Abstract
This study was undertaken to address the effects of fetal mesencephalic tissue transplantation on the serotonin system in a rat model of Parkinson's disease (PD) while also investigating the usefulness of 4-[18F]-ADAM (a serotonin transporter imaging agent) coupled with micro-PET for imaging serotonin transporters (SERTs). A PD model was induced by unilateral injection of 6-hydroxydopamine (6-OHDA) into the right medial forebrain bundle of the nigrostriatal pathway, while cell transplantation was performed via intrastriatal injection of mesencephalic brain tissue dissected from embryonic (E14) rats. The 4-[18F]-ADAM/micro-PET scanning was performed following both 6-OHDA lesioning and transplantation. Immunohistochemistry (IHC) studies were also performed following the final PET scan, and the results were compared to show a 17–43% decrease in the specific uptake ratio (SUR) and a 23–52% decrease in serotonin transporter immunoreactivity (SERT-ir) within various brain regions on the lesioned side. The number of methamphetamine-induced rotations also decreased significantly at the 4th week postgraft. In addition, striatal SUR and the SERT-ir levels were restored to 77% and 83% 5 weeks postgraft. These results suggest that Parkinson's disease also affects the serotonergic system, while both the dopaminergic and serotonergic systems can be partially restored in a rat model of PD after E14 mesencephalic tissue transplantation. In addition, we have also determined that 4-[18F]-ADAM/micro-PET can be used to detect serotonergic neuron loss, monitor the progress of Parkinson's disease, and oversee the effectiveness of therapy.
Introduction
Parkinson's disease (PD) is characterized by a progressive loss of neurons within the subcortical monoaminergic nuclei, particularly the substantia nigra. Its clinical symptoms include rigidity, resting tremor, bradykinesia, and postural instability (15,20,38). While considered primarily a disease of dopaminergic neurons, some symptoms of PD may result from loss of non-dopaminergic neurons including serotonergic neurons in the median raphe, noradrenergic neurons in the locus coeruleus, cholinergic neurons in the nucleus basalis, as well as other pigmented and brainstem nuclei (2,6). Recently, in order to better elucidate the various aspects of PD, positron emission tomography (PET) and single photon emission computed tomography (SPECT) have provided powerful noninvasive methods for examining changes in monoaminergic systems and monitoring the progress of disease, as well as the effectiveness of therapies (9,25,29,34).
The involvement of the dopaminergic system in PD has been well documented by several studies using both presynaptic (12,13,31,33,44,45) and postsynaptic (9,24) dopaminergic radioligands. Nonetheless, relatively little is known about the involvement of the serotonergic system in PD, although postmortem studies have revealed that serotonergic neurons are affected by PD (28), while a recent SPECT imaging study has found reductions in [123I]-2-([2-({dimethylamino}methyl)phenyl]thio)-5-iodophenylamine ([123I]ADAM) uptake in the thalamus and striatum of parkinsonian monkey brain (30). PET and SPECT imaging using trans-(+)-1,2,3,5,6,10b-Hexahydro-6-(4-([11C]methylthio)-phenyl)pyrrolo-(2,1-a)-isoquinoline ([11C](+) McN5652), 11C-labeled 3-amino-4-(2-dimethylaminomethyl-phenylsulfanyl)-benzonitrile ([11C]DASB), and the no-selective SERT imaging agent 123I-labeled 2b-carbomethoxy-3b-(4-[123I]-iodophenyl)tropane ([123I]-b-CIT) have also shown a reduction in serotonin transporter (SERT) density in the caudate and putamen of PD brains (18,25,27). However, SERT density in the midbrain region may not be affected in the early stages of PD (27). To investigate the involvement of serotonergic and other monoaminergic neurons in PD, Thakur et al. (51) and Wang et al. (52) used rat models and micro-PET imaging with either SERT-selective or vesicular monoamine transporter 2 (VMT2)-selective imaging agents. Currently, [11C]DASB (53) is one of the few SERT imaging agents available for human studies. However, with its short half-life (T1/2 = 20 min), it has limited clinical value. Hence, we used the 18F-labeled SERT imaging agent N,N-dimethyl-2-(2-amino-4-18F-fluorophenylthio)-benzylamine (4-[18F]-ADAM; T1/2 = 110 min) (7,21,29,35,43,47,48) together with micro-PET to monitor the effectiveness of neural transplantation in the parkinsonian rat model and to evaluate the validity of this technique by comparing the results to those found via behavioral testing and immunohistochemistry.
