Abstract
Oxidative stress is a major cause of islet damage and loss during the islet isolation process. The Nrf2 pathway plays a critical role in protecting the cells against oxidative stress. The aim of this study was to investigate the effect of an Nrf2 activator (dh404) on islet isolation and transplantation in a rodent model. Islet isolation was conducted using Nrf2-deficient and wild-type mice and vehicle-treated and Nrf2 activator (dh404)-treated rats. Islet yield, viability, and Nrf2 pathway activity were determined. An in vivo islet potency test was done. Islet yield and viability in Nrf2-deficient mice was significantly lower compared to wild-type (p < 0.05) mice. Furthermore, administration of dh404 to normal Sprague–Dawley rats enhanced nuclear translocation of Nrf2 and elevated HO-1 expression in the pancreas. Islet yield and viability in dh404-treated rats was significantly higher compared to the vehicle-treated group (p < 0.05). The diabetes cure rate in nude mice with chemically induced diabetes was significantly greater in those transplanted with islets from the dh404-treated group (6/9) than vehicle-treated rats (2/9, p < 0.05). The Nrf2 pathway plays a significant role in protecting islets against stress caused by the isolation process. Pharmacological activation of the Nrf2 pathway significantly increased HO-1 expression, improved islet yield, viability, and function after transplantation.
Keywords
Introduction
Diabetes mellitus has become a critical disorder with major costs and complications worldwide. Successful islet transplantation is a promising therapy for select type 1 diabetes (T1DM) patients, due to substantial improvements of islet processing procedures and immunosuppressive medications (22,44,46). However, a major obstacle in this therapeutic approach is the insufficient yield of islets from deceased donor pancreata. An average islet isolation generally yields approximately 50% of the estimated over 1 million islets present in an adult human pancreas (26). Therefore, more than one donor pancreas is often needed for insulin independence after clinical islet transplantation (46,48).
It has been reported that oxidative stress is increased in patients with type 1 and type 2 diabetes. Studies have shown that biochemical markers of oxidative stress are higher in tissue samples and in the pancreas of patients with diabetes (45). When reactive oxygen species (ROS) are generated in excess for prolonged periods of time, they cause chronic oxidative stress. Antioxidant enzymes contained within host tissue are the first line of defense against excessive levels of ROS. A vital consideration is the level of antioxidant host defense within the islet. It has been reported that the islet is among the least well-endowed tissue in terms of intrinsic antioxidant enzyme expression and activity (16,52). In addition, overexpression of antioxidant enzymes in insulinoma cells results in significantly increased resistance to cytokine-mediated toxicity (3). Transgenic mice that exhibit β-cell-targeted overexpression of an antioxidant enzyme were also shown to be resistant to autoimmune and streptozotocin-induced diabetes (19).
Pancreatic islets contain a very low level of antioxidant enzymes, such as catalase, superoxide dismutase, and glutathione peroxidase (30). Pancreatic islet isolation includes mechanical disruption, the chemical effects of collagenase, warm and cold ischemic injury, all of which increase ROS generation. These factors degrade islets and capillary membranes, which causes an additional release of free radicals leading to apoptosis. Oxidative stress has been shown to play a major role in cell injury and β-cell dysfunction and death during islet isolation and transplantation (18,36,55).
Nuclear erythroid 2-related factor 2 (Nrf2) is a basic region leucine-zipper transcription factor, which binds to the antioxidant response element and regulates the expression of genes involved in the cellular antioxidant and anti-inflammatory defense. Nrf2 influences sensitivity to physiologic and pathologic processes affected by oxidative and electrophilic stress. The Nrf2-Kelch ECH associating protein 1 (Keap1) signaling pathway plays a significant role in protecting cells from various stresses including environmental agents/drug, inflammatory stresses, and chronic exposures to cigarette smoke and other carcinogens (8,12,13,27,28,31,32,38,51)
The aim of the present study was to investigate the effect of Nrf2 pathway activation on islets using a rodent islet isolation and transplant model. We hypothesize that activation of the Nrf2 pathway will contribute to the protection of islet cells against oxidative stress during islet isolation and following transplantation.
