Abstract
Mesenchymal stem cells (MSCs) and especially those derived from fetal tissues exert a potent immunosuppressive effect that can be enhanced under inflammatory conditions. This study aimed to explore the immunosuppressive properties of human umbilical cord mesenchymal stem cells (HUCMSCs). We found that HLA-G, the nonclassical HLA allele with strong immune-inhibitory properties, was much more expressed on the HUCMSCs than on MSCs of other origins. Flow cytometry revealed that 90.8% of the HUCMSCs expressed HLA-G. RT-PCR revealed expression of HLA-G1, HLA-G5, and HLA-G7 in all of four HUCMSC lines. In a mixed lymphocyte reaction assay, the HUCMSCs inhibited the proliferation of lymphocytes by 35 ± 3% and could be reversed by treatment with an HLA-G blocking antibody. Upon coculture with the HUCMSCs, peripheral blood mononuclear cells expressed lower levels of proinflammatory mediators such as IL-6, TNF-α, and VEGF-α. This immunosuppressive effect was enhanced when the HUCMSCs were pretreated with IFN-γ, such that the expression of HLA-G was highly activated and HLA-DR diminished. The same phenomenon was not observed in MSCs derived from bone marrow or the placenta. In a xenograft rejection assay, the HUCMSCs survived in immunocompetent mice, whereas primary fibroblasts did not survive. This study confirms the HLA-G-related immunosuppressive property of HUCMSCs, which is more potent than MSCs of other origin. A good tolerance of this mesenchymal stem cell in allogeneic transplantation can thus be anticipated.
Keywords
Introduction
Graft-versus-host disease (GVHD) is a life-threatening complication of allogeneic hematopoietic stem cell transplantation, a therapeutic approach that relies on elimination of the recipient's hematopoietic system and replacing it with hematopoietic cells from a healthy donor. Donor T cells in the transplant can cause GVHD (38); however, they are also associated with beneficial effects in preventing graft rejection (25). To prevent GVHD, patients generally receive immunosuppressive regimens; however, these treatments are only partially effective, and some patients still develop uncontrolled and even lethal GVHD.
Mesenchymal stem cells (MSCs) are pluripotent progenitor cells that contribute to the maintenance and regeneration of a variety of connective tissues, including bone, cartilage, adipose, and muscle, and are regarded as a potential source for tissue repair and regenerative medicine (6,7,29). Several reports have shown that human and murine MSCs exert potent immunosuppressive effects in vitro (8,18) and in vivo (4,8,11,18,22,32,41) and suggested a therapeutic potential for T-cell-dependent pathologies. One advantage of human MSCs as a therapeutic tool is the relative ease of production under good manufacturing practice conditions (14). More importantly, because of the poor immunogenicity and the fact that their immunosuppressive effect is major histocompatibility complex independent (8,15), it may not be necessary to isolate specific MSCs for each patient, and a “universal” pool of immunosuppressive MSCs may suffice for all patients.
To date, clinical reports of MSC-based therapy for GVHD in humans have produced contrasting results. Initially, no GVHD-preventive effect was shown when MSCs were grafted together with hematopoietic stem cell transplantation (16). In contrast, a multicenter randomized trial demonstrated a significant reduction in the incidence of GVHD when MSCs were cotransplanted (28). Importantly, a recent article described a case in which MSCs were successfully used to treat a young patient with grade IV (severe) GVHD (17).
Recent phenotypic experiments on both fetal and adult MSCs have reported a specific expression of HLA-G, a nonclassical HLA class I molecule (13). During implantation of the embryo, HLA-G has been found in trophoblasts, where it plays a key role in maternal tolerance for the fetus. MSCs are able to inhibit immune reactions by increasing HLA-G expression resulting in 1) alloproliferation of T-cells, 2) cytolysis capacity of T cells and natural killer (NK) cells, and 3) differentiation of dendritic cells (21,30).
