Abstract
High pancreatic islet sensitivity to hypoxia is an important issue in the field of pancreatic islet transplantation. A promising strategy to improve islet oxygenation in hypoxic conditions is to leverage the properties of hemoglobin as a natural carrier of oxygen. Studies using human or bovine hemoglobin have failed to demonstrate efficacy, probably due to the molecule being unstable in the absence of protective erythrocytes. Recently, marine worm hemoglobins have been shown to be more stable and to possess higher oxygen carrier potential, with 156 oxygen binding sites per molecule compared to four in humans. Previous studies have shown the beneficial effects of two marine worm hemoglobins, M101 and M201, on nonhuman pancreatic islets. However, their effects on human islets have not been tested or compared. In this study, we assessed the impact of both molecules during human islet culture in vitro under hypoxic conditions. Human islets were exposed to both molecules for 24 h in high islet density-induced hypoxia [600 islet equivalents (IEQ)/cm²]. M101 and M201 reduced the release of hypoxic (VEGF) and apoptotic (cyt c) markers in the medium after 24-h culture. Human islet function or viability was improved in vitro in the presence of these oxygen carriers. Thus, the utilization of M101 or M201 could be a safe and easy way to improve human islet oxygenation and survival in hypoxic conditions as observed during islet culture prior to transplantation or islet encapsulation.
Introduction
Pancreatic islet transplantation is an effective cell therapy to replace deficient insulin secreting cells in case of extreme type 1 diabetes (T1D), pancreatitis, or pancreatic cancer 1–3. Regrettably, few patients benefit from this therapy due to the scarcity of human islet donors and the need for lifelong immunosuppressive treatment4,5. In addition to free islet transplantation, which is done routinely in several countries around the world, islet encapsulation (ie, micro- or macro-encapsulation) is an intense field of research that could potentially enable the use of virtually unlimited cell sources (ie, stem cell-derived or xenogeneic cells) and remove the need for immunosuppression6–9. However, in both free and encapsulated islet transplantation, one of the biggest challenges is the very high sensitivity of human islets, and especially beta cells, toward hypoxia10,11. In vivo, the high vascularization of islets results in oxygen tension around 40 mmHg throughout the islets, allowing beta cells to remain fully functional 12 . However, once islets are isolated and cultured in medium, oxygen diffusion through the liquid medium becomes the only way to supply oxygen to cells, leading to the creation of a hypoxic gradient which is accentuated in bigger islets and can create necrotic zone in the islet core 13 . Hypoxia is an even more critical issue in case of islet encapsulation due to the combination of high islet seeding density and physical barrier limiting diffusion in transplantable devices 14 .
Different oxygenation strategies have been developed over the last two decades to improve islet function and survival. A group used oxygen-permeable plates (made of silicone rubber) to culture human islets at high seeding density (between 2,000 and 4,000 IEQ/cm²) without significant loss of viability compared to classic gas-impermeable plates15,16. With a similar strategy using perfluorocarbons (PFCs) mixed in polydimethylsiloxane (PDMS) to fabricate gas-permeable plates, another group showed that targeting a physiological oxygen tension (oxygen in the incubator at 8–12.5% versus 21%) improved even more overall islet quality 17 . One limitation of these gas-permeable plates is the lack of control over oxygen tension within the culture medium, which could lead to nonhomogeneous distribution of oxygen, especially with bigger islets. To overcome this issue, interesting candidates are oxygen-generating materials like solid peroxides that generate oxygen through self-degradation over a long period of culture18–20. However, one major issue of such oxygen-generating materials is the release of reactive oxygen species (ROS) that can impair cell function. PFCs, which increase oxygen solubility by at least 20 times and has the ability to decrease hypoxia during islet culture21–23, also represents a potential improvement for medium oxygenation. However, when higher islet density is needed, we previously showed that culture of rat islets in the presence of PFCs did not maintain islet function 24 . In the same study, we showed that M201, a natural hemoglobin found in marine worms, reduced inflammation markers and maintained function of rat islets cultured under confinement (600 IEQ/cm²) and hypoxic conditions 24 .
