Abstract
Early destruction of donor islet grafts due to an instant blood-mediated inflammatory reaction (IBMIR) remains a major obstacle in islet transplantation. Thrombomodulin plays an important role in limiting coagulation and inflammatory events through a variety of effects. In this study, we investigated the ability of thrombomodulin (TM), when reconstituted as a liposomal formulation, to enhance early syngeneic islet engraftment by minimizing or abrogating the IBMIR. Administration of TM significantly improved early engraftment of syngeneic islets after intraportal transplantation in diabetic mice. In the absence of treatment, conversion to euglycemia was observed among 46.6% (7/15) of recipients. In contrast, administration of TM led to euglycemia in 93.3% (14/15) of recipients (p = 0.0142). Recipients that received TM exhibited a lower incidence of primary nonfunction and better glucose control over a 30-day period after transplantation. Fibrin deposition (p < 0.05), neutrophil infiltration (p < 0.05), expression of TNF-α and IL-β mRNA (p < 0.05), as well as NF-κB activity (p < 0.05) were significantly reduced in the liver of islet recipients having been treated with liposomal TM. These data demonstrate that TM significantly improves early syngeneic islet engraftment through effects that target both coagulation and inflammatory pathways.
Introduction
Although three decades have passed since the first human islet transplantation was performed for type 1 diabetes mellitus, clinical outcomes remain suboptimal (63,72). In a recent report only 12% of islet recipients remain insulin free 1000 days after transplantation, despite implementation of the Edmonton Protocol (76). As many as 50—75% of islets fail to engraft following intraportal islet transplantation (13). Additionally, primary nonfunction (PNF) and early nonimmune islet destruction, which has been observed both in animal models and in clinical trials, remain major hurdles in islet transplantation (17,18).
Unsatisfactory clinical outcomes of human islet transplantation have been attributed to the instant blood-mediated inflammatory reaction (IBMIR). Korsgren and colleagues demonstrated that IBMIR is initiated upon intraportal infusion of islets (55,61). Specifically, in animal models and in recent clinical reports, marked activation of coagulation has been noted after islet infusion, despite the presence of heparin in the infusate, as indicated by increases in thrombin—antithrombin (TAT) complexes, prothrombin activation fragments, and fibrinopeptide A. Notably, thrombin is a direct mediator of inflammation, acting as a chemoattractant for neutrophils and monocytes and stimulating endothelial cells to express monocyte chemoattractant protein-1 (MCP-1) and other chemokines. Thrombin also induces endothelial cell expression of ICAM-1, VCAM-1, E-selectin, and P-selectin, as well as platelet-activating factor, all of which lead to further recruitment of platelets and leukocytes to the graft site (12). Likewise, by-products of the thrombin response, including fibrinogen degradation products and fibrin, also act as chemoattractants and serve to localize this inflammatory response by adhesion-dependent processes. Furthermore, thrombin-activated endothelial cells and leuokocytes express oxygen free radicals, IL-β, TNF-α, IFN-γ, and iNOS, which can damage islets, inducing either functional impairment or death (15).
Thrombomodulin (TM) is a multiple domain-containing proteoglycan that is expressed endothelial cells (60). The endothelial growth factor-like repeats of thrombomodulin provide cofactor function for effective activation of protein C by thrombin (69). Activated protein C (APC) has been reported to play an important role in the downregulation of coagulation and inflammatory reactions (20—22,24,25,28,32,44,52,57,59,66,68). Specially, APC preserved functional islet mass after intraportal transplantation (13). Furthermore, the soluble lectin-like domain of TM also plays a direct role in modulating inflammation (14,34,45,65,74). However, the potential application of exogenously administered TM in islet transplantation has not been investigated. Focusing on the unique biological properties of thrombomodulin, we hypothesized that administration of TM would enhance islet engraftment.
In this study, we examined the capacity of exogenously administered TM to increase islet engraftment in a murine syngeneic model of intraportal islet transplantation. TM improved islet engraftment in vivo, which was associated with a marked reduction in intraportal fibrin formation, neutrophil infiltration, and cytokine production.
