Abstract
The ability of intraspinal bone marrow stromal cell (BMSC) transplants to elicit repair is thought to result from paracrine effects by secreted trophic factors including brain-derived neurotrophic factor (BDNF). Here we used gene therapy to increase or silence BDNF production in BMSCs to investigate the role of BDNF in BMSC-mediated neuroprotection. In a spinal cord organotypic culture, BMSC-conditioned medium significantly enhanced spinal motoneuron survival by 64% compared with culture medium only. Only conditioned medium of BDNF-hypersecreting BMSCs sustained this neuroprotective effect. In a rat model of spinal cord contusion, a BDNF-dependent neuroprotective effect was confirmed; only with a subacute transplant of BDNF-hypersecreting BMSCs were significantly more spared motoneurons found at 4 weeks postinjury compared with vehicle controls. Spared nervous tissue volume was improved by 68% with both control BMSCs and BDNF-hypersecreting BMSCs. In addition, blood vessel density in the contusion with BDNF-hypersecreting BMSCs was 35% higher compared with BMSC controls and sixfold higher compared with vehicle controls. BDNF-silenced BMSCs did not survive the first week of transplantation, and no neuroprotective effect was found at 4 weeks after transplantation. Together, our data broaden our understanding of the role of BDNF in BMSC-mediated neuroprotection and successfully exploit BDNF dependency to enhance anatomical spinal cord repair.
Keywords
Introduction
Improved functional recovery after transplantation of bone marrow stromal cells (BMSCs) in the injured spinal cord (1,11,12,24,25,37,43,46,54) is, at least in part, due to their neuroprotective abilities leading to nervous tissue sparing (1,24,25,46). It is thought that BMSCs exert their repair-supporting effect through paracrine actions (3,7,9,11,22,35,39,44). At present, it is incompletely understood which BMSC-secreted molecules contribute to tissue sparing in the injured spinal cord.
Transplanted BMSCs secrete brain-derived neurotrophic factor (BDNF) (9,22,35), which may be involved in neurotrophic (13,23,29,33,50) and angiogenic (8,15,19,28,40,42,53) effects. BDNF has trophic effect on neural cells in vitro and in vivo (13,23,29,33,50). Treatment of injured spinal cord with BDNF promotes neuronal sparing (4,5,32,42). Overexpression of BDNF by BMSC transplants promotes neural protection and axon regeneration in models of spinal cord injury and stroke (30,31,34,49,52).
BDNF was shown to regulate endothelial cell survival (15) in part through the tyrosine kinase receptor trkB (15,28). BDNF and trkB are upregulated in vascular smooth muscle cells following injury (16) and ectopic BDNF overexpression results in increased capillary density (15). The effects of BDNF on vascular events may contribute to nervous tissue sparing (20,27). Together, these findings and its potential dual role in angiogenesis and neuroprotection warrant investigations of the role of BDNF in BMSC-mediated spinal cord repair.
We hypothesized that BDNF secreted by transplanted BMSCs elicits angiogenesis and neuronal protection and that BDNF hypersecretion enhances these effects. Gene therapy was used to silence and upregulate BDNF production in BMSCs prior to transplantation. We used a spinal cord organotypic culture system to assess the effect of conditioned medium from the modified BMSCs on motoneurons in vitro and an adult rat model system of spinal cord contusion (45-47) to assess the effect of modified BMSCs on motoneurons, nervous tissue volume, blood vessel density, and transplant survival.
Materials and Methods
BMSC Culture and Lentiviral Transduction
BMSCs were harvested from femurs from adult female Sprague–Dawley rats (200-250 g; Charles River Laboratories, Wilmington, MA, USA) and cultured as previously described (2). After one passage, BMSCs were transduced with lentiviral vectors (LV) encoding for BDNF and green fluorescent protein (GFP) (LV-BDNF/GFP) to upregulate BDNF production and express GFP (IRES linked); LV encoding for short hairpin (sh) BDNF and GFP (LV-BDNFshRNA/GFP) to silence BDNF production and express GFP (IRES linked); five different shBDNF/GFP constructs were used (Cat. No. RMM4532-NM-012513, Open Biosystems, Dharmacon, Pittsburgh, PA, USA) to produce five different LV-shBDNF/GFP, and all were used at the same time in the LV-BDNFshRNA/GFP group, or LV encoding for GFP (LV-GFP) for control cells expressing GFP. The LVs were produced by the Viral Vector Core Facility of the Miami Project to Cure Paralysis (University of Miami Miller School of Medicine, Miami, FL, USA) and used at a multiplicity of infection of 100, which resulted in transduction ratios of 8 × 101%. Transduced BMSCs were grown in Dulbecco's modified Eagle's medium (DMEM; Sigma-Aldrich, Milwaukee, WI, USA) with 10% fetal bovine serum (Mediatech, Manassas, VA, USA) and 1% penicillin/streptomycin (Invitrogen, Grand Island, NY, USA) and passaged every 3 days. Passage 4 (P4) BMSCs were used for transplantation.
