Abstract
There is conflicting evidence favoring both the use of human serum (HS) and of human serum albumin (HSA) in human islet culture. We evaluated the effects of HS versus HSA supplementation on 1) in vitro β-cell viability and function and 2) in vivo islet graft revascularization, islet viability, β-cell death, and metabolic outcome after transplantation. Islets isolated from 14 cadaveric organ donors were cultured for 3 days in CMRL 1066 medium supplemented with HS or HSA. After 3 days in culture, β-cell apoptosis was lower in HS group (1.41 ± 0.27 vs. 2.38 ± 0.39%, p = 0.029), and the recovery of islets was 77 ± 11% and 54 ± 1% in HS- and HSA-cultured groups, respectively. Glucose-stimulated insulin secretion (GSIS) was higher in HS group (29.4, range 10.4-99.9, vs. 22.3, range 8.7-70.6, p = 0.031). In vivo viability and revascularization was determined in HS-and HSA-cultured islets transplanted into the anterior chamber of the eye of Balb/c mice (n = 14), and β-cell apoptosis in paraffin-embedded mouse eyes. Islet viability and β-cell apoptosis were similar in both groups. Revascularization was observed in one graft (HS group) on day 10 after transplantation. Islet function was determined in streptozotocin (STZ)-diabetic nude mice (n = 33) transplanted with 2,000 IEQs cultured with HS or HSA that showed similar blood glucose levels and percentage of normoglycemic animals over time. In conclusion, human islets cultured in medium supplemented with HS showed higher survival in vitro, as well as islet viability and function. The higher in vitro survival increased the number of islets available for transplantation. However, the beneficial effect on viability and function did not translate into an improved metabolic evolution when a similar number of HSA- and HS-cultured islets was transplanted.
Introduction
Clinical islet transplantation has improved substantially, and a majority of patients achieve stable and near normal glycemic control in the absence of severe hypoglycemic events with improved quality of life and beneficial effects on chronic complications (36). Nevertheless, the function of transplanted islets declines progressively, and after several years most patients show a relapse of hyperglycemia. Several factors may contribute to the progressive loss of graft function, such as chronic rejection, recurrence of the autoimmune process, and β-cell toxicity of immuno suppressive treatment (27). The long-term outcome of islet transplantation is also determined by the amount and quality of the initially transplanted tissue (44). A critical number of islets is required to attain glycemic control (39), but also to maintain it in the long term as shown in animal models (9,45) and in clinical human islet transplantation (26,44). Despite the improvement in islet isolation procedure, the current yield of a single islet isolation is usually insufficient to achieve normoglycemia after transplantation to diabetic patients. The isolation process results in substantial islet injury and death (8), and an additional islet loss takes place in the short-term 2- to 3-day culture usually performed before islet transplantation (13,31). This pretransplantation culture offers several advantages, among them the planning of the transplantation procedure with patient pretreatment, the assessment of islet quality, and islet shipping to a transplant center, and it has become a standard procedure in clinical islet transplantation. However, it has been generally associated with a decline in islet viability, islet mass (20,25,42), and β-cell function (34,41), even though improved viability and function has also been reported (22).
The addition of bovine serum to the culture media is a well-established procedure in rodent islet cultured to improve islet viability and function. Serum contains albumin, growth factors, extracellular matrix components, and it has an inhibitory action on endogenous (38) and exogenous enzymes (4). However, xenogeneic bovine serum cannot be added to culture media when human islets are to be used for clinical transplantation, and the culture media is then usually supplemented with human serum albumin (HSA) (19,21). It has been hypothesized that supplementation with human serum (HS) may offer some advantages over HSA, and some studies have reported increased islet viability, insulin content, and insulin secretion with HS (2,16,17,24,41). Nevertheless, other groups have found a negative effect of bovine serum (7) and HS (29) supplementation on human islets and better results with serum-free medium (46). Thus, there is no clear consensus on the potential advantages of HS over HSA for human islet culture. Moreover, there is very limited information on the impact of pretransplantation culture with HS or HSA on the outcome of islets if they are subsequently transplanted (3), since the vast majority of studies have been performed in vitro at the end of the culture period. The aim of our study was to determine whether supplementation of culture medium with HS could increase the viability and function of human islets and improve the outcome of islet transplantation.