Previously, neurons derived from embryonic stem cells or fetal mesencephalic tissues have been used in cell replacement therapy for PD (26,31,33). For this study, we used mesencephalic tissue from embryonic rats, making sure to include the substantia nigra and raphe nuclei, which contain the cell bodies of both dopamine and serotonin neurons (10), respectively. We predicted that both dopamine- and serotonin-containing cells will be able to engraft within the striatum, resulting in the recovery of SERT levels, in addition to the recovery of locomotor function and dopaminergic neuron levels.
Materials and Methods
Radiopharmaceutical
The 4-[18F]-ADAM was synthesized as previously reported (43). Briefly, nucleophilic fluorination of the dinitro precursor with K[18F]/K2.2.2 (Sigma-Aldrich, St. Louis, MO, USA) followed by reduction with NaBH4/Cu(OAc)2 (Sigma-Aldrich) and purification with high-performance liquid chromatography (HPLC; Waters Chromatography Division, Millipore Corporation) produced the desired compound with ~5% radiochemical yield (EOB) in a synthesis time of 120 min from the end of bombardment (EOB). The radiochemical yield of 4-[18F]-ADAM was increased to ~15% if using a different precursor (21). The chemical and radiochemical purities were >95% and the specific activity was >3 Ci/μmol.
Animals
All animal study protocols were approved by the institutional animal care and use committee. Eight-week-old male Sprague-Dawley rats (BioLASCO Taiwan Co., Ltd., Taipei, Taiwan) weighing between 280 and 300 g (mean: 290 ± 10 g) were housed in the animal facilities at the National Defense Medical Center (Taipei, Taiwan) at a constant temperature of 23 ± 2°C and a controlled light/dark cycle (light from 7:00 AM to 7:00 PM). Rats were maintained on a complete pellet diet and tap water for 1 week prior to the studies.
The Parkinsonian Rat Model
The Parkinson's disease (PD) model (n = 18) and sham control rats (n = 6) were generated as previously reported (19,40,46,50,55,56). Briefly, male Sprague–Dawley rats were anesthetized with 7% chloral hydrate (0.5 ml/kg, IP; Riedel-de Haën, Seelze, Germany) followed by unilateral injection of 6-hydroxydopamine (6-OHDA, 20 μg in 4-μl saline containing 0.02% ascorbic acid; both Sigma-Aldrich) into the ascending nigrostriatal pathway (4.4 mm posterior to bregma, 1.2 mm lateral to the midline, and 7.8 mm below the dura). Sham control rats received a similar injection containing only 0.02% ascorbic acid.
Behavioral Testing
To examine the locomotor effect of 6-OHDA lesions, rats were injected with methamphetamine (5 mg/kg, IP; Sigma-Aldrich) 2 weeks following 6-OHDA lesions as well as 4 weeks following transplantation (22). The rats were then subjected to at least 100 min of rotation test using an automated rotometer system (MED Associates, Inc., St. Albans, VT, USA). Following the initial test, only those rats showing >300 turns/h were used in subsequent experiments (PET imaging, immunohistochemistry, and mesencephalic tissue transplantation).