M201 is a large respiratory pigment (3,600 kDa) composed of 156 oxygen binding sites, which makes it a remarkable oxygen carrier as it can carry 39 times as much oxygen molecules as human hemoglobin (four binding sites). An interesting property of hemoglobins is their p50, a value reflecting their affinity for oxygen and corresponding to the oxygen tension at which 50% of the oxygen binding sites are saturated 25 . The lower the p50 value, the higher the affinity for oxygen. Having a p50 at 37 mmHg (~5% pO2), which is close to the oxygen tension found in islets in vivo (ie, ~40 mmHg), makes this molecule suitable for islet culture as it will release half of its bound oxygen at physiological oxygen tension. M201 improved oxygenation of hypoxic areas in 3D-allogenic bone scaffolds, and promoted mesenchymal stem cell proliferation and differentiation 26 . M201 has also been encapsulated in a gel with chimeric antigen receptor (CAR) T cell with IL-15 to enhance CAR T cell survival and proliferation under hypoxic conditions 27 . M101 is a similar oxygen carrier with a p50 at 7 mmHg (~1% pO2), meaning that it releases its oxygen in more hypoxic conditions, a property that can be advantageous for organ oxygenation. M101 is already involved in clinical trials for human kidney preservation 28 and has shown improvement of organ preservation during cold ischemia (CI) of the kidneys29–31, heart 32 , lungs33,34, liver 35 , and pancreas 36 in preclinical studies. These hemoglobins are not glycosylated and therefore do not induce mutagenic, immunogenic or allergenic responses. They are degraded into polypeptide chains and heme37,38.
Therefore, this study aims at assessing the effects of both hemoglobins M101 and M201 on human islets in an in vitro hypoxic model that, if successful, could be translated to islet culture before transplantation. We tested their effect on human islets cultured for 24 h at high-seeding density (600 IEQ/cm²), which induce a stressful environment by creating competition between the islets for access to oxygen and nutrients39,40.
Materials and Methods
Hemoglobins
M201 is an oxygen carrier extracted from the worm Nereis virens and is sold by HEMARINA (
Antibodies
Primary antibodies against GAPDH (anti-rabbit, 1:1,000; Cell Signaling, Ozyme, St. Quentin en Yvelines, France) and SIRT2 (anti-rabbit, 1:1,000; Abcam, Cambridge, UK) were used. Anti-rabbit and anti-mouse horseradish peroxidase-conjugated secondary antibodies were purchased from Sigma-Aldrich (A0545 and A9044).
Human Islets
The use of human tissues in the study was approved by a French regulatory body (Biomedicine Agency, Authorization number PFS12-013). Pancreases were kept in preservation solution at 4°C. Islets were isolated from the pancreases of brain-dead organ donors using collagenase NB1 and neutral protease mix (Serva, Germany). Human islet isolation was performed according to the method described by Ricordi et al 42 .
Experimental Design
After isolation, human islets were cultured in CMRL1066 (Sigma-Aldrich) media supplemented with 2% human albumin serum and 1% amphotericin B/penicillin/streptomycin (Gibco). Human islets were cultured at a high seeding density (600 IEQ/cm²) in 24-well plates (surface area: 2 cm²/well) with 800 µL of culture medium per well to induce hypoxia and provide a confined area 24 . For the experiments comparing the impact of low (150 IEQ/cm²) versus high (600 IEQ/cm²) seeding density, two groups were studied. For the experiments with M101 and M201, three groups were studied at 600 IEQ/cm²: (1) ‘600 IEQ/cm²’; a control group with culture medium without hemoglobin, (2) “600 IEQ/cm² + M101” where the medium was supplemented with the hemoglobin M101 (1 g/l), and (3) “600 IEQ/cm² + M201” where the medium was supplemented with the hemoglobin M201 (1 g/l). Then, the islets were cultured for 24 h at 37°C and 5% CO2.