Materials and Methods
Human Islet Isolation and Culture Conditions
Pancreatic islets were isolated from 14 adult cadaveric organ donors (mean age 57.3 ± 14.6 years, seven females and seven males) by collagenase digestion (Collagenase NB1 Premium Grade with Neutral Protease NB; Serva Electrophoresis GmbH, Heidelberg, Germany) using the Ricordi method (37) with some modifications (17). The islets were purified on a refrigerated COBE 2991 cell processor (COBE BCT, Lakewood, CO, USA) with a continuous density gradient. Islet yield was quantified by dithizone staining (Sigma-Aldrich, St. Louis, MO, USA) in duplicate using a standard islet diameter of 150 mm as one islet equivalent (IEQ). Immediately after purification, islets were seeded in 25-cm2 culture flasks (Sarstedt AG & Co, Nümbrecht, Germany) and cultured free floating in Connaught Medical Research Laboratories 1066 (CMRL1066) medium (5.6 mM glucose) (Mediatech Inc., Corning Cellgro, Manassas, VA, USA) supplemented with 2 mM L-Glutamine (Life Technologies, Grand Island, NY, USA), 10 mM HEPES (Biological Industries, Kibbutz BeitHaemek, Israel), 40 μg/ml gentamycin (Laboratorios Normon, Madrid, Spain), 0.25 μg/ml amphotericin B (Fungizone; Life Technologies), 20 μg/ml ciprofloxacin (Fresenius-Kabi, Barcelona, Spain), 10 mM nicotinamide (Sigma-Aldrich), and with 0.5% HSA (Grifols, Barcelona, Spain) (HSA group) or with 10% ABO-compatible HS (Blood and Tissue Bank, BST, Barcelona, Spain) (HS group) at 37°C and 5% CO2 in a humidified incubator for 3-4 days. Culture media was changed every 2 days.
Study Design
HS- and HSA-cultured islets were compared 1) in vitro after 1 and 3 days in culture to determine islet viability and β-cell apoptosis and function; 2) after transplantation into the anterior chamber of the eye of Balb/c mice to determine islet cell viability and graft revascularization in vivo, as well as β-cell apoptosis; 3) after transplantation under kidney capsule of immunodeficient nude mice treated with streptozotocin (STZ) (Sigma-Aldrich) to induce diabetes, to determine islet function in vivo. The experiments were approved by the local Ethics Committee and the Animal Ethics Committee.
In Vitro Studies in Cultured Islets
Islet Cell Viability and β-Cell Apoptosis
Islets were cultured in groups of 2,800 IEQs in 25-cm2 nontreated flasks. After 1 and 3 days in culture, islet cell viability was determined by acridine orange/propidium iodide (AO/PI) staining (Sigma-Aldrich) (5). A 50-islet aliquot was transferred in phosphate-buffered saline (PBS) (Biological Industries) to a 10 × 35-mm Petri dish (Greiner Bio-One, Frickenhausen, Germany). Acridine orange and propidium iodide stock solutions were added at a final concentration of 0.67 μM and 75 μM, respectively. Acridine-positive cells (green) were identified as viable cells and propidium iodide-positive cells (red) as dead cells. Islet cell viability was expressed as percentage of viable cells versus the total (viable and nonviable) islet cells.
β-Cell apoptosis was determined in islets cultured for 3 days. Islets were fixed overnight in 4% paraformaldehyde (Merck KGaA, Darmstadt, Germany) at 4°C, embedded in paraffin, and then sectioned and stained for immunofluorescence. Sections were double stained for apoptotic nuclei with the terminal deoxynucleotidyl transferase biotin-dUTP nick-end labeling (TUNEL) technique (In Situ Cell Death Detection Kit, ApopTag; Millipore, Temecula, CA, USA), and for insulin with a rabbit polyclonal anti-insulin antibody (Santa Cruz Biotechnology Inc., Dallas, TX, USA) (final dilution 1:100). For insulin labeling, the secondary antibody was a donkey anti-rabbit IgG conjugated with Alexa Fluor 555 (Life Technologies) (final dilution 1:400). Nuclei were stained with 300 nM DAPI (Life Technologies).
β-Cells and TUNEL-positive β-cells were counted using a Leica DM4000B microscope connected to a digital camera (Leica DFC 310FX) (Wetzlar, Germany) with a color monitor. β-Cell apoptosis was expressed as percentage of TUNEL-positive β-cells. A minimum of 1,200 cells per sample were counted; the sections were systematically sampled, and when needed, a second section was included.
