, was the highest for the C57BL/6 (fibrotic score index 3.9 ± 0.2) with virtually no reaction in the immunodeficient NOD/SCID mice (fibrotic score index 0.2 ± 0.02) and an intermediate response in the BALB/c mice (fibrotic score index 2.0 ± 0.1) (Fig. 1C). The data are further supported by measurement of the DNA content on retrieved microcapsules with those values being significantly higher for C57BL/6 compared to BALB/c and DNA content being the least in NOD/SCID (Fig. 1D). Microcapsules retrieved from all the groups were free floating and intact, with no evidence of breakage confirming the stability of barium alginate microcapsules (Table 2). Together, these data showed that the C57BL/6 mouse was highly reactive to barium alginate microcapsules compared to BALB/c or NOD/SCID and supported the use of this aggressive animal model for subsequent transplantation studies. This is consistent with a previous study that demonstrated that C57BL/6 mice elicited a significantly higher fibrotic response to alginate microcapsules compared to BALB/c mice (23).
Morphological Assessment of Microcapsules Retrieved From the Peritoneal Cavity of Different Mouse Strains
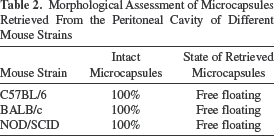

Comparison of fibrotic scores for empty alginate microcapsules retrieved from different strains of mice. (A) Representative images of empty alginate microcapsules retrieved from the peritoneal cavity of C57BL/6, BALB/c, and NOD/SCID mice after 3 weeks posttransplantation (scale bars: 500 μm). (B) The degree of PFO on retrieved microcapsules was assessed using a scoring system and (C) the distribution of the fibrotic scores for the different mouse strain represented as fibrotic score index. (D) Quantification of PFO on retrieved microcapsules from different mice strains measured as DNA content. Values are mean ± SEM (n = 4 for each mouse strain). ***p < 0.0001 for both fibrotic score index and DNA content (ng/μl), respectively, where C57BL/6>BALB/c>NOD/SCID (ANOVA with post hoc Duncan's multiple comparison test).
Differentiation of Mesenchymal Stem Cells
The multipotent potential of mMSCs was confirmed by the outcomes of the differentiation assays that were established in culture on the day of cell implantation for animal trials. mMSCs cultured using the osteogenic regime developed mineralized deposits that stained with Alizarin red (Fig. 2A) and von Kossa (Fig. 2B), consistent with their differentiation into cells of osteogenic lineage. mMSCs cultured using the adipogenic regime developed intracellular fat droplets that stained with Oil red O, consistent with their differentiation into cells of adipocytic lineage (Fig. 2C). Control MSCs grown in standard mMSC medium did not stain for Alizarin red, von Kossa, or Oil red O (Fig. 2D–F). The multipotency of hMSCs has been demonstrated previously and confirmed by their differentiation into bone, cartilage, and fat (3).

Multipotency of MSCs confirmed by differentiation under different culture regimes. Representative images of mMSCs differentiated for 28 days in osteogenic culture regime and stained with Alizarin red (A) and von Kossa (B); adipocyte culture regime and stained with Oil red O (C); controls were mMSCs cultured in usual culture regime and stained negative for Alizarin red (D), von Kossa (E), and Oil red O (F), respectively (scale bars: 100 μm).
