Abstract
A human embryonic stem cell (HESC) line, H1, was studied after differentiation to a dopaminergic phenotype in vitro in order to carry out in vivo studies in Parkinsonian monkeys. To identify morphological characteristics of transplanted donor cells, HESCs were transfected with a GFP lentiviral vector. Gene expression studies were performed at each step of a neural rosette-based dopaminergic differentiation protocol by RT-PCR. In vitro immunofluorescence revealed that >90% of the differentiated cells exhibited a neuronal phenotype by β-III-tubulin immunocytochemistry, with 17% of the cells coexpressing tyrosine hydroxylase prior to implantation. Biochemical analyses demonstrated dopamine release in culture in response to potassium chloride-induced membrane depolarization, suggesting that the cells synthesized and released dopamine. These characterized, HESC-derived neurons were then implanted into the striatum and midbrain of MPTP (1-methyl-4- phenyl-1,2,3,6-tetrahydropyridine)-exposed monkeys that were triple immunosuppressed. Here we demonstrate robust survival of transplanted HESC-derived neurons after 6 weeks, as well as morphological features consistent with polarization, organization, and extension of processes that integrated into the host striatum. Expression of the dopaminergic marker tyrosine hydroxylase was not maintained in HESC-derived neural grafts in either the striatum or substantia nigra, despite a neuronal morphology and expression of β-III-tubulin. These results suggest that dopamine neuronal cells derived from neuroectoderm in vitro will not maintain the correct midbrain phenotype in vivo in nonhuman primates, contrasted with recent studies showing dopamine neuronal survival using an alternative floorplate method.
Keywords
Introduction
Cellular transplantation of pluripotent stem cell-derived neurons has been demonstrated as a viable option for replacement of dopaminergic neurons in Parkinson's disease. Intracerebral injection of dopaminergic cells directly into the substantia nigra (SN) or the striatum has become a heavily targeted therapeutic approach to provide exogenous dopamine and enhance transmission within the basal ganglia. Several groups have reported functional integration of pluripotent stem cell-derived dopaminergic neurons in both mouse and rat models of Parkinson's disease, utilizing a variety of in vitro differentiation techniques (1,2,5,10,11,14,25,28,29,40).
To date, it remains unclear which differentiation strategy will provide functionally active populations of dopamine neurons in vivo. Neuralization of pluripotent human embryonic stem cells (HESCs) into primary neuroepithelial rosettes followed by application of fate- specific maturation factors has been the standard methodology for generating tyrosine hydroxylase (TH) immunoreactive (ir) dopamine neurons in vitro. Unfortunately, these neurons typically assume an anterior neuroectoderm fate and lack expression of several key transcription factors critical to maintenance of the midbrain dopaminergic phenotype. We therefore hypothesized that exposure to wingless-related mouse mammary tumor virus integration site (Wnt) signaling, an important developmental regulator during midbrain neurogenesis (12,13,15,34), would enhance the generation and survival of midbrain fate TH-ir neurons in vivo.
In order to determine the therapeutic potential of HESC primary neuroectoderm-derived neurons in a clinically relevant model of Parkinson's disease, we differentiated the H1 HESC line into dopaminergic neurons via a rosette-based strategy in vitro. The WA-H1 HESC line was subjected to a modified neural induction and dopaminergic maturation differentiation strategy (38,41) and then characterized in vitro by gene expression studies and biochemical analysis to validate maturation to the dopaminergic phenotype. Differentiated cells were then transplanted into the striatum and SN of asymptomatic, but dopamine-depleted, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) exposed St. Kitts green monkeys (Chlorocebus sabaeus).
Here we report incorporation of predifferentiated, transplanted HESC-derived neurons into the monkey host circuitry. Six weeks posttransplantation, β-III-tubulin-ir neurons extended polarized fibers and incorporated into the host neuronal circuitry. In addition, the monkeys' health and behavior remained stable and showed no abnormalities. Interestingly, the predifferentiated cells displayed no immunoreactivity to TH after 6 weeks despite strong TH expression in vitro. Our results demonstrate that pluripotent stem cell-derived neurons retain the capacity to robustly survive and respond to endogenous cues within the primate brain; however, the absence of TH expression in vivo indicates that other methods may be necessary to produce and maintain the proper midbrain dopaminergic phenotype in vivo.
Materials and Methods
HESC Cultures
HESC line H1 (WA01, passages P44–P54; male; WiCell Research Institute, Madison, WI, USA), previously labeled with a self-inactivating lentivirus vector (Mach7) expressing green fluorescent protein (GFP), provided by Dr. Jun Liao (Yale Stem Cell Center, New Haven, CT, USA), were cultured in an undifferentiated state in feeder-free and serum-free conditions in medium containing Dulbecco's modified Eagle's medium (DMEM)/F12 (Invitrogen Corporation, Carlsbad, CA, USA), N2 (Invitrogen Corporation), B27 (Invitrogen Corporation), l-glutamine (200 mM) (Sigma, St. Louis, MO, USA), nonessential amino acids (NEAA; 0.1 mM) (Gibco, Gaithersburg, MD, USA), penicillin/ streptomycin (P/S; Invitrogen), bovine serum albumin (BSA; 50 mg/100 ml; Sigma), monothioglycerol (MTG; 12 M; Sigma), and basic fibroblast growth factor (bFGF; 50 ng/ml; Peprotech, Rocky Hill, NJ, USA). Differentiated colonies were physically removed using a Pasteur pipette for passaging. The undifferentiated state of ESCs was confirmed by typical morphology and positive immunostaining for octamer-binding transcription factor-4 (Oct-4; see Table 1 for antibody).
