Abstract
Lymphedema is an intractable disease with few effective therapeutic options. Autologous mesenchymal stem cell (MSC) transplantation is a promising therapy for this disease. However, its use is limited by the cost and time for preparation. Recently, xenotransplantation of porcine MSCs has emerged as an alternative to autologous MSC transplantation. In this study, we aimed to clarify the usefulness of neonatal porcine bone marrow–derived MSC (NpBM-MSC) xenotransplantation for the treatment of lymphedema. One million NpBM-MSCs were xenotransplanted into the hind limbs of mice with severe lymphedema (MSC transplantation group). The therapeutic effects were assessed by measuring the femoral circumference, the volume of the hind limb, the number and diameter of lymphatic vessels in the hind limb, and lymphatic flow using a near-infrared fluorescence (NIRF) imaging system. We compared the effects using mice with lymphedema that did not undergo NpBM-MSC transplantation (negative control group). The condition of the transplanted NpBM-MSCs was also evaluated histologically. The femoral circumference and volume of the hind limb had been normalized by postoperative day (POD) 14 in the MSC transplantation group, but not in the negative control group (P = 0.041). NIRF imaging revealed that lymphatic flow had recovered in the MSC transplantation group by POD 14, as shown by an increase in luminance in the hind limb. Histological assessment also showed that the xenotransplantation of NpBM-MSC increased the proliferation of lymphatic vessels, but they had been rejected by POD 14. The xenotransplantation of NpBM-MSCs is an effective treatment for lymphedema, and this is mediated through the promotion of lymphangiogenesis.
Keywords
Introduction
Lymphedema is characterized by lymphostasis in subcutaneous tissue, owing to impairment of the lymphatic system. The prevalence of this disease is increasing annually 1 , and approximately 200 million people currently have lymphedema worldwide2,3. Severe lymphedema contributes to a poor quality of life of the patients. For instance, foot lymphedema impedes walking 4 and is a risk factor for refractory cellulitis and gangrene5,6. Furthermore, long-term lymphedema can induce angiosarcoma, a malignant tumor derived from vascular and lymphatic endothelial cells (LECs) 7 . The current standard therapy for lymphedema is a combination of decompression therapies, including skin care, manual lymphatic drainage, compression using elastic stockings, and exercise 8 . However, these therapies are only palliative, and the therapeutic effects are unsatisfactory 9 .
Recently, mesenchymal stem cells (MSCs) have been determined to be promising cellular resources for regenerative medicine because they have useful characteristics, including multipotency; the potential for self-renewal 10 ; and the production of growth factors and cytokines that promote angiogenesis and tissue regeneration and prevent fibrosis, apoptosis, and inflammation11–14. In recent years, clinical trials of the use of MSC transplantation for the treatment of cardiovascular, cartilage, and neurological diseases have been performed15–17.
MSCs also have a role in the promotion of lymphangiogenesis through differentiation into LECs and the production of growth factors 18 . This might render them suitable for the treatment of lymphedema. Indeed, a systematic review of the use of clinical autologous MSC transplantation for the treatment of lymphedema showed their usefulness and safety 19 . However, the use of autologous MSCs derived from lymphedema patients is limited by their poor quality, compared with those from healthy donors 20 . In addition, autologous MSC transplantation has other limitations associated with their preparation. First, the acquisition of autologous MSCs is invasive because it involves bone marrow harvest and liposuction. Second, the preparation of autologous MSCs is expensive and time-consuming because it takes time to culture and prepare a sufficient number of MSCs for transplantation21–23, as well as the requirement to prepare induced pluripotent stem (iPS) cell-derived biomaterials 24 . Therefore, the establishment of alternative donor sources for MSCs is important to facilitate such treatment. We have focused on the use of neonatal porcine bone marrow–derived MSCs (NpBM-MSCs) as a promising candidate.
The pig is a promising donor candidate because of its similarity to humans and the established breeding methods 25 . The immune response is the principal limitation of xenotransplantation, but recent progress in gene-editing technology might enable this limitation to be overcome and encourage the development of this approach. A recently reported case of porcine heart xenotransplantation showed that the innate immune response can be successfully avoided26–28 and indicates that porcine xenotransplantation might be feasible in the near future. Porcine MSCs can be stored in a tissue bank, which enables them to be provided on demand. Furthermore, the neonatal pig is superior to the adult as a donor in terms of cost, scale, the retention of cell viability after culture, and cellular proliferation 29 . Therefore, we consider that the development of NpBM-MSC xenotransplantation should contribute to the treatment of lymphedema.
In the present study, we aimed to clarify the usefulness of NpBM-MSC xenotransplantation for the treatment of lymphedema using a murine model of lymphedema that we have previously developed 30 .
Materials and Methods
Study Design
The design of the study is shown in Fig. 1. In brief, one million NpBM-MSCs were xenotransplanted into a hind limb of mice with severe lymphedema (MSC transplantation group), then the effects were assessed by measurement of thigh circumference, the volume of the hind limb, number and diameter of the lymphatic vessels in the hind limb, and the lymphatic flow using a near-infrared fluorescence (NIRF) imaging system. Measurements of the femoral circumference were taken at three points that were considered to be the maximum diameter of the femoral region, and the maximum diameter of the three was used. And a piece of silk thread was wrapped around the femoral circumference, and its length was measured. Hind limb volume was measured by the water displacement method from the toes to the femoral region where the circumferential skin incision was performed. This method has been done in our previous and other research5,7,30,31. The results were compared with those achieved in untreated mice with lymphedema (negative control group).
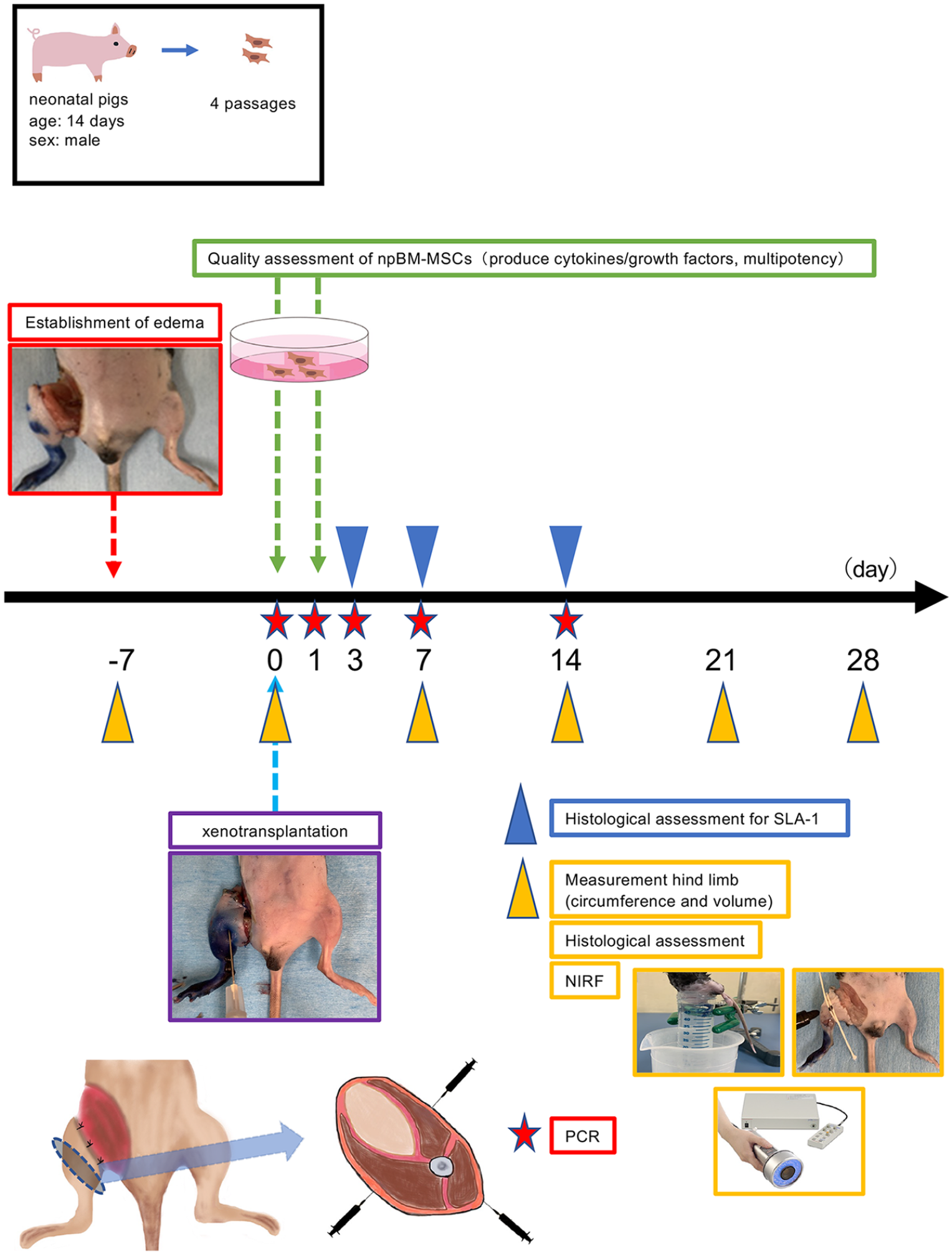
Study design.