Effect of Coencapsulation with hMSCs on PFO in Intraperitoneal Transplants
In this experiment, we examined the effect of MSCs sourced from a different species (human) to the recipient C57BL/6 mice and tested the impact of coencapsulated hMSCs on PFO. Specifically, we tested a hMSC/HUH7 ratio of 1:1 against control groups of encapsulated HUH7 alone, hMSCs alone, and empty alginate capsules, all as detailed in Table 1. The viability of encapsulated cells was tested using live/dead staining prior to transplantation and found to be similar for each individual cell type encapsulated alone and when coencapsulated. Viabilities of encapsulated HUH7 and hMSCs before transplantation were 92.4 ± 0.6% and 91.6 ± 0.9%, respectively. Coencapsulation of HUH7 with hMSCs at a 1:1 ratio did not affect cell viability at 91.2 ± 0.9% before encapsulation (Fig. 3A). The microcapsules in each treatment group were infused into the peritoneal cavity of C57BL/6 mice and retrieved after 3 weeks and then evaluated for PFO. The majority of microcapsules retrieved from mice transplanted with encapsulated HUH7 alone displayed considerable fibrosis with a score of 4+ (~80.3%) followed by a score of 3+ (~9%), a score of 2+ (~3.3%), a score of 1+ (~1.3%), and a score of 0 (~6%), respectively (Fig. 3B, C). Clumping of microcapsules was seen, and some microcapsules were found attached to abdominal organs (such as liver, omentum, or fat pad) due to extensive fibrosis. The extent of PFO as measured by fibrotic score index was the highest for encapsulated HUH7 alone with a value of 13.8 ± 0.6, consistent with the majority of microcapsules having a fibrotic score of 4+ (Fig. 3D). This contrasted to the outcome with encapsulated hMSCs alone, which had low fibrotic scores with ~57.6% of microcapsules having a fibrotic score of 4+ leading to a significantly low fibrotic score index of 11.2 ± 0.3 compared to HUH7 alone (Fig. 3C, D). Coencapsulation of hMSCs with HUH7 in a ratio of 1:1 reduced PFO by ~1.4-fold (fibrotic score index 9.7 ± 0.8) which, although still relatively high, was significantly different from the HUH7 alone (Fig. 3D). The percentage of microcapsules with a fibrotic score of 4+ dropped significantly to ~51% when coencapsulated with hMSCs (Fig. 3C). The control empty microcapsules also elicited a low-grade PFO in this C57BL/6 mouse model, with a fibrotic score index of 3.4 ± 0.5 (Fig. 3C, D), which was consistent with the outcome for the C57BL/6 mouse group in the preliminary experiment that tested empty microcapsules in the three different mouse strains. The outcomes of this experiment are tabulated in Table 3.

Effect of coencapsulation with human MSCs (hMSCs). (A) Representative viability images (6-CFDA: green—live cells and PI: red—dead cells) of varied treatment groups prior to transplantation into the peritoneal cavity of C57BL/6 mice (scale bars: 500 μm). (B) Representative images of microcapsules from each treatment group retrieved from the peritoneal cavity of C57BL/6 3 weeks posttransplantation (scale bars: 500 μm). (C) The degree of PFO on retrieved microcapsules was assessed using a scoring system and (D) the distribution of the fibrotic scores for the varied treatment groups represented as fibrotic score index. Values are mean ± SEM (n = 3 for each treatment group); ***p < 0.0001 for fibrotic score index where hMSCs/HUH7 (1:1) and hMSCs < HUH7 and empty capsules < HUH7; nsp > 0.05 for fibrotic score index between hMSCs and hMSCs/HUH7 (ANOVA with post hoc Duncan's multiple comparison test).
Assessment of Grafts Coencapsulated With Human MSCs (hMSCs) Retrieved From the Peritoneal Cavity of C57BL/6 Mice
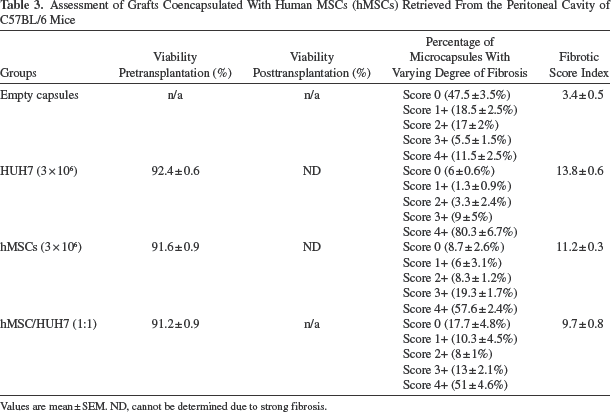
Values are mean ± SEM. ND, cannot be determined due to strong fibrosis.