Although MSCs reside predominantly in the bone marrow, they are also distributed throughout many other tissues and serve as local sources of dormant stem cells (42,43). Previously, we successfully derived MSCs from human umbilical cord matrix (6) and showed that they had a neuroplastic ability and were able to cure stroke rat. In addition, we found that they could be retained in the brain stroke area for more than 4 months and exhibited good immunomodulation ability (7).
The goal of this study was to characterize the immunosuppression activity and expression of HLA-G and related immunomodulating cytokines in human umbilical cord mesenchymal stem cells (HUCMSCs). We found that the HUCMSCs readily expressed different alleles of HLA-G that could be further induced by IFN-γ. The HUCMSCs also suppressed the proliferation of peripheral blood mononuclear cells (PBMCs). When cocultured with PBMCs, the expressions of the inflammatory cytokines were suppressed. In vivo xenotransplantation revealed that the HUCMSCs could tolerate xenorejection for at least 72 h.
Materials and Methods
Isolation and Expansion of HUCMSCs and other MSCs
The detailed protocol for deriving HUCMSCs can be found in our previously published paper (6). Briefly, human umbilical cord samples (n = 4, about 20 cm in length, 20 g in weight) were collected in sterile boxes containing Hank's balanced salt solution (HBSS; Gibco, Grand Island, NY, USA), and separation of the Wharton's jelly (WJ) from the vessels and amniotic membrane was conducted within 24 h. The protocols for sampling human umbilical cords were approved by the Institutional Review Board of Tzu-Chi University Hospital, and written informed consent was obtained from each mother before labor and delivery of their infant.
The collected human umbilical cord tissues were washed three times with Ca2+ and Mg2+-free phosphate-buffered saline (PBS; Biowest, Nuaille, France). They were then cut using scissors in a midline direction, and the vessels of the umbilical artery, vein, and outlining membrane were dissociated from the WJ. The jelly was then extensively cut into pieces smaller than 0.5 cm3, treated with collagenase type I (Sigma-Aldrich, St. Louis, MO, USA), and then incubated for 30 min at 37°C in a 95% air/5% CO2 humidified atmosphere. The explants were then cultured in Dulbecco's modified Eagle medium (DMEM; Invitrogen, Carlsbad, CA, USA) containing 10% fetal bovine serum (FBS; Biological Industry, Kibbutz, Israel) and antibiotics at 37°C in a 95% air/5% CO2 humidified atmosphere. They were left undisturbed for 5-7 days to allow for migration of the cells from the explants, and four HUCMSC lines were derived.
Bone marrow MSCs (BMMSCs) (n = 1, 30-year-old, female) were provided by the Bone Marrow Bank of Tzu Chi General Hospital. They were cultured with a-minimum essential media (MEM; Gibco) supplemented with 10% FBS (Biological Industry).
Placenta-derived mesenchymal stem cells (PMSCs) were derived from one term (38-40 weeks of gestation, n = 1, male) placenta from a healthy donor mother obtained with informed consent according to the approved procedures of the IRB. Placental tissue was dissected using mechanical and enzymatic treatments, and the homogenate was cultured in complete medium consisting of DMEM (Invitrogen) supplemented with 10% of FBS (Biological Industry) and antibiotics. PMSCs between passage 5 and 8 were used for the experiments.
Human fibroblasts (n = 1, age 40 years old) were derived from vaginal mucosa obtained from surgical specimens with informed consent. The vaginal mucosa was mechanically cut using scissors into small pieces and treated with trypsin/EDTA. The explants were then cultured in DMEM supplemented with 10% FBS and antibiotics.
Flow Cytometry of the HUCMSCs
The specific surface molecules of the HUCMSCs in 4-8 passages of culture were characterized by flow cytometric analysis. The cells were detached with 2 mM EDTA (Gibco) in PBS, washed with PBS containing 2% bovine serum albumin (BSA; Sigma-Aldrich) and 0.1% sodium azide (Sigma-Aldrich), and incubated with the respective antibody conjugated with fluorescein isothiocyanate (FITC) or phycoerythrin (PE) including CD34, CD44, CD45, CD56, CD73, CD90, HLA-ABC, and HLA-DR (1:200; BD, Pharmingen, Franklin Lakes, NJ, USA). Related isotype antibodies were used as the control. Thereafter, the cells were analyzed using a Becton Dickinson flow cytometer (Becton Dickinson, San Jose, CA, USA).