After 24 h of culture, the medium was frozen at −80°C for analysis of islet-secreted molecules. Cytoplasmic proteins were extracted to analyze protein content. Functionality and viability of islets were assessed using a glucose-stimulated insulin secretion test.
Islet Functionality, Counting, and Viability
Islet functionality was assessed as previously described with 2.8 mM glucose under basal conditions and 16.7 mM glucose under stimulatory conditions 36 . Islets were counted by measuring IEQ, using the consensus reported by Ricordi et al 43 . wherein one IEQ is equal to a pancreatic islet with a diameter of 150 μm. The viability of islets after 24 h of culture under the different conditions was analyzed using fluorescein diacetate (FDA) and propidium iodide (PI) staining (Sigma-Aldrich). Representative images were obtained using a Nikon Eclipse 50i microscope and Nis-Element-BR software (Nikon, Amstelveen, The Netherlands).
Protein Extraction
Cytoplasmic proteins were extracted from the islets in ice-cold commercial lysis buffer (NE-PER™ Nuclear and Cytoplasmic Extraction Reagents, Thermo Fisher Scientific) supplemented with a mixture of protease and phosphatase (1/100) (HALT™ protease and phosphatase inhibitor cocktail, Thermo Fisher Scientific) and preserved at −80°C. Protein concentration was determined using the bicinchoninic acid assay (Thermo Fisher Scientific).
Protein Analysis
Medium culture analysis
Apoptosis was assessed by measuring the amount of cytochrome (cyt) c released by the islets after 24 h of culturing in medium. The release of cyt c by cells is involved in initiating apoptosis, leading to caspase activation 44 . Cyt c levels was quantified using an enzyme-linked immunosorbent assay (ELISA) (Thermo Fisher Scientific, Waltham, USA), and the results were expressed as fold-increase or decrease in relation to the control.
Western blot
20 µg of total protein extraction from human islets were mixed with sample buffer (BioRad, Hercules, USA) and β-mercaptoethanol (1:100 dilution; Sigma-Aldrich) for each sample. Samples were heated at 95°C for 5 min and loaded and separated by electrophoresis on a polyacrylamide gel (Criterion XT, 4–12% Bis-Tris, BioRad). A semi-dry transfer process was used to transfer proteins onto nitrocellulose membranes (Trans-Blot® Turbo™ Transfer System, BioRad). Membranes were blocked for 1 h at 37°C in TBS-T 5% bovine serum albumin (Sigma-Aldrich) and were probed with primary antibodies overnight at 4°C. Blots were revealed using SuperSignal™ West Femto Maximum Sensitivity Substrate (Thermo Fisher Scientific) and recorded with a Chemidoc® instrument (BioRad). Densitometry analysis was performed with BioRad software, and the results were expressed relative to GAPDH.
Statistical Analysis
Statistical analyses were performed using the GraphPad Prism software (San Diego, USA). Results comparing the effect of 600 to 150 IEQ/cm² were analyzed by a nonparametric Mann–Whitney test. Results comparing the effect of 600 IEQ/cm² + M101 and 600 IEQ/cm² + M201 to 600 IEQ/cm² were analyzed by a nonparametric two-way analysis of variance (ANOVA) (Kruskal–Wallis test). The results are expressed as the mean ± standard error of mean (SEM).
Results
Culture of Human Islets at High-Seeding Density (600 IEQ/cm²) Induced a Moderate Impact on Overall Islet Quality After 24 h (21% Oxygen Tension)
Insulin secretion in response to glucose was not significantly altered in human islets cultured at high-seeding density (600 IEQ/cm²) compared with 150 IEQ/cm² (Fig. 1A). However, stimulation index of those islets showed a tendency to be decreased compared to islets cultured at low-seeding density (150 IEQ/cm²) (Fig. 1B, P = 0.06). This decrease in islet function could be related to the apparent increase in insulin release in low glucose condition (Fig. 1A). No impact of high seeding density was observed on overall islet viability after 24 h (Fig. 1C and D).