Animals and Ethics
Eight-week-old male C57BL/6J mice (Charles River Laboratories, Yokohama, Japan) were purchased, carefully monitored, and provided with a normal chow diet ad libitum. The animals were cared for, and the experimental procedures were performed in accordance with the Principles of Laboratory Animal Care of the Guide for the Care and Use of Laboratory Animals, 8th edition (National Research Council, 2011). The experimental protocol was approved by the Animal Care and Use Committee of Fukuoka University (approval number: 2113110). Regarding pain and distress categories, this study was classified as category C in our institution because animals used in this study had lymphedema for 4 weeks but prevented from severe stress with life-threatening complications.
Induction of Hind Limb Lymphedema
Lymphedema was induced in the mice using our previously published method 30 . In brief, the mice were anesthetized using isoflurane (5% for induction, 2.4% for maintenance; Fujifilm Wako Pure Chemical Corporation, Osaka, Japan). After labeling the lymph vessels and nodes of the right hind limb by the subcutaneous injection of Evans blue (Fujifilm Wako Pure Chemical Corporation), a circumferential skin incision was made at the femoral region, and the labeled sub-iliac, popliteal, and sciatic lymph nodes and lymphatic vessels were dissected. The central side of the transected skin was cauterized, and the peripheral side was fixed to the myofascia using 6-0 nylon sutures (Johnson & Johnson, New Brunswick, NJ, USA) at four different sites. Irradition therapy was not included for making this model because of preventing damage of hind limb except lymphatic vessels.
Preparation of NpBM-MSCs
Primary NpBM-MSCs were kindly provided by Otsuka Pharmaceutical Factory, Inc. (Tokushima, Japan). These were obtained from nonclinical-grade neonatal pigs (age, 14 days; sex, male; body mass, 3.2 kg; Kadoi Ltd., Ibaraki, Japan) by Dr. Masuhiro Nishimura. We used three batches of the NpBM-MSCs in this study. They were used in different examinations including xenotransplantation, in vitro quality assessments (differentiation test and enzyme-linked immunosorbent assay [ELISA] for released growth factors), and assessment of LEC proliferation. The NpBM-MSCs were maintained in minimum essential medium α (Gibco, Waltham, MA, USA) containing 10% fetal bovine serum (Gibco), 100 U/mL penicillin and 100 µg/mL streptomycin (Sigma-Aldrich, St. Louis, MO, USA), and 1 ng/mL human fibroblast growth factor-basic (Sigma-Aldrich; MSC culture medium) in 75-cm2 flasks (Thermo Fisher Scientific, Inc, Waltham, MA, USA). The NpBM-MSCs were used at four passages.
Quality Assessment of the NpBM-MSCs
Before the transplantation of MSCs, we assessed the stem cell characteristics of the cells, which should be multipotent and able to produce cytokines and growth factors that have paracrine effects. Regarding multipotency, we attempted to induce their differentiation into adipocytes or osteoblasts using an adipocyte differentiation kit (Bio Future Technology, Tokyo, Japan) or an osteoblast differentiation kit (Bio Future Technology). Adipocytes and osteoblasts were identified using oil red O and Alizarin Red S (Bio Future Technology) staining, respectively. The culture medium used to incubate NpBM-MSCs was collected after 24 h for the assessment of vascular endothelial growth factor (VEGF)-A, VEGF-C, and transforming growth factor beta (TGF-β) secretion. The number of seeded cells and the volume of culture medium were 1 × 105 cells and 2 mL per well, respectively. The VEGF-A, VEGF-C, and TGF-β1 concentrations in the medium were measured before culture to determine the basal amounts of cytokine present in the medium. These concentrations were measured by ELISA using a Porcine Vascular Endothelial Growth Factor A ELISA Kit (MyBioSource, Inc, San Diego, CA, USA), a Porcine Vascular Endothelial Growth Factor C ELISA Kit (MyBioSource, Inc), a Mouse VEGF-A ELISA kit (Novus Biological, Centennial CO, USA), a Mouse VEGF-C ELISA kit (Novus Biological), and porcine and murine TGF-β1 ELISA kits (Proteintech Japan, Tokyo, Japan). The basal concentrations in the media were subtracted from the postincubation concentrations, and the resulting concentrations were normalized to the number of cells present at the time of sample collection. The data are expressed per 1 × 105 cells.
Assessment of LEC Proliferation
C57BL/6J-derived LECs (Cell Biologics, Inc, Chicago IL, USA) at seven passages were seeded into wells in NpBM-MSC–conditioned medium, alongside wells containing nonconditioned medium. NpBM-MSC–conditioned medium was prepared using 10 mL of endothelial cell basal medium (Lonza, Basel, Switzerland) containing 0.5% fetal bovine serum (FBS) by culturing with NpBM-MSCs at 1.0 × 106 cells per 75-cm2 flask for 24 h. Each well of a six-well plate was seeded with 1.0 × 105 cells, and after 24 h, the cells were trypsinized, and the total number of cells and number of dead cells were counted.
Three-Dimensional Culture of LECs
LECs were cultured using an EGM-2 Endothelial Cell Growth Medium-2 Bulletkit (Lonza) and used for in vitro experiments after seven passages. The formation of three-dimensional capillary-like structures was assessed using a Matrigel-based tube formation assay 32 . LECs were embedded in growth factor–reduced Matrigel (Corning, Corning, NY, USA) (5 × 104 cells/50 µL) and incubated (MSC medium group). As a control, LECs cultured in the absence of Matrigel were prepared as medium-only group. The cells were incubated at 37°C in a 5% CO2-containing atmosphere for 14 days. Tube formation was assessed using a BZ-X700 fluorescence microscope (Keyence, Itasca, IL, USA) and Image J v1.54e software (NIH, Bethesda, MD, USA; available at https://imagej.net) in five random fields every other day. To quantify this, the sum of the long-axis length of all the LECs and the number of branching points per field at ×100 magnification were measured.