Materials and Methods
Animals
Male C57BL/6J (B6) mice (8 weeks old, Jackson Laboratory Bar Harbor, ME) were used as islet recipients and donors. All animal studies followed local guidelines at Emory University. The B6 mice were made chemically diabetic by intraperitoneal (IP) injection of 200 mg/kg streptozotocin in citrate-buffered saline and screened as previously described for the development of diabetes. Mice whose nonfasting blood glucose was over 250 mg/dl on two consecutive measurements were considered diabetic (16).
Preparation of TM Vesicles
Large unilamellar vesicles (LUV) were prepared from a lipid solution of 12 mM 1-palmitoyl-2-oleoyl-snglycero-3-phosphocholine (POPC) (Avanti Polar Lipids, Inc., Alabaster, AL) in PBS (80 mM Na2HPO4, 20 mM NaH2PO4, 100 mM NaCl, pH 7.4) by four successive freeze/thaw/vortex cycles using liquid N2 and a 45°C water bath. A total of 20 μg of rabbit thrombomodulin (TM) was added to 100 μl of the lipid solution and mixed gently for 1 h at room temperature before it was extruded 21 times, each through two back-to-back 600-nm and then 100-nm polycarbonate filters (42).
Islet Isolation
B6 mouse pancreata were removed after distension with collagenase P (1 mg/ml, Roche, Indianapolis, IN) through the common bile duct. Following digestion, islets were purified by a Ficoll discontinuous gradient (1.108, 1.096, and 1.037; Mediatech Inc, Herndon, VA). Isolated islets were cultured for 48 h in RPMI-1640 supplemented by 10% FCS, l-glutamine (2 mM), and penicillin (100 U/ml), streptomycin (100 μg/ml), and amphotericin B (0.25 μg/ml) (Mediatech Inc., Herndon, VA). Viability was evaluated using a Live/Dead Cell Viability/Cytotoxicity Kit (Molecular Probes, Inc., Eugene, OR) and only isolations with >90% viability were used for transplantation.
Islet Transplantation
Diabetic B6 mice underwent intraportal islet transplantation after receiving ketamine (95 mg/kg) and xylazine (5 mg/kg) IM for anesthesia. In brief, 250 or 500 B6 islets were infused in a total volume of 200 μl into the recipient liver through the portal vein using a 27-gauge insulin syringe, as previously described (49).
Experimental Groups
Mice undergoing islet transplantation were monitored by measuring nonfasting blood glucose daily for 2 weeks by using an ACCU-CHECK glucose monitor. Euglycemia was defined as nonfasting blood glucose less than 200 mg/dl on 2 consecutive days. B6 diabetic mice were randomly assigned into one of two groups (n = 15 mice/group) that received either islets alone or islets along with TM at 1 mg/kg administered intravenously (IV) 2 min before transplantation.
Histopathologic Examination
Livers were harvested 6 h after intraportal islet transplantation from both TM-treated and nontreated groups, fixed in 10% formalin, and embedded in paraffin. Step sections of 5 μm at 100-μm intervals were obtained. Five consecutive sections were collected from each step section for histological examination. Neutrophil (PMN) and fibrin staining was performed after deparaffinization and rehydration using standard immunohistochemical procedures. A commercially available kit (91-C, Sigma-Aldrich) was used to stain for neutrophils using the naphthol AS-D chloroacetate esterase procedure. Biotinylated goat anti-mouse fibrinogen antibody (Accurate Chemical & Scientific Corporation) was used to stain for fibrin staining after quenching endogenous peroxidase activity using Dual Endogeneous Enzyme Block (Dako), blocking endogenous avidin and biotin with the Biotin Blocking System (Dako), and limiting nonspecific binding using 10% normal goat serum (Vector). At least 50 islets from each liver sample were examined for fibrin and neutrophil accumulation. Fibrin deposition around each islet was scored using a semiquantitative scheme (0: no fibrin; 1: mild; 2: moderate; and 3: severe fibrin deposition). Neutrophil infiltration was characterized by counting PMNs around individual islets.