Primary and Secondary Antibodies Used
Enrichment of Neural Precursors From HESCs
Using the methods of Xia and Zhang (38), H1 HESC colonies were detached from Matrigel-coated plates (BD Biosciences, Bedford, MA, USA) by treatment with 0.2 mg/ml of dispase (StemCell Technologies, Vancouver, BC, Canada) and grown as floating cell aggregates (embryoid bodies) for 18 days in medium containing DMEM/F12, B27 (50×), Glutamax (200 mM; Invitrogen), and Normocin (100 μg/ml; InvivoGen, San iego, CA, USA). Controlled differentiation of the HESCs was achieved by supplementation with the growth and differentiation-inducing factors Noggin (100 μg/ml) (Peprotech) and bFGF (25 μg/ml; Peprotech), mimicking neuroepithelial induction during development. Media was changed every 2 days. In addition, FGF8 (50 ng/ml; Peprotech) and sonic hedgehog (SHH; 100 ng/ml; R&D Systems, Minneapolis, MN, USA) were added at 12 days. After 18 days of differentiation, neuralized precursor cells were plated onto Matrigel-coated dishes. Within 2 days, cultured cells exhibited columnar morphology and organized into a rosette form. For expansion, the selected neural structures were isolated with accutase (Millipore, Temecula, CA, USA) and cultured in suspension for 1 week in a neural induction medium supplemented with FGF8 (50 ng/ml), SHH (100 ng/ml), 2% B27 supplement and ascorbic acid (AA; 200 μM; Sigma) to generate neurospheres. During this culture period, the spheres were broken into smaller clusters by mechanical pipetting (100–200 μm). The neural induction medium consisted of DMEM/F12, N2, NEAA (0.1 mM), and heparin (2 mg/ ml; sodium salt from porcine intestinal mucosa; Sigma). A schematic and immunochemistry illustrating this method appears in Figure 1.

Neuralization of HESCs into neural progenitor cells. Human embryonic stem cells (HESCs) were differentiated into embryoid bodies (EBs; A) with Noggin and basic fibroblast growth factor (bFGF) to induce primary neutralization that expressed octamer-binding transcription factor 4 (E). Sonic hedgehog (SHH) and fibroblast growth factor 8 (FGF8) were added to ventralize cells toward an isthmic fate. Neuroepithelial (NE) cells formed columnar rosettes (B and C) expressing paired box 6 (Pax6) (F) followed by sex-determining region Y box 1 (Sox1) (G). Neuroectodermal progenitor (NP) cells were selected and expanded as aggregated spheres (D) expressing nestin (H). Scale bars: 20 μm. AA, ascorbic acid; DAPI, 4×,6-diamidino-2-phenylindole.
Dopaminergic Neuron Differentiation
To differentiate the HESC-derived neuroepithelial cells into dopamine neurons, we utilized an accepted methodology (39,41) with slight modification. Neurospheres were dissociated with accutase and plated on Matrigel-coated dishes at a density of 200,000 cells/ml, as illustrated in Figure 2. Cultures were grown for 4 weeks in a neuronal differentiation medium containing neurobasal medium (Gibco), N2 (Gibco), NEAA (0.1 mM), FGF8 (50 ng/ml), SHH (100 ng/ml), B27, AA (200 μM), cyclic adenosine monophosphate (cAMP; 1 μM), laminin (1 μg/ml; from human placenta; Sigma), transforming growth factor type β3 (TGF-β3; 1 ng/ml; R&D Systems), brain-derived neurotrophic factor (BDNF; 20 ng/ml; Peprotech), glial-derived neurotrophic factor (GDNF, 50 ng/ml; Peprotech), and Normocin (100 μg/ml). After 1 week, FGF8 was withdrawn from the neuronal differentiation medium. After 2 weeks, SHH was decreased to 10 ng/ml. Wnt1 (50 ng/ml) and Wnt5a (100 ng/ml) (R&D Systems) were added during the first 3 weeks of differentiation (from day 24 through day 45) to promote the proliferation of specified dopaminergic progenitors (3,12,13,15,34). Media was changed every 2 days.

Maturation of dopaminergic neurons from NE cells. NE rosettes expressed nuclear receptor-related protein 1 (Nurr1) (A) and β-III-tubulin (E) by 30 days. At 52 days, most differentiated cells retained low nestin expression (B), were 95% [±0.92 (SEM)] β-III-tubulin-ir (D–F, H), and 17% [±1.59 (SEM)] tyrosine hydroxylase (TH)-ir (D, G, H). All TH-ir cells colabeled with β-III-tubulin (D), while no differentiated cells expressed dopamine β hydroxylase (DBH; C). Scale bars: 20 μm. cAMP, cyclic adenosine monophosphate; TGFb3, tumor necrosis factor-β3; BDNF, brain-derived neurotrophic factor; GDNF, glial-derived neurotrophic factor. Wnt1, wingless-related mouse mammary tumor virus integration site 1; GFP, green fluorescent protein.
