Abstract
Desferoxamine (DFO), an iron chelator, mimicked hypoxia by inhibiting HIF-1α degradation and upregulated angiogenic factors. In this experiment, we elucidated the effect of DFO on CD34-positive cell migration and neovascularization. CD34-positive cells were cultured in media with DFO or an inhibitor and subjected to in vitro tubule formation and the expression of factors. Nude mice were randomly divided into five groups of 12: control, CD34, CD34-DFO, CD34-DFO-AMD (AMD3100, CXCR4 inhibitor), and CD34-DFO-LY (LY294002, the PI3K inhibitor) groups. Limb perfusion and in vivo imaging was evaluated by laser speckle imaging (LSI) and bioluminescence imaging (BLI). Capillary density was examined 14 days after surgery, and the relevant mechanism was also explored. In vitro, DFO significantly increased the tube formation and expression of angiogenic factors in CD34-positive cells, which were blocked by the PI3K inhibitor, LY294002. DFO enhanced blood flow, the function of the ischemic hindlimb, and the levels of VEGF. Further, p-eNOS and p-Akt increased in response to the ischemia. BLI showed that DFO increased the number of CD34-positive cells targeted to the ischemic sites. Immunohistofluorescence revealed that the capillary density in the ischemic hindlimb was significantly higher in the DFO treatment group compared with the other groups. However, all of these effects were diminished by LY294002. DFO treatment enhanced CD34-positive cell targeting and improved neovascularization via the PI3K/Akt signal transduction pathway in an ischemic hindlimb.
Introduction
Neovascularization plays a key role in ischemic disease (23). Postnatal neovascularization, also known as angiogenesis (15), has been defined as the growth of new capillaries from preexisting blood vessels. However, this process is limited and dependent on the ability of existing endothelial cells to respond to the angiogenic growth factor (27). In contrast, vasculogenesis is defined as the incorporation of endothelial progenitor cells to the site of ischemia and results in the formation of new blood vessels (2,8,33). Recent animal studies have shown that transplantation of EPCs improves neovascularization in ischemic tissue (1,26). CD34 is selectively expressed within hematopoietic systems on stem and progenitor cells (44). CD34-positive cells have properties of EPCs (13,46). CD34-positive endothelial progenitor cells have been widely used clinically and in animal experiments (2,30,35). The targeting and retention rate of exogenously administered stem cells within the injury site remains low (34), which suggests that administered stem cells alone were inadequate. Chemoattraction may facilitate recruitment of more cells to the ischemic site. In 2004, Ceradini et al. discovered that recruitment of progenitor cells to injured tissue is mediated by hypoxic gradients via hypoxia inducible factor 1 (HIF-1)-induced stromal cell-derived factor 1 (SDF-1) expression (9).
Hypoxia is one of the most potent stimuli to activate HIF-1α, which regulates the expression of SDF-1 and vascular endothelial growth factor (VEGF) (5,17,36), which affects signaling pathways and cell functions. The downstream gene SDF-1 and its ligand CXCR4 stimulate the recruitment and proliferation of stem cells and modulate the trafficking of stem cells to accelerate the neovascularization of ischemic tissue (9,32). Studies of many cell types suggested that the PI3K/Akt signal transduction pathway also plays a key role in cellular proliferation, migration, and survival (4,43). Recently, Zheng et al. reported that the PI3K/Akt/eNOS signal transduction pathway plays a crucial role in SDF-1α-induced stem cell migration (47). The PI3K signaling also mediated VEGF expression and activated eNOS in angiogenesis (14,25).
Desferrioxamin (DFO), an iron chelator used to treat excess iron, inhibits prolyl hydroxylation, causing HIF-1 stabilization (42). Several studies have suggested that DFO accelerates neovascularization in animal models of ischemic tissue (11,21). Further work is necessary to determine whether DFO improved the mobilization of CD34-positive cells, recruited more CD34-positive cells to the ischemic site and augmented vasculogenesis and angiogenesis at the injury site.
In this study, we showed that DFO stimulated SDF-1/CXCR4 via the PI3K/Akt signal transduction pathway and enhanced the number of migrating cells, thus accelerating cell targeting. DFO augmented local neovascularization via proliferation, differentiation, and secretion of angiogenic growth factors in response to ischemia.
Materials and Methods
Animals
We studied 6- to 8-week-old male nude mice (n = 50) and male transgenic rats (n = 10). Nude mice were from Shanghai SLAC Laboratory Animal Company (Shanghai, China). Luciferase transgenic LEWIS rats from Jichi Medical University were used. All of the animals were housed in groups of three, with free access to standard chow and drinking water, in a 12-h light cycle. All of the animal experiments in this study were performed in accordance with the Shanghai Jiaotong University Medical Center Institutional Animal Care and Use Committee.
Isolation of CD34-Positive Cells by Magnetic Cell Sorting
Mononuclear cells were derived from the bone marrow obtained from the long bones of the Luciferase LEW rats. Femurs and tibiae were aseptically removed from the euthanized animals (200 mg/kg pentobarbital IP) (Sigma-Aldrich, St. Louis, MO, USA). Bones were harvested by flushing with cold phosphate-buffered saline solution (PBS). The cells were then washed two to three times with PBS (Sigma-Aldrich). The cells were isolated by density gradient centrifugation with Histopaque-1083 (Sigma-Aldrich) for 25 min at 500 x g. Mononuclear cells were collected and washed three times in PBS with 2% FBS (Invitrogen Corporation, Grand Island, NY, USA). Cells were incubated with a monoclonal anti-CD34 antibody (1:100; Santa Cruz Biotechnology, Santa Cruz, CA, USA) for 30 min at 4°C. Additional incubation with anti-pan mouse IgG-conjugated microbeads (10 μl, Cat. No. 115.31D, Dynal; Invitrogen), for 30 min at 4°C, was followed by processing through a MACS magnet (Invitrogen) to obtain CD34-positive cells (16).