Effect of Coencapsulation with mMSCs on PFO in Intraperitoneal Transplants
In this experiment, we examined the effect of MSCs sourced from the same species and strain as the recipient C57BL/6 mouse and tested the impact of using different proportions of mMSCs on PFO. Specifically, we tested mMSC/HUH7 ratios of 1:1, 2:1, and 1:2 against control groups of encapsulated HUH7 alone, mMSCs alone, and empty alginate capsules, all as detailed in Table 1. The viability of encapsulated cells was tested using live/dead staining prior to transplantation and found to be similar for each individual cell type and when mixed. Viabilities of individual encapsulated HUH7 (3 × 106), HUH7 (1.5 × 106), and mMSCs (3 × 106) were 87.3 ± 0.7%, 86.7 ± 1.03%, and 87.6 ± 0.6%, respectively. Coencapsulating mMSCs and HUH7 at different ratios (mMSC/HUH7) did not alter the viability, with ratios 1:1, 2:1, and 1:2 having viabilities of 86.4 ± 0.7%, 85.4 ± 1.02%, and 86.8 ± 1%, respectively (Fig. 4A). In each case, the microcapsules in each treatment group were infused into the peritoneal cavity of C57BL/6 mice and retrieved after 3 weeks and then evaluated using the fibrotic scoring system. Consistent with the previous experiment, encapsulated HUH7 (3 × 106) alone elicited a strong fibrotic response with the majority (~99.4%) of capsules having a score of 4+ (Fig. 4B, C) giving a high fibrotic score index of 15.9 ± 0.1 (Fig. 4D). Further, to determine the effect of HUH7 numbers on immune response, a reduced number of encapsulated HUH7 alone (1.5 × 106) was transplanted into C57BL/6 mice and evaluated for PFO. Reducing HUH7 numbers by half to 1.5 × 106 did not reduce PFO but rather had the highest fibrotic score index of 16 (Fig. 4D), similar to that seen with encapsulated 3 × 106 HUH7 with 100% of microcapsules having a fibrotic score of 4+ (Fig. 4B, C). The fibrotic response seen to both 1.5 × 106 and 3 × 106 encapsulated HUH7 alone was so strong it sometimes led to clumping of the microcapsules and attachment to abdominal organs (such as liver, omentum, or fat pad). Further, viabilities of retrieved encapsulated HUH7 (1.5 × 106 and 3 × 106) could not be measured due to the strong PFO response, which created an opaque and impermeable barrier around the microcapsules and cells (Fig. 4E). On the other hand, encapsulated mMSCs alone retrieved from the peritoneal cavity developed very little PFO with ~61.5% of capsules having no fibrosis and a score of 0 (Fig. 4B, C), thereby having a least fibrotic score index of 2.4 ± 0.3, which was equivalent to that for empty microcapsules (3.7 ± 0.3) (Fig. 4D). The postretrieval viability of encapsulated MSCs could be measured in this case and was found to be 78.8 ± 0.9% (Fig. 4E).

Effect of coencapsulation with mMSCs. (A) Representative viability images (6-CFDA: green—live cells and PI: red—dead cells) of varied treatment groups prior to transplantation into the peritoneal cavity of C57BL/6 mice (scale bars: 100 μm). (B) Representative images of microcapsules from each treatment group retrieved from the peritoneal cavity of C57BL/6 3 weeks posttransplantation (scale bars: 500 μm). (C) The degree of PFO on retrieved microcapsules was assessed using a scoring system and (D) the distribution of the fibrotic scores for the varied treatment groups represented as fibrotic score index. (E) Viability of encapsulated cells retrieved from the peritoneal cavity of C57BL/6 mice at 3 weeks posttransplantation (scale bars: 500 μm). Viability could be estimated only for mMSCs, mMSCs/HUH7 (2:1), and mMSCs/HUH7 (1:1) treatment groups, but not for mMSCs/HUH7 (1:2), HUH7 (1.5 × 106), and HUH7 (3 × 106) groups due to the presence of extensive PFO. Values are mean ± SEM (n = 6–13); ***p < 0.0001 for fibrotic score index where mMSCs/HUH7 (2:1) and mMSCs/HUH7 (1:1) < mMSCs/HUH7 (1:2), HUH7 (1.5 × 106), HUH7 (3 × 106); *p < 0.05 for fibrotic score index where mMSCs/HUH7 (2:1) < mMSCs/HUH7 (1:1); nsp > 0.05 for fibrotic score index between empty capsules and mMSCs and when compared between mMSCs/HUH7 (1:2), HUH7 (1.5 × 106), HUH7 (3 × 106) (ANOVA with post hoc Duncan's multiple comparison test).