Micro-PET Imaging
Both 3 weeks after 6-OHDA injection and 5 weeks after mesencephalic tissue transplantation (shown below), 4-[18F]-ADAM (14.8–18.5 MBq; 0.4–0.5 mCi) was injected intravenously into the tail veins of PD rats for PET imaging. Micro-PET image acquisition was performed according to previous reports (29,35) with minor modifications. Briefly, rats were anesthetized by passive inhalation of a mixture of isoflurane/oxygen (5% isoflurane for induction and 2% for maintenance). One hour after tracer injection, PET image acquisition was started using a micro-PET R4 scanner (Concorde MicroSystems, Knoxville, TN, USA). The energy window was set at 350–650 keV. The Fourier rebinning algorithm and two-dimensional filtered back-projection (ramp filter with cutoff at Nyquist frequency) were employed to reconstruct the images. All images were analyzed using ASIPro VM 6.3.3.1 software (Concorde MicroSystems). Volumes of interest (VOIs) for the frontal cortex, striatum, thalamus, hypothalamus, midbrain, and cerebellum were drawn manually from the reconstructed PET images with the use of a rat brain atlas (42) and magnetic resonance images as described previously (29,35). The specific uptake ratio (SUR) was expressed as (target region – cerebellum)/cerebellum (29,35).
Mesencephalic Tissue Preparation and Transplantation
Six out of eighteen PD rats were transplanted with rat mesencephalic tissue that was dissected from embryonic day 14 (E14) fetal brains (50), with the dissection area selected according to the protocols of Dunnett et al. (10), with minor modifications (a larger selecting area than that of Dunnett et al.). Care was taken to ensure that the dissection tissue blocks contained the ventral mesencephalon and partial ventral pontine raphe, which contain both dopaminergic and serotonergic cell bodies, respectively. The tissue blocks were maintained in Hank's buffered salt solution (HBSS; Gibco, Grand Island, NY, USA) during the transplantation procedure. The grafting tissues were cut into small pieces (55) and then transplanted into the ipsilateral striatum (0.5 mm posterior to bregma, 2.5 mm lateral to the midline, and 4.5 mm below the dura) using a glass microtube. Each PD rat received graft from a single embryonic mesencephalic tissue sample. All rats received behavioral testing and PET imaging at the 4th and 5th week after mesencephalic tissue transplantation. They were then sacrificed for immunohistochemistry (IHC) studies the day after PET imaging.
Immunohistochemistry
One day after final micro-PET imaging, sham control (n = 6), PD (n = 6), and the grafted rats (n = 6) were sacrificed for IHC studies, performed as previously reported (16,50,55,56). Briefly, the rats were deeply anesthetized with chloral hydrate prior to perfusion with normal saline followed by 4% paraformaldehyde (Sigma-Aldrich). The brains were removed followed by postfixation and cryo-protection. Coronal sections (30 μm) were first sliced with a cryostat (Leica CM 3050; Leica Microsystem, Taipei, Taiwan), then rinsed with PBS and treated with 1% H2O2 (Calbiochem, Torrey Pines, CA) in PBS for 0.5 h. After incubation in blocking solution [0.5% Triton X-100 (Sigma-Aldrich) and 3% normal goat serum (Vector, Burlingame, CA, USA) in PBS], sections were incubated with either rabbit anti-tyrosine hydroxylase (TH) or rabbit anti-serotonin transporter (SERT) antibody (both at 1:2,000 dilution; Millipore Corporation, Billerica, MA, USA) at 4°C overnight. Thereafter, sections were rinsed with PBS and further incubated with goat anti-rabbit biotinylated IgG (1:200; Vector) for 1 h. Sections were then incubated with avidin–biotin complex (1:200; Vectastain ABC kit, Vector) for an additional 1 h. Subsequently, sections were visualized using 0.05% 3,3-diaminobenzidine (DAB; Sigma-Aldrich) for 6 min (TH) or 2.5 min (SERT), washed three times with PBS, and finally mounted on gelatin-coated glass slides.
Three IHC sections were performed on every other section from six consecutive sections of each brain region for optical density (OD) measurements. Photographs of target regions and the reference region (corpus callosum) (14,23,41) were collected using a color CCD camera attached to the microscope [OPTIPHOT-2 (10×) or MICROPHOT-FXA (100× and 200×), Nikon, Tokyo, Japan]. The photographs were then converted into an 8-bit gray scale (0–255 gray levels) for semiquantification (37,39). The OD values of SERT-ir and TH-ir were determined using image analysis software Image-Pro Plus v. 6.0 (Media Cybernetics, Inc., Bethesda, MD, USA). The OD ratio of the target region relative to the reference region was calculated and expressed as OD ratio = (OD of target region – OD of corpus callosum)/OD of corpus callosum.