Isolated CD34-positive cells were plated onto six-well plates (BD, San Diego, CA, USA) coated with fibronectin (Invitrogen) and cultured in EBM-2 (Clonetics; Lonza, Walkersville, MD, USA) with the Bulletkit supplement (5% FBS, hydrocortisone, gentamycin, VEGF, human FGF, human EGF, R3-IGF, and ascorbic acid) (Clonetics; Lonza). The medium was changed every 3 days, and the culture was maintained through 7 days.
Ac-LDL Incorporation
Endothelial progenitor cells have the capacity to uptake Ac-LDL. CD34-positive cells were incubated in a medium containing 10 μg/ml of DiI-labeled Ac-LDL (Molecular Probes, San Diego, CA, USA) for 4 h at 37°C. After staining, cell imaging was visualized with an inverted fluorescent microscope (Olympus, Tokyo, Japan), in which five randomly selected microscopic fields were used to calculate the number of positive cells.
Flow Cytometry
Isolated bone marrow-derived CD34-positive cells were subjected to flow cytometry to examine surface markers. Cells (2 × 105) were incubated with monoclonal antibodies against CD34 (1:100; Santa Cruz Biotechnology), CD133 (1:200; Abcam, Cambridge, MA, USA), and KDR (1:200; Abcam) for 30 min at 4°C. The cells were then stained with the Alexa-Fluor 488 goat anti-mouse IgG and the Alexa-Fluor 488 goat anti-rabbit IgG (1:500; Invitrogen) for 30 min at 4°C. The cells were washed three times with PBS, resuspended, and filtered. Quantitative FACS was performed on a flow cytometer (BD). The data were analyzed by FCS Express 4.0.
Tube Formation in Matrigel
CD34-positive cells have the ability to differentiate and organize into capillary-like structures in a gel matrix (2). Growth factor-reduced Matrigel (BD) was pipetted onto the 24-well plates (BD Pharmingen). The cells were seeded on 24-well plates at 5 × 104 cells/well in the presence of 100 μM DFO (Desteral; Novartis Pharma Stein AG, Stein, Switzerland), with and without AMD3100 (5 μg/ml; Sigma-Aldrich), with or without LY294002 [20 μM, dissolved in dimethyl sulfoxide (DMSO; both Sigma-Aldrich)], and incubated for 8 h at 37°C, according to the manufacturer's instructions. The tube networks were observed with an inverted microscope (Olympus), and five random fields per group were taken. The averages of the total areas were measured by Image-Pro Plus 6.0 (Media Cybernetics, Silver Spring, MD, USA).
Quantitative Real-Time Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
Total cellular RNA was extracted from CD34-positive cells that were pretreated with LY294002 (20 μM) or AMD3100 (5 μg/ml) alone for 1 h before stimulation with DFO by the TRIzol reagent (Invitrogen). Reverse transcription was performed using the M-MLV kit (Promega, Corporation, Madison, WI, USA) as described by the manufacturer (Table 1). The transcribed cDNA was then used for PCR amplification to estimate the expression of VEGF, HIF-1α, and SDF-1. The reaction was then cycled at 95°C for 5 min, 40 cycles at 95°C for 5 s, and then 60°C for 30 s. All of the reactions were performed in triplicate. The PCR products were quantified with MxPro-Mx3000P system (Stratagene, Foster, CA, USA).
Primers Used for Real-Time Polymerase Chain Reaction Amplification
Cell Culture Assays for Luciferase Activity
We demonstrated the linear relationship of the photon emission and viable cell count by plating the CD34-positive-luciferase cell suspension into 96-well plates (BD Pharmingen) in triplicate in a series ranging from 37,500 cells to 1 × 106 cells diluted in culture media. After adding 100 μl of D-luciferin substrate (Promega), the cells were incubated for 5 min and then imaged for 1 min at the same region of interest used to measure the luminescent signals (22). Bioluminescence per well was quantified in photons per second.
CD34-Positive Cell Transplantation Model and Surgical Procedures
The surgical procedure for the ischemic hindlimb was previously described (29). Athymic nude mice were anesthetized with 50 mg/kg pentobarbital IP for operation and meloxicam (Boehringer Ingelheim, Shanghai, China) (1 mg/kg, IP) for analgesia. The adequacy of anesthesia was monitored from the disappearance of righting reflex or pedal withdrawal reflex. A longitudinal incision in the middle of the hindlimb exposed the left femoral artery. The proximal end of the common femoral artery was ligated twice using a 10-0 nylon suture (Johnson & Johnson, New Brunswick, NJ, USA). To examine the cell population of CD34-positive cells targeting the ischemic tissue, 2 × 105 cells (100 μl of cell suspension in PBS) were injected into the common femoral artery with a 29-gauge needle attached to a 1-ml syringe. The distal end of the femoral artery proximal to the popliteal artery was ligated. The artery was dissected free. The incision site was repaired, and the mouse was returned to the cage after surgery. Nude mice were injected with CD34-positive cells. Mice without CD34-positive cell injections were treated as controls. To detect the effect of DFO on cell targeting, the mice injected with CD34-positive cells were treated with DFO 14 days (200 μl, 100 mg/kg/day, IP) after cell implantation. Some of these mice were treated either with or without inhibitor AMD3100 14 days (100 μl, 10 mg/kg/day, IP) beginning the day before cell implantation. Other mice were treated with or without LY294002 14 days (100 μl, 20 mg/kg/day, IP) also beginning the day before cell implantation. The experiment was divided into five groups: Control, CD34, CD34-DFO, CD34-DFO-AMD, and CD34-DFO-LY.