Species Selection, MPTP Administration, and Behavioral Outcome
Adult male St. Kitts/African green monkeys (Chlorocebus sabaeus) were systematically treated with MPTP (Sigma) via intramuscular injections administered over a period of 5 days, totaling a dose of 2 mg/per kg of body weight. Four doses were administered with a spacing of approximately 12 h between each dosing for the first 3 days, with the fifth dose being administered on the morning of the fifth day. This standard dose of MPTP leads to variable levels of parkinsonism, but all monkeys exhibit significant reductions in dopamine concentration (26). For these preliminary methodological studies, we selected two asymptomatic monkeys because they do not require special nursing care or feeding and have no pain, disability, or stress from being symptomatic. The behavior of the dopamine-depleted but asymptomatic MPTP-exposed monkeys was observed after cell injections using our standard methods (26). During this time, they appeared to be healthy and showed normal unchanged behaviors from their previous state prior to the injections. They were sacrificed after 6 weeks for postmortem brain analysis. The experimental protocols were approved by the institutional animal care and use committee of Axion Research Foundation, which is AAALAC accredited and has assured compliance (A4384) with the office of Laboratory Animal Welfare of the NIH. The use of human embryonic stem cells was approved by the Embryonic Stem Cell Research Oversight committee of Yale University.
Stereotactic Surgical Injection of HESC-Derived Neurons in Monkey Brain
Using sterile techniques, a 22-gauge needle connected to a Hamilton syringe (Reno, NV, USA) was lowered to the desired depth in properly anesthetized monkeys and left in situ for 2 min pre- and postinjection as previously described (27,30,36); HESC-derived cells, 12.5 μl, at day 57 of dopaminergic differentiation were injected into monkey Y787 and 9 μl into monkey Y880 at a concentration of 5 × 105 cells/μl into each site at 1 μl/min. The immunosuppressants azathioprine (Sigma), cyclosporine A (Sandiummume; Henry Schein Animal Health, Pittsburgh, PA, USA), and prednisolone (Henry Schein Animal Health) were administered from before the injections until sacrifice, according to our previous report (27).
Immunocytochemistry and Cell Quantification
At different stages of differentiation, cells were cultured on glass chamber slides (Becton Dickinson, Franklin Lakes, NJ, USA), fixed in 4% paraformaldehyde (Sigma), and processed by standard immunocytochemisty as previously described (27). The primary and secondary antibodies used are listed in Table 1. Cell nuclei were stained with 4×,6-diamidino-2-phenylindole (DAPI; 1.5 μg/ml; Roche, Indianapolis, IN, USA). Staining was visualized with a Nikon fluorescence microscope (Melville, NY, USA). Brain tissue preparation and immunostaining of sections were performed as previously described (27). Brain sections from adult rats and normal monkeys were used as positive controls. Omitting the primary or secondary antibodies in the immunostaining procedures also set negative controls. Quantitative analysis was performed at randomly selected visual fields. In each field, images at separate channels (DAPI, 488, 594, merge) were acquired at 40× magnification on a fluorescence microscope using Axiovision image capture equipment and software (Carl Zeiss, Thornwood, NY, USA). Ten visual fields per coverslip were analyzed and counted from each of eight replicate coverslips. The total number of TH-ir and β-III-tubulin-ir cells was plotted as the percentage of total DAPI-ir cells. Data were represented as mean ± SEM.
Electron Microscopy
Tissue blocks containing the areas of interest were retrieved from previously frozen sections, cut (100 μm) on the vibratome (Warner Instruments, Harvard Apparatus, Hamden, CT, USA), osmicated, and embedded in Araldite (Huntsman Advanced Materials, Basel, Switzerland). Ultrathin sections were analyzed using a Tecnai™ electron microscope (FEI, Hillsboro, OR, USA).
Reverse Transcription-PCR
Total RNA was extracted from HESCs at different time points concurrent with maturation of dopamine neurons. The time points were day 0 for undifferentiated HESCs, day 24 for differentiated neural progenitors, day 38 and day 45 for maturing neurons, and day 52 for differentiated dopamine cells. Total RNA was captured using the RNeasy purification kit (Qiagen, Germantown, MD, USA; cat# 74104) followed by the treatment with DNase I (Thermo Fisher Scientific, Madison, CT, USA) to remove genomic contamination. Synthesis of cDNA was carried out with the SuperScript II First-Strand Synthesis System for RT PCR (Invitrogen, cat# 18080-051) according to the manufacturer's instructions. PCR amplification was performed using a standard procedure with Platinium Pfx DNA polymerase during 30 cycles with denaturation at 94°C for 15 s, annealing temperature at 55°C for 30 s, and elongation at 68°C for 1 min. The first-strand cDNA was further amplified by PCR using individual primer pairs for specific genes. The RT-PCR primer sequences and PCR product sizes for Oct-4, paired box 6 (Pax6), nestin, β-III-tubulin, nuclear receptor-related 1 protein (Nurr1), TH, dopamine transporter (DAT), and glyceraldehyde 3-phosphate dehydrogenase (GADPH) are listed in Table 2. Amplicons were electrophoresed through 1.0% Tris base, acetic acid, and ethylenediaminetetraacetic acid (EDTA) (TAE; Thermo Fisher Scientific) agarose gels (American Bioanalytical, Natick, MA, USA), stained with ethidium bromide (Fisher Scientific, Fair Lawn, NJ, USA), and visualized on a UV transilluminator (M-20E; UVP, Upland, CA, USA).
