Abstract
The possibility of enhancing endogenous brain repair following neurological disorders, such as Parkinson's disease (PD), is of considerable recent interest. One such mechanism may exist in the striatum as an upregulated population of tyrosine hydroxylase (TH)-immunoreactive neurons that appear after 1-methyl-4-phenyl-1,2,3,6-tetra-hydropyridine (MPTP) lesions in nonhuman primates as well as in humans with PD. An intriguing possibility is that these endogenous neurons reflect a compensatory mechanism to mitigate the loss of striatal DA due to progressive destruction of the nigrostriatal pathway. The possibility of enhancing the number and function of this population is attractive; however, it is crucial to gain further information about these cells in order to comprehend more fully their possible therapeutic potential. The current research was designed to investigate the fate of this endogenous population in African green monkeys rendered parkinsonian by MPTP lesions. Specifically, we assessed changes in the numbers of striatal neurons expressing TH at differing stages of the toxin-induced behavioral disability and discovered a close relationship with symptom severity and striatal DA neuron numbers. Increased numbers of striatal TH-positive neurons were associated with MPTP treatment that produced parkinsonian symptoms compared to numbers of these neurons in MPTP-treated asymptomatic animals and untreated controls. Expression of striatal DA neurons peaked at the manifestation of symptoms in mild/moderate animals and remained stable in animals that were severely parkinsonian. Furthermore, in severely debilitated animals that improved after fetal dopaminergic grafts, we discovered a return to control levels of the endogenous population. Taken together, our results further support the concept that this population of DA neurons responds to variations in striatal DA tone and may serve as a compensatory mechanism to restore striatal DA levels in the context of significant depletion. Artificially manipulating this endogenous population could prove beneficial for PD treatment, especially for individuals in early disease stages.
Keywords
Introduction
The 1-methyl-4-phenyl-1,2,3,6-tetra-hydropyridine (MPTP) nonhuman primate model serves as an important tool for Parkinson's disease (PD) research. This neurotoxin primarily destroys dopaminergic (DA) neurons in the nigrostriatal pathway while sparing other DA systems, resulting in principal Parkinson-like symptoms (i.e., delayed movement initiations, motor freezing, resting tremor, and others) [for review, see (19, 22, 25)]. Since the initial description in 1983 (6), this model has been employed to investigate potential therapeutic treatments, including studies in our laboratories showing a reversal of MPTP-induced PD symptoms following intrastriatal grafts of fetal ventral mesencephalic DA neurons (27), as well as implantation of human neural stem cells (26) in the African green monkey.
During these investigations, we and others have reported a significant upregulation of endogenous neurons in the striatum, immunoreactive for tyrosine hydroxylase (TH), an accepted DA marker, in animals treated with MPTP (4, 8, 27). This endogenous population under normal conditions can be found in both nonhuman primates and humans in small numbers, primarily residing along the dorsal and rostral borders of the caudate nucleus (Cd) and putamen (Pt) (the striatum) (4, 5, 8, 18, 23, 24). The majority of these TH+ cells are small, uni- or bipolar aspiny neurons with diameters of 6-15 μm, noticeably smaller than substantia nigra (SN) neurons (4, 8, 24, 27). In addition to TH, these neurons also are immunoreactive for the DA transporter (DAT), supporting their status as DA neurons (4, 9, 24). The function of this population is currently unknown; however, the upregulation following a nigrostriatal lesion may represent a compensatory mechanism to restore DA balance to the striatum. Furthermore, how this population responds to the progression of nigrostriatal DA loss is not well understood.
One advantage of the MPTP model is the ability to induce varying severities of the parkinsonian syndrome, which is useful for studying different host responses as a function of progressive nigrostriatal DA loss. For example, we previously examined the correlation between striatal DA function and degree of parkinsonism, in which animals were assigned to different severity classes based on their responses to MPTP treatment (11). These groups were characterized as “asymptomatic, ” “mild, ” “moderate, ” and “severe ” (11). The current study utilized these different categories of behavioral deficits to examine the relationship between the endogenous striatal TH+ neuron population and symptom severity in MPTP-treated African green monkeys. Additionally, we investigated the fate of these TH+ neurons in animals with striatal grafts of fetal ventral mesencephalic tissue.