As with the hMSCs, the mMSCs had a beneficial effect in reducing the PFO when coencapsulated with HUH7 at different ratios (Fig. 4B). A 1:1 ratio of HUH7 and mMSCs caused a significant reduction in fibrosis, compared to HUH7 alone, with a fibrotic score index of 10.4 ± 1 with ~61% of microcapsules having a fibrotic score of 4+ (Fig. 4C, D). The greatest reduction in PFO was seen by increasing the number of mMSCs, and the greatest effect in our study was evident when the ratio of mMSCs and HUH7 was 2:1 with a reduced fibrotic score index of 7.8 ± 1.4 that was significantly less than that for the encapsulated HUH7 alone (Fig. 4D). Furthermore, the number of microcapsules with a score of 4+ was reduced by half to ~45% compared to ~99.4% for the HUH7-alone group (Fig. 4C). The majority of microcapsules from both the 1:1 and 2:1 group were free floating with no signs of clumping or adherence to abdominal organs compared to the HUH7-alone group. Viability on retrieved coencapsulated cells for the 1:1 and 2:1 groups was found to be 75.8 ± 1.4% and 79.3 ± 0.6%, respectively, which was similar to retrieved mMSCs (Fig. 4E, Table 4). Conversely, when the proportion of HUH7 cells was increased to 1:2 in the coencapsulated mMSC and HUH7 group, the fibrotic score index increased significantly (14.4 ± 1.6) to a level similar to that for encapsulated HUH7 alone (15.9 ± 0.1) (Fig. 4B–D). Viability of cells in retrieved capsules from the mMSC/HUH7 ratio of 1:2 could not be estimated due to extent of the PFO (Fig. 4E). Together, these outcomes showed that the increased proportion of MSCs contributed to the beneficial effect in reducing PFO with the mMSC/HUH7 ratio of 2:1 showing a considerably lower fibrotic score index than that for 1:1 ratio. The outcomes of this experiment are tabulated in Table 4.
Assessment of Grafts Coencapsulated With Mouse MSCs (mMSCs) Retrieved From the Peritoneal Cavity of C57BL/6 Mice

Values are mean ± SEM. ND, cannot be determined due to strong fibrotic overgrowth.
Effect of Coencapsulation with mMSCs on PFO in Subcutaneous Transplants
The impact of transplantation site on the occurrence of PFO was tested by infusing the same treatment groups of microcapsules subcutaneously and comparing to those implanted IP, as detailed in Table 1. In each case, the microcapsules in each treatment group were infused into the peritoneal cavity of C57BL/6 mice, retrieved after 3 weeks, and evaluated using the fibrotic scoring system. Outcomes of the experiment, which evaluated the PFO response to capsules delivered subcutaneously, are presented in Figure 5. Contrary to IP transplant outcomes, subcutaneous transplantation of encapsulated HUH7 (3 × 106) alone resulted in significantly less fibrosis with only ~1.8% of capsules having the highest fibrotic score of 4+ (Fig. 5A). The majority (~52.8%) of retrieved microcapsules in the HUH7 