Materials and Methods
Bardoxolone Methyl Analog (dh404)
RTA dh404 was provided by Reata Pharmaceuticals, Inc. (Irving, TX, USA). The chemical name for RTA dh404 is CDDO-9,11-dihydro-trifluoroethyl amide (CDDO-dhTFEA) (2,11,33,49,54,57).
Animals and Experiment Design
All animals were purchased from Charles River (Wilmington, MA, USA) or were received from collaborators and housed at our animal facility in accordance with the Institutional Animal Care and Use Committee of the University of California, Irvine (Irvine, CA, USA). Animals were maintained under 12-h light/dark cycles and allowed water and food ad libitum. Nrf2-deficient mouse (KO) on C58BL6; 129SV mix background have been described previously (5). To confirm the genotype from each animal, DNA was extracted from the tail and analyzed by PCR using the following primers: 3′-primer, 5′-GGAATGGAAAA-TAGCTCCTGCC-3′: 5′-primer, 5′-G CCTGAGAGCTGTAGGCCC-3′: lacZ primer, 5′-GGGTTTTCCCAGTCACGAC-3′. Nrf2-/- and Nrf2++/+ mice exhibited one band at 200 and 300 bp, respectively. Nine to 12-week-old mice (five mice in each group) were used in this study.
In a separate study, male Sprague–Dawley rats (250-300 g) were supplemented with oral Nrf2 activator, dh404. Rats were randomly divided into vehicle- and dh404-treated groups (10 rats in each group). Dh404was dissolved in sesame oil (Sigma-Aldrich, St. Louis, MO, USA) and administered once a day by gavage at a dose of 0.6 mg/kg body weight for 3 days prior to islet isolation. The control animals were fed vehicle (sesame oil) instead.
Streptozotocin (STZ)-Induced Diabetic Rat Model
Production of oxygen-free radicals and lipid peroxidation has been considered the main mechanisms by which STZ causes β-cell damage. To determine the protective effect on islets of dh404 against oxidative stress and optimize the dose of dh404, an STZ-induced diabetes rat model was used. Dh404 was orally given to rats via gavage at a dose range from 0.1 to 2 mg/kg (five rats in each group). Briefly, the male Sprague–Dawley rats were administrated with dh404 at a dose range of 0.1-2 mg/kg body weight for 3 days prior to STZ treatment. STZ (Sigma-Aldrich) at a dose of 50 mg/kg body weight was dissolved in a freshly prepared citrate buffer (0.05 mol/L; pH 4.5; Sigma-Aldrich) and given to rats via an intraperitoneal injection.
Islet Isolation
Under isoflurane (Phoenix Pharmaceuticals, Inc., Burlingame, CA, USA) anesthesia, midline abdominal incisions were performed, and the pancreases were exposed. The bile duct at the level of the duodenum was located and clamped. Animals were then sacrificed under deep sedation by cutting the abdominal aorta. A 23-gauge needle (rats) or 30 gauge (mice) attached to a 10-cc syringe was used to puncture and intubate the common bile duct. The pancreas was slowly distended by intraductal injection of precooled Hank's balanced salt solution (HBSS; Mediatech, Herndon, VA, USA) containing 1.0 mg/ml collagenase type V (Sigma-Aldrich) at a dose of 1.5-2 ml for mice and 7-8 ml for rats. After distension, the whole pancreas was harvested and incubated in a 50-ml conical tube at 37°C for 18 min. Subsequently, the digestion was stopped by the addition of 20 ml of cold HBSS, and the conical tube was shaken vigorously. The suspension filtrated through a metallic filter net was washed with HBSS three times and centrifuged (150 x g, 2 min, 4°C). The islets were purified using a Ficoll density gradient (Mediatech, Inc.). Islets were then collected and washed in HBSS. The crude number of islets in each diameter class was determined by counting after diphenylthionarbazon (DTZ; MP Biomedicals, Santa Ana, CA, USA) staining using an optical graticule. This number was then converted to the standard number of islet equivalents (IEQs; diameter standardizing to 150 μm) (15). The purity of islet was also calculated during islet counting using DTZ staining. To minimize the bias, the islet number and purity were counted by double-blind evaluations.