In Vitro Differentiation Assay of the HUCMSCs
The detailed method of in vitro differentiation of HUCMSCs has previously been described (6). The differentiation potential for osteogenesis was assessed by the mineralization of calcium accumulation by Alizarin red S staining (Sigma-Aldrich). For adipogenic differentiation, intracellular lipid droplets were observed by microscopy and confirmed by Oil red O staining (Sigma-Aldrich) (19). Chondrogenesis was confirmed by Alcian blue (Fluka, Chemie GmbH, Buchs, Germany) (37). Briefly, for chondrogenesis, the HUCMSCs were seeded in a 48-well plate (Corning, Corning, NY, USA) at density of 1.5 × 107 cells/cm2 and grown in chondrogenic media consisting of DMEM, 10% FBS, 100 ng/ml of insulin growth factor-1, 5 ng/ml of fibroblast growth factor-2, 10 ng/ml of growth hormone, 50 μg/ml of ascorbic acid-2-phospate, and 6.25 μg/ml of transferrin (all Sigma-Aldrich), with the media being changed every 3 days. The cells were cultured in this chondrogenic media at 37°C with 5% CO2 for 3 weeks, and then fixed in paraformaldehyde (Bionovas, Toronto, Canada), mounted on slides, and stained using standard Alcian blue staining protocols. For osteogenesis and adipogenesis, the HUCMSCs were cultured in osteogenic medium [DMEM supplemented with 10% FBS, 0.1 μmol/L dexamethasone (Sigma-Aldrich), 10 mmol/L b-glycerol phosphate (Sigma-Aldrich), and 50 μmol/L ascorbate (Sigma-Aldrich)] and adipogenic medium [DMEM supplemented with 10% FBS, 1 μmol/L dexamethasone, 5 μg/ml insulin, 0.5 mmol/L isobutylmethylxanthine (Sigma-Aldrich), and 60 μmol/L indomethacin (Sigma-Aldrich)] for 3 weeks.
HLA-G Immunoassays
We used the mouse anti-HLA-G1/G5 MEMG/9 FITC antibody (Abcam, Cambridge, MA, USA) at 1:500 final concentration for flow cytometry analysis (45). The intracellular expression of HLA-G in HUCMSCs was revealed by using the Cytofix/cytoperm kit (BD, Pharmingen) according to the recommended conditions. Isotypic control was also included.
For intracellular HLA-G immunofluorescence stain, cells were fixed for 20 min in 4% paraformaldehyde (Bionovas) and permeabilized with 0.3% Triton X-100 (J. T. Baker, Phillipsburg, NJ, USA) with 1% BSA blocking for 45 min at room temperature. For live cell immunostain, cells were blocked with 10% goat serum and 3% bovine serum albumin (BSA) for 20 min at 4°C and stained with anti-HLA-G1/G5 MEMG/9 FITC antibody for 60 min at 4°C. After the primary antibody reactions in both intracellular and live cell stain, the cells were incubated with the secondary fluorescent antibody diluted at 1:100 for 1 h. To visualize the nuclei, the slides were mounted with 10 μl of 4′,6-diamidino-2-phenylindole (DAPI) antifade solution (Millipore, Billerica, MA, USA) and visualized by confocal microscopy.