High islet-seeding density-induced hypoxia (600 IEQ/cm²) induced moderate impact on overall human islet quality after 24 h culture, 21% oxygen tension. (A) Insulin secretion of human islets (n = 10) was measured in basal (2.8 mM glucose) and stimulated (16.7 mM glucose) conditions by ELISA. (B) Corresponding index stimulation (n = 10) was calculated. (C) Viability pictures of human islets (n = 6) were evaluated by FDA/PI staining and (D) estimated% of viability was assessed. Results are expressed as mean ± SEM. Statistical analysis was performed with a Mann–Whitney test and significance is relative to control (150 IEQ/cm²).
M101 and M201 Both Increased Human Islet Function and Viability After 24 h Culture at 600 IEQ/cm² (21% Oxygen Tension)
A significant increase in insulin released in response to glucose stimulation was observed when islets were cultured with M101 (P = 0.05) (Fig. 2A) but not M201. This result was correlated to an increase in human islet stimulation index with M101 (p = 0.0411) and not M201 (Fig. 2B). No impact on total insulin content was observed (data not shown). Cyt c released (a pro-apoptotic molecule) by islets was measured in the medium after 24 h culture. A significant decrease in cyt c released by islets was observed only with M101 (P = 0.048) (Fig. 2C). An increase in overall viability of human islets was found to be significant only in the presence of M201 (P = 0.04); still no increase in cell death was observed for M101 (Fig. 2D and E)

M101 and M201 both increased human islet function in vitro and increased protection against apoptosis (cyt c release) and global cell death (viability staining) after 24 h culture at 600 IEQ/cm², 21% oxygen tension. (A) Insulin secretion of human islets (n = 9) was measured in basal (2.8 mM glucose) and stimulated (16.7 mM glucose) conditions by ELISA. (B) Corresponding index stimulation (n = 9) was calculated. (C) Islet release of cyt c was assessed in human islet-conditioned medium after culture by ELISA (n = 6). (D) Viability pictures of human islets were evaluated by FDA/PI staining and (E) estimated% of viability was assessed (n = 7). All results are expressed as mean ± SEM of the measure rationalized with control. Statistical analysis was performed using a Kruskal–Wallis test and the significance is relative to the control (600 IEQ/cm²). **P < 0.01; *P < 0.05.
M101 and M201 Both Protected Human Islets From Hypoxia (VEGF) While Only M201 Provided Protection Against Oxidative Stress (SIRT2) After 24 h Culture at 600 IEQ/cm² (21% Oxygen Tension)
VEGF secretion decreased significantly in presence of both oxygen carriers, but a more important decrease versus the control was observed for M101 (Fig. 3A, P = 0.0001) compared with M201 (Fig. 3A, P = 0.05). SIRT2 expression, a marker of oxidative stress (a deacetylase that increases in the presence of oxidative stress 45 ), was studied in total protein extract of those human islets. A decrease in SIRT2 expression in human islets was observed only in the presence of M201 (Fig. 3B, p = 0.025).