Glucose-Stimulated Insulin Secretion (GSIS)
After 1 and 3 days in culture, the islets were washed with Krebs–Ringer bicarbonate buffer (NaCl, NaHCO3, CaCl2, 2H2O from Sigma-Aldrich and KCl, MgCl2 from Merck KGaA) supplemented with HEPES (Sigma-Aldrich) (KRBH) and with 2.8 mM glucose (Sigma-Aldrich). Then triplicate groups of 10 islets were incubated in borosilicate vials containing 1 ml KRBH with 2.8 mM glucose (Sigma-Aldrich) for 1 h in a shaking water bath at 37°C (stabilization period). The medium was gently removed and replaced by 1 ml KRBH containing 2.8 mM glucose, and the islets were incubated again for 1 h (basal secretion). The medium was collected and replaced by KRBH 20 mM glucose, and islets were incubated for an additional hour (stimulated secretion). The medium was collected and stored at −80°C until insulin quantification by ELISA (Mercodia, AB, Uppsala, Sweden).
Insulin and DNA Content
After the GSIS assay, islets were washed with phosphate-buffered saline (PBS) (PAA Laboratories GmbH, Pasching, Austria) and sonicated in 1 ml bidistilled water. For insulin content, an aliquot of the homogenate was extracted with acid-ethanol solution and stored at −80°C until insulin quantification by ELISA. The remaining homogenate was stored at −80°C, and DNA content was measured in 0.1 ml of homogenate with a fluorimetric assay using 0.1 μg/ml Hoechst 33258 (Sigma-Aldrich) (excitation wave length 356 nm and emission wave length 448 nm) on a fluorescence spectrophotometer (F-2000; Hitachi Ltd, Tokyo, Japan).
Islet Transplantation Into the Anterior Chamber of the Eye
Islets were transplanted into the anterior chamber of the eye to determine the effect of islet culture with HS or HSA on islet cell death and on graft revascularization. Human islets from three different isolations, cultured for 4 days with HSA or HS as described above, were labeled with a nontoxic fluorescent cell tracer (Vybrant CFDA SE; Molecular Probes, Life Technologies) (10 μM) for 15 min, incubated overnight at 37°C/CO2 5% in a humidified incubator, and transplanted into the anterior chamber of the eye of normoglycemic Balb/c mice (Harlan Laboratories, Horst, The Netherlands) weighing approximately 20 g (male, age range 7-13 weeks old) (n = 13). For transplantation, groups of 150-200 islets were collected into a polyethylene tube (Smiths Medical International, Kent, UK) that was connected to a Hamilton syringe and were carefully injected into the anterior chamber of the eye of anesthetized animals through a corneal incision previously performed with a 27 gauge needle (BD, Madrid, Spain) (40). The anterior chamber of the eye had been considered an immunoprivileged site (40), and immunosuppressant was not administered to transplant recipients.
Graft Revascularization and Islet Cell Death
Ten to 14 days after transplantation, rhodamine B isothiocyanate-dextran (Sigma-Aldrich) (2 mg), propidium iodide (Life Technologies) (50 mg), and Hoescht (Life Technologies) (250 mg) were injected intravenously in each mouse for in vivo assessment of transplanted islet vascularization and β-cell death quantification. Simultaneous two-photon LSM (TPLSM) (Leica Microsystems, Wetzlar, Germany) was used to capture optical sections at different depths in transplanted islets. To determine vascularization, the presence of functional blood vessels, labeled with rhodamine B isothiocyanate-dextran (red), was identified in Vybrant-labeled islets (green), and the number of transplanted islets with functional vessels was assessed. Islet cell death was determined by counting the nuclei labeled with propidium iodide, and results were expressed as number of dead cells per square mm of islet graft area. Mice were subsequently sacrificed, and the eyes harboring the transplanted islets were harvested, fixed overnight in 4% paraformaldehyde at 4°C, and processed for paraffin embedding. Eye sections were double stained for apoptotic nuclei with the TUNEL technique (In Situ Cell Death Detection Kit, ApopTag), and for insulin as described above. Total β-cells and TUNEL-positive β-cells were counted using a Leica DM4000B microscope connected to a digital camera (Leica DFC 310FX) with a color monitor. β-Cell apoptosis was expressed as percentage of TUNEL positive β-cells.