Real-Time RT-PCR
Liver samples (n = 4—6 livers per group) were harvested 24 h after islet transplantation and subjected to total RNA extraction to determine levels of TNF-α and IL-1β mRNA. Three to four tissue segments (~3 mm3 cube/segment) were obtained from visibly ischemic regions of each liver and processed using Trizol reagent (Invitrogen). RNA was digested using RNase-free DNase (Qiagen) and extracted using an RNeasy Mini kit (Qiagen). cDNA was synthesized from 1 μg of total RNA using a High Capacity cDNA Reverse Transcription kit (Applied Biosystems). TNF-α and IL-1β expressions were investigated using appropriate primers and probes (Taqman Universal PCR MasterMix, Taqman Gene Expression Assays, Applied Biosystems) with 50 ng of reverse transcribed total RNA in a total volume of 20 μl. PCR was run in a 7900HT Sequence Detection System (Applied Biosystems). The relative amount of TNF-α and IL-1β mRNA was normalized to the expression level of 18S and to the corresponding mRNA level in normal livers that did not receive islets or TM. The relative fold increase was calculated using the comparative threshold cycle method.
Preparation of Nuclear Extract and Determination of NF-κB Activation
Nuclear extracts from recipient liver tissue (100 mg) were prepared using a nuclear extraction kit, as described by the manufacturer (Active Motif). Protein concentrations in nuclear extracts were determined using the Bradford assay (Bio-Rad). The TransAM NF-κB p65 transcription factor assay kit (Active Motif) was used to monitor NF-κB activation in liver tissues. The kit consists of a 96-well plate in which oligonucleotides containing the NF-κB consensus site (5′-GGGACTTTCC-3′) have been bound. The active form of NF-κB in the nuclear extract binds to this oligonucleotide and is recognized by a primary antibody specific to the activated form of p65 of NF-κB. An HRP-conjugated secondary antibody provides a colorimetric readout, which was quantified spectrophotometry (Multiskan Spectrum with Skanlt software, Thermo Electron Corporation). Results were expressed as fold increase over the sham operation control group.
Statistical Analysis
All data are expressed as mean ± SD. Comparison between groups was performed by a Student's t-test. Statistical significance was established at p < 0.05. Analysis of eugylcemic conversion over time was performed by Kaplan-Meier method with a Logrank test to assess statistical significance (Prism Software, GraphPad, Inc.).
Results
Liposomal Formulations of TM Improve Engraftment of Syngeneic Islet Grafts After Intraportal Transplantation
To investigate the effect of liposomal TM on islet early engraftment, a suboptimal mass of syngeneic islets (250 islets/mouse) was transplanted intraportally. In the control group, nonfasting blood glucose levels decreased transiently within 3 days after transplantation, but soon rebounded to hyperglycemic levels (>250 mg/dl) 4 days after transplantation (Fig. 1A). In the TM-treated group, euglycemia was achieved on average 5 days after transplantation, which was maintained throughout a 30-day observation (Fig. 1A). Euglycemic conversion was achieved in 46.6% (7/15) of recipients that had received 250 islets in the absence of TM, while treatment with TM increased the proportion of mice that attained euglycemia to 93.3% (14/15; p = 0.0142) (Fig. 1B). No instances of late graft failure were observed in the TM treatment group (Fig. 1B). As demonstrated in Figure 1C, primary nonfunction was observed among eight mice in the control group, but in only one recipient in the TM group (p = 0.0142). Nonetheless, among those mice that became euglycemia, the time to conversion was similar in both groups.