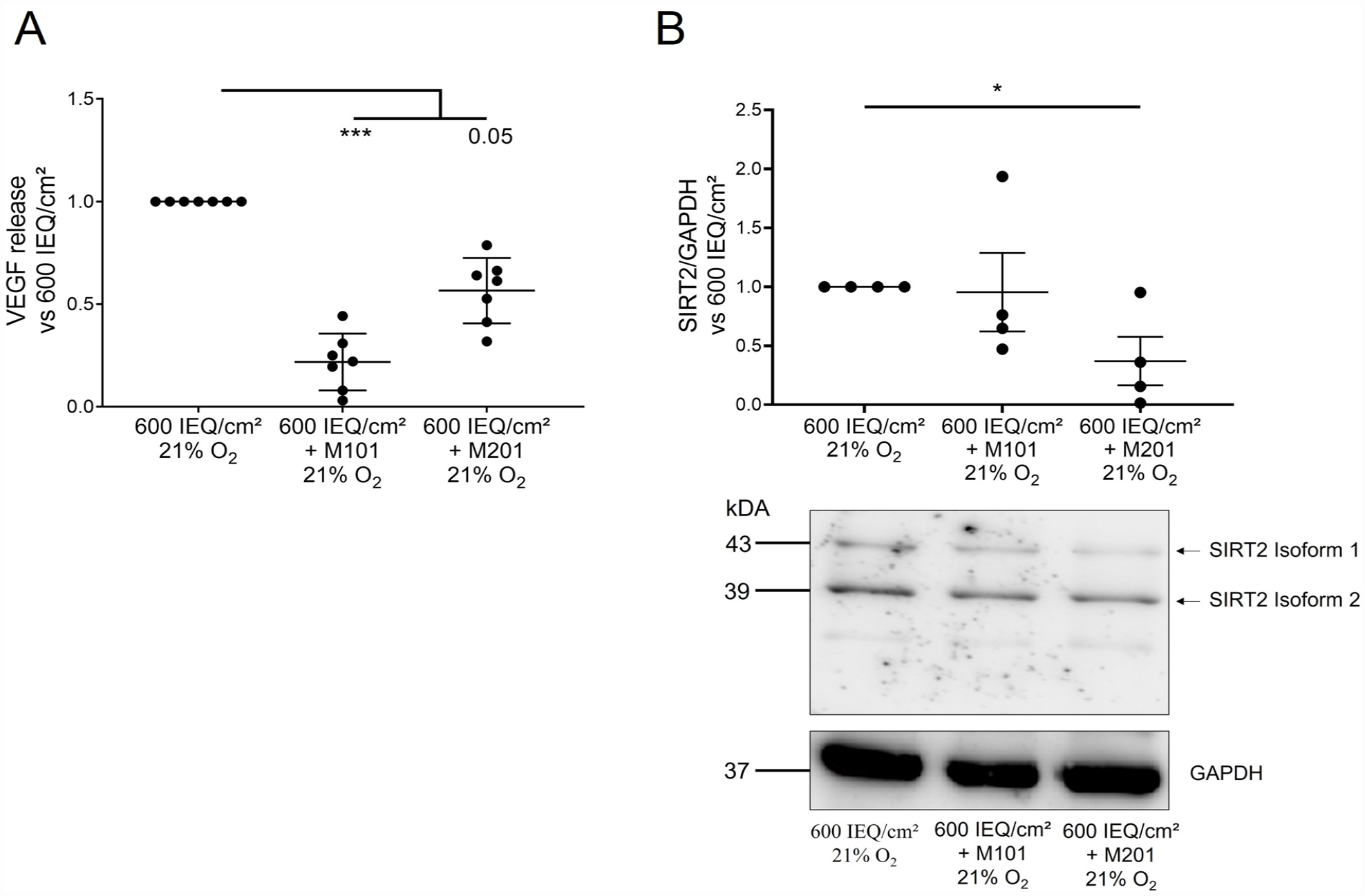
M101 and M201 protected human islets from hypoxia (VEGF) and oxidative stress (SIRT2) after 24 h culture at 600 IEQ/cm², 21% oxygen tension. (A) Islet release of VEGF was assessed in human islet-conditioned medium after culture by ELISA (n = 7). (B) SIRT expression was measured in islet protein extract by western blot (n = 4). Blots are representative of independent experiments. All results are expressed as mean ± SEM of the measure rationalized with control. Statistical analysis was performed using a Kruskal–Wallis test and the significance is relative to the control (600 IEQ/cm²). *** P < 0.001; ** P < 0.01; * P < 0.05.
Both Oxygen Carriers M101 and M201 Showed a Tendency to Improve the Ability of Human Islets to Respond to Glucose Challenge at High-Seeding Density and Low Oxygen Tension (2% Oxygen Tension)
As human islets cultured at 600 IEQ/cm² and at atmospheric oxygen tension (21%) were still considered as functional (stimulation index > 2), we repeated the same experiment at lower oxygen tension (2%) to see if both oxygen carriers could still be beneficial.