Quantitative Real-Time Reverse Transcription-Polymerase Chain Reaction (qRT-PCR) and RT-PCR
Sequence of Primers and Conditions Used in Real-Time Polymerase Chain Reaction and RT-PCR

Primer Set of Various HLA-G Used in RT-PCR

Mixed Lymphocyte Reaction Assay (MLR)
The immunomodulation activity of the HUCMSCs was demonstrated with MLR. Human PBMCs from two different donors (male, 25 and 40 years old) who provided informed consent were isolated from heparinized blood by gradient centrifugation with Ficoll solution (Sigma-Aldrich) at 400 × g for 20 min at room temperature. Stimulator PBMCs were prepared by treatment with mitomycin (Sigma-Aldrich) at 25 μg/ml for 30 min at 37°C. Cell count and viability were assessed by trypan blue (Biowest) dye exclusion and then used directly in the MLR. The HUCMSCs were plated in triplicate at passage 2 into U-bottomed 96-well plates (Corning) at 105 cells/ml in 100 μl of FCS-DMEM and allowed to adhere to the plate for 1 to 2 h. Human responder (105 PBMCs) and an equal number of stimulator PBMCs were added to the wells in 100 μl of RPMI-1640 (Invitrogen) in 10% inactivated FCS (Sigma-Aldrich). The cultures were incubated at 37°C in 5% CO2 for 5 days, and the suspended responder PBMCs were then counted using the 2,3-bis-(2-methoxy- 4-nitro-5-sulfophenyl)-2H-tetrazolium-5-carboxanilide (XTT) assay kit (Biological Industries). To test the role of HLA-G in MLR, 3 μg/well of HLA-G-neutralizing antibodies (87G Abs) or control antibody (20 μg/ml) (Exbio Antibodies, Praha, Czech Republic) was added on the first day of the MLR cultures.
Coculture of the HUCMSCs with PBMCs
At day 0, PBMCs were cultured for 7 days with the HUCMSCs at a 5:1 ratio in a Transwell coculture system (Coring Inc.) with a 0.4-μm-pore-sized membrane. The HUCMSCs were cultured in the bottom well, and the PBMCs were kept in the inserts. On day 7, the PBMCs were collected, and the expressions of IL-6, TNF-α, and VEGF-α were analyzed by real-time PCR.
Interferon Stimulation
To assess the effects of interferon-γ (INF-γ), HUCMSC, PMSCs, and BMMSCs were pretreated with 100 ng/ml (R&D Systems Inc., Minneapolis, MN, USA) for 48 h prior to flow cytometry. The cell surface expressions of HLA-G and HLA-DR were evaluated.
Xenotransplantation
All animal experiments were performed with the approval of the Institutional Animal Care and Use Committee. The surgical procedures were performed under anesthesia induced by intraperitoneal injections of 0.1 ml of a mixture consisting of ketamine (5 mg/kg; Ubiasia, Taipei, Taiwan) and xylazine (15 mg/kg; Bayer, Leverkusen, Germany). Immunocompetent NMRI mice (female, 6-8 weeks old, purchased from Tzu Chi University Animal Center) were used to assay the survival of the HUCMSCs in vivo under the condition of acute rejection. Ready-to-use highly efficient lentivirus was purchased from Cellexium Biomedica Incoporation (Taipei, Taiwan). The lentiviral rGFP-tagged HUCMSCs and rRFP-tagged primary human fibroblasts were cultured at an identical seeding density of 1 × 106/graft. The cells were transplanted as a monolayer on porous gelfoam (Ethicon Inc., Somerville, NJ, USA) to allow for easy visualization. The gelfoam was made from porcine gelatin and had the consistency of a sponge-like material. The gelfoam sponge contained many porous structures, which allowed the cells to grow in a 3D manner (26). A 1-2-cm incision was made on the dorsal skin, and one piece of gelfoam (0.5 × 0.5 × 0.5 cm with 1 × 106 cells) was grafted. The grafts were removed 72 h later to assess cell survival under fluorescent microscopy.
Statistical Analysis
All statistical analyses were performed using SPSS version 15 (IBM, Armonk, NY, USA). Statistical significance was calculated using t-tests, and significance was set at p < 0.05. All values were expressed as the mean and standard deviation (SD). Three different HUCMSC samples were used to evaluate differences in the percentage of HLA-G in MSC/MLR (using ANOVA with post hoc Bonferroni test). The percentage of HLA-G-positive HUCMSCs was determined from up to three successive in vitro passages from three independent samples.