MSC Xenotransplantation and Animal Groups
MSC xenotransplantation was performed in mice 7 days after the induction of lymphedema. NpBM-MSCs (1 × 106 cells) suspended in 100-µL phosphate-buffered saline (PBS, Gibco) were injected at three locations (cranial, lateral, and caudal aspects, Fig. 1) in the femoral muscle using a 27-gauge needle under general anesthesia induced and maintained using isoflurane. Mice in the MSC xenotransplantation group were administered NpBM-MSCs, and mice in the negative control group were injected with the same volume of PBS. We also prepared a normal group consisting of mice that were untreated.
Measurements of Femoral Circumference and Hind Limb Volume
The degree of lymphedema of the mice was evaluated by measuring their femoral circumference and the volume of their hind limbs using the water displacement method5,7,31. Successful induction of lymphedema was accepted if there was an increase in the circumference or volume of the hind limb of >10% vs those before surgery.
Histological Assessment of the Hind Limb
Histological assessments of the number, maximum short-axis diameter, and total area of lymphatic vessels of the hind limb were conducted and measured using a BZ-X700 fluorescence microscope (Keyence) and software (Hybrid Cell Count) of the BZ-X700 fluorescence microscope (Keyence). The data acquired by Keyence are able to automatically measure number, maximum short-axis diameter, and total area. Femoral samples were excised from some of the mice in the normal, negative control, and MSC transplantation groups on postoperative day (POD) 0 (the day of MSC transplantation) and PODs 7, 14, 21, and 28. Three slides of the largest section of the mouse hind limb femoral region (n = 10) were used for histological assessment. The samples were fixed using 10% formaldehyde solution, embedded in paraffin, and cut into 3-µm-thick sections. They were then subjected to heat-induced epitope retrieval, blocking by incubation in Blocking One Histo (Nacalai Tesque, Kyoto, Japan) for 30 min at room temperature, and incubation with primary antibodies. The primary antibodies were purified hamster anti-podoplanin (Pdp, 1:100; BioLegend, San Diego, CA, USA), for the identification of lymphatic vessels, and mouse anti-pig swine leukocyte antigen (SLA) class I (1:100; Bio-Rad Laboratories Inc., Hercules, CA, USA), for the identification of transplanted NpBM-MSCs 23 . The secondary antibody was goat anti-hamster IgG (H + L) Alexa Fluor 546 (Invitrogen, Carlsbad, CA, USA). Samples were incubated with primary antibodies for 2 h at room temperature and with secondary antibody for 2 h in the dark at 37°C. Images were acquired using a BZ-X700 fluorescence microscope (Keyence). Pdp-positive cells were regarded as being part of lymphatic vessels, and the number, diameter, and area of each were calculated as the mean values for five different fields of view at ×100 magnification using BZ-X700 software (Hybrid Cell Count).
Measurement of Lymphatic Flow Using NIRF Imaging
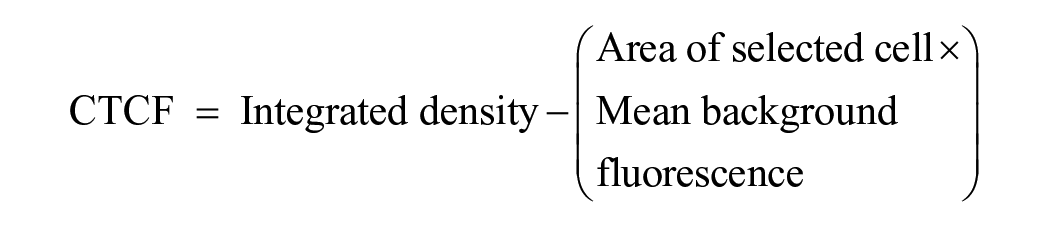
The subcutaneous tissue of a hind limb of each mouse in both the MSC and negative control groups was injected with 10 μl of indocyanine green (ICG, 2.5 mg/mL; Daiichi Sankyo, Tokyo, Japan) under general anesthesia using isoflurane. The lymphatic flow in the hind limb was then measured using a NIRF imaging system (Pde-neo; Hamamatsu Photonics, Shizuoka, Japan). The measurement conditions (external light, such as fluorescent light, and the brightness and contrast of the photodynamic eye [PDE]) were standardized before commencing these measurements. The lymphatic flow was measured weekly from the date of induction of lymphedema to the fourth week following NpBM-MSC xenotransplantation as luminance using image J v1.54e software (NIH). Luminance was calculated as the corrected total cell fluorescence (CTCF) using the following formula24,25,33,34:
However, we modified this by including the noncell fluorescence in the equation. The mean relative luminance was determined by dividing the CTCF by the area of fluorescence, as follows:
Real-Time Quantitative Polymerase Chain Reaction
The hind limbs of some of the mice in the three groups were collected on PODs 0, 7, 14, 21, and 28 for the quantitative polymerase chain reaction (qPCR) analysis of the hind limb tissues other than bone. After removing the bones, the tissues were homogenized together using a homogenizer (T10 basic; IKA Japan, Osaka, Japan) in TRIzol reagent (Invitrogen), then RNA was extracted. The concentrations of the mRNA samples obtained were measured using a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Inc) and then equalized. cDNA was synthesized from this RNA using a PrimeScript Plus RT Kit (Takara Biotechnology Co., Ltd., Kusatsu, Japan), in accordance with the manufacturer’s instructions.
qPCR was performed using the StepOnePlus Real-time PCR System (Applied Biosystems; Thermo Fisher Scientific, Inc, Waltham, MA, USA) and a SYBR green assay. The porcine and murine primers used for the qPCR are shown in Table 1. Relative quantification of target mRNAs was performed using StepOne Software v.2.3 (Applied Biosystems, Inc), and the results were normalized to the expression of the reference gene Actb. All the reactions were performed in triplicate, and quantification was performed using the 2−ΔΔCT method.
Primer Information.

Statistical Analysis
Data are presented as mean ± standard error of the mean. Statistical comparisons of two groups were made using Student’s t-test; and ANOVA, followed by Tukey’s HSD test, was used for multiple groups. P < 0.05 was considered to represent statistical significance. All analyses were performed using SPSS v.24 (IBM Corp., Armonk, NY, USA).
Results
NpBM-MSCs Promote the Proliferation of LECs Via the Secretion of VEGFs
First, we characterized the NpBM-MSCs. The NpBM-MSCs were able to differentiate into osteocytes and adipocytes (Fig. 2A, B). They also secreted porcine VEGF-A and VEGF-C into the culture medium (113.8 ± 6.9 pg/ mL vs 0.0 ± 0.0 pg/ mL [P < 0.0001] and 110.4 ± 1.9 pg/ mL vs 0.0 ± 0.0 pg/ mL [P < 0.0001], respectively, vs cell-free medium; Fig. 2C, D). However, neither murine VEGF-A nor VEGF-C was detected in the NpBM-MSC–conditioned medium (Fig. 2F, G). The cells also secreted porcine TGF-β1 into the culture medium (139.2 ± 4.6 pg/ mL vs 0.0 ± 0.1 pg/ mL, P < 0.0001, vs cell-free medium; Fig. 2E). Furthermore, the proliferation of LECs was promoted by culture with the NpBM-MSC–conditioned medium (15.3 ± 0.4 × 104 cells vs 20.2 ± 1.4 × 104 cells after 24 h, P < 0.01; Fig. 2H). Tube formation, which was defined as the presence of a tube-shaped structure consisting of LECS that had proliferated and budded, was also promoted by incubation with the NpBM-MSC–conditioned medium (Fig. 2I). Both the total length of the long axes of the LECs and the number of branching points per field were significantly larger in cells incubated in NpBM-MSC–conditioned medium (4,970.0 ± 354.7 vs 3,358.6 ± 312.7 μm/field after 72 h, P < 0.01; and 29.8 ± 1.8 vs 20.7 ± 1.7 points/field after 72 h, P < 0.001; respectively; Fig. 2J, K).