Islet Transplantation Into Nude Mice
Human islets were transplanted under the kidney capsule of nude mice to investigate the effect of pretransplantation islet culture with HS or HSA on the metabolic outcome of the graft. Islets isolated from four different donors were cultured in parallel for 3-4 days with HS (n = 17) or with HSA (n = 16) in nonadherent platelet culture bags (Fenwal Europe SPRL, Mont Saint Guibert, Belgium). After 3-4 days in culture, 2,000 IEQs were counted and transplanted under the kidney capsule of athymic nude-Foxn1nu mice (male, age range 7-23 weeks old) (Harlan Laboratories, Horst, The Netherlands) housed under specific pathogen-free conditions. To determine the potential impact of culture conditions (HSA vs. HS) on the endocrine and nonendocrine cell populations, a sample of cultured cells was collected as for transplantation at the end of the culture period (n = 4 in each group), and it was pelleted by centrifugation, fixed in 4% paraformaldehyde (Merck KGaA), and processed for paraffin embedding. Sections of the pelleted cell preparations were stained with antibodies for insulin (Santa Cruz Biotechnology, Inc.), a cocktail for glucagon (Cell Signaling Technology, Inc., Leiden, The Netherlands), somatostatin (Dako, Glostrup, Denmark), and pancreatic-polypeptide (PP) (Chemicon, Merck Millipore, Darmstadt, Germany), and for amylase (Sigma-Aldrich) and cytokeratin 19 (Dako). Stained cells were counted under the microscope, and the results were expressed as percent number of each cell type over total cell number.
Ten to 14 days after transplantation, mice were intraperitoneally injected with five consecutive daily doses of STZ (Sigma-Aldrich), 70 mg/kg body weight (total dose of 350 mg/kg), freshly dissolved in citrate buffer (Sigma-Aldrich) (pH = 4.5), as previously reported (28). A control group of nontransplanted mice was also injected with STZ. Human islets are known to be exceedingly resistant to STZ (48), and in preliminary experiments we confirmed that this dose of STZ did not induce β-cell damage (data not shown). Thus, after STZ injection that selectively depleted the rodent pancreas from β-cells but preserved the STZ-resistant human β-cells of transplanted islets (48), glucose control was dependent on the function of transplanted human islets. Blood glucose was determined between 9:00 and 11:00 A.M. in nonfasting conditions. Blood was obtained from the snipped tail, and glucose was measured with a portable meter (Glucocard Memory; A. Menarini Diagnostics, Barcelona, Spain). Animals were considered hyperglycemic when blood glucose values were >11.1 mM on two consecutive measurements. Grafts were harvested 60 days after STZ injection. After graft harvesting, blood glucose was determined to assess the appearance or worsening of hyperglycemia.
Statistical Analysis
Statistical analysis was performed using Prism 5.0 (GraphPad, La Jolla, CA, USA). Results are expressed as mean ± SEM (islet cell viability and β-cell apoptosis) and as medians with range (glucose-stimulated insulin secretion results and cell death in islet grafts after transplantation into the anterior chamber of the eye). In figures, mean ± SEM is given to describe the data as it is a better known and more widely used descriptor. Differences between groups were analyzed using the Student's t-test, the Wilcoxon test for paired samples, the Mann–Whitney test for nonpaired samples, and the chi-square test, as appropriate. For multiple comparisons, de Kruskal–Wallis one-way analysis of variance was used, and if statistically significant the post hoc Dunn's test for multiple comparison was subsequently used to adjust for multiple testing. The Kaplan–Meier method was used to analyze the metabolic evolution after transplantation, and the differences among the survival curves were compared by the log-rank test. A value of p < 0.05 was determined as significant.
Results
Islet Cell Viability and β-Cell Apoptosis in Cultured Islets
Islet cell viability was similar after 1 day in culture with media supplemented with HS (80.8 ± 4.4%) or with HSA (75.2 ± 4.5%) (Fig. 1A). In contrast, after 3 days in culture, viability was higher in islets cultured with HS than with HSA (91.7 ± 1.9% vs. 75.3 ± 5.6% p = 0.049), and β-cell apoptosis was lower (1.41 ± 0.27% vs. 2.38 ± 0.39%, p = 0.029) (Fig. 1B).