Statistical Analysis
The SURs of PET and relative OD ratio from IHC-stained images in different brain regions were compared by Student's t test for two independent groups or oneway ANOVA (analysis of variance, Bonferroni posttest) for multiple comparisons. A value of p < 0.01 was considered statistically significant. Data are presented as mean ± SD.
Results
The 4-[18F]-ADAM/micro-PET imaging (Fig. 1) of normal rat brain showed that the 4-[18F]-ADAM uptake was the same in both hemispheres (Fig. 1A). In addition, the PET imaging of the sham group also showed no difference between the sham-operated and contralateral hemispheres (Fig. 1B). In the PD rat, PET images showed obviously lower 4-[18F]-ADAM uptake in the 6-OHDA lesioned hemisphere compared to the contralateral side, which was seen both overall (Fig. 1C, D) and for various brain regions within the lesioned side (p < 0.01) (Fig. 2). Specifically, the average SUR values of 4-[18F]-ADAM in various brain regions of the lesioned hemisphere were 17–43% lower than what was found for the corresponding regions in the contralateral side (Table 1).

PET images of 4-[18F]-ADAM uptake in rat brain. Transverse images collected 60–90 min after injection of N,N-dimethyl-2-(2-amino-4-18F-fluorophenylthio)-benzylamine (4-[18F]-ADAM) show that 4-[18F]-ADAM uptake (which was reduced on the lesioned side of the brain) is restored after transplantation of embryonic mesencephalic tissue into the striatum. The 4-[18F]-ADAM uptake shows no difference between (A) normal and (B) sham-operated rat brains. The arrows in (C) and (D) indicate the lesioned side, and the arrow in (E) indicates the grafted side. PET, positron emission tomography; 6-OHDA, 6-hydroxydopamine; SERT, serotonin transporter.

Analysis of serotonin transporter levels via PET imaging. The uptake of 4-[18F]-ADAM expressed by specific uptake ratios (SURs) was used to compare SERT levels on the 6-OHDA-treated and contralateral hemispheres of rat brains 3 weeks following lesions. ∗∗p < 0.01; p value refers to ipsilateral versus contralateral in the 6-OHDA lesion group.
SURs of 4-[18F]-ADAM/PET Images and OD Ratios of SERT-ir of 6-OHDA-Lesioned Parkinsonian and Sham-Operated Rats

Values are mean ± SD; n = 6.
p < 0.01, the p value of Student's t test refers to ipsilateral versus contralateral in the 6-OHDA lesion group. SUR refers to the specific uptake ratio of 4-[18F]-ADAM; OD ratio refers to the optical density ratio of SERT immunohistochemistry. Decrement (%) presents the data comparison between the contralateral and ipsilateral sides in 6-OHDA-lesioned rats. The 4-[18F]-ADAM/PET studies were performed at 3rd week after 6-hydroxydopamine (6-OHDA) lesion. The next day, following the N,N-dimethyl-2-(2-amino-4-18F-fluorophenylthio)-benzylamine (4-[18F]-ADAM)/positron emission tomography (PET) studies (over 24 h), the immunohistochemistry studies were carried out. SERT, serotonin transporter.
Immunocytochemistry results showed decreased tyrosine hydroxylase immunoreactivity (TH-ir) in both the striatum and substantia nigra of the 6-OHDA lesioned side compared to those of the contralateral side (Fig. 3). Similarly, there was also an obvious decline in serotonin transporter immunoreactivity (SERT-ir) in brain regions within the lesioned side compared to those within the contralateral side (Fig. 4). Analysis of signal density found decreases of 23–52% in the OD ratios of the frontal cortex, striatum, thalamus, hypothalamus, and midbrain within the lesioned relative to those of the contralateral side in 6-OHDA lesioned rats (p < 0.01) (Table 1; Fig. 5). However, in the sham group, there was no reduction in TH-ir within the control injection side compared to the contralateral (Fig. 3C, D), and SERT-ir levels in various brain regions of the sham-operated side (Fig. 4K–O) was comparable to that of the control side (Fig. 4F–J).