In Vitro Analysis of BMSCs
Lentiviral transduction of BMSCs in culture was confirmed by verifying stable GFP expression for seven passages. For characterization of BMSCs, P4 BMSCs were cultured on PDL-coated eight-well chamber glass slides (2,000 BMSCs/well; BD Falcon; BD Biosciences, Bedford, MA, USA) at 37°C/5% CO2, fixed with 4% paraformaldehyde (Sigma-Aldrich), incubated with 5% normal goat serum (NGS; Vector, Burlingame, CA, USA) for 30 min and incubated for 2 h with antibodies against CD90 (1:100; Cat. No. MAB1406; Millipore, Temecula, CA, USA), CD105 (1:50; Cat. No. 05-1424; Millipore), CD45 (1:100; Cat. No. 05-1413; Millipore), TrkB (1:2,000; Cat. No. AB9872; Millipore), P75 (1:100; Cat. No. 07-476; Millipore), or no antibody for a negative control. After washing twice in 1x PBS (Sigma-Aldrich), cells were incubated with secondary antibody Alexa Fluor 594 (1:200; Invitrogen) for 2 h at room temperature and stained with 4′,6-diamidino-2-phenylindole (DAPI; 2 μl/ml; Sigma-Aldrich). Slides were covered with glass slips in fluorescent mounting medium (Dako, Carpinteria, CA, USA) and stored at 4°C.
The amount of secreted BDNF was quantified in medium from confluent P4 BMSC cultures (2 × 106 cells) collected after 24 h using enzyme-linked immunosorbent assay (ELISA; Promega, Madison, WI, USA) according to the manufacturer's guidelines. Unconditioned culture medium was used as a negative control. Three biological replicates were quantified per condition, and each biological replicate was the average of two technical replicates.
Motoneuron Survival in Spinal Cord Organotypic Cultures
The effect of culture medium conditioned by transduced BMSCs on motoneuron survival was determined using a spinal cord organotypic culture system prepared from neonatal (postnatal days 1-1) B6.Cg-Tg(Thy1-YFP)16Jrs/J male and female transgenic mice (Jackson Laboratories, Bar Harbor, ME, USA) in which spinal motoneurons express (Thy1-driven) yellow fluorescence protein (YFP). The spinal cord was dissected and transferred into dissection medium containing Hank's balanced salt solution (Gibco, Carlsbad, CA, USA) with 4.3 mM NaHCO3 (Sigma-Aldrich), 10 mM HEPES (J.T. Baker, Swedesboro, NJ, USA), 33 mM glucose (Sigma-Aldrich), 0.03% BSA (Sigma-Aldrich), 0.15% MgSO4-7H2O (Sigma-Aldrich), 100 U/ml penicillin (Invitrogen), and 100 U/ml streptomycin (Invitrogen). After removal of the dura, the spinal cord was transversely sectioned at 350 μm on a Mcllwain tissue chopper (Stoelting Co., Wood Dale, IL, USA). Five sections were kept in inserts with collagen-coated Transwell membranes (3.0 μm pore size; Corning, Lowell, MA, USA). Inserts were placed in six-well tissue culture plates (Corning) and incubated with BMSC-conditioned culture medium or spinal cord organotypic culture medium containing 50% MEM (Gibco), 25% Hank's balanced salt solution (Gibco), 25% heat-inactivated horse serum (Thermo Scientific, Rochester, NY, USA), 25 mM HEPES (J.T. Baker), 200 mM L-glutamine (Gibco), 3.5 g/L D-glucose (Sigma-Aldrich), 100 U/ml penicillin (Invitrogen), and 100 U/ml streptomycin (Invitrogen). Sections were kept for 5 days in vitro (DIV) in a humidified chamber with 5% CO2 at 37°C. Medium was replaced every second day. After 5 DIV, the sections were fixed with 4% paraformaldehyde (Sigma-Aldrich) in 0.1 M PBS (Sigma-Aldrich) for 2 h at room temperature, washed three times for 10 min with PBS, and stored at 4°C until imaging. Motoneuron survival in the spinal cord sections was determined under the fluorescent microscope by quantifying YFP-positive motoneurons at 3 and 5 DIV. The numbers of all sections per group were averaged.
Spinal Cord Contusion and BMSC Transplantation
A total of 40 adult female Sprague–Dawley rats (200-250 g; Charles River Laboratories, Wilmington, MA, USA) were anesthetized with an intraperitoneal injection of ketamine (60 mg/kg; Butlerschein, Dublin, OH, USA) and Dexdomitor (0.5 mg/kg; Pfizer, New York, NY, USA) (47). The backs were shaved, and Lacrilube ophthalmic ointment (Fischer, Hanover Park, IL, USA) was applied to the eyes to prevent drying during and after surgery. The skin and underlying muscles were incised, and a laminectomy of the ninth thoracic (T9) vertebra was performed. The underlying spinal cord was contused using the Infinite Horizon Impactor (Precision Systems and Instrumentation, LLC, Versailles, KY, USA) at an impact force of 200 kDyne. Subsequently, the muscles were sutured separately, and the skin was closed with metal Michel wound clips (Butlerschein). Rats were given 1.5 mg/kg of Antisedan (Pfizer) subcutaneously to reverse the sedative effects of Dexdomitor (Pfizer), 6 mg/ml of gentamicin (VWR, Radnor, PA, USA) intramuscularly, and 10 ml of Ringer's solution subcutaneously (Butlerschein). Rats received daily injections of 5 ml Ringer's (Butlerschein) subcutaneously, 6 mg/kg gentamicin (VWR) intramuscularly, and 5 mg/kg of Rimadyl (Pfizer) subcutaneously until BMSC transplantation (47). Bladders were manually emptied twice daily until reflex voiding recovered at about 2 weeks postinjury.