While no significant difference was observed on insulin released by islets upon glucose stimulation due to the very low number of human islets preparations (Fig. 4A), stimulation index of three islet preparation out of six were improved in presence of M201 (Fig. 4B). This was associated with restoration of a classic low–high–low insulin secretion in response to glucose challenge (Fig. 4A). No impact on total insulin content was observed (data not shown).

M101 and M201 both reduced variability of islet response to glucose challenges in vitro after 24 h of culture at 600 IEQ/cm², 2% oxygen tension. (A) Insulin secretion of human islets (n = 6) was measured in basal (2.8 mM glucose) and stimulated (16.7 mM glucose) conditions by ELISA. (B) Corresponding index stimulation (n = 6) was calculated. All results are expressed as mean ± SEM of the measure rationalized with control.
Discussion
In this study, we demonstrated that the oxygen carriers M101 and M201 reduce hypoxia-induced damages to human pancreatic islets. We showed that both oxygen carriers improved insulin secretion in response to glucose challenge, probably due to better oxygenation as indicated by a decrease in VEGF released by islets in presence of these molecules. In addition, M101 specifically reduced apoptotic markers while M201 improved oxidative stress status and overall cell viability.
Despite representing only 1% of the total pancreatic mass, pancreatic islets receive between 15% and 20% of the total pancreatic blood flow46–48, providing a constant oxygen tension close to 40 mmHg around islets 12 . As a result of islet vascularization disruption during the islet isolation process, oxygen diffusion from atmospheric air through the culture medium becomes the only source of oxygen for islets. It has been shown that even a very short ischemic period induced by high-density culture decreases function and viability of human islets 49 . Brandhorst et al 39 showed that human islets cultured at 600 IEQ/cm² for 24 h (21% oxygen tension) had drastically impaired ability to respond to glucose stimulation in vitro and decreased viability compared to lower seeding (150 IEQ/cm²). In our hands, the same conditions (ie, 600 vs 150 IEQ/cm², 24 h culture at 21% oxygen tension) resulted in moderate impact on function and no impact on cell viability. Because of the high variability between islet isolation centers (variations due to different protocols, reagents, expertise, etc.), this difference is not so surprising. It is likely that longer time of culture (48 h or more) would have a greater deleterious impact on islets. Of note, in rat islets cultured in the same conditions, function and viability were significantly and negatively impacted at 600 compared with 150 IEQ/cm² culture for 24 h (data not shown). Nevertheless, a tendency for the stimulation index to decrease was observed, showing that human islets were stressed by the high-seeding density. As it is well-demonstrated that human islets are very sensitive to hypoxia even at low seeding density and normoxic conditions, we chose to perform our experiments culturing human islets at 600 IEQ/cm² for 24 h at 21% oxygen tension, a condition that would allow for increased oxygen depletion during culture. Due to their high ability to bind oxygen and their specific p50, both marine oxygen carriers M101 and M201 represent an interesting opportunity to bring an optimal oxygen gradient in the culture before islet transplantation.
VEGF is a hypoxic marker that can stimulated angiogenesis when produced by cells in response to a hypoxic environment50,51. It has been shown that VEGF release by human islets in culture is enhanced with increasing seeding density 39 (82.4 vs 1,014 pg VEGF/pg DNA at 150 vs 600 IEQ/cm² at 21% oxygen tension), probably as a cell response to the lack of oxygen. In our hands, both oxygen carriers decreased VEGF release by human islets at high-seeding density compared with control. It is to note that M101 allowed a stronger decrease compared with M201, with a similar trend observed on rat islets. This difference is likely related to the different p50 between both oxygen carriers (M101: 7 mmHg, M201: 37 mmHg). In static culture condition, islets are relying only on diffusion for their oxygen supply, creating an oxygen gradient throughout the culture medium and within the islets that can result in a necrotic zone in the center of islets 13 . In these conditions, it seems that M101 (releasing its oxygen molecules more effectively in hypoxic conditions than M201), would be more efficient at high islet density to provide adequate oxygenation to maintain islet function. This could be verified by measuring and comparing oxygen tension after culture with or without the oxygen carriers. The decreased expression of cyt c, a pro-apoptotic molecule released by mitochondria to initiate apoptosis, was observed only in the presence of M101, again underlying the different kinetic of oxygen release between both molecules due to their different p50. However, even without significant decrease in apoptosis with M201, the presence of M201 improved the overall cell viability. As no decrease in cell viability was observed in presence of M101, it still indicates the safety of M101 on human islets. In addition, M201 induced a significant decrease in the necrotic marker high–mobility group box 1 (HMGB1) in rat islets (see supplemental data S1). This reduction of necrosis with M201 was linked to improved function of the rat islets in vivo, supporting previous results showing a correlation between HGMB1 and graft failure52,53. Further validation on human islets would be needed, but it suggests that both molecules can reduce cell death pathways.