Reverse Transcription-PCR Primers, Sequences, Tm, and Size

GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Depolarization-Evoked Dopamine Release and Intracellular Dopamine Content
At the end of the dopaminergic neuronal differentiation protocol, activity-dependent dopamine release from the cultured cells was measured using reverse-phase high-performance liquid chromatography (HPLC; LC-10ADVP; Shimadzu Scientific Instruments, Kyoto, Japan) (20). Differentiated cells were incubated in neural differentiation medium + 200 μM ascorbic acid (AA) or in neural differentiation medium + 200 μM AA + 56 mM KCl (evoked; Sigma) for 30 min at 37°C. The media was then collected and stabilized with 0.1 M perchloric acid (J. T. Baker, Phillipsburg, NJ, USA) + 0.1 mM EDTA (American Bioanalytical), and the samples were stored at −80°C until they were analyzed. Dihydroxybenzylamine (Sigma) was added to each sample to serve as internal standard, then pH was adjusted to 8.2, and alumina (Sigma) added to the mixture. Catechols were adsorbed to alumina by vortexing the sample. The alumina fraction was recovered and transferred to a small column, which allowed an efficient wash of alumina with water and elution of catechols from alumina in a small volume (0.1 ml) of perchloric acid. An aliquot was separated by isocratic reverse-phase HPLC, and dopamine and internal standard were detected in each sample by electrochemical detection. The ratio of dopamine and internal standard signals was calculated, and dopamine level in each sample was quantified with respect to external standards.
Results
In Vitro Neuralization of HESCs Into Neural Precursor Cells
Building on well-established protocols (22,38–41), we modified an efficient differentiation method to produce a large percentage of neurons from HESCs for transplantation. To generate dopaminergic neurons in vitro, two stages of differentiation were used: an initial rosette-based neuralization phase to derive synchronized primitive anterior neuroepithelia (NE) (38,41) (Fig. 1), followed by a secondary neuronal maturation step to generate TH-ir dopaminergic neurons (39,41) (Fig. 2).
HESC line WA01 was previously labeled with a lentivirus vector expressing GFP to track cell fate in vivo. HESC colonies expressing Oct-4 (Fig. 1E) were dissociated, and aggregates of cells were grown in the presence of bFGF and Noggin. Noggin, a bone morphogenic protein antagonist, has been reported to induce neural precursor development as well as increase the yield of dopaminergic neurons from HESCs (5,31). After 6 days, dissociated cells formed embryoid bodies (EBs) (Fig. 1A) and were replated onto laminin-coated dishes to induce attachment to the surface. Neural progenitor selection began at day 12 with the addition of developmentally critical morphogens, FGF8 and SHH (12), to enhance ventralization of early precursor cells toward the isthmic identity as previously demonstrated (39). Attached neuroectoderm columnar cells formed small adherent colonies around day 12 and expressed Pax6, indicative of early NE (Fig. 1B, F). By day 18, the differentiated cells organized into rosettes, expanded, and expressed Sox1, a transcription factor marking definitive NE cells (Fig. 1C, G). After day 18, the cells were detached physically and expanded in suspension to form proliferative neurospheres (Fig. 1D). At day 24, NE precursor cells expressed the standard neural precursor cell marker, nestin, when cultured under adherent conditions for imaging (Fig. 1H).
In Vitro Maturation of HESC-Derived NE Cells Into Dopaminergic Neurons
Upon completion of the neuralization process into definitive NE, the cells were differentiated toward the dopaminergic phenotype utilizing a slight modification of a well-accepted methodology (39–41) in neural differentiation medium supplemented with FGF8, SHH, AA, TGF-β3, BDNF, (Fig. 2, schematic). In addition, Wnt1 and Wnt5a, important developmental components to dopaminergic neurogenesis, were added to the medium in substitute for Wnt3a-conditioned medium to enhance maturation into the midbrain fate (12,13). Within 2–3 days, cells displayed typical neuronal morphologies with elongated processes and expressed the prodopaminergic transcription factor Nurr1 (Fig. 2A) as well as the neurofilament β-III-tubulin (Fig. 2E). After 52 days of differentiation, the percentage of β-III-tubulin-ir neuronal cells reached 95.68 ± 0.92% (SEM) (Fig. 2D, F), and the proportion of TH-ir cells (Fig. 2B–D, G) increased to 17.13 ± 1.59% (SEM) of the entire population of cells (Fig. 2H). These results indicated that the neuroepithelia rosette-based dopaminergic differentiation protocol represented an efficient method to generate neuroectoderm and derive TH-ir neurons from pluripotent HESCs.