Three days after the contusion, rats were anesthetized as described above, and the contused spinal cord was reexposed. Next, 5 × 105 BMSCs in DMEM or DMEM alone (total volume=5 μl; n = 10/group) was injected into the contusion epicenter using a glass pulled needle on a Hamilton syringe held within a micromanipulator (47). After injection, the needle was kept in place for 3 min and then retracted slowly over a 2-min period to prevent back leakage of the injectate. The muscles and skin were closed and the rats treated postoperatively as described above. Daily injections of Ringer's and Rimadyl for 3 days and gentamicin for 4 days were given as described above (47). There were four experimental groups: BMSC, BMSC-BDNF, BMSC-BDNFshRNA, and DMEM (n = 10 per group).
Histological and Immunocytochemical Procedures
One week (n=3 per group) or 4 weeks (n=7 per group) after injection, rats were deeply anesthetized with an intraperitoneal injection of ketamine (90 mg/kg; Butlerschein) and Dexdomitor (0.75 mg/kg; Pfizer) and transcardially perfused with 250 ml of 0.1 M PBS (Sigma-Aldrich) followed by 400 ml of 4% paraformaldehyde (Sigma-Aldrich). Spinal cords were dissected out, postfixed overnight in the same fixative, and then cryoprotected for 24 h in phosphate-buffered 30% sucrose (Fischer) for 24 h. Ten-millimeter-long segments centered on the contusion epicenter were sectioned horizontally (20 μm) on a Leica CM 1950 cryostat. Every 12th section was stained with cresyl violet (Sigma-Aldrich) for evaluating the cytoarchitecture.
Sections were immunoblocked and permeabilized in 5% NGS (Vector) and 0.3% Triton (Sigma-Aldrich) for 30 min at room temperature. Subsequently, sections were incubated with primary antibodies for 2 h at room temperature and then overnight at 4°C. The used primary antibodies were rabbit polyclonal anti-glial fibrillary acidic protein (GFAP; 1:200; Dako) to stain reactive astrocytes, and mouse monoclonal rat endothelial cell antibody (RECA-1, 1:100; Abd Serotec, Raleigh, NC, USA) to identify blood vessels. After washing twice in 1x PBS, sections were incubated with secondary antibody Alexa Fluor 594 or 488 (Invitrogen) at a concentration of 1:200 for 2 h at room temperature. Sections were stained with DAPI (2 μl/ml; Sigma-Aldrich) for 3 min to stain nuclei, covered with glass slips in fluorescent mounting medium (Dako), and stored at 4°C.
Quantification of Motoneurons and Blood Vessels
At 4 weeks posttransplantation, the number of motoneurons in the ventral horn was determined in cresyl violet-stained sections in blinded fashion. Motoneuron number was expressed as a percentage of the number of motoneurons in a comparable uninjured spinal cord segment.
At 4 weeks posttransplantation, blood vessels were quantified in RECA-1-stained sections using MATLAB® Software (MathWorks, Natick, MA, USA) (17). Per animal, three samples of 320 μm x 420 μm were taken in the three middle sections of one series (240 μm apart) at 20x magnification in the gray matter 0.6 mm caudal to the injury epicenter. All RECA-1-positive structures were outlined, and the area fraction of the outlined structures was determined in each sample and averaged per rat and per group (n=7 per group) (46).
Assessment of GFP-Positive BMSC Number and Spared Tissue Volume
One week and 4 weeks postinjection, the number of GFP-positive BMSCs in the contusion was determined in DAPI-stained sections using StereoInvestigator (MicroBrightField Inc., Colchester, VA, USA). In each section, the area with GFP-positive cells was outlined manually at 2.5x magnification and covered with a 250 × 250-μm grid. At 60x magnification with oil immersion, GFP-positive cells were marked with the optical fractionator using a 60 × 60-μm counting frame (38). Results were averaged per group and expressed as a percentage of the number of injected cells.
The volume of spared nervous tissue 4 weeks posttransplantation was determined in cresyl violet-stained sections using the Cavalieri estimator function in StereoInvestigator® Software (MicroBrightField Inc.). In each 12th section, the volume of spared tissue in a 3.4-mm-long segment centered on the contusion epicenter was determined. Beyond the rostral and caudal borders of this segment, cavities were absent. Within the analyzed segment, tissue was considered spared if it lacked cavities, areas with densely packed small cells resembling infiltrated neutrophils and lymphocytes, and neurons with darkly stained cytoplasmic Nissl bodies (38). The Gundersen coefficient of error was <0.05 for all measurements. The volume of spared tissue was expressed as a percentage of the average volume of a comparable uninjured spinal cord segment and displayed as mean ± SEM.