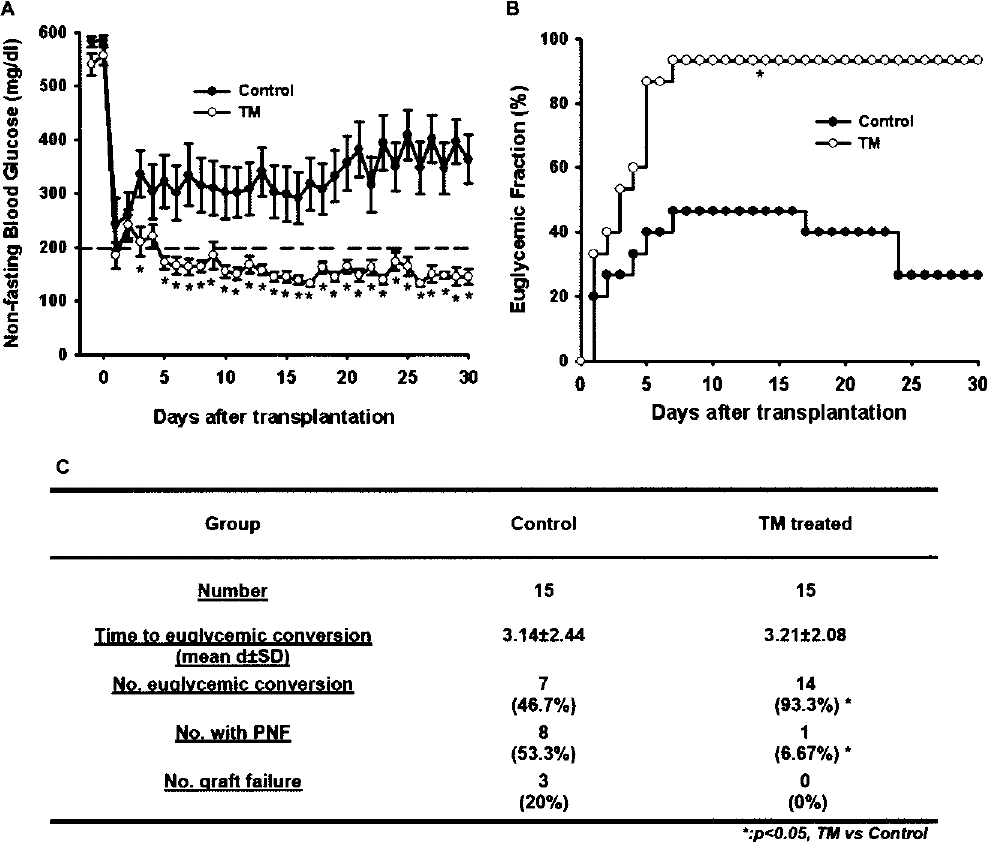
TM improved the engraftment of syngeneic islet grafts after intraportal transplantation. (A) Pancreatic islets from B6 mice were transplanted into male B6 mice that were rendered diabetic by streptozotocin (200 mg/kg, IP). Diabetic animals received either 250 islets alone (n = 15) or 250 islets along with TM/lipid vesicles (n = 15) by intraportal injection. In the group receiving TM/lipid vesicles, TM at a dose of 1 mg/kg was administered IV via the jugular vein 2 min prior to islet infusion. Nonfasting blood glucose levels (mean ± SE) are plotted as a function of time after transplantation for all mice enrolled within each group (*p < 0.05 by Student's t-test for 250 islets vs. 250 islets + TM). (B) Serial blood glucose levels were measured and conversion to euglycemia was defined as glucose levels <200 mg/dl for >2 consecutive days (*p < 0.05 by chi-square analysis). (C) Outcome of syngeneic islet graft (*p < 0.05 by chi-square analysis).
TM Reduces Fibrin Deposition and Neutrophil Infiltration After Intraportal Islet Transplantation
Livers were harvested and examined for both fibrin and neutrophil infiltration in the vicinity of intraportal islets 6 h after transplantation. Using a semiquantitative scoring scheme (Fig. 2A), we observed that treatment with TM reduced peri-islet fibrin deposition from an average score of 2.4 ± 0.2 to 1.1 ± 0.2 (p < 0.0001) (Fig. 2B). Likewise, neutrophil infiltration was reduced from 55.8 ± 8.5 PMNs/islet to 20.6 ± 3.1 PMNs/islet among animals receiving an infusion of liposomal TM (p < 0.0001) (Fig. 3B).