RT-PCR Analysis of HESC-Derived TH-ir Neurons
To track dopaminergic development, HESCs and their neural derivatives were characterized by RT-PCR at each step of the differentiation process. Oct-4, a marker for undifferentiated HESC, was detected at the pluripotent stage but not after the initiation of neuralization (Fig. 3A). At day 24, cells expressed the neural progenitor markers Pax6 and nestin. By day 38, expression of the proneuronal marker β-III-tubulin and transcription factor Nurr-1 was also prominent. Importantly, by day 52, cells expressed transcripts for the prodopaminergic neuronal marker TH (rate-limiting enzyme in dopamine synthesis), as well as weak expression of the dopamine transporter (DAT). Collectively, the RT-PCR results coincide with immunostaining data, indicating maturation of HESC into dopaminergic neurons in vitro, utilizing the modified NE rosette-based protocol.

Gene expression and potassium-stimulated dopamine release. (A) Gene expression of proteins involved in dopaminergic differentiation from day 0 (D0) to day 52 (D52) from the undifferentiated state (ES) confirmed the immunohistochemistry. HESCs turned off Oct-4 and expressed nestin followed by Pax6 and β-III-tubulin. Neural precursor (NP) cells matured into dopaminergic neurons expressing Nurr-1, TH, and some dopamine transporter (DAT) (composite illustrative figure taken from multiple gels; *these time points were not run). (B) Dopamine was present in medium at day 52 and day 72. Depolarization with 56 mM KCl for 30 min increased dopamine compared with parallel cultures from 30-min control, suggesting activity-dependent release. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as a loading control.
Biochemical Analysis of HESC-Derived TH-ir Neurons
To explore whether the HESC-derived TH-ir dopaminergic neurons produced dopamine, analyses were performed on duplicates of cultured neurons at days 52 and 72 by measuring dopamine release in response to membrane depolarization. No dopamine could be detected in samples of media alone. Under control conditions (cultures in cell media), dopamine levels were high enough to be detected in the media (Fig. 3B). Depolarization of the cultured neurons by exposure to 56 mM KCl increased the amount of dopamine (+39%) in the media. In parallel cultures, the magnitude of the induced release appeared greater in day 72 cells compared with day 52 cells (+275%). This observation suggests that, in addition to expressing TH, day 52 and day 72 cells are able to synthesize and release dopamine in an activity-dependent manner.
HESC-Derived Cell Transplants Incorporate Into the Host Brain Circuitry
In an initial preclinical effort to test the therapeutic potential of HESC-derived dopamine neurons, we injected differentiated neurons at day 57 into MPTP-exposed vervet monkeys. Cells were injected into two sites, the substantia nigra and the caudate, of immunosuppressed monkeys and evaluated after 6 weeks.
High-density striatal grafts of HESC-derived neuronal cells were stained with the human-specific cytoplasm antibody (STEM121) and imaged in coronal section with darkfield illumination (Figs. 4–7). Only grafted human cells are labeled with this species-specific antibody illuminating several morphologically distinct characteristics including 1) beaded fibers coursing parallel to the corpus callosum (CC) (Fig. 7), 2) neurites that penetrate the neuropil of the internal capsule (IC) reaching the putamen (Pt) (Fig. 5A, B), 3) delicate parallel fibers that penetrate the griseum pontis caudato lenticulares (Fig. 5C, D), and 4) fibers that course toward the ependymal lining of the lateral ventricle (V) (Fig. 7).

Robust survival of HESC-derived neuronal grafts in the monkey striatum. Human cytoplasm antibody (Hu Cyto) and darkfield illumination revealed extensive striatal graft survival with four unique areas of interest (A). The first (1) appears as parallel arrays of linear profiles of beaded fibers among the myelinated fibers of the corpus callosum (CC). The second (2) are dense collections of neurites that penetrate the neuropil between the fascicles of the internal capsule (IC) to reach the adjacent putamen (Pt). They appear to course laterally from the implant site, but not medially, and are interpreted as long-distance fibers that would be destined to integrate into major fiber tracts in the nervous system. The third (3) also penetrates the neuropil between the fascicles, gray matter that has been termed the griseum pontis caudato lenticulares, but these fibers appear as delicate strands of parallel fibers in contrast to those described for area (2) and are seen to advantage in (B). The fourth (4) area is seen medial to the implant site (*) and consists of numerous linear profiles. (B) Arrows extend toward the lateral ventricle (V), some in proximity to the ventricular ependymal cell lining. Scale bars: (A) 200 μm, (B) 100 μm.
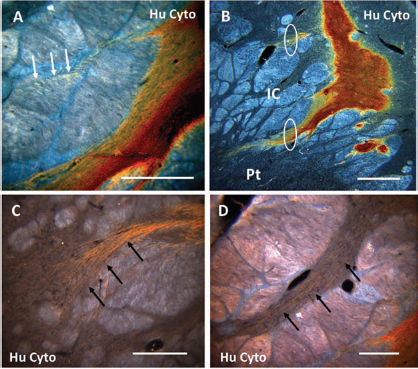
Extensive integration of HESC-derived neurons in the primate striatum. HESC-derived neuronal grafts project through the internal capsule to the putamen. Dense collections of neurites penetrated the neuropil of gray matter that appears to spate the separate fascicles of the internal capsule (IC) to reach the adjacent putamen (Pt). Although the majority of these bundles coursed laterally (B, white ovals), some turned caudally in a neuritic genu to descend toward the mesodiencephalic junction (not shown) and are seen in the form of short fibers (arrows in A) instead of linear profiles. (C) Fibers in area (2, Fig. 4) appeared to spread in a fan-like formation as they approached the medial boundary of the putamen. (D) Fibers of the third area coursed laterally toward the putamen between the fascicles of the internal capsule but were not as densely packed as those that gave the appearance of early stages of long tract formation. Scale bars: (A, C, D) 100 μm, (B) 200 μm.