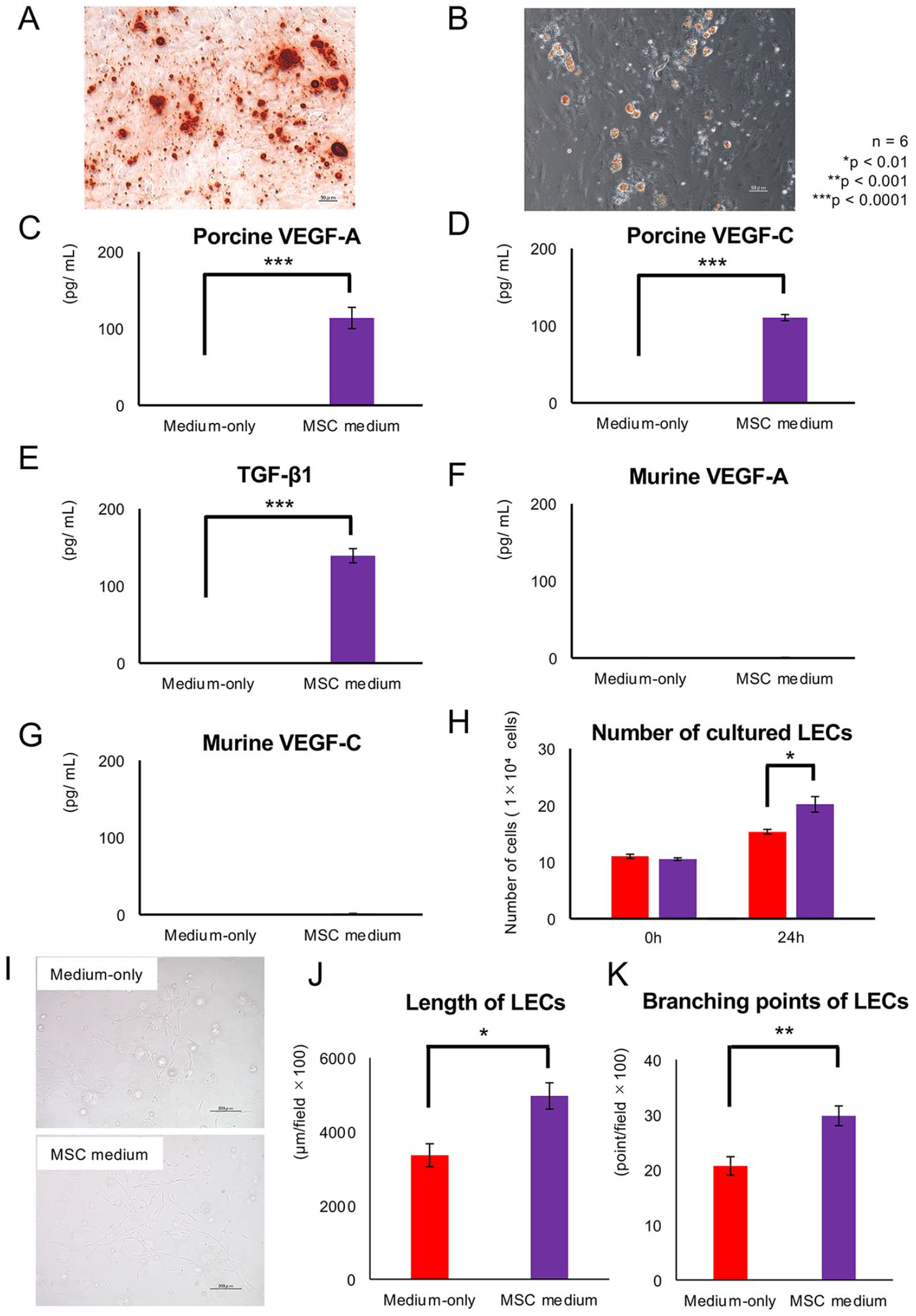
Characteristics of NpBM-MSCs as promoters of lymphangiogenesis. Multipotency of the NpBM-MSCs: differentiation into osteoblasts (A) and adipocytes (B). Concentrations of porcine VEGF-A (C), VEGF-C (D), and TGF-β1 (E) in the NpBM-MSC medium after 24 h of culture. Concentrations of murine VEGF-A (F) and VEGF-C (G) in the NpBM-MSC medium. (H) Number of cultured LECs after incubation with the NpBM-MSC–conditioned medium. The number of seeded cells was 1 × 104 cells. (I) LECs after 3D culture with nonconditioned medium (upper panel) and NpBM-MSC–conditioned medium (lower panel) for 72 h. (J) The sum of the long-axis lengths of the LECs per field (×100). (K) Branching points per field (×100). Scale bars = 50 µm (A and B), 200 µm (I). Nonconditioned medium (red) and MSC-conditioned medium (purple). n = 6 per group. *P < 0.01, **P < 0.001, ***P < 0.0001.
NpBM-MSC Xenotransplantation Ameliorates Lymphedema Via the Promotion of Lymphangiogenesis and a Recovery of Lymphatic Flow
Next, we evaluated the potential therapeutic effects of NpBM-MSCs using the lymphedema model. Fig. 3A shows the changes in hind limb lymphedema that occurred following NpBM-MSC transplantation. All the mice in both the negative control and MSC transplantation groups developed lymphedema, showing increases in hind limb circumference and volume 7 days after the induction, at the time of MSC transplantation (POD 0). In the MSC transplantation group, significant amelioration of the lymphedema was apparent on POD 7 (Fig. 3A), and the femoral circumference and volume of these mice were significantly smaller (had been normalized) on POD 14. In contrast, the lymphedema was worse in the negative control group on POD 7 (Fig. 3A). This gradually improved with time, but neither the femoral circumference nor volume normalized. The mean femoral circumferences on POD 14 were 46.7 ± 1.1 vs 39.8 ± 0.7 mm, respectively (P < 0.01; Fig. 3B), and the hind limb volumes on the same day were 203.2 ± 7.5 vs 128.2 ± 4.7 mm³, respectively (P < 0.001; Fig. 3C). Both the circumference and volume of the hind limbs of the MSC transplantation group remained smaller than those of the negative control group (Fig. 3B, C).