Islet cell viability and apoptosis. Islet cell viability (A) and β-cell apoptosis at day 3 (B) in human islets cultured with CMRL media supplemented with human serum albumin (HSA, open columns) or with human serum (HS, black columns). Islet cell viability is expressed as percentage of viable cells (acridine orange-positive/propidium iodide-negative cells) over total cells within each islet. Values are mean ± SEM of six different islet isolations with islets from each donor cultured in parallel in both conditions. Paired Student t-test, *p = 0.049 (A) and *p = 0.029 (B).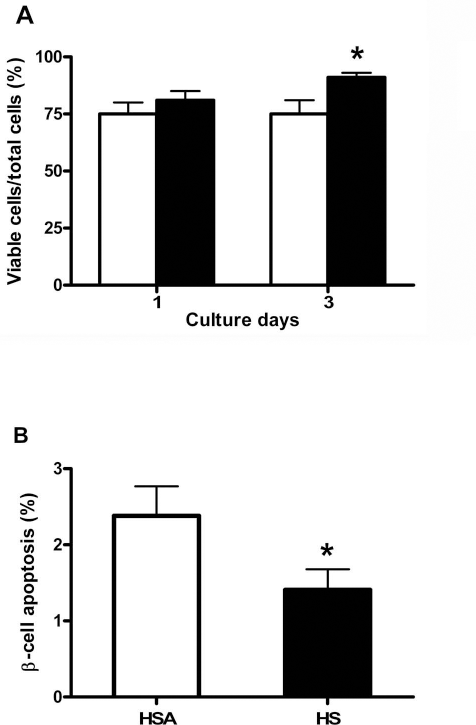
Glucose-Stimulated Insulin Secretion
Glucose stimulated insulin secretion and stimulation index (ratio of insulin secretion at 20 mM vs. 2.8 mM glucose) was similar in islets cultured with HS and with HSA after 1 day in culture. In contrast, GSIS was higher in islets incubated with HS after 3 days in culture: 29.4 (range: 10.4-99.9) versus 22.3 (range: 8.7-70.6) ng of insulin/μg of DNA, p = 0.031 (Fig. 2A). Stimulation index was reduced in HSA-cultured islets on day 3 compared to both day 1 HSA- and day 1 HS-cultured islets (Kruskal–Wallis, p = 0.0063, post hoc Dunn's test for multiple comparison: *p < 0.05) (Fig. 2B). Insulin and DNA content was similar in HS- and HSA-cultured islets on days 1 and 3 (Fig. 2C, D).
Islet function in culture. Islet function in human islets cultured with CMRL media supplemented with human serum albumin (HSA, open columns) or with human serum (HS, black columns). (A) Glucose stimulated insulin secretion. *p = 0.031 (Wilcoxon test). (B) Stimulation index (ratio between insulin secretion at 20 mM and at 2.8 mM glucose). Kruskal–Wallis one-way analysis of variance p = 0.0063, with post hoc Dunn's test for multiple comparison: *p < 0.05 between HSA day 1 and day 3 groups, and between HS day 1 and HSA day 3 groups. (C) Islet insulin content. (D) Islet DNA content. Values are mean ± SEM of four to six different islet isolations, with islets from each donor cultured in parallel in both conditions. Insulin secretion experiments were performed in triplicate.
Islet Vascularization and Cell Death After Transplantation Into the Anterior Chamber of the Eye
Functional new vessels were identified in one of four grafts of HS-cultured islets and in none of the seven HSA-cultured islet grafts. In the vascularized graft, functional vessels were observed in six out of eight evaluated islets (Fig. 3A–C).