Laser Speckle Imaging
The laser speckle imaging (LSI) device (Moor Instruments Co. Ltd., Devon, UK) was used to measure cutaneous blood flow in the ischemic and contralateral limbs of the mice. Perfusion analyses were performed under pentobarbital anesthesia before surgery, immediately after, and at 3, 7, and 14 days after surgery. The LSI (Moor Instruments Co. Ltd.) measurements were performed in a quiet environment. The CCD camera was positioned 25 cm above the tissue surface of the hindlimb. The height and system parameters remained constant in every case, with values expressed in BPU. To prevent measurement biases, images were recorded (red denoted high perfusion, blue signified low perfusion), and calculated perfusion was expressed as a ratio of the ischemic hindlimb to the contralateral normal limb.
In Vivo Bioluminescent Imaging
We longitudinally tracked the cell grafts in vivo, by subjecting the nude mice to bioluminescence imaging 0, 3, 7, and 14 days after transplantation. Bioluminescence imaging (BLI) was performed using the IVIS200 (Xenogen, Alameda, CA, USA) system, and signal intensity was quantified by photon flux in units of photons/s/cm2/steradian in the region of interest using Living Image 3.0 software (Caliper Life Sciences, Alameda, CA, USA), as previously described (38). Recipient nude mice were anesthetized with isoflurane. D-Luciferin was administered at a dose of 150 mg/kg and placed in the imaging chamber of a CCD camera system. All of the images were collected 15 min after D-luciferin IP injection. Regions of interest were drawn on the ischemic hindlimb. To determine the threshold for a positive BLI signal, animals without cell injections were imaged, and the results were quantified.
Immunohistochemistry
The nude mice were sacrificed with 200 mg/kg pentobarbital IP, and the ischemic hindlimb muscles were removed. The samples were then covered with OCT (Tissue-Tek, Torrance, CA, USA). The 7-μm frozen sections were prepared for immunohistochemical analysis and fixed in methanol (Sigma-Aldrich) at room temperature. Immunofluorescence staining was performed with the following antibodies: mouse anti-CD31 (1:100; Abcam), rabbit anti-α-SMA (1:200; Abcam), rabbit anti-Ki-67 (1:200; Abcam), Desmin (1:100; Abcam), rabbit anti-CXCR4 (1:100; Abcam), rabbit anti-luciferase and rabbit anti-eNOS (1:50; Abcam). The secondary antibodies for each immunostaining were as follows: Alexa Flour 488-conjugated goat anti-mouse, Alexa Flour 555-conjugated goat anti-rabbit, and Alexa Flour 555-conjugated goat anti-mouse (1:500; Invitrogen). DAPI (1:1,000; Chemicon, Temecula, CA, USA) was used to stain the cell nuclei in all of the tissue samples. The fluorescence signals were visualized with a laser scanning confocal microscope (LSM 710; Leica, Wetzlar, Germany). Within each section, five random vision areas were chosen. Images were analyzed by Image-Pro Plus (Media Cybernetics, Silver Spring, MD, USA).
Terminal Deoxynucleotidyl Transferase-Mediated Deoxyuridine Triphosphate Nick End Labeling (TUNEL) Staining
Apoptotic cells in the tissue sections were analyzed by TUNEL staining using the In Situ Cell Death Detection TMR red (Roche, Basel, Switzerland) following the manufacturer's protocol. The sections were fixed by 4% paraformaldehyde (Sigma-Aldrich) for 10 min at room temperature and permeabilized with 0.3% Triton X-100 (Sigma-Aldrich) for 10 min. The sections were incubated with the TUNEL reaction mixture at 37°C for 1 h. Subsequently, the samples were covered with mounting medium with DAPI (Chemicon). The sections were washed three times in PBS. Five random fields per section were selected to count the number of TUNEL-positive cells.
Protein Extraction and Western Blot
The total extracted protein from the adductor muscles in the ischemic hindlimb was separated with 10% SDS-PAGE (Sigma-Aldrich), transferred to a polyvinylidene difluoride membrane (Sigma-Aldrich), and blocked with 5% milk (Meiji, Kanagawa, Japan) Tris-buffered saline (Sigma-Aldrich) with Tween (Sigma-Aldrich). The primary antibodies were used for Western blot against the following proteins: p-Akt (1:500; Abcam), AKT (1:500; Abcam), VEGF (1:1,000; Santa Cruz Biotechnology), SDF-1 (1:1,000; Abcam), HIF-1(1:500; Abcam), p-eNOS (1:500; Abcam), eNOS (1:500; Abcam), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH; 1:1,000; Santa Cruz Biotechnology). These solutions were stored overnight at 4°C. The membranes were then washed and probed with HRP-conjugated secondary antibodies (1:500). The labeled bands were visualized using ECL (Amersham Pharmacia Biotech, Piscataway, NJ, USA), and protein expression was semiquantified using a Tanon imager and analysis system (Tanon, Shanghai, China).