group had a fibrotic score of 2+ followed by scores of 3+ (~28.2%), 1+ (~14.6%), and 0 (~2.5%), respectively (Fig. 5A). This resulted in a low fibrotic score index of 4.9 ± 0.5 (Fig. 5B), which was significantly lowered by ~3.2-fold when compared to HUH7 transplanted IP (fibrotic score index of 15.9 ± 0.1) (Fig. 5C). Similarly, the PFO on encapsulated mMSCs was very low with ~99.4% of capsules having a fibrotic score of ≤2+ (Fig. 5A), resulting in a low fibrotic score index of 1.1 ± 0.3, which was equivalent to that of empty microcapsules transplanted subcutaneously (0.7 ± 0.2) (Fig. 5B). As seen with HUH7, implanting mMSCs and empty microcapsules subcutaneously significantly reduced fibrotic score index by ~2.2- and ~5.3-fold, respectively, compared to respective IP transplants (Fig. 5C). As was noted previously with IP-implanted capsules, PFO was significantly reduced in the subcutaneous tissue transplants when coencapsulated with mMSCs. Coencapsulating mMSCs with HUH7 at a 1:1 ratio significantly reduced fibrosis with the majority of microcapsules having a fibrotic score of ≤2+ (Fig. 5A) with a fibrotic score index of 3.4 ± 0.8 compared to HUH7 (4.9 ± 0.5) (Fig. 5B). Increasing the proportion of mMSCs by increasing the mMSC/HUH7 ratio to 2:1 further reduced PFO with a fibrotic score index of 2.3 ± 0.3, which was similar to that of empty and mMSC microcapsules transplanted subcutaneously (Fig. 5B). Again, compared with IP transplant counterparts, the fibrotic score index for coencapsulated mMSCs/HUH7 at ratios of 1:1 and 2:1 was significantly reduced by ~3.1- and ~3.4-fold, respectively, when transplanted subcutaneously, demonstrating the effect of transplant site on PFO formation (Fig. 5C).
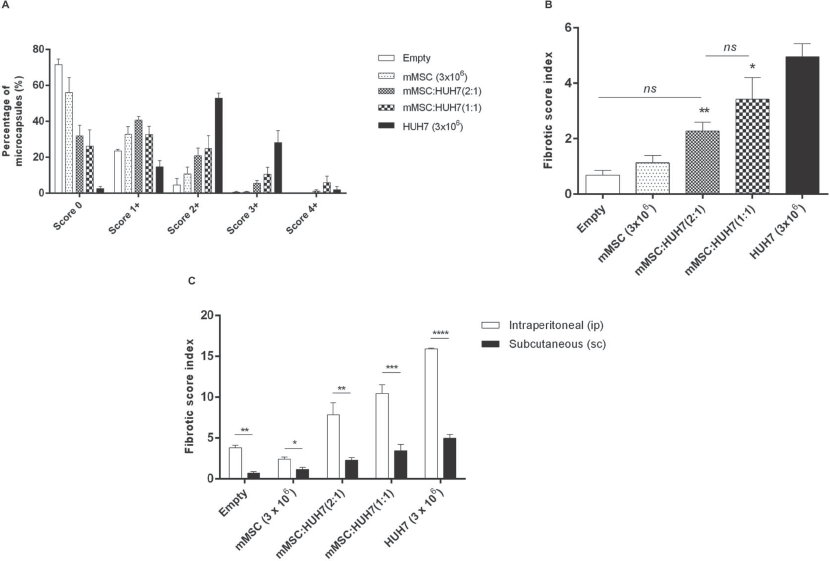
Effect of implant site. The impact of delivery site on the fibrotic response was tested by infusing microcapsules subcutaneously into C57BL/6 and comparing to that of IP transplant outcomes. (A) The degree of PFO on retrieved microcapsules was assessed using a scoring system, and (B) the distribution of the fibrotic scores for the varied treatment groups transplanted subcutaneously represented as fibrotic score index. Values are mean±SEM (n = 6 for each treatment group). **p < 0.01 for fibrotic score index where mMSCs/HUH7 (2:1)<HUH7 (3× 106); *p < 0.05 for fibrotic score index where mMSCs/HUH7 (1:1)<HUH7 (3× 106); nsp > 0.05 for fibrotic score index between empty capsules and mMSCs and when compared between empty capsules, mMSCs, and mMSCs/HUH7 (2:1) (ANOVA with post hoc Duncan's multiple comparison test). (C) Comparison of fibrotic score index between IP and subcutaneous transplants. Values are mean±SEM; *p < 0.05, **p < 0.01, ***p < 0.001, and ****p < 0.0001 for fibrotic score index when compared between IP and subcutaneous transplants between the different treatment groups (Student's t-test).