Materials and Methods
MPTP Treatment and Transplantation
Adult male African green monkeys (Cercopithecus sabaeus; 4-5 years of age) were obtained from wild and bred populations in St. Kitts and housed at the St. Kitts Biomedical Research Foundation (St. Kitts, West Indies). All animals were used in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and approved by the Institutional Animal Care and Use Committee. Control animals (n=4) (Table 1) did not receive MPTP injections or tissue transplantations. All other subjects (n=21) (Table 1) received intramuscular injections of MPTP HCl (four to five daily injections over a 5-day period; RBI/Sigma-Aldrich, Natick, MA, USA), depending on the level of acute toxicity that developed over the treatment period. The resulting cumulative dose for each monkey varied between 1.6 and 2.5 mg/kg (11). Because dose of MPTP may not correspond closely to degree of parkinsonism, monkeys were assigned to severity groups based on their response to the drug during the first month after treatment (11). Experienced observers, blinded to the treatments, scored the behavior of each animal twice a day, 5 days a week. This entailed scoring the frequency and severity of certain behaviors such as prostrate immobility, head and limb tremor, motor freezing, and delayed initiation of movement (30, 31). A parkinsonian summary score (PSS) was derived from these assessments. Monkeys with scores between 0 and 5 were labeled “asymptomatic ” (n=4), 5-12 were “mild, ” 12-10 were “moderate, ” and >40 were “severe ” (n=4) (11, 30, 31). Mild and moderate animals were combined to form a mild/moderate group (n=4). The majority of monkeys used in the symptom severity groups were treated only with MPTP; however, some animals subsequently received control operations (i.e., sham implants or cerebellum tissue implant) following MPTP treatment. Detailed investigations of subjects receiving these control procedures demonstrated no effect on changes in DA or DA metabolite levels, DAT binding, or behavior (9, 10, 29, 32). Additionally, animals in these groups did not differ significantly from other group members in striatal TH+ neuron counts. Time from MPTP treatment to brain removal was 6 to 12 months for all treated animals.
List of Animals Used in Each Group and Subsequent Treatments

The dose range of MPTP (mg/kg) is the cumulative amount over a 4- to 5-day period.
After the initiation of MPTP treatment, eight subjects showing signs of severe parkinsonism received multiple implants of fetal ventral mesencephalic tissue [derived from embryonic (E40-14) Cercopithecus sabaeus donors] into the Cd [procedure described in (27)]. Mean time between MPTP treatment and tissue transplantation was approximately 208 days, and time between transplantation and death was approximately 175 days for all animals. Morphological analysis of grafted cell survival in the host striatum stained for TH revealed five grafted animals with high survival of donor cells and three with low survival of donor cells (i.e., grafts either void of DA neurons or with few DA neurons). Animals were euthanized by a lethal dose of sodium pentobarbital (Lundbeck, Inc., Deerfield, IL, USA). Brains were fixed in paraformaldehyde (Sigma-Aldrich, St. Louis, MO, USA), stored in 30% sucrose (Sigma-Aldrich), and prepped for sectioning.
Immunohistochemistry, Cell Quantification, and Statistics
Frozen brains were sectioned serially into 50-μm coronal sections using a sliding blade microtome. Every fourth section was processed for TH to identify DA neurons. Sections were incubated overnight in primary antibody (TH; Chemicon, Temecula, CA, USA; 1:1,000 in 5% goat serum; Sigma-Aldrich; 0.3% Triton X-100 in phosphate-buffered saline; Sigma-Aldrich) and visualized using diaminobenzidine-nickel chromagen (DAB-nickel; Sigma-Aldrich). Sections to be analyzed were matched for anatomical levels between all subjects and taken at 150-μm intervals beginning at the rostral pole of the head of the Cd at the level of the genu of the corpus callosum and extending through the level where the Pt is prominent and the nucleus accumbens is present. This encompassed a rostral to caudal distance of ~2.5 mm. All TH+ neurons in the Cd containing one or more processes were quantified using unbiased stereology, dissector analysis (7), using the computer software cellSens (Olympus America, Inc., Center Valley, PA, USA) for measuring the reference volume by an investigator blinded to the behavioral score of the animal. Final cell counts were expressed as cells/mm2. A one-way ANOVA with a Fisher LSD post hoc test was used to determine statistical difference between groups at p < 0.05 (Prism; GraphPad Software, San Diego, CA, USA).