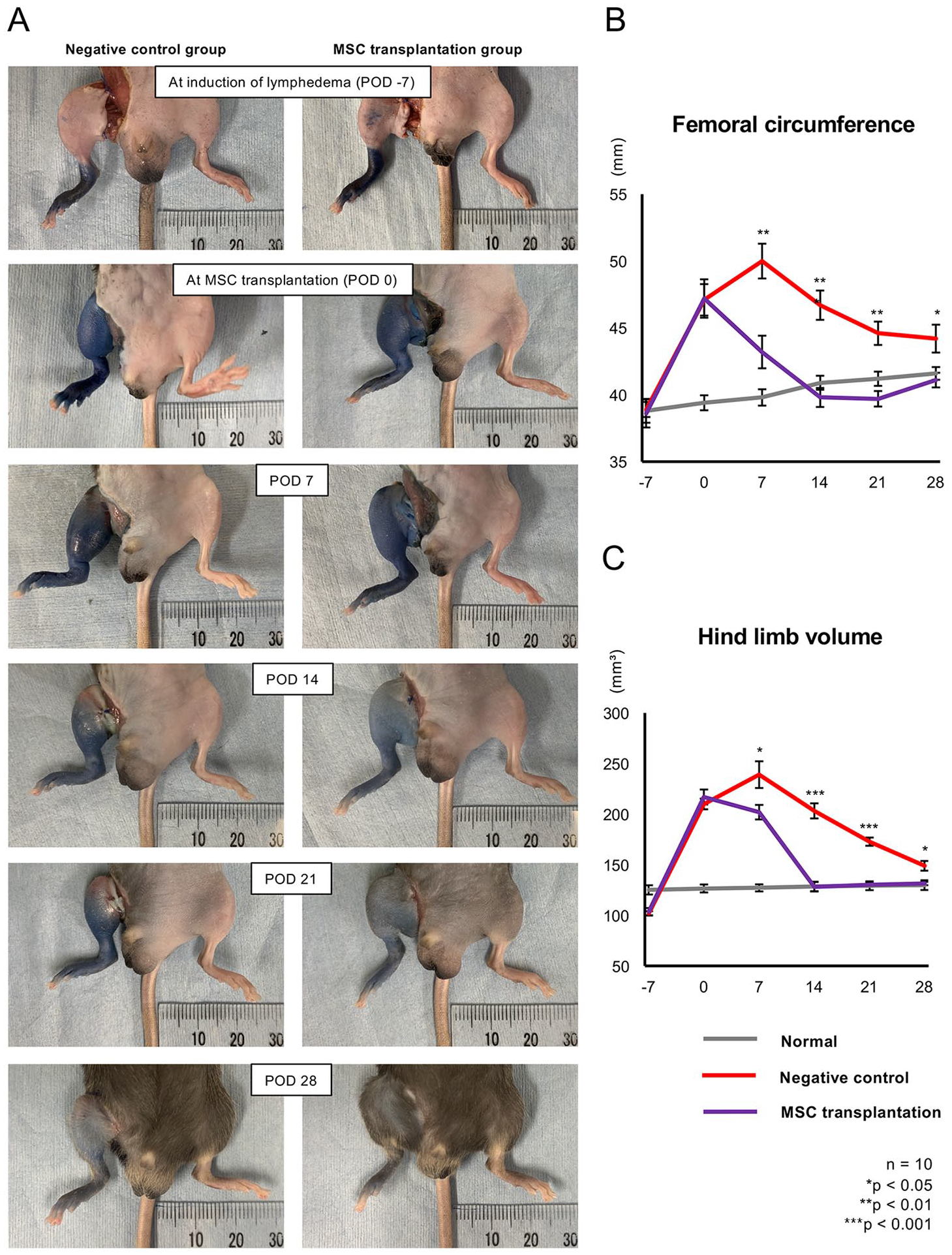
Assessment of mouse hind limbs following NpBM-MSC xenotransplantation. (A) Changes in the hind limbs of mice following the induction of lymphedema and NpBM-MSC xenotransplantation. Left panel: negative control group; right panel: MSC transplantation group. The date of induction was defined as postoperative day (POD) −7, and the date of MSC transplantation as POD 0. Thigh circumference (B) and hind limb volume (C) of the negative control (red), MSC transplantation (purple), and normal (gray) groups. n = 10 per group. *P < 0.05, **P < 0.01, ***P < 0.001.
Next, we assessed the lymphatic flow in the hind limbs of the mice to evaluate the therapeutic effects of NpBM-MSC transplantation using the NIRF imaging system. In this way, we visualized the ICG-enhanced lymphatic vessels and lymph nodes in the hind limbs of the mice in the normal group (Fig. 4A). In the MSC transplantation group, the lymphatic flow was visualized on the central side of the excised area of skin on POD 14. In addition, with reference to the commonly used classification of the severity of lymphedema, the negative control group showed a stardust or diffuse pattern on POD 14 (Fig. 4B), which is considered to reflect severe lymphedema. However, the MSC transplantation group showed a linear pattern, indicative of the recovery of lymphatic flow and amelioration of the edema (Fig. 4B). On POD 28, the luminance at the site was maximal. On POD 28 in particular, the MSC transplantation group showed not only greater but also more extensive luminance than the negative control group (Fig. 4B). The CTCF/area was significantly higher after POD 14, and peaked at POD 28, in the MSC transplantation group (10.3 ± 1.0 vs 3.9 ± 0.6, P < 0.01; Fig. 4C).
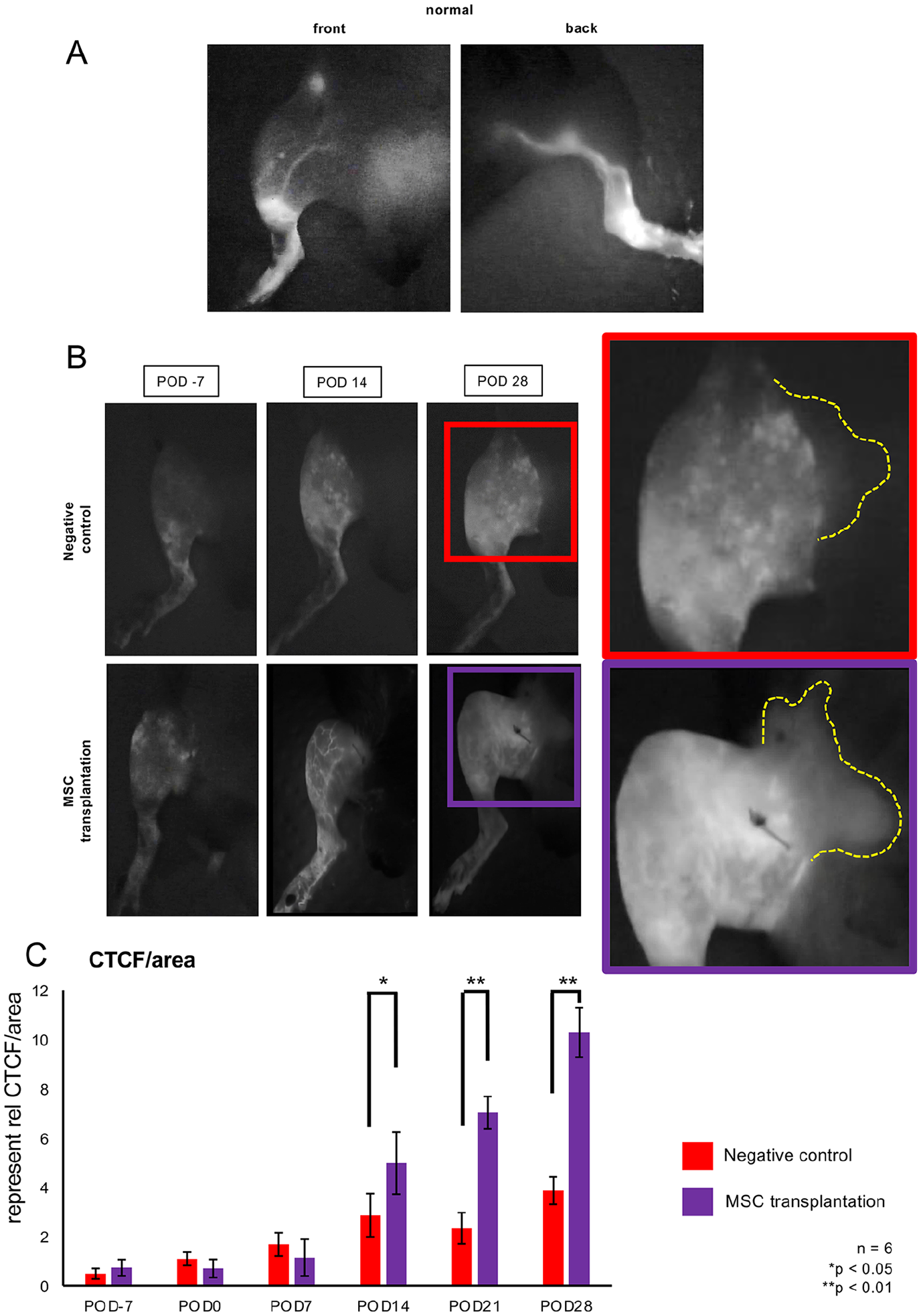
Assessment of lymphatic flow in the hind limbs of the mice using near-infrared fluorescence (NIRF) imaging. (A) NIRF images of a hind limb from the normal group. Left panel: cranial aspect; right panel: caudal aspect. (B) NIRF images of a hind limb on PODs −7, 14, and 28 from the negative control (upper panel) and MSC transplantation (lower panel) groups. (C) Quantification of fluorescence in the hind limbs of negative control and MSC transplantation groups using ImageJ, as CTCF/area (integrated density − (area of selected region × mean fluorescence of five background readings)/area of the selected region). *P < 0.05, **P < 0.01.