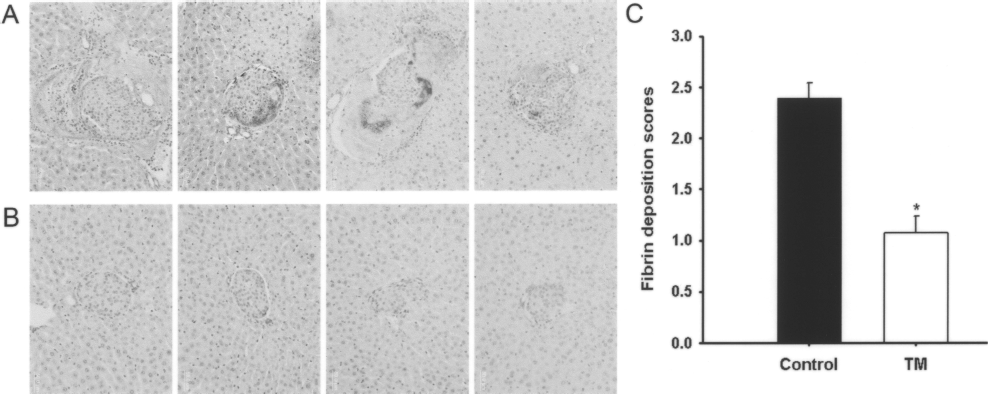
Fibrin deposition after intraportal transplantation of syngeneic mouse islets. Images of islets infused intraportally in the absence (A) or presence (B) of TM/lipid vesicles (original magnification 20x). (C) Distribution of fibrin scores 6 h after intraportal transplantation of syngeneic mouse islets (500 islets/mouse) with or without TM (n = 5). Fibrin scores for each group expressed as mean ± SD, *p < 0.05 (n = 5).

Neutrophil infiltration of syngeneic islets after intraportal transplantation. Images of islets infused intraportally in the absence (A) or presence (B) of TM/lipid vesicles (original magnification 20x). The accumulation of neutrophils was identified by naphthol AS-D chloroacetate esterase staining. (C) Neutrophils/islet for each group are expressed as mean ± SD, *p < 0.05 (n = 5).
TM Decreases Hepatic mRNA Expression of TNF-α and IL-1β
Administration of TM significantly decreased the expression of proinflammatory cytokines, TNF-α and IL-1p, as revealed by an analysis of mRNA in recipient livers 24 h after transplantation (Fig. 4). Transplantation of islets, without TM administration, resulted in a three-to fourfold increase in TNF-α and IL-1β mRNA expression. In contrast, infusion of TM significantly reduced the expression of TNF-α and IL-1β mRNA to levels observed in normal livers that had not received islet grafts. Statistical analysis confirmed that the reduction in the TNF-α mRNA expression was statistically significant (p < 0.05).

Thrombomodulin suppressed local inflammatory responses after intraportal islet transplantation. Five hundred B6 mouse islets were transplanted with or without TM/vesicles. Livers were harvested 24 h after transplantation. The mRNA expression of TNF-α and IL-β was quantified by real-time RT-PCR. Data are presented as mean ± SD, *p < 0.05 (n = 4—5).
TM Reduces Hepatic NF-κB Activity
The activity of NF-κB in liver of islet recipients was increased by nearly threefold compared to the activity measured in the liver of sham operated controls. In contrast, infusion of TM significantly reduced NF-κB activity (p < 0.05) (Fig. 5).

Effect of TM on NF-κB activation in nuclear extracts of recipients' liver tissue. Five hundred B6 mouse islets were transplanted with or without TM/vesicles. Livers were harvested 24 h after transplantation and examined for NF-κB activity. Results are expressed as mean ± SD, *p < 0.05 TM versus control (n = 4—6).
Discussion
There are extensive two-way interactions between coagulation and inflammation pathways. Coagulation not only affects inflammation activity, but also inflammation markedly leads to activation of coagulation. The main points of interaction linking coagulation and inflammation are the tissue factor pathway, thrombin, the protein C system, and the fibrinolytic system. Activated coagulation proteases and physiological anticoagulants or components of the plasminogen—plasmin system can modulate inflammation by activation of specific cell surface receptors and, in return, proinflammatory cytokines and chemokines can influence prothrombotic responses (12,19—23,26,27,29,53,58,73).
The intensive “cross-talk” between coagulation and inflammation contributes to the pathogenesis of early islet graft loss following intraportal transplantation and has been referred to as the IBMIR (5). Marked activation of coagulation has been noted after islet infusion, despite the presence of heparin in the infusate, which leads to significant levels of early islet destruction, as well as overt and subclinical episodes of portal vein thrombosis (33,64). Prothrombotic triggers include the expression of tissue factor either by transplanted islets or by locally injured endothelial cells (55,61). As a consequence of thrombin generation, activated platelets bind to the islet surface and further amplify the coagulation cascade. Furthermore, nonspecific activation and dysfunction of intrahepatic endothelial cells, characterized by upregulation of intracellular adhesion molecule (ICAM)-1 and P-selectin, along with production of proinflammatory cytokines and other factors, such as TNF-α, IL-1β, INF-γ, and nitric oxide (NO), have been demonstrated in vitro and in vivo after islet transplantation. These effects contribute to the complex network of events that ultimately lead to early graft loss (3,4,8,10,30,31,47,50,54,77).