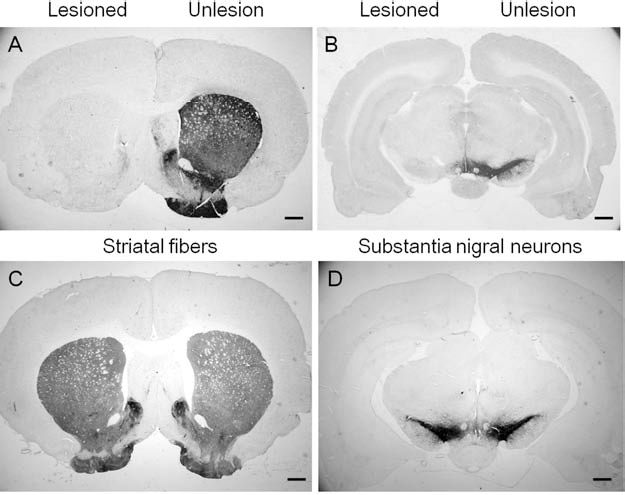
Tyrosine hydroxylase immunoreactivity (TH-ir) in both 6-OHDA-lesioned brains and vehicle injection controls. TH-ir in both the striatum (A) and substantia nigra (B) are depleted after 6-OHDA lesioning. In the sham control group, the TH-ir of sham-operated side in striatum (C) and substantia nigra (D) was the same as the contralateral side. Scale bars: 1 mm.

Serotonin transporter immunoreactivity (SERT-ir) in 6-OHDA-lesioned or sham-operated brain. The depletion of serotonin fibers in 6-OHDA-lesioned brains is shown on the lesioned (A–E) compared to the unlesioned side (F–J), with sham controls shown in (K–O) (scale bars: 100 μm). (A, F, K) Frontal cortex; (B, G, L) striatum; (C, H, M) thalamus; (D, I, N) hypothalamus; (E, J, O) dorsal raphe nucleus.

Results of the analysis of OD ratios of SERT-ir signals in 6-OHDA-lesioned brain. Compared with the contralateral side, 23–52% fewer serotonergic fibers were observed on the lesioned side. ∗∗p < 0.01; p value refers to ipsilateral versus contralateral in the 6-OHDA lesion group. OD, optical density.
At the 4th week following E14 embryonic mesencephalic tissue transplantation, the number of methamphe tamine-induced rotations in the grafted rats was significantly reduced to 71 ± 54 turns/h relative to the 442 ± 130 turns/h shown in the pregraft test (p < 0.01) (Fig. 6), while the sham group displayed 2 ± 2.90 turns/h. Furthermore, PET imaging of the PD rats at 8 weeks after 6-OHDAlesioning found significantly lower uptake of 4-[18F]-ADAM in the lesioned striatum compared to the contralateral side (p < 0.01) (Figs. 1D and 7; Table 2), indicating that decreases in SERT remain at least 8 weeks following 6-OHDA lesioning. However, in the grafted rat group, the uptake of 4-[18F]-ADAM was significantly increased within the grafted striatum at the 5th week posttransplantation (p < 0.01) (Figs. 1E and 7; Table 2), showing recovery of SERT from 58% (8th week after lesion) to 77% (5th week postgraft) of that found in the corresponding striatums of the contralateral hemisphere.

Methamphetamine-induced rotation behavior of 6-OHDA-lesioned rats both prior to and 4 weeks following transplantation of embryonic mesencephalic tissue. Sham control rats showed 2 ± 2.90 turns/h in the rotation behavior test. ∗∗p < 0.01.

The specific uptake ratio (SUR) of 4-[18F]-ADAM in the control, 6-OHDA-lesioned and embryonic mesencephalic tissue-grafted striatum in PD rats. Comparison of the specific uptake ratio (SUR) of 4-[18F]-ADAM in the 6-OHDA-lesioned striatum in rats 5 weeks following embryonic mesencephalic tissue transplantation to 6-OHDA-lesioned rats that did not receive transplants. ∗∗p < 0.01.