Fig. 5A shows the histological images of the lymphatic vessels in the hind limbs of the mice, stained for Pdp. These showed that the number of lymphatic vessels in the MSC transplantation group gradually increased with time and was significantly larger than that in the negative control group after POD 14 (Fig. 5A, B). Regarding the maximum short-axis diameter of the lymphatic vessels, which is indicative of the dilation of the lymphatic vessels, it became larger in the negative control group than in the normal group. The diameters of the MSC transplantation group were also significantly larger than those of the negative control group on PODs 7 and 14 (P < 0.05 and < 0.001, respectively; Fig. 5A and C). We consider this dilation was reflected on the promotion of lymphatic drainage and removal of internal pressure in the hind limb with lymphedema. Furthermore, the total area of the lymphatic vessels was significantly larger in the MSC transplantation group than that in the negative control group on the same days (P < 0.001; Fig. 5A, D).
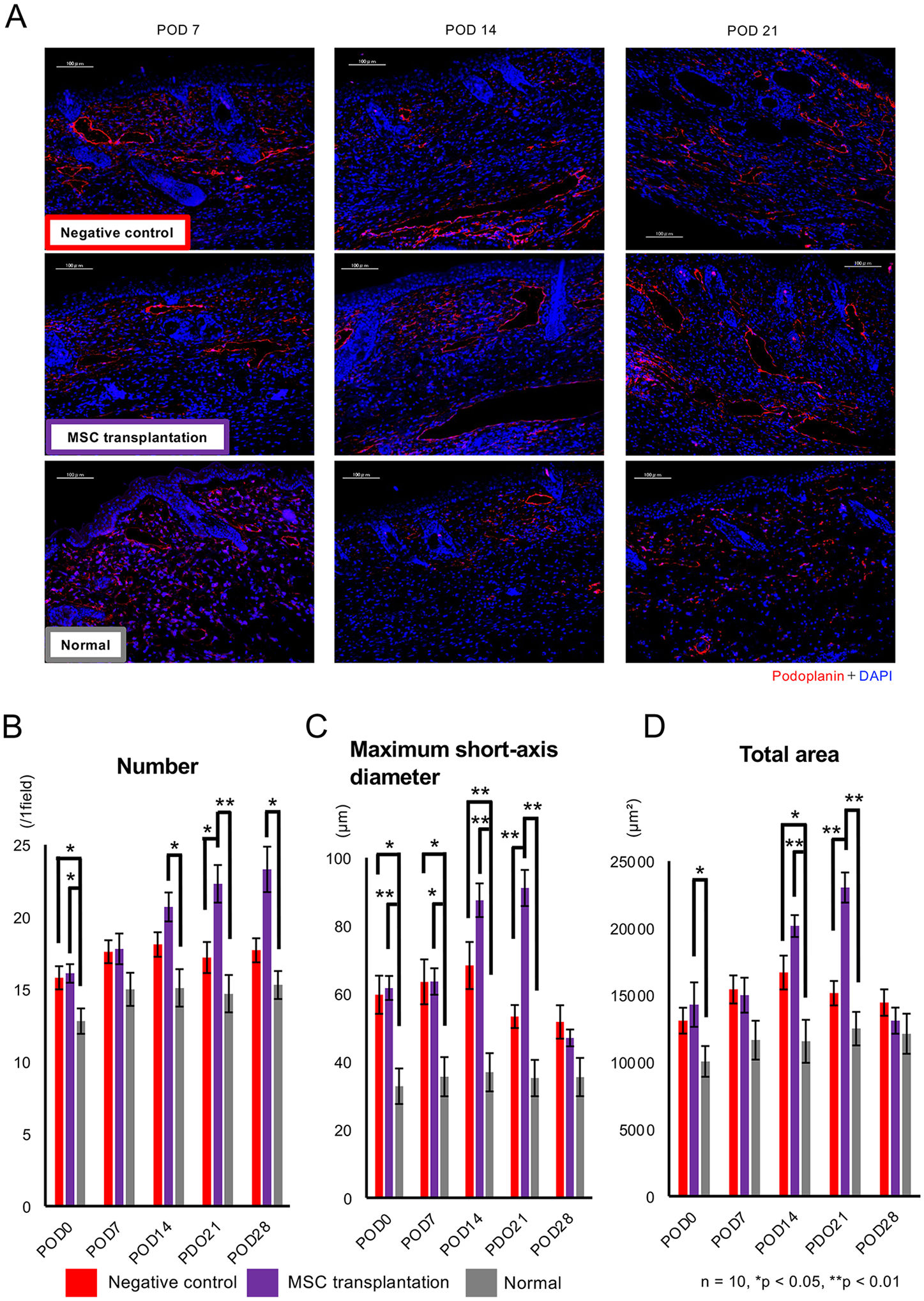
Histological assessment of mouse lymphatic vessels after MSC transplantation. (A) Immunofluorescence staining for podoplanin (red) in thigh samples obtained from mice in the negative control (upper), MSC transplantation (middle), and normal (lower) groups on PODs 7, 14, and 28. Scale bars: 100 µm. The number (B), maximum short-axis diameter (C), and total area (D) of lymphatic vessels in the negative control (red), MSC transplantation (purple), and normal (gray) groups. n = 10 per group. *P < 0.05, **P < 0.01.
These data indicate that NpBM-MSC xenotransplantation ameliorates lymphedema by promoting lymphangiogenesis in the hind limbs of mice, facilitating the recovery of lymphatic flow at the site of the intervention.
Xenotransplanted NpBM-MSCs Increase the Expression of Both Porcine and Murine VEGFs Until They Are Rejected
We next assessed the condition of the NpBM-MSCs following xenotransplantation. Fig. 6A shows the transplanted NpBMs, stained for SLA class I, in the hind limb. NpBM-MSCs were identified on both POD 3 and 7, but there were fewer on POD 14. However, there were no SLA class I-positive cells in the hind limbs of the negative control group (Fig. 6B). Therefore, we speculate that the NpBM-MSCs were viable and facilitated lymphangiogenesis by POD 7 but had been rejected by POD 14.