Results
Characteristics of HUCMSCs Derived From Human Umbilical Cords
In primary explants of WJ tissue pieces, spread cells showed a spindle-shaped morphology (Fig. 1A). These cells divided rapidly with a doubling time of 28 h. They were cultured for more than 25 passages, which is equivalent to over 40 population doublings, without spontaneous differentiation. They were negative for CD34, CD45, CD56, and HLA-DR, and positive for CD44, CD73, CD90, and HLA-ABC (Fig. 1B), similar to other MSCs, and were defined as HUCMSCs.
Characterization of human umbilical cord mesenchymal stem cells (HUCMSCs, representative images of WJ28). (A) Primary cells growing from explants of umbilical cord Wharton's jelly from four donors showed fibroblast-like, spindle-shape morphology (different magnification). (B) Flow cytometry analysis of representative (WJ28) HUCMSCs was negative for CD34, CD45, CD56, and HLA-DR, and positive for CD44, CD73, CD90, and HLA-ABC. (C) Upon adipogenic differentiation, the cells formed neutral lipid vacuoles and contained numerous Oil red O-positive lipid droplets (left). In osteogenic medium, the cells broadened to form a mineralized matrix, which was strongly stained with Alizarin red S (middle). Upon chondrogenic differentiation, the cells were positively stained with Alcian blue (right). In all the experiments, the undifferentiated control was done in parallel to the differentiation-induced cells. Undiff, uninduced control. (D) Expression of genes specific for adipogenic (PPARγ), osteogenic (osteopontin), and chondrogenic (SOX9, COL2a1, ACAN) differentiation was shown by RT-PCR, with GAPDH as a positive control. Scale bar: 1,000 μm in (A) left panel, 100 μm in (A) right two panels and (C).
In Vitro Differentiation of HUCMSCs to Osteocytes, Adipocytes, and Chondrocytes
Adipogenic differentiation of the HUCMSCs was apparent 1 week after incubation with an adipogenic medium supplement. Changes in cell morphology were evident with the formation of Oil red O-positive neutral lipid vacuoles in almost all of the cells (Fig. 1C, left). Similarly, the induction of differentiation in the osteogenic medium caused the treated cells to grow rapidly and contain mineralized matrices, which were strongly stained by Alizarin red S, indicating the deposition of calcium after 2 weeks of cultivation (Fig. 1C, middle). Chondrogenic differentiation of the HUCMSCs after 3 weeks of differentiation was characterized by positive Alcian blue staining (Fig. 1C, right), and the expressions of adipogenic (PPARγ), osteogenic (osteopontin), and chondrogenic (ACAN, SOX9, COL2A1) genes (Fig. 1D).
Expression of the HLA-G Molecule in the HUCMSCs
Expression of HLA-G mRNA was evident in the HUCMSCs from passage 3 to passage 9 with the most abundant expression noted in passage 6 (Fig. 2A). Flow cytometry of the HLA-G molecule revealed 90.82% of HUCMSCs expressed the HLA-G molecule intracellularly (Fig. 2B). Live cell immunostaining further revealed HLA-G protein was also expressed on the cell surface (Fig. 2C2). The intracellular HLA-G molecules are supposed to be the soluble isoforms 5-7, which are lacking in the transmembrane domain and are designated to be secreted. In addition, the surface expression is supposed to be the membrane isoform 1. HLA-G has been predicted to have seven protein isoforms by alternative splicing, with isoforms 1-4 to be membrane bound and isoforms 5-7 to be soluble/secreted (3). In RT-PCR analysis, all four HUCMSC lines studied expressed HLA-G isoform 7 strongly and isoforms 1 and 5 with varying intensity. One HUCMSC line (WJ29) expressed isoform 3, two lines (WJ28, 29) expressed isoform 2/4, and one (WJ29) expressed isoform 6 (Fig. 2D).
HLA-G was highly expressed on the HUCMSCs. (A) Quantitative real-time PCR revealed a steady expression of HLA-G mRNA in the HUCMSCs. The highest level was noted at passage 6 (p6). (B) Flow cytometry showed that 90.82% of the HUCMSCs were positive for HLA-G intracellularly. (C) Immunofluorescence revealed intracytoplasmic (C1) and cell membrane (C2) expression of HLA-G in the HUCMSCs. The nuclei were stained by DAPI. Scale bar: 10 μm (C1), 20 μm (C2). (D) The HUCMSCs derived from four different donors expressed varying amounts of HLA-G isoforms 1 and 5, and a universal abundance of isoform 7, as revealed by RT-PCR.