Determination of Mouse Islet Cell Viability
Fluorescein diacetate (FDA) and propidium iodide (PI) staining were performed after mouse islet isolation to determine islet cell viability. Briefly, 50 islets were incubated with 20 μl PI (1:10; Invitrogen Corp., Grand Island, NY, USA) for 10 min kept from light at room temperature. After washing with phosphate-buffered saline (PBS; Fisher Scientific, Pittsburgh, PA, USA) three times, 20 μl FDA (1:10; Sigma-Aldrich) was added and counted under fluorescence microscope (Nikon Eclipse TS100; Nikon Instruments Inc., NY, USA). Dead cells were stained red, and viable cells were stained green. The percentage of viable cells in each islet was assessed, and the average was taken from 50 islets per mouse (4).
Glucose-Stimulated Insulin Secretion (GSIS) Test
To determine the in vitro potency of isolated rat islets, the insulin secretory response to glucose was measured using a modified method previously described (47). Briefly, 20-30 islets were randomly transferred to a cell culture insert (Millipore Ltd., Billerica, MA, USA) with Krebs buffer (115 mM NaCl, 5.0 mM KCl, 2.3 mM CaCl2, 1.0 mM MgCl, 1.2 mM KH2PO4, 25 mM NaHCO3, pH 7.4), 25 mM HEPES, 0.1% BSA fraction V (Sigma-Aldrich) containing 2.8 mM glucose, and incubated at 37°C for 1 h as preincubation. Thereafter, they were suspended three times for 60 min at 37°C in Krebs buffer with the addition of various glucose concentrations (basal I: 2.8 mM, stimulation: 28 mM, basal II: 2.8 mM, respectively). The supernatants were collected and stored at −20°C. The insulin level was measured using a Rat Insulin Enzyme-Linked Immunosorbent Assay kit (Mercodia Inc., Winston Salem, NC, USA). The stimulation index (SI) was calculated as the insulin released into the stimulation medium divided by insulin released into the basal I medium.
In Vivo Islet Potency Test
Islet transplantation into diabetic nude mice was performed as an in vivo assessment of islet potency (n=9). Male nude mice (8 weeks old) were used as recipients of islet transplantation. Diabetic nude mice were made by intraperitoneal injection of STZ (Sigma-Aldrich) at 200 mg/kg body weight. The mice were considered diabetic if their nonfasting blood glucose levels exceeded 350 mg/dl for 3 consecutive days. Diabetic nude mice were anesthetized by isoflurane, and the left kidney was exposed through a lumbar incision. A breach was made in the kidney capsule, and a polyethylene catheter was introduced through the breach and advanced beneath the kidney capsule to generate a subcapsular space. To minimize the influence of purity on islet transplantation, purified islets were handpicked under the microscope to remove remaining acinar tissue to obtain islets with 95% purity for transplant. After handpicking, the islet samples were retaken and recounted, 200 IEQs were slowly injected through the PE50 tube (Fisher Scientific) into the subcapsular space. After removing the PE50 tube, the opening was cauterized, and the kidney was repositioned, followed by suturing of muscle and skin. Daily blood glucose levels were obtained via tail vein prick using a contour glucometer (Bayer Healthcare LLC, Whippany, NJ, USA). At 4 weeks after transplantation, the nephrectomy with islet grafts was performed to confirm the graft function. Success of transplantation was defined as maintenance of normoglycemia (<200 mg/dl) for at least 3 consecutive days and return of hyperglycemia (>250 mg/dl) after nephrectomy.