Sensorimotor Function Assessment
Sensorimotor function was assessed before contusion to acclimatize the rats and 4 weeks after transplantation by having the rats walk across a horizontal ladder and counting the number of slips of the foot, part of the lower leg, or the full leg. Rats crossed the horizontal ladder three times, and the average number of slips of these three crossings was expressed as a percentage of the total number of steps taken (45).
Statistical Analysis
For all results, one-way ANOVA with Tukey's post hoc analysis was used to compare differences among the experimental groups using GraphPad Software (La Jolla, CA, USA). Differences were accepted as statistically significant at p < 0.05.
Ethics and Surgical Approval
Rats used in this study were housed according to the National Institutes of Health and the United States Department of Agriculture guidelines. Rats were kept within a double-barrier facility, air in the cages was continuously refreshed, and water and food were available ad libitum. All animal procedures were approved by the Institutional Animal Care and Use Committee at the University of Pittsburgh.
Results
In Vitro Characterization of Modified BMSCs
We characterized the transduced BMSCs in vitro, which were found to have stable GFP expression for at least seven passages. Compared with BMSCs transduced with LV-GFP (Fig. 1A-E), transduction of BMSCs with LV-BDNF/GFP (Fig. 1F, G) or LV-BDNFshRNA/GFP (Fig 1K-O) did not affect the presence of typical BMSC surface markers CD90 (Fig. 1A, F, K, respectively) and CD105 (Fig. 1B, G, L, respectively) or the absence of the hematopoietic marker CD45 (Fig. 1C, H, M, respectively). BMSCs transduced with LV-BDNF exhibited an increase in staining for the BDNF receptors TrkB (Fig. 1I) and P75 (Fig. 1J), while BMSCs transduced with LV-BDNFshRNA showed a decrease in staining for these receptors (Fig. 1N, O, respectively) compared with BMSCs transduced with LV-GFP (Fig. 1D, E, respectively). Omitting the primary antibody resulted in no staining (data not shown). ELISA was used to determine the effectiveness of the used LVs in P4 BMSCs. BMSCs transduced with LV-BDNF/GFP secreted 60.4 ± 8.3 pg BDNF per milliliter of medium in 24 h, which was over sixfold higher than that secreted by BSMCs transduced with LV-GFP (9.4 ± 0.3 pg/ml/24 h, p < 0.05) (Fig. 1P). BMSCs transduced with LV-BDNFshRNA secreted an amount of BDNF below the linear range of the ELISA (<7.8 pg/ml) (Fig. 1P). DMEM does not contain BDNF.

Characterization of lentivirally transduced BMSCs. BMSCs transduced with LV-GFP (BMSC; A-E) express the BMSC surface markers CD90 (A) and CD105 (B) and show absent staining for hematopoietic marker CD45 (C). In addition, BMSCs express BDNF-signaling receptors TrK (D) and P75 (E). BMSCs transduced with LV-BDNF/GFP (BMSC-BDNF; F-J) and LV-BDNFshRNA/GFP (BMSC-shBDNF; K-O) show similar staining for CD90 (F and K, respectively), CD105 (G and L, respectively), and CD45 (H and M, respectively). BMSC-BDNF show increased expression of BDNF receptors TrkB (I) and P75 (J) and BMSC-shBDNF show decreased staining for these receptors (N, O), compared with control BMSCs. ELISA revealed that BMSC-BDNF secreted over six times more BDNF than BMSC (P). The amount of BDNF secreted by BMSC-shBDNF and that present in standard (unconditioned) culture medium (CM) were below the linear range of the ELISA (arrows). Error bars = SEM. *p < 0.05. Scale bar (A): 30 μm in (A-O).
Increased BDNF Sustains BMSC-Mediated Neuroprotection
We used a spinal cord organotypic culture system to examine the neuroprotective potential of the modified BMSCs. Figure 2 shows examples of 350-μm-thick spinal cord sections with YFP-positive ventral horn motoneurons in conditioned medium from control BMSCs (Fig. 2A, B), BDNF-hypersecreting BMSCs (Fig. 2C, D), BDNF-hyposecreting BMSCs (Fig. 2E, F), and with incubation in the defined spinal cord organotypic culture medium (Fig. 2G, H). Incubation of the 350-μm-thick spinal cord sections with conditioned medium from BMSCs transduced with LV-GFP, LV-BDNF, or LV-BDNFshRNA for 3 days resulted in a 58% (M=62.6 ± 6.4), 77% (M=70.1 ± 6.2), and 57% (M=62.1 ± 3.6) increase, respectively, in YFP-positive motoneurons compared with incubation with spinal cord organotypic culture medium (p < 0.05) (Fig. 2I). This result showed that the BMSCs, regardless of their respective genetic modification, secreted trophic factors leading to a similar degree of motoneuron sparing and that BDNF is not a necessary factor for motoneuron protection in vitro. Interestingly, at 5 days in vitro the protective effect on motoneurons was sustained with conditioned medium from BDNF-hypersecreting BMSCs (M = 66.8 ± 5.6; 87% increase over incubation with spinal cord organotypic culture medium), but not with conditioned medium from BDNF-hyposecreting BMSCs (M = 35.6 ± 4.4) or control modified BMSCs (M = 49.8 ± 3.6) (Fig. 2J). The data demonstrated that increased BDNF levels sustain spinal motoneuron sparing in vitro.