Vascularization, viability, and apoptosis of human islets transplanted into the anterior chamber of the eye. In vivo imaging of graft vascularization, islet viability, and β-cell apoptosis in pancreatic islets transplanted into the anterior chamber of the eye. (A–C) Noninvasive in vivo imaging of islet vascularization. (A) Vybrant-labeled islets are shown in green. (B) Blood vessels labeled with rhodamine B isothiocyanate-dextran in red. (C) Merged image with Hoescht-labeled nuclei in blue (25×). Simultaneous two-photon LSM (TPLSM) was used to capture optical sections at different depths in the engrafted islets. (D) In vivo islet cell viability. Results are expressed as number of dead cells per islet area (mm2). Values are mean ± SEM of human serum albumin (HSA)-cultured islets grafts (n = 7) and human serum (HS)-cultured islet grafts (n = 4), (Mann–Whitney test). (E) β-Cell apoptosis in islet grafts. Section was double stained for TUNEL (green nuclei) and insulin (red) (magnification 40×).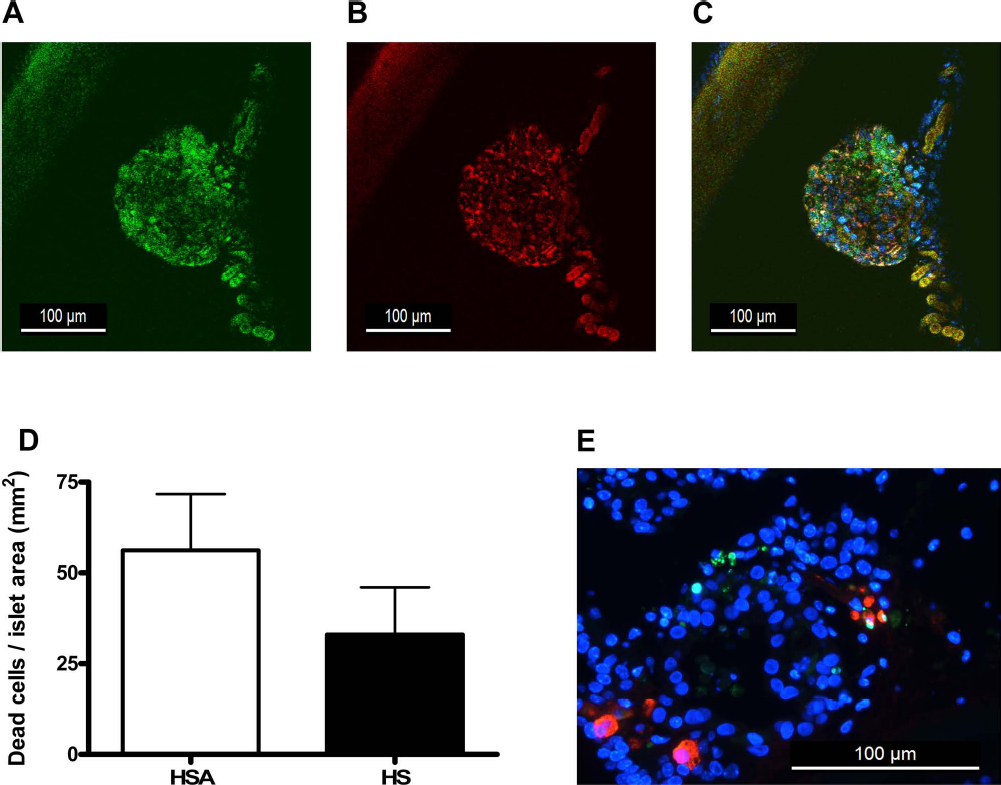
Cell death was similar in HS- and HSA-cultured islet grafts: 43.4 (range: 0-277.3) versus 0 (range: 0-113.8) dead cells/mm2 (Fig. 3D). β-Cell apoptosis was also similar in HS-cultured (15 out of 2,017 β-cells) and HSA-cultured transplanted islets (6 out of 1,087 β-cells) (p = 0.76).
Islet Transplantation Into Nude Mice
The recovery of islets after 1 day in culture was 91 ± 9% and 86 ± 11%, and on day 3 it was 77 ± 11% and 54 ± 1% in HS- and HSA-cultured groups, respectively. Thus, the initial number of islets required to collect 2,000 IEQs after 3 days in culture was 2,597 and 3,704 IEQs for HS- and HSA-cultured islets, respectively. Pellets of cells collected for transplantation at the end of the culture period with HS and HSA showed similar endocrine and nonendocrine cellular composition. The cell composition in HS and HSA groups was β-cells 42.4 ± 3.81% versus 39.3 ± 3.00%, endocrine non-β-cells 25.3 ± 4.44% versus 20.4 ± 4.57%, ductal cells 11.5 ± 1.70% versus 15.8 ± 2.37%, and acinar cells 14.8 ± 3.48% versus 14.0 ± 2.79%, respectively. Blood glucose was significantly lower after human islet transplantation (log-rank test, p < 0.05), a reduction that is in agreement with previous indications that human islets transplanted to rodents regulate the recipient glycemia at the human set point. These lower glycemic levels were maintained after STZ injection. In contrast, STZ injection resulted in the rapid development of severe hyperglycemia and weight loss in the control nontransplanted group, and mice were sacrificed for ethical reasons 2 weeks after STZ injection. Mice transplanted with human islets remained normoglycemic in the initial days after transplantation, and none of them died during the 60-day follow-up after STZ injection (Fig. 4A). Throughout the study, mean blood glucose was similar in mice transplanted with HS- or HSA-cultured islets. Mild to moderate nonfasting hyperglycemia developed progressively in mice transplanted with HS- and with HSA-cultured islets. Hyperglycemia was detected at days 14 (range: 0-53) and 11 (range: 7-48) after STZ injection in HS- and in HSA-cultured groups, respectively, and when grafts were harvested 60 days after STZ injection, 88% and 69% of transplanted mice showed nonfasting hyperglycemia, respectively (Fig. 4B). After graft harvesting, blood glucose increased acutely, indicating a partial function of the graft, concordant with the survival of all transplanted mice and the moderate levels of hyperglycemia in both groups.