However, although the PFO reaction to the individual microcapsules implanted subcutaneously was lower than equivalent implants delivered IP, all microcapsule treatments implanted subcutaneously were contained in a pouch by the 3-week time point (Fig. 6A). The majority of microcapsules were found free floating within the fibrotic pouch. Histological evaluation of the pouches showed them to be formed by uninterrupted multiple layers of vascularized fibrous tissue in every case. Where microcapsules contained cells, the fibrotic tissue was infiltrated with immune cells (Fig. 6B). The viability of cells contained in retrieved microcapsules within these pouches was still relatively high for mMSC-containing groups at 3 weeks. Encapsulated mMSCs alone had a viability of 85.7 ± 0.4% compared to HUH7, the viability of which had dropped significantly to 51.5 ± 0.7%. Coencapsulating with mMSCs significantly improved viability with mMSCs/HUH7 at a ratio of 1:1, demonstrating a viability of 79.1 ± 0.8% compared to HUH7. Increasing the proportion of mMSCs to 2:1 in the mMSC/HUH7 group significantly improved viability of retrieved cells to 83.1 ± 0.6% at 3 weeks postimplantation (Fig. 6C, Table 5). The outcomes of this experiment are tabulated in Table 5.

Evaluation of fibrotic pouch formation and viability of encapsulated cells in subcutaneous transplants. (A) Macroscopic view of typical pouch that formed around subcutaneous infusion of microcapsules retrieved from mouse at 3 weeks posttransplantation. Majority of microcapsules were found free floating within the pouch. (B) Histological analysis of the pouches showed that they were comprised of multiple layers of vascularized (arrows) fibrous tissue containing mixed infiltrate of immune cells (scale bars: 100 μm). (C) Viability images (6-CFDA: green—live cells and PI: red—dead cells) of encapsulated cells retrieved free floating from the fibrotic pouch at 3 weeks posttransplantation (scale bars: 500 μm).
Assessment of Grafts Coencapsulated With Mouse MSCs (mMSCs) Retrieved From the Subcutaneous Space of C57BL/6 Mice
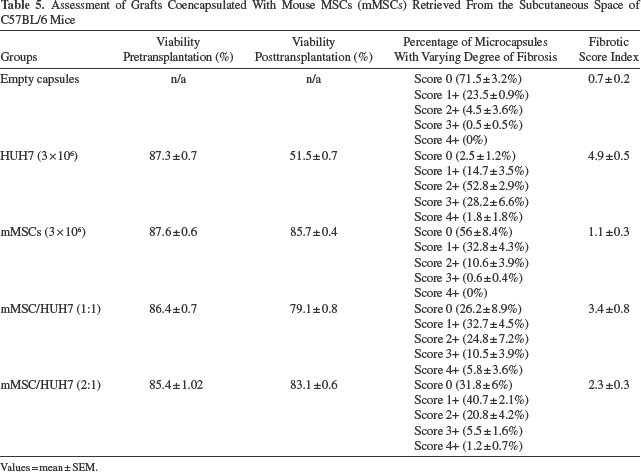
Values = mean ± SEM.
Discussion
PFO is one of the major reasons for the limited graft survival of alginate microencapsulated cells and, as such, is a major impediment to the clinical success of encapsulated cellular therapies (44). PFO is either seen as a foreign body reaction against the microcapsules and/or an immunological response to antigens shed by the encapsulated cells (8,9,18). The severity of PFO is highly variable in animal models (23) and hence requires a careful selection of the suitable in vivo model to test the varied antifibrotic strategies. In this study, we initially assessed the nature of PFO in response to alginate microcapsules transplanted into different mouse strains and then selected the most aggressive mouse model to test the antifibrotic strategy of coencapsulation with immunomodulatory MSCs.