Determination of Rat Islet Cell Viability
Cell viability was determined with previously reported methods (20,21). Briefly, 500-800 rat islets were incubated in 1 ml accutase solution (Sigma-Aldrich) at 37°C for 10 min. Accutase was deactivated with cold fetal bovine serum (Sigma-Aldrich). Washed cells were transferred into filtered FACS tubes (BD Falcon, Franklin Lakes, NJ, USA) to remove undigested tissue. Cells were resuspended in PBS and stained with tetramethylrhodamine ethyl ester perchlorate (TMRE; Life Technologies, Grand Island, NY, USA) for 30 min at 37°C. This was followed by 7-aminoactinomycin D (7-AAD; Life Technologies) stain on ice immediately prior to the FACS analysis. TMRE stain indicates mitochondrial membrane potential, and 7-AAD is DNA-binding dye. The percentage of 7-AAD+ cells was recorded for dead cells, and further analysis for TMRE+ (viable cells) and TMRE- (apoptotic cells) was performed after their exclusion (gating out). Fifty thousand cells from each sample were assessed by flow cytometry (Accuri C6 cytometers; BD Biosciences, Ann Arbor, MI, USA). Software (Accuri C6 software; BD Biosciences, San Jose, CA, USA) was used for analysis (20).
Western Blot Analysis
Total protein from rat pancreases (both vehicle- and dh404-treated groups) were obtained with CelLytic™ NuCLEAR™ Extraction Kit (Sigma-Aldrich) using the manufacturer's instructions. In brief, pancreatic tissue was homogenized and lysed on ice with lysis buffer including DTT and protease inhibitors for 20 min. The pancreatic lysate was centrifuged at 11,000 x g for 11 min. The cytoplasmic protein concentration was measured using Bio-Rad DC Protein Assay (Bio-Rad Laboratories, Hercules, CA, USA). The primary antibodies used were rabbit antibodies against Heme oxygenase-1 (HO-1; 1:1,000 dilution; Abcam Inc., Cambridge, MA, USA) and GAPDH (1:5,000 dilution; Sigma-Aldrich). Aliquots containing 15 μg proteins were loaded on NuPAGE 4-12% Bis-Tris gel (Life Technologies) and then were transferred to a PVDF membrane (Pall Life Science, Ann Arbor, MI, USA). The membrane was blocked in TBS-T (Thermo Scientific, West Palm Beach, FL, USA) containing 5% blocking-grade nonfat dry milk (Bio-Rad Laboratories), and incubated with primary antibodies overnight at 4°C. After washing, the membrane was further incubated with HRP-conjugated goat anti-rabbit secondary antibody (1:3,000 dilution; Cell Signaling, Danvers, MA, USA) at room temperature for 2 h. Immunoreactive bands were visualized using an enhanced chemiluminescence detection system (Thermo Scientific, Rockford, IL, USA). Densitometric measurements were done with Image Quant (GE Healthcare Life Sciences, Pittsburgh, PA, USA). GAPDH bands, as a housekeeping protein, were used as an internal control.
Immunofluorescence and Confocal Microscopy
Rat islets were fixed in 10% formalin (Fisher Scientific) and embedded in optical cutting temperature compound (Sakura Finetek Inc., Torrance, CA, USA) and frozen; then 10-μm-thick sections were cut, rinsed in PBS and 5% BSA, and incubated with Universal Blocker Reagent (Biogenex, San Ramon, CA, USA) for 30 min in a humidified chamber at room temperature. Thereafter, sections were incubated with primary antibodies, rabbit anti-Nrf2 (1:100 dilution; Abcam Inc.) overnight at 4°C in a humidified chamber. After PBS wash, Alexa Fluor 488 antibody (1:100 dilution; Molecular Probes, Carlsbad, CA, USA) and 4′,6-diamidino-2-phenylindole (DAPI; Invitrogen Corp.) were applied to the slides and incubated at room temperature for 2 h in the dark. The slides were analyzed under a confocal laser scanning microscopy (Zeiss LSM700; Carl Zeiss, Jena, Germany), and the images were analyzed by ZEN 2011 (Carl Zeiss, Jena, Germany).