Increased level of BDNF sustains in vitro protection of spinal motoneurons. Pictures represent spinal cord organotypic cultures using conditioned medium from BMSCs transduced with LV-GFP (BMSC; A and B), LV-BDNF/GFP (BMSC-BDNF; C and D), and LV-BDNFshRNA/GFP (BMSC-shBDNF; E and F), or defined spinal cord organotypic culture medium only (DCM; G and H) at 5 days in vitro. Analysis revealed motoneuron protection by all types of conditioned medium at 3 days in culture (I). This protection was only seen in 5-day-old cultures with conditioned medium from BMSC-BDNF (J). Error bars = SEM. *p < 0.05. Scale bar (A): 500 μm in (A, C, E, G). Scale bar (B): 125 μm in (B, D, F, H). V denotes the ventral side of the cord.
BDNF Hypersecretion Enhanced Motoneuron Survival in a Contused Spinal Cord
Our data showed that BDNF has protective effects on spinal motoneurons in vitro. We next examined whether manipulation of BDNF secretion in BMSCs transplanted in a spinal cord contusion would affect spinal motoneuron survival (Fig. 3). Figure 3A shows an illustration of a 10-mm spinal cord segment centered on the contusion epicenter in which ventral horn motoneurons were quantified. Relative to the number in a comparable uninjured spinal cord segment, 80.6 ± 5.3% motoneurons were present in rats with transplants of BMSCs transduced with LV-BDNF (Fig. 3C) and 60.7 ± 1.6% in rats with DMEM injections (Fig. 3E), which represents a 33% increase in motoneuron number in rats that received BDNF-hypersecreting BMSCs (p< 0.05) (Fig. 3F). Transplantation of BMSCs transduced with LV-GFP (Fig. 3B) or LV-BDNFshRNA (Fig. 3D) did not significantly change the number of motoneurons (M= 73.9 ± 5.6, M= 63.9 ± 4.3, respectively) (Fig. 3F).

BDNF hypersecretion enhances BMSC-mediated motoneuron survival in a contused spinal cord. (A) Illustration of spinal cord section showing motoneurons in the ventral horn. Ventral motoneurons in the entire contused segment were quantified. (B-E) Motoneurons were present in the ventral spinal cord of rats injected with BMSCs transduced with LV-GFP (BMSC; B), LV-BDNF/GFP (BMSC-BDNF; C), LV-BDNFshRNA/GFP (BMSC-shBDNF; D), or with DMEM only (DMEM; E). (F) An uninjured thoracic spinal cord segment for comparison. (G) Rats that received BMSC-BDNF had 33% more motoneurons spared than rats that received DMEM. Error bars = SEM. *p < 0.05. Scale bar (B): 200 μm in (B-E).
BDNF-Hypersecreting BMSCs Augment BMSC-Mediated Blood Vessel Protection
Previously, we have demonstrated that BMSC transplantation results in increased blood vessel numbers in a contused spinal cord (46). Here we examined the role of BDNF in this protective effect by determining the density of blood vessels near the contusion (Fig. 4A). Blood vessels were present in rats with BMSCs transduced with LV-GFP (Fig. 4B), LV-BDNF (Fig. 4C), LV-BDNFshRNA (Fig. 4D), and in rats with DMEM injections (Fig. 4E). Quantification revealed a significant sixfold increase (p < 0.05) in blood vessel density in rats with BDNF-hypersecreting BMSCs compared with control (DMEM-injected) rats (Fig. 4G). Rats with BMSCs transduced with LV-GFP had a significant fourfold increase in blood vessel density compared with DMEM-injected control rats (Fig. 4G). Overexpressing BDNF in a BMSC transplant resulted in a 35% increase and silencing BDNF expression in a 76% decrease in blood vessel density compared with control BMSCs (Fig. 4G). Rats with control or BDNF-hypersecreting BMSCs have higher blood vessel density than uninjured rats (Fig. 4F and dotted line in Fig. 4G), suggesting angiogenesis accounts for at least part of the increase in blood vessel density.