Results
Endogenous TH+ Neurons and Symptom Severity
We found many small, uni- and bipolar endogenous TH+ neurons in the Cd following MPTP treatment (Fig. 1). In order to investigate whether these cells represent a possible compensatory mechanism due to striatal DA loss, we analyzed this population through the progression of the syndrome and determined if it correlated with symptom severity. We discovered that subjects showing no signs of symptoms (asymptomatic) contained numbers of TH+ neurons equivalent to untreated control animals (control, 80.9 ± 8.5 vs. asymptomatic, 80 ± 12.48; p ≥0.05) (Fig. 2). In groups where symptoms were apparent (mild/moderate and severe), a significantly greater number of TH+ neurons were measured compared to control and asymptomatic individuals (p ≤0.05) (Fig. 2). There was an equivalent expression of TH+ neurons between mild/moderate and severe groups (mild/moderate, 135.9 ± 11.96 vs. severe, 121.6 ± 17.27; p >0.05) (Fig. 2). The greater number of endogenous TH+ neurons were most evident around the dorsolateral and ventromedial borders of the Cd (Fig. 3).
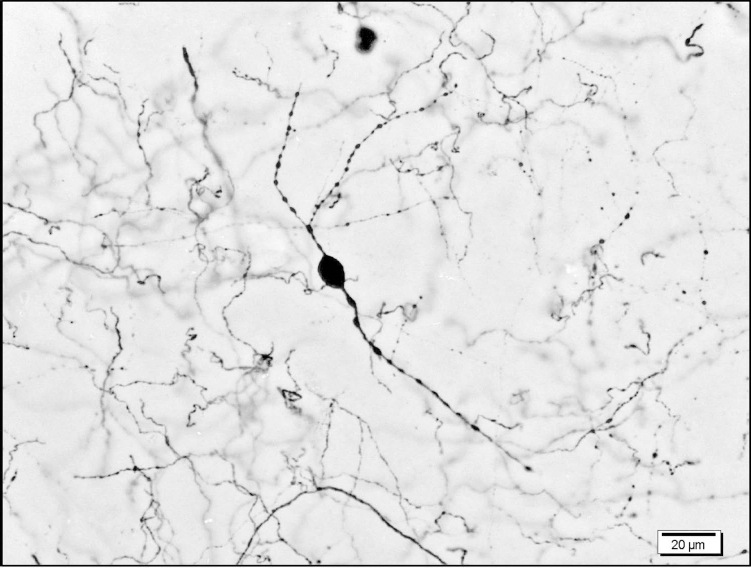
Endogenous TH+ neuron. Representative TH+ neuron in the caudate nucleus after MPTP treatment showing bipolar, beaded neurites, and a dense stain, 40×.

Neuron counts and behavioral symptoms. There was a significant difference in the number of TH+ neurons between control and symptomatic groups, F(3, 12)=4.850, p <0.05. The number of TH+ neurons becomes elevated significantly upon onset of symptoms in mild/moderate animals and remains at a constant high level in severe group animals (mean ± SEM).
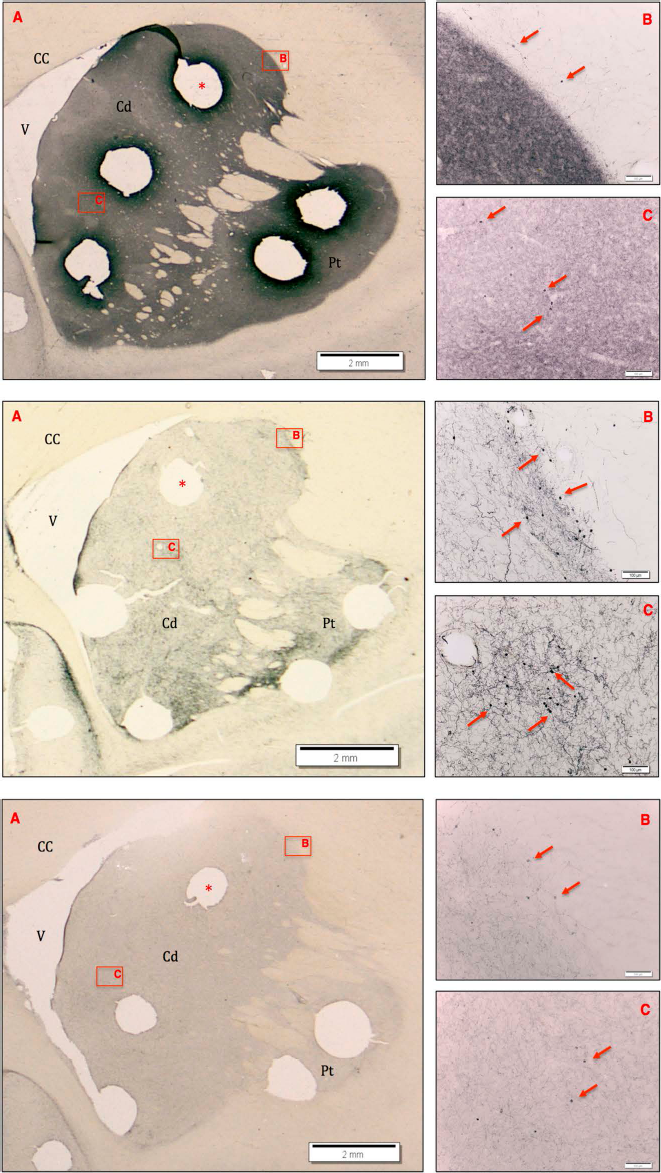
TH+ neuron distribution. Representation of the endogenous TH+ neuron distribution in control (top), MPTP (middle), and fetal graft subjects (bottom panels). Middle panel: (A) Coronal section of striatum illustrating greater number of TH+ neurons following MPTP treatment compared to control (top) and grafted (bottom) animals (scale bar: 2 mm). The two areas of interest (B and C) are seen at higher magnification on the right and display a greater number of cells along the periphery and in the head of the Cd compared to control and grafted animals (scale bar: 100 μm). The same regions are displayed in top and bottom panels for comparison. Arrows indicate TH+ neurons; *one of five micropunches removed for biochemical analysis. CC, corpus callosum; Cd, caudate nucleus; V, lateral ventricle; Pt, putamen.
Endogenous TH+ Neurons and Fetal Tissue Grafts