Statistical Analysis
The data were processed using SPSS software (SPSS Incorporated, Chicago, IL, USA). Values were reported as means ± SD. Statistical analysis among multiple groups at each time point was performed by ANOVA (SNK's test). A value of p < 0.05 was considered statistically significant. Analysis of correlation between cell number and the photon intensity was performed by linear regression.
Results
Bone Marrow-Derived CD34-Positive Cells From Luciferase Transgenic Rats
The CD34-positive cells that were isolated and cultured for 7 days turned into spindle cells. To evaluate the biological characteristics of CD34-positive cells, the cells were tested for uptake of DiI-ac-LDL. From these cells, 88.75 ± 1.2% of them incorporated DiI-ac-LDL. The CD34-positive cells were analyzed by flow cytometry. The CD34-positive, CD133-positive, and KDR-positive fractions were 64.95 ± 4.06%, 90.5 ± 2.27%, and 30.79 ± 1.93%, respectively. To visualize the fate of the CD34-positive cells, luminescence sensitivity was determined. The cells exhibited a robust correlation between firefly luciferase expression and the number of CD34-positive cells (R2=0.994).
DFO Treatment Activates the HIF-1 Pathway and the PI3K/AKT/eNOS Signaling Pathway In Vitro
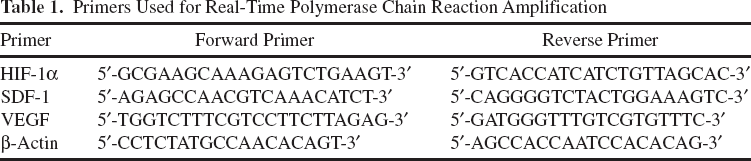
To evaluate whether the SDF-1/CXCR4 and the PI3K/Akt signal transduction pathways were activated by DFO, CD34-positive cells were pretreated for 1 h with CXCR4 inhibitor AMD3100 (5 μl/ml) and PI3K inhibitor LY294002 (20 μM). DFO at 1-100 μM increased the expression of HIF-1α, SDF-1, and VEGF in CD34-positive cells, especially at 100-200 μM (p < 0.05). The expression of HIF-1 a, SDF-1, and VEGF significantly increased due to DFO, compared with other groups, and activated downstream genes VEGF and SDF-1. A corresponding increase in HIF-1α, SDF-1, VEGF, p-Akt, and p-eNOS protein expression was observed. The upregulated expression was abolished by incubation with AMD3100 or LY294002 (p < 0.05), suggesting that activation of the factors occurred through CXCR4-dependent pathways, and eNOS phosphorylation activated via a PI3K/Akt signal transduction pathway (Fig. 1).
DFO treatment activates the HIF-1 pathway and the PI3K/Akt/eNOS signaling pathway in vitro. (A) Dose-dependent changes in VEGF, SDF-1, and HIF-α after DFO stimulation. (B) Expression of targeted genes by DFO (100 μM) incubated with or without AMD3100 (5 μl/kg) and LY294002 (20 μM). HIF-1, SDF-1, VEGF, Akt, and eNOS expression increased due to DFO treatment. LY294002 and the CXCR4 antagonist, AMD3100, blocked the upregulation of these genes (p<0.05; two-way repeated-measures ANOVA; n = 6). (C) The expression of targeted protein was seen in different experimental groups. (D, E) Quantitative analysis of Western blots for HIF-1α, SDF-1, VEGF, p-Akt, Akt, p-eNOS, and eNOS protein in different groups (p<0.05; n = 6).
DFO Enhances Tube Formation In Vitro
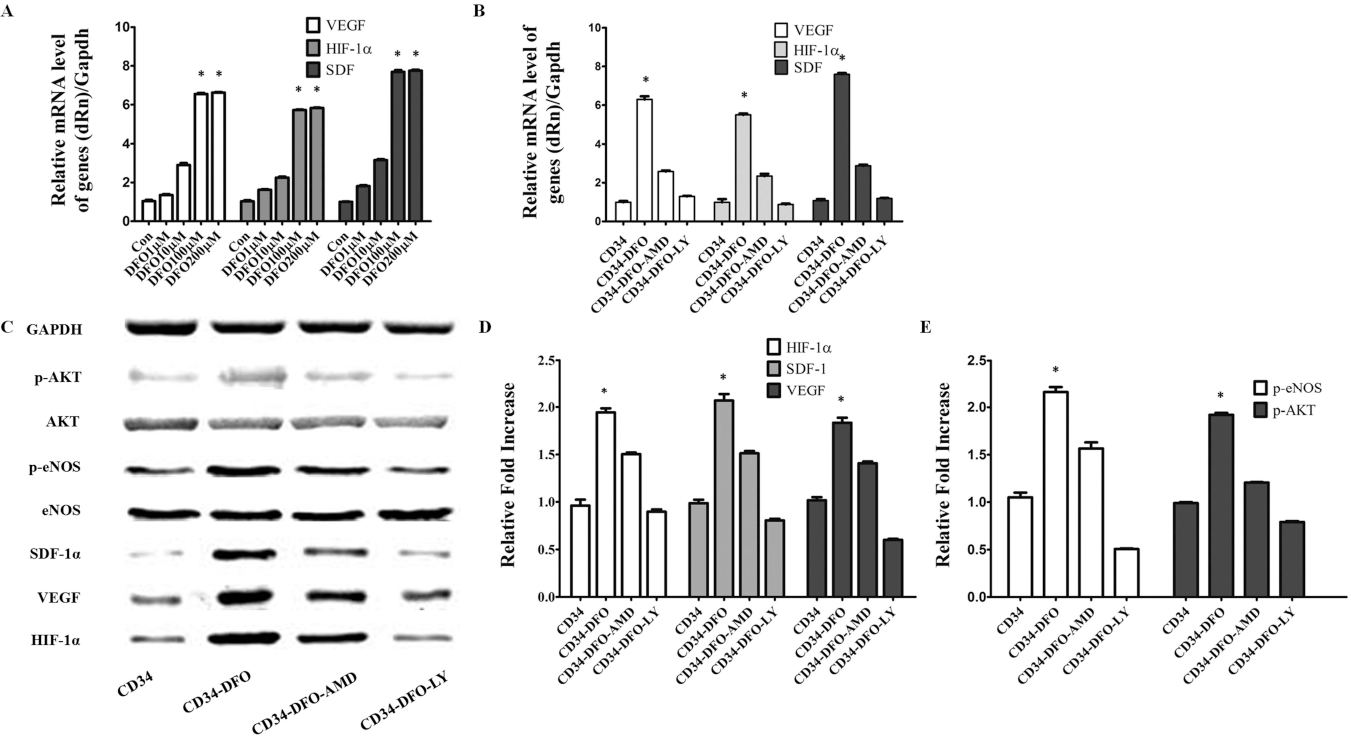
To assess whether DFO activates endothelial function, in vitro tube formation by CD34-positive cells was measured. In the tube formation assay, the area of complete tubes was analyzed. DFO-treated CD34-positive cells induced a significantly larger tube formation area than other groups (DFO 100 μM 209 ± 12.73 vs. Con 31.5 ± +2.12, p<0.05). AMD3100 (CXCR4 inhibitor) and LY294002 (PI3K inhibitor) suppressed DFO-induced tube formation (Fig. 2). The results indicated that DFO promotes tube formation through PI3K/Akt mediated signaling.