BDNF hypersecretion augments BMSC-mediated blood vessel formation in a contused spinal cord. (A) Illustration of spinal cord section showing blood vessels. All RECA-1-positive blood vessels present in the boxed area were quantified. (B-E) RECA-1-positive blood vessels (red) were present in gray matter near the contusion in rats injected with BMSCs transduced with LV-GFP (BMSC; B), LV-BDNF/GFP (BMSC-BDNF; C), LV-BDNFshRNA/GFP (BMSC-shBDNF; D), or with DMEM only (DMEM, E). (F) Gray matter density in an uninjured spinal cord for comparison. (G) Rats that received BMSC-BDNF exhibited a sixfold increase and rats that received BMSC a fourfold increase in blood vessel density compared with rats without injury (dotted line) and rats receiving DMEM only. Rats receiving BMSC-shRNA had a lower blood vessel density compared with rats without injury (dotted line) and rats receiving BMSC. Error bars = SEM. *p < 0.05. Scale bar (B): 30 μm in (B-E).
Tissue Sparing 4 Weeks After BMSC Transplantation
Previous research showed that intraspinal BMSC transplants result in larger volumes of spared nervous tissue (38,46). Here we examined the role of BDNF in BMSC-mediated neuroprotection in the contused spinal cord. At 4 weeks postinjection, the volume of spared tissue in rats that received BMSCs transduced with LV-GFP or LV-BDNF was 68% larger (p < 0.05) in both groups compared to that in rats with DMEM injections (Fig. 5A). Spared tissue volume in rats with BDNF-hyposecreting BMSCs was similar as in rats with DMEM injections (Fig. 5A). Assessment of sensorimotor function recovery at 4 weeks posttransplantation showed no differences between groups (Fig. 5B).
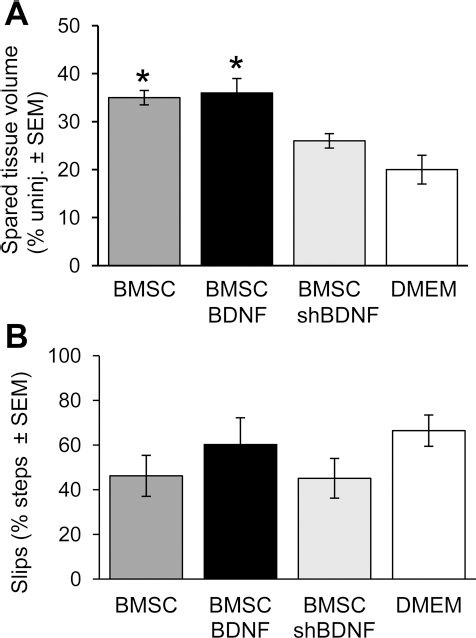
Nervous tissue sparing and functional recovery 4 weeks after BMSC transplantation into the injury epicenter 3 days after a contusion. Four weeks after transplantation, the volume of spared nervous tissue was larger in rats that received BMSCs transduced with LV-GFP (BMSC) or LV-BDNF/GFP (BMSC-BDNF) compared with rats that received BMSCs transduced with LV-BDNFshRNA/GFP (BMSC-shBDNF) and DMEM only (DMEM). There was no difference between the volumes of spared nervous tissue in rats that received BMSC-shBDNF or DMEM (A). Assessment of sensorimotor function recovery at 4 weeks posttransplantation showed no differences in the number of slips made when walking across a horizontal ladder between groups (B). Error bars = SEM. *p < 0.05.
BDNF Hyposecretion Affects BMSC Survival in a Spinal Cord Contusion
Survival of a BMSC transplant in damaged nervous tissue is limited (38). We examined whether modified production and secretion of BDNF would affect BMSC survival in a contusion environment (Fig. 6). One week after transplantation into a 3-day-old spinal cord contusion, transplanted BMSCs transduced with LV-GFP (Fig. 6A) or LV-BDNF (Fig. 6B) appeared to be mostly spindle shaped, which was in agreement with our previous study (38). Stereology-based quantification showed that 13.7 ± 2.2% of the transplanted BMSCs transduced with LV-GFP and 17.2 ± 4.9% of BMSCs transduced with LV-GFP were present in the contusion (Fig. 6D). Transplanted BMSCs transduced with LV-BDNFshRNA were not found in the contusion (Fig. 6C). The data showed that BDNF is necessary for BMSC survival in a spinal cord contusion, but overexpressing BDNF does not lead to improved survival. Four weeks after transplantation into a 3-day-old spinal cord contusion, no cells were found in any group.

BDNF silencing affects BMSC survival in a contusion. One week after transplantation, BMSCs (green) were present in the contusion in rats transplanted with BMSCs transduced with LV-GFP (BMSC, A) or with LV-BDNF/GFP (BMSC-BDNF, B) but not with LV-BDNFshRNA/GFP (BMSC-shBDNF, C). The spinal cord sections in (A-C) were stained with antibodies for glial fibrillary acidic protein to detect astrocytes (red), which enabled delineating the contusion. (D) The number of surviving BMSCs in the contusion in rats that received BMSC or BMSC-BDNF was similar (D). Error bars = SEM. Scale bar (A): 500 μm in (A-C).