The HLA-G Molecule Is Crucial for the Inhibitory Effect of HUCMSCs on Allogenic PBMC Proliferation
The immunomodulation activity of the HUCMSCs was analyzed by MLR. After adding HUCMSCs to the MLR, the mean percentage of proliferation of responder PBMCs was significantly lower than that without adding the HUCMSCs (65 ± 3% vs. 100%), and this reduction was reversed by adding the HLA-G-neutralizing antibody (Fig. 3). This suggests that HLA-G can mediate the immune inhibitory effect of HUCMSCs.
HLA-G in the HUCMSCs was responsible for suppressing the mixed lymphocyte proliferation reaction (MLR). The effect of HUCMSCs and HLA-G-neutralizing antibody on MLR was studied. Using an XTT assay, the average suppression of lymphocyte proliferation after addition of HUCMSCs to the PBMC culture was 35% (p < 0.05). This inhibition was fully restored in the presence of HLA-G blocking antibody (87G) (p < 0.05). In contrast, the human fibroblast control did not suppress the MLR. The results are presented as mean ± SD of three separate experiments, each performed in triplicate. Statistical analysis was performed using ANOVA with post hoc Bonferroni test. *p < 0.05.
Expression of Proinflammatory Genes in PBMCs Was Decreased in the Presence of HUCMSCs
To further study the inflammatory gene expression of PBMCs with or without coculture with HUCMSCs, the observed immunosuppressive effect of the HUCMSCs on PBMCs was further characterized by cytokine and growth factor analyses in a Transwell coculture system. The expressions of proinflammatory cytokines including IL-6, TNF-α, and VEGF-α by the PBMCs was significantly lower in the presence of HUCMSCs (Fig. 4).
Suppression of inflammatory cytokine expression in PBMCs upon coculture with HUCMSCs. Expressions of IL-6, TNF-α, and VEGF-α were analyzed by real-time polymerase chain reaction analysis in the PBMCs (MSC) after coculture with the HUCMSCs for 7 days in a Transwell coculture system. Gene expression levels were normalized to the concentration of GAPDH. PBMCs, peripheral blood mononuclear cells; MSC, HUCMSCs. *p < 0.05 by Student's t-test.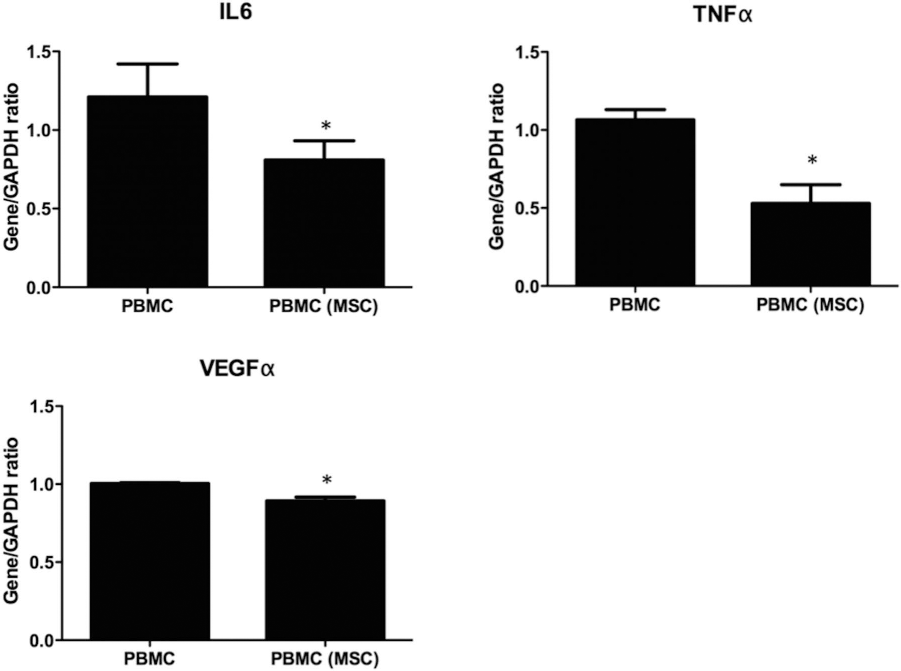
IFN-γ Stimulated the Expression of HLA-G in HUCMSCs but not other MSCs
IFN-γ is a potent inducer of major histocompatibility complex (MHC) class II molecules and β2-microglobulin, which is coexpressed with MHC class I. After 2 days of IFN-γ stimulation, the HUCMSCs expressed a higher level of HLA-G; however, the expression of the MHC class II molecule HLA-DR remained low (Fig. 5). This is in contrast to the PMSCs and BMSCs, where IFN-γ stimulation resulted in activation of HLA-DR but not HLA-G.