SURs of 4-[18F]-ADAM and OD Ratios of SERT-ir and TH-ir in the Striatum in the Three Groups

n=6 in each group, mean ± SD. Control side refers to the unlesioned side 5 weeks since embryonic mesencephalic tissue was transplanted.
p < 0.01, lesion side at the 8th week after lesion compared with the control side;
p < 0.01, graft side at the 5th week postgraft compared with the lesion side at the 8th week after lesion. TH, tyrosine hydroxylase.
After 4-[18F]-ADAM/micro-PET imaging, the grafted (5th week postgraft) and PD (8 weeks postlesion) rats were sacrificed for immunochemistry staining. In the PD rats, the OD ratios of TH-ir were 0.07 ± 0.03 in the lesioned striatum compared to 0.74 ± 0.08 in the contralateral side (Table 2). In the grafted rats, however, the OD ratio of TH-ir had increased to 0.41 ± 0.04 in the grafted striatum (Table 2; Fig. 8A). These results suggest a dopaminergic recovery from 9% (8th week after lesion) to 55% (5th week postgraft) relative to the corresponding contralateral striatum (Figs. 8 and 9). Similarly, 5 weeks following transplantation, the SERT OD ratio was 0.38 ± 0.11 in the lesioned striatum of PD rats, compared to 0.75 ± 0.06 in the contralateral striatum, while increasing to 0.62 ± 0.07 in the grafted striatum (Table 2; Fig. 8C), indicating a recovery from 50% (8th week after lesion) to 83% (5th week postgraft) relative to the contralateral value (Fig. 9). Photomicrographs of SERT-ir in the striatum at the 5th week after transplantation (Fig. 8C) showed abundant SERT-positive fibers with interspersion of some SERT-positive cell bodies among the grafted area (Fig. 8D, E). On the contrary, SERT-positive fibers in sites away from the grafted area (Fig. 8G) were sparser compared to the corresponding contralateral sites (Fig. 8F, H).

Immunohistochemical photomicrographs of both tyrosine hydroxylase immunoreactivity (TH-ir) and serotonin transporter immunoreactivity (SERT-ir) in 6-OHDA-lesioned striatum 5 weeks following embryonic mesencephalic tissue transplantation. (A) TH-ir in the grafted area. (B) TH-ir cell bodies and TH-ir fibers in the grafted area. (C) SERT-ir in the grafted area. (D–H) SERT-ir in striatum of parkinsonian rats 5 weeks after embryonic mesencephalic tissue transplantation. The SERT-ir is higher within (D) and near (E) the grafted area than on the control side (F). The SERT-ir at a site far from the grafted area (G) is less than on the control side (H). White arrows (D) point to SERT-positive cell bodies within the grafted area. Scale bars: 1 mm (A, C); 50 μm (D); 100 μm (B, E–H).

Tyrosine hydroxylase immunoreactivity (TH-ir) and serotonin transporter immunoreactivity (SERT-ir) following lesion and transplantation. Comparison of tyrosine hydroxylase immunoreactivity (TH-ir) and serotonin transporter immunoreactivity (SERT-ir) between 8 weeks following 6-OHDA lesions and 5 weeks following embryonic mesencephalic transplantation. SERT-ir and TH-ir signals in the striatums of PD and grafted rats were expressed as the OD ratios (n = 6; ##,∗∗p < 0.01).
Discussion
In this study, we found that a single dose of 6-OHDA injected into the rat brain resulted in SERT decrease in various brain regions both 3 and 8 weeks following treatment, which suggests that SERT expression does not recover following a 6-OHDA lesion, similar to what has been seen in the dopaminergic system (11), which is that in addition to long-term decreases in DA levels. In our previous study, the [123I]ADAM uptake is markedly lower than normal value within the effected brain regions both 2 (34) and 6 (30) years postlesion in monkey. We did not see any changes in SERT levels in the sham control group, which only received an injection of 0.02% ascorbic acid, which demonstrates that the effects of SERT reduction were not due to injection injury.