The effect of DFO on tubule formation (n = 4). (A) A representative figure showing tube formation in vitro that was induced by the absence (control) or presence of DFO, with or without 5 μg/ml AMD3100, and with or without 20 μM LY294002 (magnification: 100x). Scale bar: 100 μm. (B) A quantitative graph of an area of complete tubes (p < 0.05; two-way repeated-measures ANOVA).
DFO Accelerates Revascularization of Acute Hindlimb Ischemia
To analyze the impact of DFO on blood perfusion after femoral artery ligation and the subsequent repair of ischemic tissue in vivo, LSI imaging, the physiological status of the ischemic hindlimb, and capillary density were measured for revascularization. LSI showed that perfusion of the ischemic hindlimb decreased equally in all groups immediately after surgery.
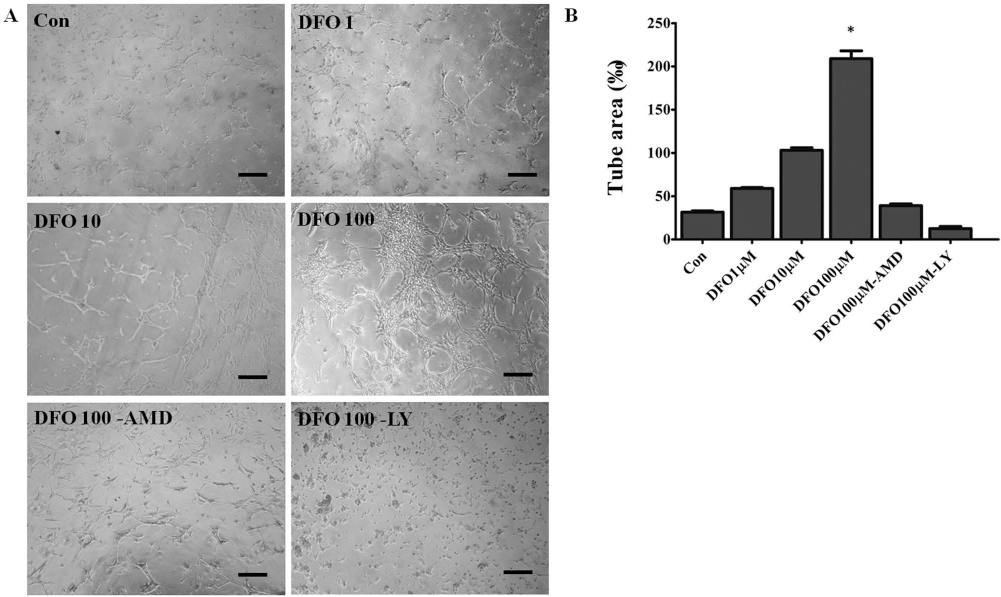
The physiological status of the ischemic hindlimb was measured by salvage, necrosis of the foot, or spontaneous amputation at 14 days. In the CD34-DFO group, 6.67% of nude mice showed autoamputation, with foot necrosis limited to 33.3%, and 60% of the mice showing salvage. In the CD34-DFO-AMD group, 46.67% of mice showed salvage and was slightly improved from the ischemic state of the control group. In the CD34-DFO-LY group, this effect was not ameliorated. The incidence of necrosis of the ischemic hindlimb in the CD34-DFO group was significantly lower than in other groups (p < 0.05) (Fig. 3A). Furthermore, we compared the efficacy of all groups in restoring blood flow to ischemic hindlimbs. Blood perfusion of the ischemic hindlimb in the CD34-DFO group increased with time and significantly recovered compared with other groups at days 7 and 14 (p < 0.05). The rates of blood perfusion at day 14 were 39.39 ± 0.03, 60.67 ± 0.03, 80.63 ± 0.03, 65.89 ± 0.05, and 34.49 ± 0.05 in the control, CD34, CD34-DFO, CD34-DFO-AMD, and CD34-DFO-LY groups, respectively. Perfusion in the CD34-DFO-AMD group was higher than in the control group, and a similar degree of improvement was observed in the CD34 group. The improvement of blood perfusion was diminished by the use of LY294002 (Fig. 3B, C). These findings were concordant with the incidence of necrosis. As shown in Figure 4A, the capillary density significantly increased in the CD34-DFO group versus the other groups (p < 0.05). The number of capillaries in the CD34 and CD34-DFO-AMD groups was similar; the values (611.23 ± 22.64 vs. 656.63 ± 24.68; p > 0.05) were not significantly different, but both groups were superior to mice in the control group (395.2 ± 48.06; p < 0.05). New vessel formation was inhibited in the CD34-DFO-LY group. To identify arterioles, α-SMA was used in tissue sections of the ischemic legs 14 days after cell delivery. DFO treatment significantly increased arterio-genesis compared with other groups (p < 0.05) (Fig. 4A). The results demonstrated that DFO stimulated the neovascularization response to hypoxia and achieved a better effect on the improvement of ischemic limb functions.