To obviate the effects of IBMIR, most centers performing allogeneic islet transplantation currently use systemic heparin at the time of transplantation. Korsgren and colleagues (6) have observed that heparin prevented islet-induced coagulation in an ex vivo model, but at an eightfold higher concentration than that used clinically (4 U/ml blood). Furthermore, despite heparin administration at 4 U/ml blood, extensive platelet and fibrin formation, as well infiltration of CD11b+ cells continued to be observed on blood-exposed islets. Finally, even if one were to accept the risk of bleeding complications to be anticipated at a dosing level of 300 U heparin/kg, systemic heparin has a half-life of 1 h and is therefore active for only a few hours. Thus, the potential therapeutic impact of intravenously administered heparin is limited both by its systemic anticoagulant activity that increases the risk of bleeding complications and short half-life.
As an alternative strategy, Contreras et al. (13) have reported that exogenous APC, at a 10-fold higher dose than that recommended for clinical use, reduces the loss of functional islet mass in a murine model of intraportal islet transplantation. Activated protein C together with its cofactor protein S inactivates two coagulation factors, Va and VIIIa, thereby preventing the generation of Xa and thrombin. As such, APC inhibits mononuclear phagocyte (MØ) activation and production of proinflammatory cytokines, such as TNF-α and IL-1β, which are known to be cytotoxic to islets (9,38,39,43), and limits neutrophil binding to selectins (9,38—40,43). APC also suppresses MØ-dependent proliferative responses of T cells, inhibits mixed lymphocyte responses of human and rat mononuclear cells, and prolongs xenograft survival in a guinea pig to rat cardiac transplant model (9). Significantly, primate studies have shown that activation of the protein C anticoagulant pathway carries little risk of bleeding (41). When activated protein C was infused at doses sufficient to prevent venous or arterial thrombosis, excessive bleeding from surgical wounds did not occur. Nonetheless, the half-life of APC is approximately 10—20 min, which would necessitate repetitive dosing for a sustained effect (7).
Beyond the APC, TM has several distinct functions. TM intervenes in thrombosis by changing the function of thrombin from a procoagulant to an anticoagulant, inhibiting the prothrombinase complex (factor Xa—factor Va—phospholipids—Ca2+) and displays AT-III-dependent heparin-like function. There are additional indirect mechanisms by which TM may exert anti-inflammatory effects. First, thrombin-activatable fibrinolysis inhibitor (TAFI), which inhibits C5a and C3a, requires the presence of TM—thrombin complex (11). Furthermore, the proinflammatory properties of thrombin are reversed when it is associated with TM. The C-type lectin-like domain of TM might also have a direct role in modulating inflammation by inhibiting leukocyte transmigration and suppressing endothelial cell activation. Finally, TM suppresses expression of ICAM-1 (75).
Several studies have documented that various forms of solubilized thrombomodulin are capable of activating protein C in vivo. For example, Kumada et al. (51) observed that TM prolonged the survival of mice in a model of thrombin-induced thromboembolism. Other studies have demonstrated that administration of TM reduces endotoxin-induced lung injury (70,71) and thrombosis from disseminated intravascular coagulation (DIC) in rats and monkeys (1,35,56), limits thrombosis in an arteriovenous shunt model in rats (2), attenuates thrombotic glomerulonephritis in rats (46), and decreases trauma-induced spinal cord injury (67). Early phase clinical studies have recently noted that soluble thrombomodulin may promote the resolution of DIC (62) and reduce the incidence of deep venous thrombosis after hip replacement (48).
Prior studies have confirmed that IBMIR is a barrier to engraftment in murine models of intraportal islet transplantation (13,36,37,78). In this study, we confirmed the effect of TM on early engraftment and long-term survival of islet grafts in a murine syngeneic model of intraportal islet transplantation. This report demonstrates that TM enhances both engraftment and prolongs long-term islet graft survival through effects that simultaneously target both coagulation and inflammation.
Footnotes
Acknowledgment
Supported by grants from the NIH and JDRF.