We hypothesized that by alleviating symptoms in severely parkinsonian animals with fetal DA cell grafts, a blunting of the TH+ neuron response would occur. Tissue slices from a group of severely parkinsonian animals that were previously used to demonstrate behavioral improvements after receiving fetal DA cell grafts were utilized to investigate the effect of fetal DA cell grafts on the number of TH+ neurons in the Cd. Each severely parkinsonian animal received bilateral DA cell grafts in the Cd from early stage (E40-E44) embryonic nonhuman primate donors. Grafted animals were separated into two groups: high donor cell survival within grafts (mean number of cells within all grafts/individual is >10,000) and subjects with low donor cell survival (mean number of cells within all grafts/individual <1,000). Grafts, stained for TH, showed a clear distinction between groups (Fig. 4). We discovered that grafted animals with high donor cell survival contained endogenous striatal TH+ neuron counts similar to untreated controls (p > 0.05) and significantly lower than MPTP-treated-only animals (p < 0.05, Fisher LSD post hoc analysis). The distribution of endogenous TH+ neurons in high-survival grafted animals was 74.6 ± 8.92 compared to 71.4 ± 2.48 for untreated controls and 174.3 ± 43.77 for sham-operated, MPTP-treated-only animals (Fig. 5).

Fetal tissue grafts. (A) Representative image of a fetal cell graft in the caudate nucleus (Cd) with high donor DA neuron survival, stained for TH. (B) Fetal cell graft with low donor DA neuron survival, stained for TH. Arrows outline grafts; *micropunch removed for biochemical analysis.

Neuron counts and fetal tissue grafts. Animals that received fetal cell grafts with high donor DA neuron survival as shown in Figure 4A had endogenous TH+ neuron counts similar to untreated control subjects, that is, significantly lower than MPTP sham-operated animals. Animals with fetal cell grafts with low donor DA neuron survival were not significantly different from MPTP sham-operated animals and approached significance when compared to controls, p = 0.06 [overall ANOVA, F(3, 12) = 4.696, p < 0.05; mean + SEM].
The sham-operated, MPTP-treated-only group was comprised of two severe and two moderately parkinsonian animals, leading to the slightly different TH+ neuron counts seen between these groups in Figures 2 and 5. This was due to the necessity of including sham-operated animals for graft comparisons; the replaced animals in the severe group did not undergo sham operations. Grafted animals with low survival of grafted DA neurons tended to have higher endogenous TH+ neuron counts similar to sham-operated, MPTP-treated-only animals; however, they were also not significantly different from control animals (p > 0.05, Fisher LSD post hoc analysis) (Fig. 5). The distribution of TH+ cells in this group was 145.9 ± 21.22.