To analyze the percentage of β-cells in islet, the fresh isolated rat islets were dissociated into single cells, and they were fixed on glass slides with 2.5% paraformaldehyde (Sigma-Aldrich). Cells were incubated with Universal Blocker Reagent (Biogenex) for 30 min, to reduce nonspecific binding. Thereafter, cells were incubated overnight at 4°C with mouse monoclonal antibody to insulin (1:200 dilution; Abcam). After washing, cells were incubated with goat anti-mouse IgG (Alexa Fluor 488 goat anti-mouse IgG, 1:200 dilution; Life Technologies). Omission of the primary antibody served as negative control. DAPI was applied to stain cell nuclei. The cellular composition was determined by manually counting the stained cells using a fluorescent microscope. β-cell content was determined by taking the number of positively stained β-cells and dividing by the total number of stained cells (20,21)
Statistical Analysis
Results were expressed as mean ± standard deviation (SD), and the statistical differences between groups were determined by an unpaired t-test. The percentage of cured diabetic nude mice between two groups was determined by Gehan–Breslow–Wilcoxon test. Values of p < 0.05 were considered significant.
Results
Islet Yield and Viability of Nrf2-Deficient Mice
To investigate the effect of Nrf2 deficiency on the pancreas, islet isolation was performed in Nrf2-deficient mice. Interestingly, the islet yield in the Nrf2-deficient group was significantly lower when compared to the wild-type group (15.6 ± 4.9 IEQs/g body weight vs. 24.2 ± 1.91 IEQs/g body weight), although islet number was comparable (Fig. 1A, B). The islet viability assessed by FDA/PI in Nrf2-deficient islets (84.5 ± 0.6 %) was also significantly lower when compared to wild-type islets (93.0 ± 3.8%) (Fig. 1C).

Islet yield and viability of Nrf2-deficient mice. (A) Islet yields. The islet yield in the wild-type group was significantly higher than that in Nrf2 KO group (*p < 0.05). (B) Islet number. There was no significant difference between the groups. (C) Islet viability. The percentage of viable cells in the wild-type group was significantly higher than that in Nrf2 KO group (*p < 0.05). Data are expressed as mean ± SD.
Adjustment of dh404 Dose Using STZ-Induced Diabetic Rat Model
An STZ-induced diabetic rat model was used to determine an optimal dose of dh404 in vivo for islet protection against oxidative stress during islet isolation. Dh404 was orally given to rats via gavage at a dose range from 0.1 to 2 mg/kg. Figure 2 shows blood glucose level after STZ injection in the rats treated at 0.1, 0.6, and 2 mg/kg body weight. The results revealed that the dose of dh404 between 0.5 and 1.5 mg /kg was beneficial for the β-cell protection from STZ. However, higher doses (2 mg/kg) of dh404 did not show protective effects in our STZ-induced diabetic rat model.

Adjustment of dh404 dose using streptozotocin (STZ)-induced diabetes model. STZ-induced diabetes model was used to determine an optimal dose of dh404. The 0.5-1.5 mg/kg dose demonstrated beneficial effects on STZ-induced diabetes. Higher doses (more than 2 mg/kg) of dh404 did not show protective effects in this model.
Pharmacological Activation of Nrf2 Pathway by dh404 in Rodent Pancreases
Nrf2 translocation to the nucleus was evaluated to confirm the upregulation of Nrf2 after dh404 oral administration. The pancreas of dh404-supplemented and vehicle-treated rats were stained with Nrf2 antibody and DAPI and then assessed by confocal microscope. Compared to the control group, Nrf2 translocation to the nucleus of pancreatic tissue in the dh404-treated rats was clearly demonstrated (Fig. 3A). In addition, the expression of HO-1, one of the target antioxidant enzymes of the Nrf2 pathway, was examined in the pancreases of the rats by Western blot. Dh404 treatment produced a 10-fold increase in HO-1 expression compared to the vehicle-treated controls (Fig. 3B).