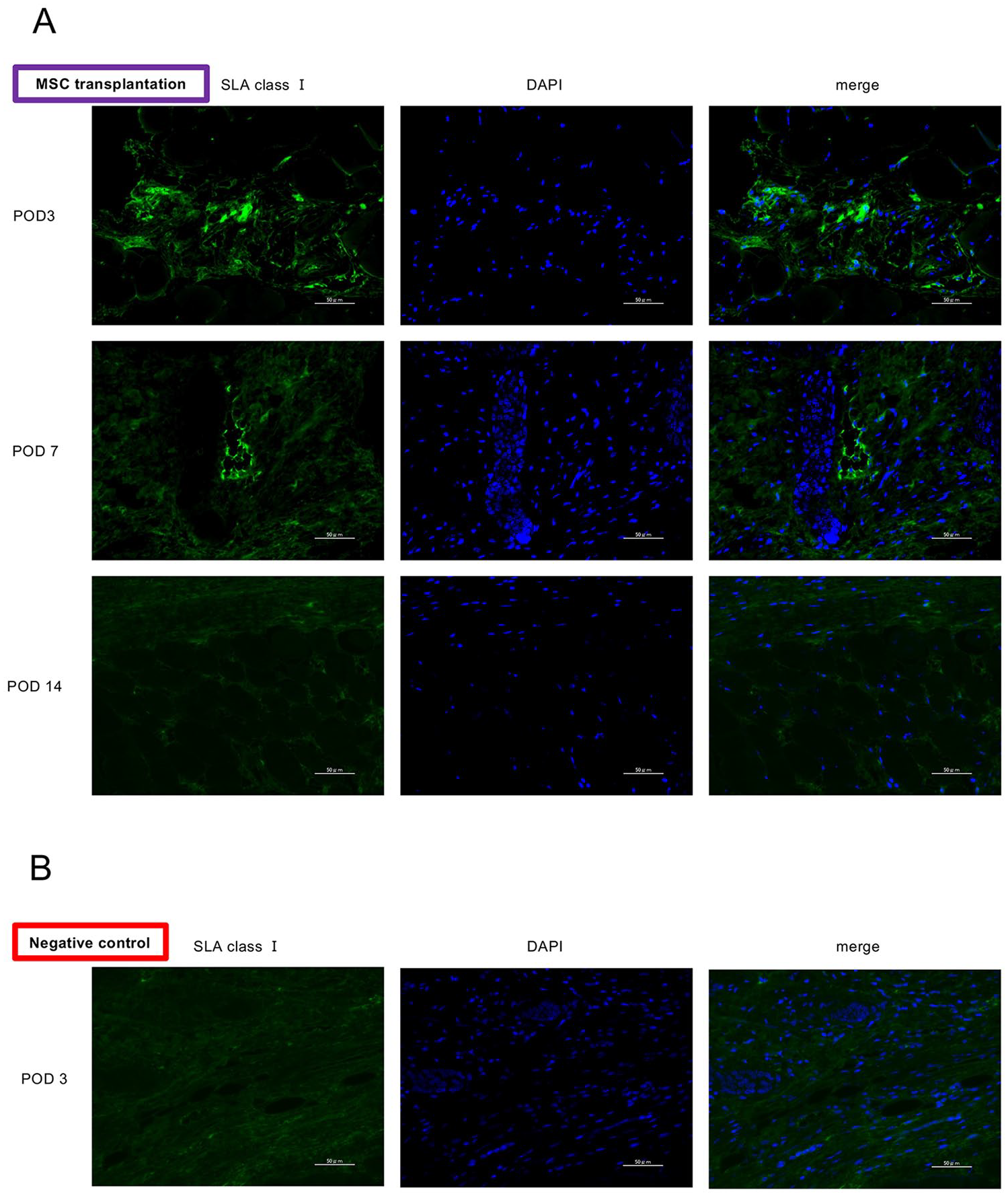
Assessment of the engraftment and rejection of xenotransplanted porcine MSCs. (A) Histological findings associated with transplanted NpBM-MSCs, immunostained for swine leukocyte antigen (SLA) class I, in the hind limbs of mice in the MSC transplantation group on PODs 3, 7, and 14. (B) Equivalent findings for the negative control group on POD3. Scale bars: 50 µm.
We next evaluated the expression of VEGF genes in the hind limbs of the mice. Fig. 7 shows the relative expression of porcine and murine VEGF-A, VEGF-C, and VEGF-D and TGF-β1 mRNAs in the hind limbs of the negative control, MSC transplantation, and normal groups. The expression of porcine Vegfa, Vegfc, Vegfd, and Tgfb1 was not detected in the negative control and normal groups (Fig. 7A–D). However, the expression of porcine Vegf and Tgfb1 was present on POD 1 in the MSC transplantation group. Although the expression of Vegfa and Vegfd was lower on POD 3 (Fig. 7A, C), that of porcine Vegfc and Tgfb1 continued to increase until POD 7 (Fig. 7B, D). Furthermore, NpBM-MSC xenotransplantation affected the expression of murine Vegf genes. Although an increase in the expression of murine Vegf genes was identified in the negative control group, the expression in the MSC transplantation group was higher (murine Vegfa: P < 0.05 on POD 1, P < 0.05 on POD 3; murine Vegfc: P < 0.01 on POD 1, P < 0.01 on POD 3; murine Vegfd: P < 0.05 on POD 3; murine Tgfb1: P < 0.05 on POD 3; Fig. 7E–H). The expression of murine Vegf genes other than Vegfc and Tgfb1 in the MSC transplantation group was lower after POD 7, and that of murine Vegfc was also low on POD 14.
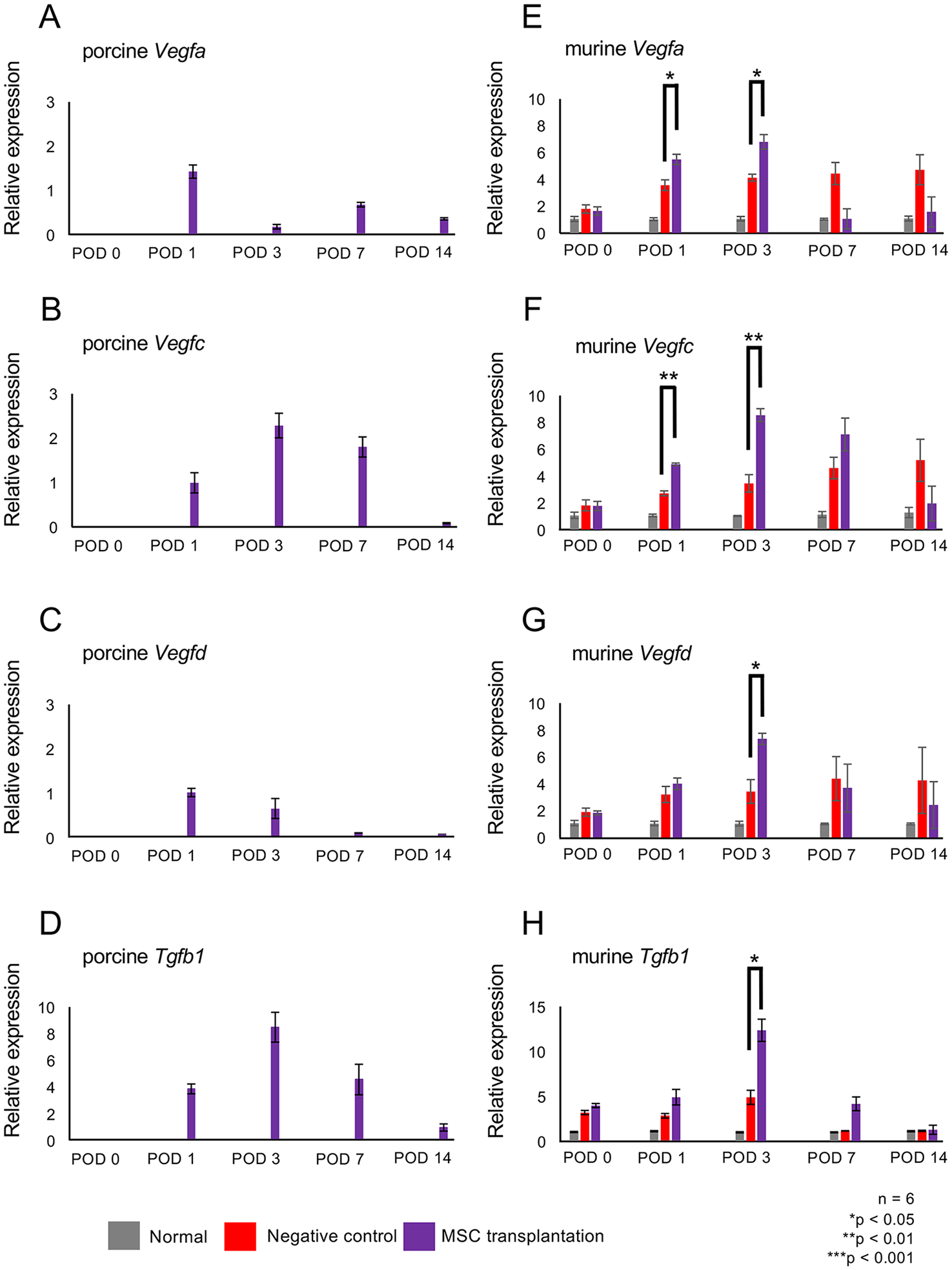
Expression of genes encoding lymphangiogenetic factors at the transplantation site. mRNA expression of porcine VEGF-A (A), VEGF-C (B), VEGF-D (C), and TGF-β1 (D). mRNA expression of murine VEGF-A (E), VEGF-C (F), VEGF-D (G), and TGF-β1 (H) in the thighs of mice in the normal (gray), negative control (red), and MSC transplantation (purple) groups on PODs 0, 1, 3, 7, and 14. Expression was quantified using the 2−ΔΔCT method, and Actb was used as the reference gene. n = 6. *P < 0.05, **P < 0.01, ***P < 0.001.