Microcapsules used in all experiments were made from the same batch of commercially purchased ultrapure alginate from Pronova (ultrapure medium viscosity 263 mPas, guluronic/mannuronic acid ratio ≥1.5, guluronic acid content 69%, protein content ≤0.3, and endotoxins 61 EU/g). The purity of alginate as measured by endotoxins and protein content is highly important as variations in alginate purity can be directly associated with the nature of the host immune response in determining the biocompatibility of alginate microcapsules being tested (7,30). Despite Pronova alginate being labeled ultrapure and being the most commonly used, a recent study found that they contain other endotoxins or pathogen-associated molecular pattern molecules such as peptidoglycan and lipoteichoic acid, which are immunostimulatory (32). The endotoxins peptidoglycan and lipoteichoic acid are well-known ligands for toll-like receptors and are involved in the innate immune response against alginate-based microcapsules (14,19). In this study, despite using the same batch of Pronova ultrapure alginate, the immune response to empty alginate microcapsules, as measured by PFO, was significantly higher in the immunocompetent C57BL/6 mice compared to the other immunocompetent strain, BALB/c, with PFO being the least in the immunocompromised NOD/SCID mice. The strongest PFO response was seen in C57BL/6 mice and was not unexpected as previous research reported the upregulation of transforming growth factor-β (TGF-β) in this mouse strain (25). This molecule is known to contribute to fibrosis by promoting myofibroblast differentiation and collagen synthesis and is involved in the host reaction against alginate-poly-L-lysine microcapsules (16,34). Furthermore, a defective toll-like receptor signaling (31) and decreased interleukin-1β (IL-1β) and tumor necrosis factor-α (TNF-α) expression (23) in macrophages of BALB/c mice might be attributed to the reduced PFO response to alginate microcapsules seen in this strain of mice compared to the C57BL/6 strain. Together, these data clearly emphasize the need for careful selection of the most appropriate animal model to test the biocompatibility of alginate microcapsules and to evaluate various antifibrotic strategies. In this study, C57BL/6 mice of the same age were purchased from the same commercial source and were used for all experiments, since the planned study needed the most discriminating mouse model for clear experimental outcomes.
The potential antifibrotic benefit of MSCs was tested by coencapsulating model target cells of human origin (HUH7) with MSCs of either human or mouse origin to examine any species effect. Different ratios of both MSC sources were tested to see if there was an empirical relationship involved in the MSC response. In both cases, the presence of MSCs with HUH7 in a ratio of 1:1 resulted in a reduction in the mean fibrotic score index by ~35% with mMSCs and ~30% with hMSCs compared with that measured for HUH7 alone. Increasing the ratio of mMSCs to HUH7 to 2:1 reduced PFO by ~51% compared to that measured with HUH7 alone. Conversely, increasing the ratio of HUH7, as seen in the mMSC/HUH7 1:2 treatment group, resulted in PFO increased to a level almost as high as HUH7 alone. Together, these data showed that coencapsulation of MSCs with target cells significantly reduced the level of PFO when microcapsules were implanted IP over a 3-week period. MSCs of both mouse and human origin showed a similarly beneficial effect in this regard when coencapsulated with target cells in a ratio of 1:1. In the case of mMSCs, PFO was further reduced by increasing the proportion of MSCs relative to target cells to 2:1, providing tangible evidence for some empirical relationship in the antifibrotic effect of MSCs in our study. This outcome is consistent with reports that the immunomodulatory benefit depends on the MSC concentration (22,38). mMSCs encapsulated alone elicited a minimal PFO score at the IP site, which would be expected given the syngeneic nature of the cells in the recipient C57BL/6 mice. Human MSCs alone caused moderate PFO after 3 weeks in the peritoneal cavity of mice, which was not unexpected due to the xenogeneic nature of the graft despite the hypoimmunogenic effects of the MSCs (27). Previous work has shown that nonencapsulated human MSCs transplanted into rodents have limited immunomodulatory effects (49) and survival (5).