DFO improved ischemic revascularization 14 days after the operation. (A) Improvement of necrosis using nude mice. The CD34 group had a slightly reduced incidence of necrosis. The CD34-DFO group had significantly reduced necrosis. The CD34-DFO-LY and CD34-DFO-AMD group had an increased incidence of necrosis (*p<0.05, x2 test). (B, C) LSI imaging of the hindlimb after CD34-positive cell delivery. (B) Representative laser speckle imaging showed blood perfusion in limb ischemia at 14 days after cell delivery. (C) The ratio of ischemic to normal tissue as visualized by LSI. The improvement in blood perfusion was significantly gradually recovered in CD34-DFO group at both days 7 and 14 (n=10 per group; p<0.05; two-way repeated-measures ANOVA).

Effect of DFO on neovascularization and fate of implanted CD34-positive cells by immunofluorescent staining. (A) Immunofluorescent staining for CD31, α-SMA, CXCR4, and DAPI (blue) in the ischemic leg. Scale bar: 50 μm. (B) A representative triple immunofluorescent staining for nuclei (blue), luciferase (green), and CD31 (red) in the ischemic limb. (C) Quantitative analysis of the number of capillaries per mm2 in the five experimental groups. (D) Quantification of arteriolar density in all groups. (E) Quantitative analysis of CXCR4-positive cells in the ischemic limb (p<0.05 vs. any other treatment groups; two-way repeated-measures ANOVA) (n = 5).
Effect of DFO on Fate of Implanted CD34-Positive Cells
To evaluate whether CD34-positive-luciferase participated in the ischemic tissue by differentiation, a double immunofluorescent analysis of the tissue sections was performed. Some CD34-positive-luciferase developed into an endothelium-like cell layer (Fig. 4B). Furthermore, to study whether DFO treatment enhanced the recruitment of cells to ischemic muscle, CXCR4 expression was measured by immunofluorescence at day 14. The cells that expressed CXCR4 were significantly different between CD34-DFO and other groups (Fig. 4A). In contrast, CD34-positive cells were rare, and vessel formation was inhibited in the CD34-DFO-LY and CD34-DFO-AMD groups. Expression of CXCR4 significantly decreased due to AMD3100 and LY294002. Overall, these results suggested that DFO enhanced the migration and retention of cells in ischemic tissue and promoted vasculogenesis by differentiation. These effects were nullified by LY294002 and AMD3100.
To track the distribution of allogenic CD34-positive cells, we used CD34-positive cells from transgenic mice that expressed luciferase. Cells delivered by the intra-arterial route primarily accumulated in the lungs and in the ischemic limb at day 0. The BLI intensity in the lung declined and disappeared over the following 3 days in all of the groups. In the CD34-DFO group, the intensity of the BLI signal in the ischemic limb was significantly higher at day 14 compared with the other groups (p < 0.05). The BLI signal was not significantly different between the CD34-DFO-LY and the CD34-DFO-AMD group (1.28 × 105 ± 2.02 × 104 vs. 1.01 × 105 ± 8.82 × 103; p > 0.05), and the BLI signal was not greater than the threshold at day 14 (Fig. 5). The results demonstrated that LY294002 or AMD3100 completely inhibited the migration or retention of CD34-positive cells in the ischemic tissue. However, the CD34-positive cells were enhanced by DFO.

Distribution and survival of CD34-positive cells in the ischemic hindlimb. Luciferase-CD34-positive cells were delivered to the ischemic limb by IA. D-Luciferin was injected by IP, and luminescence was detected using the IVIS system. (A) Luminescence was recorded at days 0, 3, 7, and 14 posttransplantation. (B) Quantification of BLI signals in the hindlimb after cell delivery. The CD34-DFO group had significantly higher luminescence than any other group at 14 days (p < 0.05; two-way repeated-measures ANOVA). (The dashed line represents the threshold for a positive BLI signal; n = 10 per group.)