In summary, NpBM-MSC xenotransplantation increases the expression of not only the porcine but also the murine Vegf genes, and especially of Vegfc, until rejection.
Discussion
In the present study, we have shown that NpBM-MSC xenotransplantation corrects lymphedema within 14 days using our mouse model of lymphedema, and this effect persists for over a month 30 . The therapeutic effects of the NpBM-MSC xenotransplantation were found to be exerted through the paracrine promotion of lymphangiogenesis. We found that NpBM-MSCs secrete angiogenic and lymphangiogenic factors, including VEGF-A, VEGF-C, and TGF-β1 and increase the expression of porcine Vegf genes and Tgfb1 at the transplantation sites, until they are rejected, at approximately POD 14. VEGF-C is a representative lymphangiogenic factor that induces both angiogenesis and lymphangiogenesis and promotes lymphatic sprouting 35 . Furthermore, the xenotransplantation of NpBM-MSCs increases the expression of genes encoding not only porcine but also murine growth factors. As a result, the resolution of intestinal edema might be caused by a recovery of lymphatic flow and an amelioration of lymphedema. Thus, MSC transplantation has beneficial effects on lymphedema in mice over a significant period of time despite their rejection approximately 2 weeks after transplantation. We made similar findings in our previous study, which revealed the beneficial effects of xenogeneic NpBM-MSCs in a model of lower-limb ischemia 36 .
The xenotransplanted NpBM-MSCs were viable at the site of transplantation for at least 7 days, produced growth factors including VEGF-C, and initiated lymphangiogenesis locally. After the initiation of lymphangiogenesis, the transplanted NpBM-MSCs were eliminated by the immune responses of the recipient animals. However, sustained lymphangiogenesis was achieved through an increase in the production of growth factors by the recipient. Indeed, as shown in Fig. 6, greater production of murine VEGFs occurred at the same time as the expression of porcine VEGFs, which might have contributed to the subsequent lymphangiogenesis 37 . We speculate that porcine NpBM-MSCs might directly promote lymphangiogenesis via the secretion of endogenous VEGFs and indirectly contribute to this lymphangiogenesis via the production of such growth factors in recipient tissues, including by fibroblasts, myocytes, and lymphatic vessels 32 . These indirect effects might be important therapeutically because the direct effects on the secretion of porcine VEGFs are transient, owing to the rejection of the transplanted cells.
Some studies have considered that porcine growth factors, including TGF-β1, induce angiogenesis through the upregulation of VEGFs38,39, which are secreted by cells at the transplantation site, thereby indirectly supporting lymphangiogenesis. The long-lasting lymphangiogenetic effect of NpBM-MSC xenotransplantation is a desirable characteristic of an agent for the treatment of lymphedema. In contrast to autologous MSCs, porcine MSCs can be administered whenever required. If the safety of porcine cell administration, such as the prevention of zoonosis owing to porcine endogenous retrovirus40,41, can be established, this approach may represent a suitable option for the treatment of lymphedema. Furthermore, the therapeutic effects of NpBM-MSCs might be prolonged by the use of gene-edited pigs in which carbohydrate antigens are eliminated, and the innate immune response is prevented 28 . For the success of the xenotransplantation of NpBM-MSCs, quality control is important, including the culture medium conditions, the transplantation technique, the age of the donor, and the duration of culture42,43. Regarding the quality control, measurement of repressor/activator protein (Rap) 1 before NpBM-MSCs transplantation can be a useful parameter. Recent studies have shown that status of MSC is tightly regulated by telomerase-associated proteins Rap1/nuclear factor-kappaB (NF-κB) signaling pathway. They revealed that this signaling pathway in MSCs regulated the paracrine effects 44 , and Rap1 deficiency attenuated paracrine effects leading to impairment of immunosuppressive potency 45 . Therefore, it would be informative to check RAP1 level in NpBM-MSCs for prediting the outcome of the transplantation. Furthermore, the extension of the lifespan of grafts will be a pivotal challenge with respect to the sustainable therapeutic effects of xenotransplanted NpBM-MSCs. And it is the limitation of this study. The extension of the graft survival without rejection would contribute to the cure of lymphedema. We consider that this might be feasible in the near future through the use of gene-edited pigs, as shown in recent clinical trials of porcine heart xenotransplantation28,46.
As the other limitation, we have no data showing the differentiation of NpBM-MSCs into lymphatic vessels. Previous studies anout MSC therapy for heart failure indicated that transplanted MSCs promoted endogenous myocardial regenerartion, angiogenesis, and immunomodulation via paracrine effects47,48. On the other hand, Lian et al. 49 showed transplanted iPS cells–derived MSCs induced vascular and muscle regeneration via both direct differentiation and paracrine mechanisms in their limb ischemia model. We predicted that xenotransplanted NpBM-MSCs were rejected before engraftment and differentiation; however, the assessment about direct differentiation will be done in future study.
We consider that these therapeutic effects of NpBM-MSCs are seen in MSCs derived from other tissue, especially adipose tissue. For example, Ogino et al. 50 attempted to elucidate the therapeutic effects of adipose-derived stem cell (ADSC) transplantation for lymphedema animal model. They showed that ADSC transplantation promoted lymphangiogenesis and compromised fibrosis in hind limb with lymphedema via upregulation of Tgfb1 and downregulation of Colla1 50 .
In conclusion, the xenotransplantation of NpBM-MSCs ameliorates lymphedema in mice. We consider that this approach will be a reliable option for the clinical treatment of lymphedema in the future.
Footnotes
Acknowledgements
Data availability statement
The data that support the findings of this study are available from the corresponding author (NS and SK), upon reasonable request.
Ethical Approval
The animals were cared for and the experimental procedures were performed in accordance with the Principles of Laboratory Animal Care of the Guide for the Care and Use of Laboratory Animals, 8th edition (National Research Council, 2011). The experimental protocol was approved by the Animal Care and Use Committee of Fukuoka University (approval number: 2113110).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by an intramural grant to SK.