Metabolic evolution after human islet transplantation to STZ-injected nude mice. Metabolic evolution of STZ-athymic nude mice transplanted with 2,000 IEQs cultured with human serum albumin (HSA, black squares) or with human serum (HS, open squares) prior to transplantation, and control nonislet transplanted mice (circles). (A) Nonfasting blood glucose. Tx indicates transplantation day, STZ indicates streptozotocin injection, and the arrow indicates the removal of the graft. Values are mean glucose value at each time point. *p < 0.05 among nontransplanted and transplanted animals (Mann–Whitney U-test). (B) Kaplan–Meier survival curve showing differences in percentage of normoglycemic animals (log-rank, χ2 = 41.17, p = 0.0001). There were no differences between human serum albumin and human serum groups (log-rank, χ2 = 0.17, p = 0.28), that showed a significantly better evolution than nontransplanted mice (HSA group log-rank, χ2 = 28.87, p = 0.0001; HS group log-rank, χ2 = 25.48, p = 0.0001).
Discussion
In this study, we have found that supplementation of CMRL culture medium with HS resulted in increased human islet viability and survival, reduced β-cell apoptosis, and higher GSIS compared with HSA supplementation. The higher in vitro survival of HS-cultured islets increased the number of islets available for subsequent transplantation. However, the beneficial effect of HS on islet viability and function did not lead to an improved metabolic evolution when a similar number of HS- and HSA-cultured islets was transplanted to immunodeficient STZ-diabetic mice, and we did not identify differences in grafted β-cell apoptosis or in islet graft vascularization after transplantation into the anterior chamber of the eye.
The addition of fetal bovine serum to culture media is a standard practice in pancreatic rodent islet culture (2). In human islet culture, the use of CMRL media supplemented with 10% fetal bovine serum is recommended (20). However, since the use of xenogeneic products must be avoided in clinical islet transplantation, the common practice is to supplement the culture media with 0.5% human albumin (14,19,21,33), a concentration similar to that of albumin in human serum, or even with 1.5% concentration (6,35). More recently, some groups have suggested that human AB serum supplementation could be superior to HSA. In these studies, the use of HS resulted in improved islet viability, lower apoptosis, increased insulin secretion or β-cell mass preservation during the pretransplantation culture (3,17,24). Serum has an inhibitory effect on pancreatic enzymes and on exogenous enzymes, such as caseinolytic enzymes (38) or collagenase, that reduce insulin secretion and activate apoptotic pathways in islets (4). Moreover, serum contains several growth factors that can activate antiapoptotic and mitogenic pathways in islets, such as growth hormone (18), prolactin (47), IGF-1 (15,30), or IGF-2 (12), providing a basis for the superior beneficial effects of serum supplementation. However, other studies have shown a detrimental effect of serum in human islet culture. It has been reported that serum induces a higher production of NO in the culture medium and has a negative impact on islet function in long-term culture (7). A lower islet recovery and insulin secretion has also been found, and it was suggested that the absence of hormones and growth factors in islets cultured with albumin keeps cells less metabolically active and less vulnerable to hypoxia (29). Our results, in well-standardized culture conditions and parallel experiments with islets isolated from the same donor pancreas, clearly show that the use of HS shows superior benefits, compared with HSA supplementation, in terms of increased islet viability and survival, reduced β-cell apoptosis, and better preservation of β-cell function after 3 days in culture, a similar time period to the one commonly used in clinical human islet transplantation.