Postmortem graft survival and host integration of HESC-derived neurons. GFP-ir neuronal cell grafts survived robustly and integrated within the host. Representative low-magnification image of (A) monkey Y787 striatal graft and (C) monkey Y880 substantia nigra (SN) graft. Higher power images (B, D, E, G) showed that grafted cells from Y787 were morphologically consistent with a healthy neuronal profile, bearing large soma and intact, punctate processes. (E) Fine neuronal processes and enlarged soma typical of grafted cells (from D). The ventral striatal graft in Y787 in (G) revealed extensive GFP-ir axons polarized and streaming in the direction of host white matter bundles (arrow). While most of the grafted GFP-ir cells were β-III-tubulin-ir (B, red), none of the grafted cells colabeled with TH. (B) and (E) are higher magnification images from the same section in (D). Higher power images from Y880 (C) are shown in (F) and (H). ic, internal capsule, opt, optic tract. Scale bars: (A, C) 1,000 μm, (B, F) 50 μm, (D, G) 100 μm, (E) 15 μm, (H) 20 μm.

Integration of HESC-derived neurons in the primate brain. The most dorsal fiber region is seen within the corpus callosum (CC) and extends both in a medial and lateral direction from the implant track (A). The parallel, linear, beaded fibers course among the interconnecting, heavily myelinated fibers of the corpus callosum and were traced for relatively short distances within the white matter of this interhemispheric tract. An overview of the region is seen in the two-panel montage to the right (B). The fourth area is seen medial to the implant site (*) and consists of numerous linear profiles (arrows) that extend toward the lateral ventricle (V), reaching near the ependymal lining. Scale bars: (A) 200 μm, (B) 500 μm.
Immunohistochemistry for GFP, TH, β-III-tubulin, and human-specific cytoplasm (hu cyto) was performed on sagittal sections to determine the identity, location, and migration of grafted cells in the SN and striatum. In agreement with results from coronal sections, robust survival of HESC-derived neuronal grafts was confirmed with staining for GFP in both the striatum (Fig. 6A, D) and SN (Fig. 6C, F) of two MPTP-exposed vervet monkeys. Grafted cells in the striatum had a morphological profile consistent with that of early stage neurons with an enlarged soma, bearing punctate fibers (Fig. 6E, G).
Long, polarized GFP-ir fibers were also found coursing toward and along host white matter tracts (Figs. 4, 5, 6). The results suggest reciprocal communication between graft and host and provide further evidence for preservation of a permissive growth environment in the dopamine-depleted primate brain. Immunohistochemistry of adjacent sections suggested that GFP-ir donor axons were integrating and migrating through spared host circuitry including the nigrostriatal tract (data not shown). Similar results were found within the midbrain, where robust GFP-ir grafts typically retained morphological features consistent with developing neurons including large soma and healthy fine punctate fibers (Fig. 6F, H). Although most cells were β-III-tubulin-ir (Fig. 6B), no grafted cells showed convincing TH expression, despite marked expression in vitro prior to transplantation. In addition, grafts within the striatum and SN lacked expression of Nurr-1, En1, and FoxA2, master regulators of midbrain dopaminergic neurogenesis (16,18,19), as well as DAT and VMAT, which are more stable markers of dopaminergic neuronal elements.
Electron microscopy on human cytoplasm-ir cells revealed that the surviving healthy cells had characteristics of immature neuroblasts. Among these cells were some dead cells, glia, and a few macrophages. We did not see host axons contacting human cytoplasm-ir stem cells or human cytoplasm-positive boutons forming contacts with host cells (Fig. 8).

Electron micrograph of human cytoplasm-ir graft in the monkey striatum. Owing to initial frozen sectioning and lack of glutaraldehyde in the fixative, the structural preservation was poor for electron microscopy. However, human cytoplasm-ir cells (C) and their processes (some of them are labeled with arrows) could be recognized. Among the healthy-looking stem cells there are some dead cells (DC). Scale bar: 2 μm.
Discussion
Parkinson's disease was one of the first neurological disorders to be studied for potential replacement of lost neurons, beginning with fetal substantia nigra dopamine precursor cells in the late 1970s. Since then, these cells have been extensively studied, including in human trials, which saw limited success and identified several problems. Neural stem cells, embryonic stem cells, and induced somatic sources have provided new avenues for creating cells to restore function in Parkinson's disease. There has also been significant progress with learning the required gene expression, growth factors, and culture conditions for differentiating cells into apparent dopamine neurons. However, with the exception of a recent report (14), dopamine neurons differentiated and characterized in culture have not produced long-lasting midbrain-specific neurons when transplanted in rodents or monkeys, and there have been only pilot reports of functional improvement.