Effect of DFO on Tissue Regeneration
To determine whether improved limb reperfusion and enhanced tissue regeneration occurred in the ischemic limb, cell proliferation and apoptosis were assessed in the ischemic tissue at day 14. Regenerated tissues were stained with Ki-67 antibody for investigation of cell proliferation. There were more positive cells in the CD34 and CD34-DFO-AMD groups than in the control group (CD34 257.65 ± 32.71 vs. Con 128.24 ± 28.89; CD34-DFO-AMD 227.13 ± 57.21 vs. Con 128.24 ± 28.89). This result was significantly increased by DFO (CD34-DFO group) and inhibited by LY (p < 0.05) (Fig. 6A, B). Apoptosis, which was measured by TUNEL-positive nuclei, significantly decreased in the DFO-CD34 group compared with other groups (DFO-CD34 9.76 ± 2.99 vs. Con 26.43 ± 3.733). The number of apoptotic cells increased in the CD34-DFO-LY groups (p < 0.05) (Fig. 6A, C). These results demonstrated that DFO treatment inhibited apoptosis and enhanced regeneration in ischemic tissue.
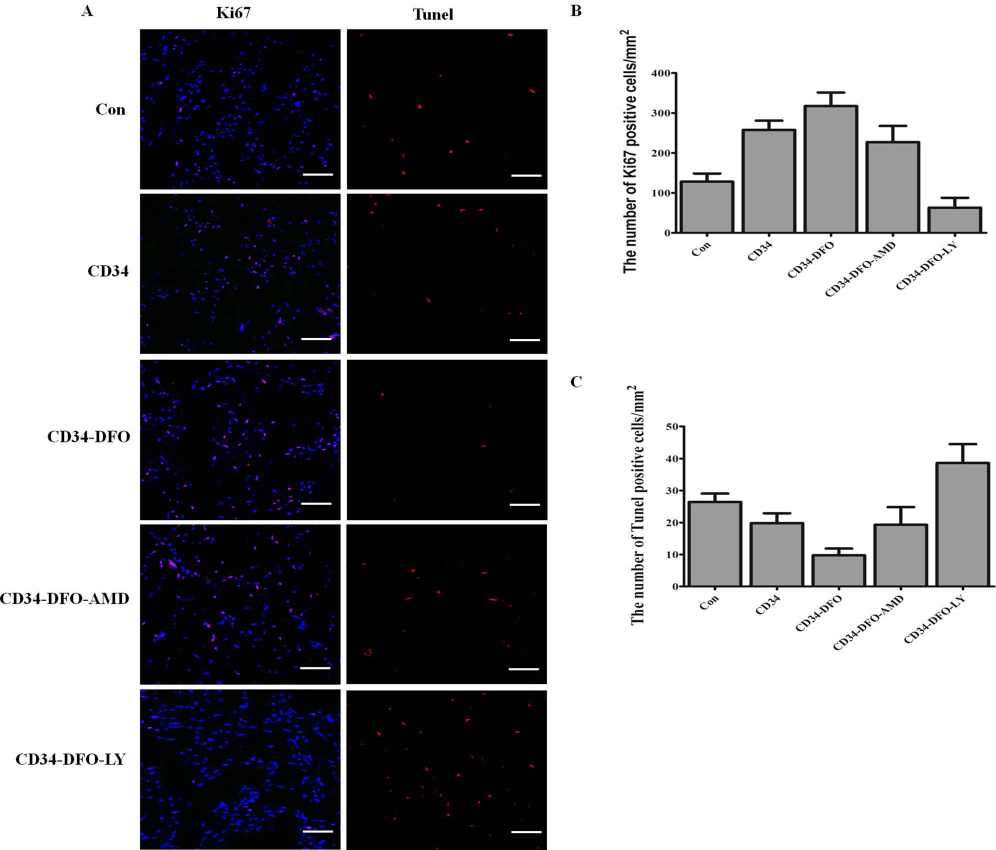
Effect of DFO on tissue regeneration. (A) Immunofluorescent staining for Ki-67 and TUNEL (red) and DAPI (blue) in the ischemic leg. Scale bar: 50 μm. (B) The number of Ki-67-positive cells was quantified. (C) Quantitative analysis of apoptotic cells in the ischemic limb (p < 0.05 vs. any other treatment groups; two-way repeated-measures ANOVA) (n = 5).
DFO Upregulation of Angiogenic Factors via Stimulation of the P13K Pathway
To address the mechanism and quantify signaling intermediates under different stimuli, we measured HIF-1α, VEGF, SDF-1α, p-eNOS, e-NOS, p-Akt, and Akt by Western blot (Fig. 7). The protein levels of HIF-1α, VEGF, SDF-1α, p-eNOS, and p-Akt was upregulated in the CD34 and CD34-DFO-AMD groups. However, the protein levels were relatively lower than in the CD34-DFO group (p < 0.05). LY inhibited protein expression. These results supported the hypothesis that DFO stimulates angiogenic expression to improve neovascularization via the PI3K/Akt signal pathway.

DFO upregulation of angiogenic factors. (A) The expression of targeted protein was seen in the ischemic muscles of mice in the five experimental groups. (B) Quantitative analysis of Western blots for HIF-1α, SDF-1, VEGF, p-Akt, Akt p-eNOS, and eNOS protein in five groups (p < 0.05) (n = 5).
Discussion
In this report, we examined the effect of DFO on CD34-positive cell targeting and neovascularization in ischemic tissue. The results demonstrated that DFO promoted vasculogenesis and angiogenesis in hindlimb ischemia by activating the PI3K/Akt signaling pathway by SDF-1 mediation, leading to enhanced CD34-positive cell migration in response to ischemia. Furthermore, DFO promoted revascularization by increasing the number of capillaries and arterioles. DFO also inhibited apoptosis via the activation of Akt and upregulation of the angiogenesis factors, VEGF and eNOS.