(A) Nrf2 translocation in pancreas. The pancreases of dh404-supplemented rats were stained with Nrf2 antibody and DAPI and then assessed by confocal microscope. Nrf2 translocation (merged blue and green) to the nucleus in the dh404-treated rats was clearly demonstrated (blue = nuclei, green = Nrf2, arrows showing Nrf2 translocation). (B). HO-1 expression in dh404-supplemented rat pancreas. Dh404-treated pancreas produces a further by 10-fold increase in HO-1 expression.
Nrf2 Pathway Activation in dh404-Treated Islets
To investigate the effect of pharmacological activation of the Nrf2 pathway on islet isolation, dh404 (0.6 mg/kg once/day) was administrated orally to rats for 3 days prior to isolation. The isolated islets were stained with Nrf2 antibody and DAPI and then assessed by confocal microscope. Nrf2 translocation to the nucleus of islets in the dh404-treated group was clearly shown (Fig. 4).

Nrf2 translocation in isolated islets. The isolated islets from dh404-supplemented rats were stained with Nrf2 antibody and DAPI and then assessed by confocal microscope. Nrf2 translocation (merged blue and green) to the nucleus of islets in the dh404-treated group was clearly demonstrated (blue = nuclei, green = Nrf2, arrows showing Nrf2 translocation).
Administration of dh404 Improves Islet Yield and Purity
The islet yield and islet number in the dh404-treated group (1522.6 ± 434.8 IEQs) was significantly higher than those in the vehicle-treated group (1192.4 ± 233.4 IEQs, p < 0.05) (Fig. 5 A, B). Moreover, the purity of islets in the dh404-treated group (91.5 ± 3.72%) was significantly higher compared to the vehicle-treated animals (85 ± 3.78%, p < 0.01) (Fig. 5C). Stimulation index in the dh404-treated group (1.77 ± 0.34) was comparable to the vehicle-treated group (1.6 ± 0.27) (Fig. 5D).

Islet isolation in dh404-treated rat. (A) Islet yield. The islet yield in the vehicle-treated group was significantly lower compared to the dh404-supplemented group (*p < 0.05). (B) Islet number. The islet number in the vehicle-treated group was significantly lower compared to the dh404-supplemented group (*p < 0.05). (C) Islet purity. Islet purity in the dh404-supplemented group was significantly higher. (D) Glucose-stimulated insulin release. There was no significant difference between groups. Data are expressed as mean ± SD.
Islet Viability and Cellular Composition
Single islet cell suspensions were incubated with 7-AAD and TMRE after dissociating freshly isolated islets. Apoptosis and necrosis in islet cells were analyzed by flow cytometer. The percentage of apoptotic cells (TMRE-) in the dh404-treated group (18.3 ± 2.1%) was significantly lower than in the vehicle-treated group (23.4 ± 2.1%, p < 0.05) (Fig. 6A). However, there was no significant difference in necrotic cells between the experimental groups (7.44 ± 3.24% vs. 5.95 ± 2.19%). Moreover, the percentage of β-cells in islets was analyzed to determine the influence of Nrf2 pathway activation on islet survival. As shown in Figure 6B, the percentage of β-cells in the dh404-treated group was significantly higher when compared with the vehicle-treated groups (70.2 ± 4.7% vs. 65.9 ± 4.0%, p < 0.05).

(A) Rat islet viability. Islets in dh404-supplemented rats showed less apoptotic cells compared to the vehicle-treated group (*p < 0.05). There was no difference in dead cells between these two groups. (B) Cellular composition in islet. Islets in dh404-supplemented rats showed a significantly higher percentage of β-cells in islet compared to the vehicle-treated group (*p < 0.05). Data are expressed as mean ± SD.