The objective of our studies has been to test HESC-derived dopaminergic neurons for long-term survival and functional benefits after stereotaxic placement into the nigrostriatal system in the nonhuman primate model of Parkinson's disease: MPTP-exposed monkeys. In the present study, we tested the survival capacity of H1 HESC-derived dopaminergic cells that were predifferentiated utilizing a modification of a well-accepted rosette-based method of neuralization to induce neuroepithelial cells from pluripotent cells (38,41). Neuroepithelial cells were then differentiated into mature dopaminergic neurons following another accepted methodology (39,41) that was previously shown to reverse functional deficits in parkinsonian rats (40). A large literature has shown that members of the Wnt family of secreted glycoproteins are expressed in the midbrain and regulate precursor proliferation and neural differentiation (24) in the nervous system. In addition, Wnt-1 and Wnt-5a increased the number of rat midbrain dopamine neurons in rat embryonic precursor cultures by increasing the proliferation of Nurr1-positive precursors and by promoting the acquisition of a dopamine phenotype in Nurr1-expressing precursors, respectively (3). Another study clearly showed that the effects of Wnt-5a overexpression could also be achieved in vitro by treatment of cultures with recombinant Wnt5a protein (100 ng/ml) during the period of differentiation (23). Therefore, we further hypothesized that addition of recombinant Wnt signaling through recombinant proteins during the dopaminergic maturation phase of differentiation would enhance specification and maintenance of the midbrain fate in vivo.
Prior to implantation, gene expression data and protein analysis by immunohistochemistry and HPLC confirmed the derivation of differentiated neurons that appeared to show a dopaminergic neuronal phenotype in vitro including Nurr1 and TH expression as well as dopamine release. Six weeks after transplantation into the dopamine-depleted primate brain, GFP-ir, HESC-derived neuronal cells showed remarkable survival, robust integration into the host, and exhibited morphological traits consistent with healthy β-III-tubulin-ir neurons in both the caudate and substantia nigra. However, although they possessed a characteristic neuronal profile and appeared to be extending axons into the nigrostriatal bundle, no TH-ir cells were detected in these grafts after 6 weeks in vivo. These results suggest that the HESC-derived dopaminergic progenitors were either not fully specified toward the midbrain lineage or lacked additional regulatory cues essential for maturation into the midbrain-specific lineage and maintenance of the dopaminergic phenotype. The present results demonstrate robust survival of HESC-derived cells, although they confirm previous reports using other stem cell sources that TH gene expression may be transient upon transplantation in vivo (21). In addition, these data raise questions about the optimal stage and environment that favor graft survival, appropriate migration, and integration within host dopaminergic systems, as well as which factors are required to sustain a replaced dopaminergic phenotype in a dopamine-depleted host.
Several possibilities have been suggested to explain the failure of continued TH expression or the survival of dopaminergic neurons that were present in vitro prior to implantation in a mature host brain, including the absence of critical factors in the adult or aged brain that are necessary for maintenance of the dopaminergic phenotype. The absence of immunoreactivity to DAT and VMAT would suggest that dopamine neurons were not merely downregulated by endogenous dopamine. A greater immunogenicity has been suggested for terminally differentiated dopamine neurons than that of cells earlier in the dopaminergic lineage. With a more robust immunosuppression regimen, our cells had better survival than shown in a primate study by Emborg et al. (8). In that 3-month study, no surviving neuroectoderm-derived cells were found in two of three monkeys, and no TH+ or NeuN+ cells were found in the third. In addition, all of the injected sites showed evidence of rejection.
Another potential mechanism for loss of grafted dopaminergic neurons might be apoptosis of the cells that fail to achieve appropriate synapses with the correct targets for dopamine neurons. During development, dopamine neurons are produced considerably in excess of those required for normal circuitry. These excess neurons undergo apoptosis, leaving the proper number of cells, which are synaptically connected. In addition, it is known from previous studies that a significant proportion of transplanted fetal dopaminergic neurons undergo apoptosis within a few days of placement in the adult host brain (33), possibly as a result of their placement in an unfavorable environment. However, in order for a similar early apoptotic event to account for the loss of TH expression in grafted cells in the current study, the dopaminergic cells would have to be selectively vulnerable, as we observed a preferential loss of TH immunoreactivity.
While these experiments were under way, a series of reports utilizing dual SMAD [sma (small body size) and mothers against decapentaplegic] inhibition (4) in combination with early stage, high-dose SHH signaling, and acidic FGF8 were shown to repress Dickkopf WNT signaling pathway inhibitor 1 (Dkk-1)-mediated specification of the anterior neuroectoderm in favor of the FoxA2-ir floorplate fate (6,9). In addition, early stage activation of WNT signaling through recombinant proteins (6,9) was demonstrated to enhance the generation of midbrain-specific FoxA2/TH-ir double-labeled cells. More recently, this powerful floorplate induction strategy was further improved with the addition of CHIR99021, a small molecule activator of Wnt signaling. This potent glycogen synthase kinase 3 β (GSK3B) inhibitor replaced the use of recombinant Wnt proteins and was shown to induce the expression of another important midbrain transcription factor, LIM homeobox transcription factor 1 α (LMX1A), and enhance the generation of true functionally active midbrain fate dopaminergic neurons (14). Furthermore, floorplate-derived dopamine neurons were shown to survive and maintain the TH-ir/FoxA2-ir midbrain identity up to 1 month posttransplantation into the striatum of two MPTP-lesioned aged rhesus monkeys (14).