Studies have shown that improving neovascularization with stem cells is one of the most effective strategies to alleviate ischemic injury (6). CD34-positive cells induce neovascularization and secretion of cytokines. Our study demonstrated the vasculogenic potential of ex vivo CD34-positive cells in vivo. Our data indicated that CD34-positive cells were functionally more efficient in restoring tissue blood flow to the ischemic hindlimb muscle than in the control group. BLI confirmed that a small number of CD34-positive cells targeted the ischemic region and differentiated into endothelial cells. Furthermore, implantation of CD34-positive cells enhanced the paracrine function.
Hypoxia stimulates vasculogenesis in adult ischemic tissue by inducing expression of chemotactic factors, such as SDF-1, which enhances the migration of stem cells (9). DFO stabilizes the HIF-1α protein and activates the expression of genes involved in the reduction of hypoxia (10). Therefore, DFO may be a new method to promote CD34-positive cell migration to ischemic tissue. To further verify this hypothesis, CD34-positive cells were transplanted with the luciferase gene in combination with BLI analysis. We then observed that the CD34-positive cells dynamically migrated to the injury site. CD34-positive cells were distributed into the lungs and the injured site 2 days after operation. A long-term analysis of the dynamic biodistribution of CD34-positive cells demonstrated the effect of DFO on CD34-positive cell migration in ischemic hindlimbs. After DFO was administered, BLI showed that the administered CD34-positive cells systemically localized to an ischemia site significantly more than the other groups. Further, histology studies confirmed significant cell migration and differentiation into the ischemic tissue following DFO treatment compared with any other group. Therefore, DFO treatment appears to be a valid strategy to drive CD34-positive cells to ischemic sites.
The chemokine stromal cell-derived factor-1 (SDF-1, also known as CXCL12) is known to play a critical role in directing progenitor cells to ischemic regions (9). Activation of the SDF-1/CXCR4 system invokes mobilization and targeting of stem cells to areas of ischemia to improve neovascularization (18,45). Akt signaling is essential in the reorganization and direction of endothelial cell migration (28). The PI3K/Akt signal transduction pathway is involved in the regulation of SDF-1a-mediated migration (20,39). The PI3K/Akt signal transduction pathway plays a crucial role in SDF-1α-induced cell migration (47). Based on these findings, we further explored the effect of DFO on the SDF-1 and PI3K/Akt signal transduction pathway and confirmed that DFO treatment stabilized HIF-1α, activated SDF-1, and increased p-Akt expression in vitro (21). The PI3K/Akt inhibitor (LY294002) and CXCR4 antagonist (AMD3100) inhibited these effects. The BLI signals were enhanced by DFO and attenuated by both AMD3100 and LY294003. Therefore, we concluded that DFO mimics hypoxia by stimulating p-Akt activation, which in turn promotes the migration of CD34-positive cells toward ischemia.
DFO has also been shown to promote angiogenesis in response to ischemia (12,21). Angiogenesis is induced by increased expression of VEGF mRNA and protein secretion and is mediated via HIF-1α activation and inhibition of prolyl4-hydroxylase (3). HIF-1α is a transcriptional factor that directly activates the expression of multiple angiogenic growth factor genes (31,37,40). VEGF activates PI3K and Akt in angiogenesis (25). PI3K/Akt signaling further increases expression of HIF-1α and VEGF (24). In the present study, DFO increased both HIF-1α and VEGF mRNA levels in vitro. DFO also increased p-Akt and p-eNOS levels, which led to enhanced tube formation. LY294002 treatment suppressed endothelial cell function. In vivo, DFO not only enhanced capillary density and improved blood flow, but also reduced cellular apoptosis, necrosis, and autoamputation. Western blot results showed that DFO treatment significantly increased VEGF, p-eNOS, and p-Akt protein expression. LY294002 completely blocked these effects, in part through AMD3100. AMD3100, which is a common CXCR4 potent antagonist, has been shown to promote angiogenesis and inhibit vasculogenesis in hindlimb ischemia (7), consistent with our results. The results showed that Akt is a key signaling molecule mediating the function of VEGF and eNOS in an ischemic hindlimb. DFO treatment promoted angiogenesis and vasculogenesis via the PI3K/Akt signal transduction pathway.
DFO is widely used in the treatment of iron overload and is considered safe. DFO decreased oxygen free radicals and preserved ischemic flap survival (19). Based on in vivo results, DFO treatment may potentially improve treatment of ischemia-related diseases. However, ophthalmic, auditory, pulmonary, and renal toxicity effects have been reported. Side effects were most common in patients receiving daily doses greater than 60 mg/kg/day (41). Therefore, DFO doses in the clinical treatment of ischemia and related diseases should be strictly limited. In the future, we will investigate the toxicity of DFO dosage in ischemia and related diseases.
In conclusion, DFO stabilized HIF-1α, increased the expression of downstream genes, and promoted CD34-positive cell homing, and improved vasculogenesis and angiogenesis mediated by activation of the Akt-dependent pathway in response to ischemia.
Footnotes
Acknowledgments
We thank Eiji Kobayashi (Division of Organ Replacement Research, Center for Molecular Medicine, Jichi Medical School, Tochigi, Japan) for luciferase transgenic Lewis rats. This work was supported by the National Natural Science Foundation of China (Nos. 30730092, 30925034) and Joint Research Project of Shanghai Municipal Level Hospital for Emerging Cutting-Edge Technology (No. SHDC12010105) as well as subprojects of The National Science and Technology Support Program (No. 2012BA11B00). The authors declare no conflicts of interest.