Oxidative phosphorylation is the preferred pathways of human islets for glucose breakdown, generation of adenosine triphosphate (ATP), and insulin secretion in response to glucose stimulation. It has been shown that human islets increase their oxygen consumption rate (OCR) during high glucose stimulation 54 , emphasizing the crucial role of oxygen in the insulin secretion process. Impairment of beta cell function is correlated to a switch from an aerobic metabolism to an anaerobic metabolism in hypoxic conditions as shown by an increase in lactate production and a decrease in insulin secretion by human islets 55 . Consequently, insulin secretion, insulin biosynthesis, and insulin total content are all negatively impacted by hypoxia. While a moderate impact on insulin secretion was observed in our study, a decrease in the stimulation index in high- versus low-density culture was still observed, likely due to an increase in the insulin secretion in basal conditions, which is a known reaction of islets in hypoxic conditions56,57. Only M101 improved the stimulation index of islets after culture while M201 increased overall islet viability, showing the beneficial impact of these oxygen carriers. Finally, as control human islets were still functional at 600 IEQ/cm² (160 mmHg, stimulation index >2), we also investigated the impact of the oxygen carriers under hypoxic conditions (15 mmHg). Even with the high variability between human islets preparation, both oxygen carriers were able to reduce variability of insulin secreted in response to low glucose stimulation. We also observed that 50% of preparations with M201 improved their stimulation index compared to control. Because OCR is a well-known predictive marker of positive clinical transplantation outcomes58,59, next experiments should take this variable into consideration. Indeed, the oxygen carriers might have a different impact on islets depending on this parameter (low vs. high OCR, for example). The potential difference in OCR in our preparations could also explain some of the variability we observed.
Other groups have shown maintenance of islet survival (ratio OCR/DNA) in gas-permeable culture plate at very high density (between 2,000 and 4,000 IEQ/cm²). Here, we use classic nonoxygen permeable culture plate combined with a lower islet density (600 IEQ/cm²) to study the impact of the hemoglobins, because a combination of very high islet density in gas-impermeable plates would result in too harsh hypoxic conditions that could hide the effects of the oxygen carriers. Even though we observed an overall improvement of islet quality, we recognize that at higher density and in the same conditions, the effect of the hemoglobins might be limited. Indeed, because we performed static incubation in gas-impermeable plates, M101 and M201 might only act as oxygen reservoirs with low possibilities for oxygen reloading. At some point, the oxygen release capacity of M101 and M201 may become too low to provide the oxygen necessary for activating survival pathways. In these conditions, the use of the oxygen carriers in gas-permeable culture plates could drastically benefit islet quality in culture by taking advantages of both strategies: release of oxygen in function of islet needs in the medium from oxygen carriers combined with efficient reloading of these molecules by the gas-permeable plates. This promising combination deserves to be tested in the future. In the case of islets facing hypoxia in an encapsulation device, another source of oxygen to reload the oxygen carriers would be needed during the first days posttransplantation to avoid cell death and loss of graft function (neo-angiogenesis around or within the encapsulation device can take several days to a few weeks in vivo). In this case, a study demonstrated that combination of hemoglobin and calcium peroxide improved the beneficial effect of M101 on porcine islet survival in an encapsulation device 19 .