The results in these combined studies suggest that the TH-ir neurons generated in vitro in the current study were not true midbrain fate-specified dopamine neurons. Despite strong expression of Nurr-1 and TH in vitro, it is likely that the critical window for derivation of the floorplate lineage was missed and that these cells were already fate specified to anterior neuroectoderm. Several key factors would likely have improved success including utilizing high-strength SHH (C25II) and acidic FGF8 much earlier during neuralization, as well as the potent effect of small molecules for dual SMAD inhibition instead of recombinant noggin. Another key missing factor was the early addition of Wnt activation, through the small molecule CHIR99021. While we modified the accepted protocol with the addition of recombinant Wnt proteins, data from several studies now suggests that early timing of CHIR99021 exposure in a dose-dependent manner is critical to bypass the anterior neuroectoderm lineage and adopt the FoxA2-ir midbrain floorplate fate. Utilizing a similar floorplate-based strategy, Xi et al. recently reported expression of engrail-1 (EN1), another important master transcriptional regulator of midbrain dopaminergic neurogenesis, in FoxA2/TH/LMX1A-ir cells, indicating further refinement toward midbrain-specific dopamine neurons (37). Furthermore, Denham and colleagues reported that specification of the floorplate fate is temporally dependent, occurring prior to the onset of Pax6 expression, at the pre-neuroepithelial stage (7). These data argue that the onset of Pax6 expression in HESC-derived cells in the current study would have already excluded the development of the floorplate and instead adopted the neuroepithelial lineage.
Conclusion and Summary
Results from the present study suggest that methodologies favoring derivation of neuroectoderm to generate dopaminergic neuronal cells from HESCs are not capable of maintenance of the midbrain dopaminergic fate in vivo in nonhuman primates. These strategies are fully capable of generating β-III-tubulin-ir, Nurr-1-ir, TH-ir, dopamine-producing cells, but these neurons lack critical synergistic transcriptional signaling cues to appropriately maintain the midbrain fate in vivo. This conclusion was obscured in the Emborg et al. (8) study by almost total loss of injected cells and evidence of immune rejection, compared with the present study with more extensive immunosuppression and robust survival of neuronal cells. In contrast to these studies, there is now considerable evidence to suggest that floorplate-based methodologies are capable of generating authentic FoxA2-ir midbrain dopamine neurons and offer considerable promise for transplantation in Parkinson's disease (14,37). Given the extremely robust survival and fibritic output of HESC-derived neuronal grafts in both the striatum and SN of vervet monkeys in the current study, as well as results from two MPTP-lesioned rhesus monkeys (14), it is likely that pluripotent cells directed with the floorplate technique will retain their midbrain fate in vivo, long term, offering a suitable alternative source for transplantation in Parkinson's disease. Longer term studies will be necessary to determine whether other mechanisms may also limit long-term functional replacements and whether these show significant reversal of parkinsonism, tumor formation, or dyskinesia (17,32,35).
Footnotes
Acknowledgments
This material is based upon work supported by the State of Connecticut under the Connecticut Stem Cell Research Grants Program (08-SCC-YSME-005 and 12-SCIDS-Yale-01). Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the State of Connecticut, the Department of Public Health of the State of Connecticut, or Connecticut Innovations, Incorporated. We thank the California Institute of Regenerative Medicine for collaborative support, especially Evan Y. Snyder, Sanford Burnham Medical Research Institute, Axion Research Foundation, and St. Kitts Biomedical Research Foundation, where the in vivo studies were carried out. Additional support for D.R.W. came from the National Institutes of Health 1-R21-NS080380-01. We thank Barbara Blanchard for assistance with histological data and the staff at St. Kitts Biomedical Research for their research activities with the monkeys. We thank Dr. Jun Liao of the Yale Stem Cell Center, (New Haven, CT, USA) for labeling cells with a GFP lentivirus. Human embryonic stem cell line (WA01-H1) was obtained under license from WiCell through the Yale Stem Cell Center. Author contributions: Dustin Wakeman: Collection of data (Figs. 4, 5, 6, 7), data analysis and interpretation, manuscript writing, final approval, financial support. Stephanie Weiss: Collection of data (Figs. 1, 2, 3A, B), data analysis and interpretation, manuscript writing, final approval of manuscript. John R. Sladek, Jr.: Assembly of data (Figs. 4, 5, 6, 7), data analysis and interpretation, manuscript writing, final approval of manuscript. John D. Elsworth: Conception and design, collection of data (Fig. 3B), data analysis and interpretation, manuscript writing, final approval of manuscript. Brian Bauereis: Assembly of data (Fig. 3A), final approval of manuscript. Csaba Leranth: Collection of data (Fig. 8), data analysis and interpretation, final approval of manuscript. Patrick J. Hurley: Collection of data (Fig. 3A), final approval of manuscript. Robert H. Roth: Conception and design (Fig. 3B), administrative support, final approval of manuscript. D. Eugene Redmond, Jr.: Conception and design, financial support, administrative support, collection of data, data analysis and interpretation, manuscript writing, final approval of manuscript. The authors declare no conflicts of interest.