The beneficial effects of HS supplementation have been essentially shown in vitro, and there is a general lack of information on whether they may translate into an improved graft outcome when islets are transplanted. To address this question, we transplanted the same number of islets previously cultured with HS or with HSA to immunodeficient mice in which diabetes was induced with STZ. A critical islet mass of 2,000 IEQs was transplanted, sufficient to initially maintain normoglycemia in most animals but insufficient to sustain it over time. It must be noted that due to the different islet survival in culture, the initial number of IEQs required to transplant 2,000 IEQs was 42.6% higher in the HSA-cultured group (2,597 vs. 3,703 IEQs). HS- and HSA-cultured islets were able to maintain normoglycemia initially after STZ injection, but mild hyperglycemia gradually developed in most animals and at a similar time and rate. When islet grafts were harvested, 60 days after STZ injection, the percentage of hyperglycemic animals and the level of hyperglycemia were similar in both groups. After islet graft harvesting, severe hyperglycemia and weight loss developed in both groups, confirming the partial function of transplanted grafts, and again, no differences were identified between groups. These results indicate that the beneficial effects of HS supplementation on islet viability and β-cell function that we had identified at the end of the culture period did not translate into an identifiable metabolic benefit if the same number of islets was subsequently transplanted.
Even though the metabolic evolution is the primary outcome of interest in islet transplantation, it does not provide an indication of the events taking place at the graft level. Thus, in order to have a better assessment of the potential effects of HS supplementation during the pretransplantation culture we determined β-cell viability, β-cell apoptosis, and islet revascularization of the grafts. Islets were transplanted into the anterior chamber of mouse eye, a transplantation site that allows the in vivo assessment of transplanted islet vascularization and viability (23,40). Functional revascularization was detected in a single graft in islets cultured with HS and in none of the HSA-cultured islets. In vivo islet cell viability, expressed as number of death cells per graft area was similar in both groups. The transplanted islets were then harvested and processed for ex vivo specific determination of β-cell apoptosis in graft sections double stained for TUNEL and insulin. In agreement with the in vivo data on islet cell viability, β-cell apoptosis was similar in islet grafts from HS- and HSA-cultured islets. Thus, the transplant experiments did not confirm the superior results achieved in vitro when islets were incubated with HS.
Strengths of the study include the use of in vitro and in vivo models to assess the effect of HSA or HS supplementation. The in vivo experiments included two experimental models, with in vivo and ex vivo evaluation of morphological and functional aspects of the graft. Even though we did not perform additional measurements in islet grafts, such as β-cell replication or islet graft mass, it is unlikely that in the absence of differences in cell viability, β-cell apoptosis, initial islet revascularization, and blood glucose evolution we would have found differences in these additional parameters. The study has also limitations. The 0.5% HSA concentration that we have tested is the one commonly used to supplement human islet culture before transplantation. However, we cannot exclude that a different concentration could yield different results (6). In addition, based on initial reports indicating that the anterior chamber of the eye was an immunoprivileged site (40), immunosuppression of the recipient was not used in these experiments. However, more recent data (1) have shown that there is only partial immunoprotection of the transplanted islets. Thus, partial rejection of xenotransplanted human islets may have had a negative impact on the survival and vascularization of the graft, reducing the possibilities to detect differences between groups transplanted with HS- or HSA-cultured islets.
In summary, the supplementation of culture media with HS provided superior benefits compared with HSA in terms of islet survival, viability, and function. However, when a similar number of HSA- or HS-cultured islets were transplanted, we were unable to confirm a benefit in vivo. We speculate that the in vitro effects of HS on islet cell viability and function may not be sufficient by themselves to have a significant impact on transplanted islets when they face the difficult conditions of islet implantation and survival after transplantation (27). We and others have shown the difficulty of reducing β-cell death in the initial days after islet transplantation (10,11,32,43). Nevertheless, the positive in vitro results indicate that supplementation of culture medium with HS optimizes the number of islets available for transplantation, and it may be expected that it could have an additional beneficial impact if used in combination with other strategies to improve islet transplantation outcome.
Footnotes
Acknowledgments
This work was supported by grants from Instituto de Salud Carlos III (PI10/00636 and PI13/00108), from Center of Regenerative Medicine in Barcelona (CMRB; Promt-0901), from Fundació La Marató de TV3 (FMTV3 121130), from FEDER (EU), and by the Sardà Farriol Research Program. CIBER of Diabetes and Associated Metabolic Diseases (CIBERDEM) is an initiative from ISCIII. Mar Pairó was supported by a grant from IDIBELL.