Discussion
The repair-supporting effects of BMSC transplants in the injured spinal cord (1,11,12,24,25,46,54) are thought to result from paracrine actions by secreted trophic factors (3,7,9,11,22,35,39,44). We used gene therapy to manipulate BDNF production by BMSCs and assess its role in BMSC-mediated neuroprotection. In our in vitro experiments using culture medium conditioned by the modified BMSCs we found that BDNF in BMSC-conditioned medium is not a necessary factor for motoneuron survival, but elevated levels of BDNF sustained motoneuron protection, thereby enhancing the therapeutic efficacy of BMSCs. In vivo, there was also an enhanced therapeutic effect mediated by BDNF overexpression in BMSCs; increased numbers of motoneurons were found 4 weeks after grafting of BMSCs overexpressing BDNF. This in vivo effect was associated with increased gray matter vascularization, but not with increased tissue volumes or improved functional recovery. Our data further revealed that BMSC survival and survival-associated neuroprotection is dependent on BDNF production. Together our data demonstrate the significance of BDNF in BMSC-mediated neuroprotection and successfully exploit BDNF dependency to enhance anatomical spinal cord repair.
Motoneuron Survival in Organotypic Cultures
With an organotypic culture system, we showed that motoneuron survival in spinal cord sections was similar in BMSC-conditioned medium with normal, increased, or decreased amounts of BDNF at 3 days in vitro. This finding indicates that low or absent levels of BDNF are sufficient to maintain survival and that there is a threshold beyond which further increases in BDNF have no increased effect on motoneuron survival at this time point. After the first days in culture, motoneuron survival was only sustained with increased BDNF amounts. Accordingly, only BDNF-hypersecreting BMSCs enhanced spinal motoneuron survival in our adult rat model of spinal cord contusion. Thus, increased BDNF is vital to maintain the motoneuron survival-promoting effects exerted by the trophic molecules secreted by BMSCs. It should be noted that different types of neurons may have been involved in our in vivo model system compared with our in vitro model system because we did not specifically stain for motoneurons, but based our analysis on location in the ventral horn. BDNF is known to exert neurotrophic actions on neurons (3,7,9,11,22,35,39,44), and treatment of injured spinal cord with BDNF (4,5,32,42) or with BDNF-hypersecreting cells has been shown to promote neuron survival (26,34,36,48,49,51,52). Possibly, BDNF sustains the survival of motoneurons by preventing or delaying apoptotic pathways (21,23).
Survival of Transplanted BMSCs
The finding that survival of BMSCs with decreased BDNF was poorer than that of BMSCs with normal or increased BDNF in a contusion environment is intriguing. To our knowledge, this is the first demonstration of transplantation of BMSCs with silenced BDNF production into the injured spinal cord. In culture, we observed no difference in survival of any of the differently modified BMSCs that were used in this study. Also, BDNF hypersecretion did not enhance BMSC survival in vitro or in vivo. How can we explain the decrease in BMSC transplant survival in the absence of BDNF? It is possible that grafted BMSCs are protected by BDNF through autocrine signaling from apoptotic death (25,50) due to injury-related events including oxidative stress, hypoxia, and/or transplant preparation-related events such as detachment-induced anoikis. It is plausible that the observed lower expression of the BDNF receptors TrkB and P75 in BDNF-hyposecreting BMSCs also played a role in the failing autocrine BDNF-mediated protection of BMSCs. In culture, injury-related events are absent, and cells are attached; thus, BDNF would not be required for their protection. Another possible mechanism of accelerated BMSC death is immune rejection. In the present study, we did not use immunosuppressants because previous studies have shown no or limited effect of cyclosporine on survival of BMSCs injected into the injured spinal cord (41,45), possibly due to hypoimmunogenicity of BMSCs (14).
Alternatively, better graft survival in the BMSC and BDNF-hypersecreting BMSC groups may be related to the observed increase in blood vessel density near the graft in these groups, which was not observed in rats that received BDNF-hyposecreting BMSCs. Consequently, BDNF-hyposecreting BMSC grafts may have been more deprived of oxygen and nutrients. Further research is needed to elucidate the role of BDNF in BMSC transplant survival in the damaged spinal cord.
Autocrine Effect of BDNF on BMSCs
Our in vitro finding that BMSCs with higher BDNF levels have upregulated expression of BDNF receptors TrKB and P75 and that BMSCs with silenced BDNF production have downregulated expression of these receptors suggests a positive autocrine feedback mechanism. The mechanism by which BDNF may exert an autocrine positive feedback mechanism has been elucidated previously in axons (10). BDNF induces cytoplasmic cAMP and protein kinase A, which leads to increased secretion of BDNF and insertion of the BDNF receptor TrkB. In addition, BDNF-TrkB signaling activates PI3-kinase, which promotes TrkB transport (10). Similar mechanisms may play a role in BMSCs and may account for our observed findings.