In addition to the promising impact of combining the hemoglobins to a constant oxygen source, it will be crucial to clearly define the optimal conditions of use of these molecules. Computational modeling has been used to predict the hypoxia level islets face in function of different parameters like islet size, oxygen transport (diffusion or convection), type of material used for culture or for encapsulation and OCR11,60,61, By taking into account the properties of oxygen release of M101 and M201, such model can be used to optimize the dose of oxygen carriers needed, the time where replenishment of those oxygen carriers is needed and the exact oxygen tension surrounding islets.
Recently, another group took inspiration from the oxygen transport strategy used by insects. Unlike in vertebrates, where oxygen supply comes from circulatory blood, insects provide tissue oxygenation through a gas-filled channel network that allows for faster gas exchange than in blood. Leveraging this natural adaptation, the group developed a biomimetic scaffold allowing gas circulation, which increased oxygen diffusion by 10,000-fold compared with hydrogels 62 . They observed a stable 40-mmHg oxygen tension in almost every part of the thick device (6.6 mm) allowing maintenance of cell survival in intraperitoneal space-mimicking conditions. Addition of oxygen carriers like M101 and M201 during the preculture step in vitro, during the encapsulation process and during the host vascularization process (3–4 weeks), would likely have a positive impact at each of these steps. Experiments using insect-inspired gas circulation and enhanced oxygen transport via the use of marine worm hemoglobin confirmed that bioinspiration is a powerful strategy to solve biological issues faced by human medicine.
In conclusion, we showed that for human islet culture, M101 and M201 displayed anti-apoptotic, anti-necrotic and antioxidant effects on human islet in vitro. This is, to the best of our knowledge, the first time that the efficiency and safety of such hemoglobins from marine worms are demonstrated on human islets. Moreover, as M101 has already been validated for use in clinical trials in the case of kidney preservation and transplantation, the utilization of this biotechnological tool during islet culture and/or encapsulation could be rapidly integrated in future clinical trials.The authors thank the entire Center Européen d’étude du Diabète team for their technical help and advice. The M101 and M201 solutions used in this research were supplied by HEMARINA. The authors are indebted to Prof. Wolf and Prof. Bachelier, the local coordination and retrieval team, and the French organ procurement agency (Agence de Biomédecine) for procuring human pancreases.
Supplemental Material
sj-docx-1-cll-10.1177_09636897231179642 – Supplemental material for Beneficial Effects of Two Marine Oxygen Carriers, M101 and M201, on Human Islet Quality in Hypoxic Culture Conditions
Supplemental material, sj-docx-1-cll-10.1177_09636897231179642 for Beneficial Effects of Two Marine Oxygen Carriers, M101 and M201, on Human Islet Quality in Hypoxic Culture Conditions by Florent Lemaire, Séverine Sigrist, Jonathan Brassard, Leila Demini, Franck Zal, Nathalie Jeandidier, Michel Pinget and Elisa Maillard in Cell Transplantation
Footnotes
Acknowledgements
The authors thank the entire Center Européen d’étude du Diabète team for their technical help and advice. The M101 and M201 solutions used in this research were supplied by HEMARINA. The authors are indebted to Prof. Wolf and Prof. Bachelier, the local coordination and retrieval team, and the French organ procurement agency (Agence de Biomédecine) for procuring human pancreases.
Author Contributions
F.L. and E.M. designed and performed the research and wrote the paper. S.S., F.Z., L.D., J.B., N.J., and M.P. provided critical advice for writing the paper. F.L. performed the technical experiments.
Availability of Data and Material
All data are available on request.
Ethical Approval
The use of human tissues in the study was approved by a French regulatory body (Biomedicine Agency, Authorization number PFS12-013).
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the National Institutes of Health and local ethical committee (CREMEAS, France) guidelines (Authorization number: C67-482-28).
Statement of Informed Consent
Written informed consent was obtained from a legally authorized representative for anonymized patient information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: F.Z. is the founder of and holds stock in Hemarina, the company that produces M101 and M201. L.D. is a collaborator of Hemarina and does not have HEMARINA stock.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by CEED and a grant for a PhD student from Société Francophone de Transplantation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