HLA-G was readily induced by IFN-γ in human umbilical cord mesenchymal stem cells (HUCMSCs) but not in other mesenchymal stem cells. The cell surface expressions of HLA-G and HLA-DR on HUCMSCs, placenta mesenchymal stem cells (PMSCs), and bone marrow MSCs (BMMSCs) with or without 2 days of treatment with IFN-γ were evaluated by flow cytometry; the percentage of positive cells is indicated.
The HUCMSCs Survived for an Extended Period After Xenotransplantation in Immunocompetent Mice
The tolerance of immune rejection was further investigated in vivo by xenotransplantation of the cells into immunocompetent NMRI mice. The HUCMSCs and HLA-G-negative human fibroblasts were stably transfected with lentiviral constructs expressing the fluorescent transgenes rRFP and rGFP, respectively. Whereas no human fibroblasts survived after xenotransplantation, a good number of HUCMSCs remained in the grafted gelfoam 72 h after transplantation (Fig. 6). Moreover, when mixed with the HUCMSCs, a few fibroblasts survived 72 h after xenotransplantation. Therefore, HUCMSCs seem to offer other human cell type protection from immune rejection.
Human umbilical cord mesenchymal stem cells (HUCMSCs) survived in xenografts in immunologically competent mice. HUCMSCs and human fibroblasts, expressing lentiviral rGFP and rRFP, respectively, were grafted to the subcutaneous layer of the dorsal skin of NMRI mice, and removed after 3 days and visualized using fluorescent microscopy. Green fluorescence indicated the presence of HUCMSCs up to 72 h after transplantation, whereas the red fluorescence of normal human fibroblast was barely detectable. Cotransplantation with HUCMSCs improved the survival of the fibroblasts in the xenotransplantation mouse model. More rRFP-positive fibroblasts were found in the mixed cell grafts than in the grafts with fibroblasts alone.
Discussion
In the present study, we confirmed the immunosuppressive function of HUCMSCs mediated by HLA-G. Compared to MSCs from adult tissues, the HUCMSCs expressed a higher level of HLA-G with active immunosuppressive function on MLR assay. The HUCMSCs also interacted better with PBMCs in immunomodulation. With IFN-γ stimulation, the HUCMSCs increased the expression of HLA-G but not HLA-DR. In contrast, the BMSCs and PMSCs responded with an increase in HLA-DR but not HLA-G. Furthermore, the HUCMSCs could suppress the expressions of proinflammatory cytokines from the PBMCs when cocultured.
HLA-G has been reported to have immunomodulatory effects on a wide range of immune cells, including dendritic cells and monocytes (2). In addition, HLA-G can disturb the cytolysis, adhesion, and migration capacities of NK cells. HLA-G has also been reported to impair the cytolysis potential of antigen-activated CD8+ cells (31,33). The expression of HLA-G in HUCMSCs has been reported in previous articles (5,23,39,40). However, these studies either focused on the two full-length isoforms (isoforms 1 and 5) or only distinguished the membrane and soluble forms of HLA-G. In comparison, the present study examined all the seven known isoforms of HLA-G in HUCMSCs. One (HLA-G1) of the four membrane bound and two (HLA-G5, -G7) of the three soluble isoforms were expressed in all the four HUCMSC lines studied. HLA-G1 has been reported to prevent dendritic cell maturation (21,30), and HLA-G5 has been repotted to play an important role in allograft acceptance through induction of immunosuppressive/regulatory T cells and a NK cytolysis effect (44). Both isomers have been shown to inhibit CD4+ T-cell proliferation, and a combination of them has been shown to have an additive effect on NK cytolysis (44). The role of isoform G7 has yet to be elucidated.