Nervous Tissue Protection
In BDNF-hyposecreting BMSCs, neuroprotective effects including nervous tissue sparing and ventral horn motoneuron protection were absent at 4 weeks after transplantation, which was likely due to decreased survival of the BMSCs, rather than absence of BDNF. The finding that tissue sparing is related to BMSC survival supports two previous observations (38,47). First, we showed that earlier transplantation times (15 min or 3 days after injury) lead to better BMSC survival and increased tissue sparing compared with later transplantation times (7 days or 21 days after injury) (38). Second, we demonstrated that transplantation within a polyurethane-based reverse thermal gel increases BMSC survival accompanied by enhanced tissue sparing (47).
In the contused spinal cord with a transplant of BDNF-hypersecreting BMSCs the volume of spared nervous tissue was similar as with control BMSCs. Our finding that a transplant of control BMSCs resulted in improved tissue sparing corroborated with data from our previous studies (38,46) as well as that from other groups (1,24,25). The lack of enhanced nervous tissue sparing with BDNF-hypersecreting BMSCs was also found in an adult rat model of complete spinal cord transection (42) but not partial, dorsal column transection (48). Possibly, the severity of the injury is a determinant in BDNF-mediated nervous tissue sparing. Alternatively, gray matter and white matter may be differentially affected by BDNF. We showed previously that tissue volume analysis is mainly a measure of white matter sparing (46). Indeed, gray matter analysis did reveal additional benefits of BDNF-hypersecreting BMSCs. The 33% increase in ventral horn motoneurons that we found may not have been reflected by a significant increase in spared tissue volumes because the volume of these individual neurons is small. The increased sparing of ventral horn motor neurons could have been a direct result of increased BDNF exposure on these neurons, as discussed previously, but could also have been mediated by the angiogenic/angioprotective effects that we observed.
Blood Vessel Density
A transplantation of BMSCs with increased BDNF production significantly augmented BMSC-mediated increases in gray matter blood vessel density near the injury epicenter. With a transplant of BMSCs with decreased BDNF and decreased survival, blood vessel sparing was decreased relative to control BMSCs. Previously we showed that BMSC transplants resulted in increased vascularization in the contused spinal cord (46). Here we expand this observation and show that BDNF-hypersecreting BMSCs can enhance this effect. Other studies have also implicated BDNF in vascular events in the injured nervous tissue (8,15,28,40,42,53), possibly through upregulation of vascular endothelial growth factor (53). It is possible that our findings that transplants of BDNF-hypersecreting BMSCs leading to increased motoneuron sparing and vascularization are related. Interestingly, the blood vessel density in control and BDNF-hypersecreting BMSCs was higher than that in uninjured control rats, suggesting that angiogenesis accounts for at least part of the increase in blood vessel density. Although enhancing blood vessel number through angiogenic/angioprotective effects may result in neuroprotection (19,20,27), angiogenesis may also contribute to further injury, since newly formed blood vessels do not have a functional blood–spinal cord barrier yet (18), which may result in increased inflammation. Future research will need to elucidate the interplay between BDNF-mediated vascular and neuronal sparing.
Functional Recovery
The differences in spared ventral horn motoneurons, tissue volumes, and vascularization observed in this study did not result in statistically significant sensorimotor recovery at 4 weeks posttransplantation. Previously, we did observe BMSC-mediated improved sensorimotor functional recovery (46,47), both with a higher dose of 1 × 106 BMSCs (46) and with the currently used dose of 5 × 105 (47). The discrepancy may be a result from the combination of the difference in observation time point with a different dose, since previous differences at 4 weeks postinjection were seen with the higher dose, and previous differences using the current dose were seen at 6 weeks postinjection (47). Since seemingly small differences in methodology can result in different outcomes, further research should focus on the extent and the limitations of the therapeutic range of BMSCs. Alternatively, the increase in ventral horn motor neuron survival in rats with BDNF-overexpressing BMSCs may have been too modest to result in functional improvements. Also, possible negative effects of angiogenesis (see discussion above) may have counteracted an enhancement in sensorimotor functional recovery.
It is possible that beneficial effects provided by BDNF-hypersecreting BMSCs only address neuron survival, but not long axon regeneration and/or synaptic reconnectivity that may be important for achieving functional recovery. Therefore, development of treatment strategies that focus on axon regeneration is necessary. However, it remains crucial to combine such regenerative strategies with neuroprotective strategies to limit degeneration of newly regenerated and spared axons.
BMSC Transplantation Versus Conditioned Medium Administration
Given the growing evidence of the current and previous studies that BMSCs exert their therapeutic effects after spinal cord injury by paracrine effects, the question arises whether transplantation of BMSCs is necessary. Although it seems plausible that continuously secreted factors by transplanted BMSC have an advantage over administration of BMSC-conditioned medium, which quickly dilutes away, there is some evidence that BMSC-conditioned medium by itself can have therapeutic effects (6). Further research is needed to directly compare the effects of transplanted BMSCs with BMSC-conditioned medium.
Footnotes
Acknowledgments
Financial support was provided by LUMC (grant No. 30229/5000), Wings for Life Foundation (grant WFL-US-015/13), Craig H. Nielsen Foundation (grant 284621), and the Department of Physical Medicine and Rehabilitation at the University of Pittsburgh. The authors declare no conflicts of interest.