In Vivo Islet Potency Test
Marginal mass of rat islets (200 IEQs/mouse) in each group were transplanted into the kidney capsule of nude mice with STZ-induced diabetes to determine the effect of dh404 on islet function in vivo (Fig. 7). The reversal rate of diabetes in the dh404-treated group was significantly higher than the vehicle-treated group (6/9 vs. 2/9, p = 0.034). The nephrectomy of the islet-grafted kidney was performed at 4 weeks after transplantation to confirm the graft function.
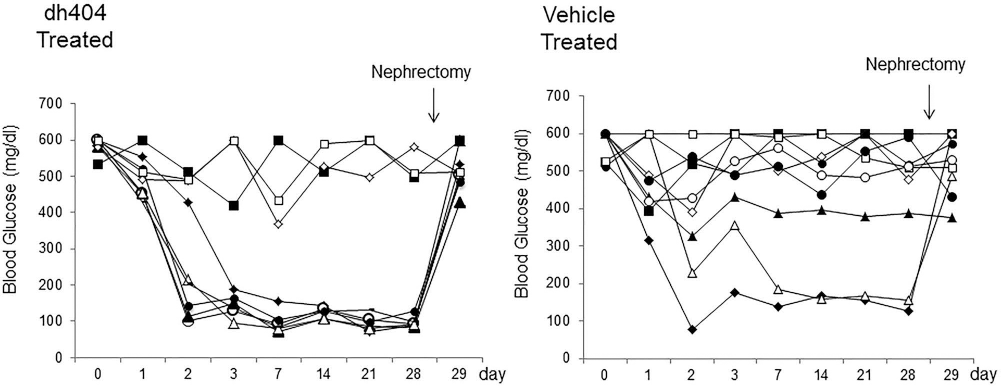
In vivo islet potency tests. Islets (200 IEQs/mouse) in each group were transplanted into kidney capsule of STZ-induced diabetic nude mice to determine the effect of dh404 on islet function in vivo. The reversal rate of diabetes in the dh404-treated group was significantly higher than the vehicle-treated group (6/9 vs. 2/9, p = 0.034). The nephrectomy of the islet-grafted kidney was performed at 4 weeks after transplantation to confirm the graft function.
Discussion
Obtaining a consistently high islet yield and viability is a prerequisite to succeed with islet transplantation. In clinical settings, only 20-50% of potential islet mass in a human pancreas are generally recovered even with the best islet isolation procedure and by well-experienced operators (44). Oxidative stress is a major factor potentially causing islet damage and loss during the islet isolation process (42,43). ROS have a great potential to directly or indirectly disturb the cellular integrity and physiological function of biomolecules like lipids, proteins, and DNA and have been implicated in various diseases, including atherosclerosis, pulmonary fibrosis, cancer, and adult respiratory distress syndrome (1,17). Pancreatic islets have low activity of free radical detoxifying enzymes when compared to other tissue (16,30,37). Several studies have shown that administration of antioxidants like curcumin, quercetin, and probucol could protect islet cells from damage by free radicals (9,14,35). In addition, as one of the strategies against oxidative stress, overexpression of genes responsible for antioxidant enzymes in islets have exhibited better protection against damage caused by free radicals (1). Nrf2 induction prevents reactive species damage in pancreatic β-cells, and the Keap1 Nrf2 system is the crucial defense pathway for the physiological and pathological protection of pancreatic β-cells (56). In the current study, we demonstrated the protective effect of Nrf2 activator dh404 on islet cells during the islet isolation procedure. Islet yield and viability were found to be significantly reduced in Nrf2-deficient pancreas. Furthermore, pharmacological Nrf2 activation by dh404 resulted in significantly better islet yields, viability, and β-cell survival, leading to higher reversal rate of diabetes in islet transplantation into diabetic immunodeficient mice. Nrf2 is a transcription factor important in the protection against oxidative stress through the antioxidant response element (ARE)-mediated transcriptional activation of several phase II detoxifying and antioxidant enzymes. It is generally thought that dh404 allows Nrf2 to translocate to the nucleus where it upregulates expression of antioxidant and cytoprotective genes (11,49,54). Nrf2 has a significant protective role against oxidative injury, through transcriptional activation of genes encoding antioxidant enzymes and related proteins (6,7). Our results clearly indicated that dh404 treatment promotes translocation of Nrf2 from the cytoplasm into the nucleus in isolated islet cells (see Fig. 4A). Moreover, it upregulated HO-1 expression in pancreatic tissue. HO-1 has been implicated as a promising therapeutic target in various diseases (10,50). A study in HO-1-deficient mice has shown HO-1 to play a critical role in the anti-inflammatory process (23). Furthermore, it has been reported that HO-1 upregulation led to protective effects on β-cells against various apoptotic stimuli including cytokines and Fas (41,53).