It has been reported that coculturing nonhuman primate or human MSCs with peripheral blood lymphocytes from allogenic donors did not stimulate their proliferation in vitro (1,18). It is possible that HLA-G of the MSCs could be mediating this suppressive effect. The present study also clearly demonstrated that HUCMSCs can inhibit MLR and that adding an anti-HLA-G-neutralizing antibody (87G antibody) could reverse this inhibition, further suggesting that HLA-G is responsible for the escape of immune recognition of HUCMSCs by allogenic peripheral blood lymphocytes.
HLA-G can induce regulatory immunosuppressive cells, cell cycle arrest, and TH2 cytokine profiles (3). PBMCs can also produce proinflammatory and growth factors such as IL-6, VEGF-α, and TNF-α, which may explain the inhibitory effect of MSCs on PBMCs (27). Cross talk between HUCMSCs and PBMCs may also result in the immune tolerance effect of PBMCs. In the present study, gene expression analysis showed that PBMCs had particularly decreased levels of IL-6, TNF-α, and VEGF-α after coculture with HUCMSCs.
The immunological properties of HUCMSCs characterized in the present study are consistent with reports on MSCs derived from other extra-embryonic fetal tissues, such as umbilical perivascular cells (10), amniotic membrane (20), and amniotic fluid (36). Many of these cells (umbilical perivascular, amniotic membrane, and amniotic fluid cells) also consistently express MHC-I but not MHC-II antigens as with HUCMSCs (35). In addition, HUCMSCs also share the ability to inhibit lymphocyte proliferation in vitro with the abovementioned cells. This common immunomodulation characteristic, in contrast to adult MSCs, may be associated with the common origin of the maternal/fetal interface, where immunotolerance is physiologically essential. As with fetal MSCs or embryonic stem cells, these extra-embryonic fetal MSCs are also immunologically naive (9,12).
In the present study, the immunomodulatory activity of the HUCMSCs was further demonstrated by their survival as xenotransplants in immunocompetent NMRI mice without immunosuppressive treatment. The HUCMSCs survived for up to 72 h under the conditions of normal xenorejection, while human fibroblasts were readily cleared. Moreover, cotransplantation of HUCMSCs prolonged the survival of the fibroblasts, suggesting that factors produced by HUCMSCs can confer non specific protection to neighboring immunogenic cell types. Previous studies have used the persistence of biochemical markers such as transplanted cell-specific protein/DNA (24) or prelabeled molecular markers (34) to demonstrated survivability. In this study, we used gelfoam as the cell carrier for the xenograft survival assay. Gelfoam is made of porcine gelatin and is used as a hemostasis agent. Cells can grow in the gelfoam in a 3D manner (26), and we showed that this readily retained the viability of the HUCMSCs in xenografts for at least 3 days. However, the possibility of retention of the immunogenic markers in dead cells or the possible internalization of cellular debris by adjacent cells cannot be ruled out.
Conclusion
In summary, we demonstrate that multipotent MSCs derived from the Wharton's jelly of human umbilical cords have a strong HLA-G-mediated immunosuppressive activity. HUCMSCs may be an attractive multipotent progenitor for therapeutic use.
Footnotes
Acknowledgments
The authors would like to thank the National Science Council of the Republic of China, Taiwan, for financially supporting this research under Contract No. NSC 98-2314-B-303-009-MY3. The Buddhist Tzu Chi General Hospital is thanked for its financial support under Contract Nos. TCRD99-12, TCRD100-55-1, and TCSP98-07. The authors declare no conflicts of interest.