The effect of transplant/delivery site on the immunomodulatory effect of MSCs on the fibrotic response was tested by infusing microcapsules containing different ratios of mMSCs and HUH7 subcutaneously in C57BL/6 mice, which were retrieved after 3 weeks. Outcomes revealed substantially reduced PFO scores when microcapsules were implanted subcutaneously. Despite the reduced PFO seen with encapsulated HUH7 alone transplanted subcutaneously, coencapsulation of mMSCs with HUH7 in different ratios resulted in a significant reduction of PFO by ~30% and ~53% for ratios of 1:1 and 2:1, respectively. This percent reduction in PFO seen with the mMSC/HUH7 coencapsulated groups transplanted subcutaneously was similar to that transplanted IP with a ~35% and ~51% reduction in PFO seen with 1:1 and 2:1 groups, respectively. These results suggest that coencapsulated MSCs at different ratios exert a similar benefit when transplanted IP or subcutaneously and that the immunomodulatory effect of mMSCs is concentration dependent but independent of the transplant site.
Interestingly, a direct comparison of the fibrotic score indices suggests that PFO was significantly reduced in all the treatment groups transplanted subcutaneously compared to the corresponding IP transplants. When the HUH7/mMSC 1:1 treatment group was implanted subcutaneously, the PFO score was reduced by ~78% compared to the equivalent group implanted IP. The HUH7/mMSC 1:2 subcutaneous group PFO score was reduced by ~86% compared with the equivalent group implanted IP. These outcomes are consistent with previous reports of reduced fibrotic scores in subcutaneous implants of microcapsules containing pig islets in rats (11). However, the formation of a fibrotic pouch surrounding the microcapsules was not reported in that study. In our study, we found that the microcapsules transplanted subcutaneously were surrounded by multiple layers of vascularized fibrous tissue with a mixed infiltrate of immune cells representing a strong secondary fibrotic response at this surgical site. Interestingly, the microcapsules inside the pouch were free floating with very little PFO and improved viability when coencapsulated with MSCs. Coencapsulation of MSCs with HUH7 at different ratios significantly improved cell survival within the fibrotic pouch when compared to HUH7 alone transplanted subcutaneously to 3 weeks. The beneficial effect of MSCs in promoting graft survival might be attributed to the trophic factors secreted by MSCs in improving viability and apoptosis (21,33) and not due to enhanced angiogenesis as microencapsulation effectively prevents revascularization. Accordingly, in this study we found that the cell viability of microcapsules containing coencapsulated mMSCs and HUH7 at a 2:1 ratio was higher compared to that of 1:1 when retrieved from the subcutaneous fibrotic pouch, again suggesting that beneficial effects of MSCs is concentration dependent (22,38).
Conclusions
In summary, we have shown that coencapsulation of MSCs with target effector cells reduced PFO when xenotransplanted into a highly aggressive immunogenic strain of mice. We have also demonstrated that the beneficial effect of MSCs in reducing PFO is highly dependent on its concentration, which might be attributed to the immunomodulatory effects mediated by anti-inflammatory cytokines (29,42) or metalloproteinase secreted by MSCs (10). Outcomes of the current study demonstrated a benefit of coencapsulation of model human cells with MSCs of either bone marrow-derived mouse or placenta-derived human origin when delivered IP. A further reduction in PFO that was evident when microcapsules were delivered subcutaneously may be negated by the secondary fibrosis, which presented as an intact fibrous pouch and perhaps might result in graft failure, but this remains to be tested. Functional studies need to be conducted to determine if the reduction in PFO seen at both IP and subcutaneous transplant sites could improve graft survival and allow the delivery of insulin in response to blood glucose levels in a diabetic animal model. A successful outcome has the potential to extend the therapeutic platform for MSCs to include encapsulated cell therapies designed to treat insulin-dependent diabetes (1), liver failure (51), and chronic neurological diseases (41).
Footnotes
Acknowledgments
The authors would like to thank the Australian Foundation for Diabetes Research and the John Ebsary Foundation for contributing to the funding that supported this research work. B.E.T. was a member of the Australian Foundation for Diabetes Research at the time of the research and currently is an Honorary Director. V. V. received salary support from the John Ebsary Foundation. We gratefully acknowledge Gail McFarland for lab support and assistance with tissue culture. The authors declare no conflicts of interest.