The mechanisms of 6-OHDA's effect on serotonergic neurons are not well defined. Initially, 6-OHDA was considered a specific dopaminergic neurotoxin that acts via the dopamine transporter (5,17), but there is now evidence that it can damage the serotonergic system as well. For example, unilateral 6-OHDA lesioning of the nigrostriatal bundle in marmosets reduced serotonin concentration in both the caudate (72%) and putamen (60%) (3). Studies utilizing [123I]ADAM/SPECT imaging have also indicated that SERT is decreased in monkey brains following 6-OHDA injections into the medial forebrain bundle (34). Other studies have shown that dopamine transporter blockers inhibit the uptake of 6-OHDA into dopaminergic cells but do not prevent neurotoxicity (1,49). This was later found to induce the generation of extracellular auto-oxidation products, causing damage to dopaminergic neurons by oxidative stress (4). Furthermore, 6-OHDA is toxic to PC12 cells that lack dopamine transporter (4,8) as well as cultured mesencephalic cells (32). Also, the addition of antioxidants to culture media containing PC12 cells could provide sufficient protection against neurotoxicity induced by high concentrations (600 μM) of 6-OHDA (8,54). This would provide a sufficient mechanism for 6-OHDA to damage serotonergic neurons despite their lack of dopamine transporters.
There are various radioligands that can be used in conjunction with PET imaging to examine the effects of PD. For example, imaging of parkinsonian rats using [18F] AV-133 has demonstrated the involvement of vesicular monoamine transporter 2 (52). Our 4-[18F]-ADAM uptake studies as well as the [18F] dihydrotetrabenazine (DTBZ) uptake study by Thakur et al. (51) suggest the involvement of SERT in parkinsonism. This is confirmed by the present immunohistochemical and behavioral studies as well as the studies by Kish (28) in humans, establishing that 4-[18F]-ADAM is an effective imaging agent for studying PD using PET.
A recent study using [18F]fluorodopa (FDOPA)/PET to monitor the long-term clinical outcome on fetal dopamine cell implantation in PD patients found that its clinical benefit, as well as graft viability, could be sustained up to 4 years after transplantation, with imaging changes reliably correlated with clinical outcome over the entire posttransplantation period (36). These results are similar to those in the present study, which demonstrate the utility of 4-[18F]-ADAM/micro-PET to detect and monitor the efficacy of embryonic mesencephalic tissue transplantation in PD rats. Four weeks after transplantation, PD rats showed a significant reduction in methamphetamine-induced rotations. At the 5th week postgraft, there was a 77% recovery of 4-[18F]-ADAM uptake and a 83% recovery of SERT-ir levels in the striatum within the 6-OHDA-lesioned hemisphere. Of particular interest was our finding of both dopaminergic and serotonergic cell bodies in the striatum of grafted rats. Since the striatum only contains the fibers of dopamine and serotonin neurons, we conclude that these cells must have been derived from the graft. Thus, our study shows that mesencephalic tissue grafts can be used to repair not only the dopaminergic but also the serotonergic system.
Our data showed that 6-OHDA affects both dopaminergic and serotonergic neurons. However, this study only examined the long-term (3th and 8th weeks after lesion) effects of 6-OHDA on serotonergic systems. Future experiments will be needed to elucidate the short-term acute effects, if any.
In this study, the lower level of 4-[18F]-ADAM uptake was found following 6-OHDA lesions, and the progressive recovery of 4-[18F]-ADAM uptake in the SERT-depleted striatum after transplantation indicates that embryonic mesencephalic tissue grafts can restore serotonergic as well as dopaminergic systems. Therefore, 4-[18F]-ADAM/micro-PET may be an alternative method for monitoring the progression of PD and the therapeutic efficacy of mesencephalic transplantation.
Footnotes
Acknowledgments
This work was supported by the National Science Council of Taiwan, Grants NSC 96-2811-B-016-004 and NSC 95-2321-B-016-001-MY2. We also acknowledge technical support from the Molecular and Genetic Imaging Core supported by the National Research Program for Genomic Medicine, National Science Council, Taiwan (NSC95-3112-B-001-009 and NSC97-3112-B-010-016). We are grateful to Mr. Chi-Jiun Peng and Ms. Yu-Yeh Kuo for their technical assistance. The authors declare no conflict of interest.