Discussion
In this study, we have described a dynamic population of endogenous DA neurons in the Cd of a nonhuman primate model of PD that increase with symptom severity and decrease after the transplantation of DA tissue. Understanding the expression patterns of this endogenous neuron population could provide valuable insight into their functional compensatory role in PD and, by extension, lead to potential advancements in therapeutics. We discovered that the size of this population is significantly greater as animals exhibit symptomatic parkinsonism. Furthermore, by investigating severely parkinsonian animals that previously had behavioral symptoms alleviated by fetal DA cell grafting, we found the number of striatal DA cells returned to control levels, suggesting a role for DA in affecting striatal TH+ cell numbers. This idea is supported further by another study that reported a reduction in striatal TH+ neurons in MPTP-treated monkeys following l-dopa treatment (16). Although the function of these cells is speculative, the correspondence between the increase in population of TH+ neurons and manifestation of behavioral symptoms lends credence to their potential compensatory function. Immunohistochemical analysis provides further support for their status as DA neurons because they stain positively for the DAT (4, 9, 24). However, a future study showing release of DA from these cells and/or synaptic connections to other neurons in the system would be beneficial.
It should be noted that the present study examined the Cd exclusively, in distinction from another study that also examined the Pt (23). However, similar levels of TH+ neurons reported by Palfi et al. were discovered in this region of our symptomatic animals and the MPTP- and GDNF-treated nonhuman primates in the prior study. We studied the Cd and not the Pt because of a) our previous work that revealed more profound changes in the Cd than the Pt following implantation of human neural stem cells into MPTP-treated primates (5) and b) the proximity of these cells to the neurogenic niche of the subventricular zone of the lateral ventricle that gives rise to the rostral migratory stream, which is their most probable source.
Future studies on the potential relationship between the endogenous population and residual DA neurons in the SN could help elucidate a compensatory mechanism. Parkinsonian symptoms do not appear until approximately 60-80% of the nigrostriatal projection is depleted in both humans and nonhuman primates (3, 14). It is reasonable to suggest that the upregulated striatal neurons might contribute in part to the stability of motor function by working in concert with remaining nigrostriatal projections (i.e., the compensatory mechanism may come in a form of delaying the onset of clinical symptoms or the progression to severe symptoms from mild/moderate symptoms). This could explain why we see similar numbers in mild/ moderate and severe animals; that is, neurons already are working at a maximum level and can no longer compensate for the continuous decline of DA in the system, thus leading to further deterioration of motor function. This would imply that the compensatory mechanism ultimately fails during the most severe stages of DA deficits in the system. An exciting advancement would be to artificially enhance this population beyond current maximum levels (pharmacologically or other) in an attempt to increase DA release, potentially acting in a supplemental manner for existing PD therapies (i.e., l-dopa).
In order to fully understand the function of these neurons and ultimately their therapeutic potential, their origin needs to be determined. One possibility is that they arise by neurogenesis following loss of nigrostriatal DA neurons. This seems particularly feasible because of their proximity to the neurogenic niche located in the subventricular zone of the lateral ventricle. This niche gives rise to neuroblasts that travel an impressive distance via the rostral migratory stream (RMS), ventrally along the rostral portion of the striatum to the olfactory bulb in the primate (1, 20). Some of these migrating neuroblasts ultimately become DA interneurons having an inhibitory effect on neural transmission in the olfactory bulb (2, 13, 15). These migrating RMS neuroblasts also share morphological and chemical similarities to the upregulated striatal TH+ neurons reported presently having small, oval, or rounded perikarya containing varicose processes, as well as both populations being immunoreactive for glutamic acid decarboxylase (GAD), a GABAergic neuronal marker, and TH (1, 4, 12, 13). Another explanation involves a possible phenotypic chemical shift from extant neurons in the striatum (4, 21, 28). Huot and Parent proposed that under normal conditions these cells might contain small or undetectable TH protein levels not measurable with current immunohistochemical techniques and may be sensitive to the microenvironment (17). Thus, when DA is reduced or removed from the system, an upregulation of TH may occur that renders the neurons immunoreactive for TH (17). Although this theory argues against neurogenesis as their source, it does not rule out the possibility of migration from the RMS to the striatum.
In conclusion, we discovered a relationship between the endogenous TH+ neuron upregulation and symptom severity in the nonhuman primate model of PD, supporting further their role as a compensatory mechanism. Understanding more thoroughly the dynamics of this population could lead to therapeutic strategies regarding endogenous brain repair as an adjunct or alternative to more invasive surgery. However, it is first crucial to understand the origins and mechanisms of this dynamic neuronal population to determine convincingly if they are a product of neurogenesis, as suspected, or a shift in a chemical phenotype.
Footnotes
Acknowledgments
This work was funded by PO 1 NS24302-15 and a research development award to J.R.S. from the University of Colorado School of Medicine. The authors declare no conflict of interest.