We performed GSIS with isolated islets to evaluate the effect of dh404 on islet function. The dh404-treated group showed no significant improvement. For clinical application, in vivo function is a most important evaluation after islet transplantation. Transplanted islets can be damaged by early inflammation at the site of implantation, which leads to ROS inducing either functional impairment or cell death (25,40). Our study demonstrated that HO-1 upregulation could improve graft function in vivo as well as another study (39). Further, the HO-1 system serves as a novel therapeutic concept in organ transplantation as the multifaceted targets of HO-1-mediated cytoprotection may simultaneously benefit both local graft function and host systemic immune response (24).
Compared to the liver, islets contain only 1% catalase, 2% GPX1, and 29% SOD1 activities. Thus, β-cells are considered to be low in antioxidant defense and are therefore susceptible to oxidative stress (29). In addition, Yogishita et al. recently reported that β-cell-specific Nrf2 conditional knockout mice strongly aggravated β-cell damage and that Nrf2 induction prevented reactive species-induced damage in β-cells (56). Therefore, the pharmacological activation of the Nrf2 pathway by dh404 significantly increased not only islet yield but also β-cell content in isolated islets, since it may be more effective in β-cells when compared to other endocrine cells.
β-cells are the primary target of diabetogenic agents, such as STZ (34). To determine if dh404 treatment would have an effect on β-cell mass after isolation, we analyzed β-cell content in isolated islets. A remarkable finding in our current study was that β-cell content was significantly increased in the dh404-supplemented group after isolation. In a human islet isolation study, authors showed β-cell mass to be directly related to the in vivo transplantation success rate (20). Our data showed that dh404 treatment had not only improved islet yield but also increased β-cell content in isolated islets after the isolation procedure.
A low dose of dh404 was administrated into rats for islet isolation in our current study. A recent study showed that dh404, unless administered acutely at low doses, is not well tolerated in rodents (58). The adjustment of the dose of dh404 in various targets may be crucial for clinical application of dh404. dh404 is not water soluble, but it can be dissolved in DMSO, which allows us to add dh404 into various media used for pancreas preservation, islet isolation, and culture. The effect of in vitro treatment of dh404 on islet isolation using pancreases from large mammals such as human, monkey, and pig will be investigated as the next step toward its potential utility in a clinical setting.
In summary, our data demonstrated that pretreatment with low-dose dh404 improved islet yield, viability, and β-cell content by promoting Nrf2 nuclear translocation and upregulating HO-1 in islet cells during the islet isolation procedure. TargetingNrf2 activation may prove to be a therapeutic strategy for improved islet isolation and transplantation outcomes.
Footnotes
Acknowledgments
This study was in part supported by grants from NIH-NCRR UL1 TR000153, KL2 TR000147; the Juvenile Diabetes Research Foundation International 17-2011-609. The authors declare no conflicts of interest.
