Abstract
Human induced pluripotent stem cells (hiPSCs) can be genetically reprogrammed to an embryonic stem cell-like state and can provide promising medical applications, such as diagnosis, prognosis, drug screening for therapeutical development, and monitoring disease progression. Despite myriad advances, traditional viral-based reprogramming for generating hiPSCs has safety risks that hinder further practical applications of hiPSCs. In the past decade, nonviral-based reprogramming has been used as an alternative to produce hiPSCs and enhance their differentiation. In addition, the efficiency of nonviral-based reprogramming is generally poor, compared to that of viral-based reprogramming. Recent studies in nanoscale-structured particles have made progress in addressing many applications of hiPSCs for clinical practice. The combination of hiPSCs and nanotechnology will actually act as the therapeutic platform for personalized medicine and can be the remedies against various diseases in the future. In this article, we review recent advances in cellular reprogramming and hiPSC-related research, such as cell source, delivery system, and direct reprogramming, as well as some of its potential clinical applications, including mitochondrial and retinal disease. We also briefly summarize the current incorporation of nanotechnology in patient-specific hiPSCs for future treatments.
Introduction
The groundbreaking discovery of cellular reprogramming by Takahashi and Yamanaka demonstrates that the enforced expression of four factors, Oct3/4, Sox2, c-Myc, and Klf-4, also known as the Yamanaka factors, can reprogram mouse embryonic fibroblasts (MEFs) to a pluripotent state in a few weeks (57). Based on the full reprogramming process, these stem-like cells were named induced pluripotent stem cells (iPSCs). Only 1 year later, human iPSCs (hiPSCs) were generated by similar transductions of defined transcription factors (56,67). This progress has opened up the possibility of autologous regenerative medicine whereby immune-matched hiPSCs could be derived from adult somatic cells (42,49). Indeed, easily accessible somatic cells reprogrammed to an embryonic state with the Yamanaka factors have recently been shown to alleviate the symptoms of disease in rodent models (6,13,32,36,45,48,55). In addition to cellular therapy, patient-specific hiPSCs may enable the establishment of genetic disease modeling and facilitate the identification of novel drugs for the treatment of these disorders, such as mitochondrial diseases or retinal diseases (18,24).
Although the iPSC technique has been widely used for decades, there are many problems that still must be addressed. First, iPSCs are pluripotent; they contribute to all three germ layers when injected into blastocytes. They also share many similarities with embryonic stem cells (ESCs) regarding cellular morphology, proliferative ability, surface antigens, gene expression patterns and profiles, epigenetic status of pluripotent cell-specific genes, and stemness signatures, as well as telomerase activity. In addition, iPSCs contribute to germ line transmission and even contribute to a more stringent tetraploid complementation (2,68). These traits of iPSCs (pluripotent potential and genetic modification) increase the risk of tumorigenesis and prohibit their usage further in clinical applications.
There are several methods of inducing pluripotency when producing iPSCs, including the use of viral vectors, nonviral vectors, small molecules accompanied by chemical treatment, and utilizing RNAs. Each method of generating iPSCs has its own advantages and disadvantages. However, the use of viral vectors that integrate into the genome run the risk of insertional mutagenesis and genomic aberrance. Recently, for iPSC generation, several nonviral-based platforms were used to deliver vectors. As an alternative, several methodological platforms have reported that use of nonviral-based vectors to deliver the cDNA or RNA materials for stimulating cellular reprogramming, including DNA or RNA by electroporation, polymer/lipid-based nanoparticles, proteins, or peptides, and gene-polymer/lipid complexes that have the ability to promote the differentiation of stem cells to specific lineages. In the past decade, nanoparticles (NPs) have been widely investigated, such as in imaging (65), targeting delivery, and drug delivery systems (11). The unique nanoscale structure of NPs provides an efficient delivery rate into adult human cells through endocytosis. In addition, because of their high surface area, NPs are capable of targeting moieties for both drug delivery and carrying therapeutic agents. The properties of NPs serve as a good carrier for various molecules or drugs. Moreover, NPs can entrap the drugs inside and protect them from degradation, while the drugs circulate in the bloodstream or in a living body.
In this article, we provide a review of recent advances in the induction of pluripotency in somatic cells with defined transcription factors and also discuss direct reprogramming. We briefly summarize current applications of nanoparticles in drug delivery, followed by the progress of recent research on patient-specific hiPSCs. We then discuss the roles of nanoparticles and hiPSCs in the processes of personalized medicine against certain diseases, such as mitochondrial and retinal disease.
Stem Cell Reprogramming and iPSCs
Stem cells, a special form of cells derived from adult tissues or embryos, are known to contain the characteristics of self-renewal, high plastic capability, and multiple lineages of differentiation. Stem cells have three types based on their abilities to differentiate. The first ranking for the definition of stem cells are “totipotent” stem cells, which can be implanted in the uterus of a living animal and give rise to a full organism. The second ranking for the definition of stem cells are “pluripotent” stem cells, such as ESCs and iPSCs. They can give rise to every cell of an organism except extraembryonic tissues. The third ranking for the definition of stem cells are “multipotent” stem cells. They belong to adult stem cells that only generate specific lineages of cells in the differentiation process. Meanwhile, adult stem cells, also known as somatic stem cells, are undifferentiated cells that can differentiate to some or all of the major specialized cell types of the tissue or organ in the living body. Wernig et al. have demonstrated that transplantation of iPSC-derived neuronal cells into the brain can improve symptoms in a rat model of Parkinson's disease (61). Previous studies have shown that subdural transplantation of iPSCs with fibrin glue leads to functional improvement in focal cerebral ischemia in rats (8). Although the therapeutic potential of iPSCs in transplantation has been demonstrated in animal models (8,61), the mechanisms underlying these therapeutic effects are not well understood.
Direct Reprogramming
Direct reprogramming to transdifferentiate somatic cells into another cell type without producing the intermediate pluripotent stem cell stage has been drawing attention in biomedical research. Traditional iPSC reprogramming techniques through a viral vector have several difficulties that hinder many practical applications. For example, permanent genome modification of reprogrammed somatic cells by exogenous genes and even fragments from delivering vectors could be very harmful. Recent progress has been made in overcoming these safety concerns by using nonintegrating gene delivery approaches or reprogramming proteins (21). Also, the process of reprogramming induced by the four Yamanaka factors generally entails a stepwise progression of nonspecific events that give rise to pluripotent cells as one of many possible destinations (38), resulting in poor efficiency and slow kinetics.
Recently, direct transdifferentiation in vivo was achieved with ectopic expression of lineage-specific transcription factors (TFs) in situ. Similar to the process of iPSCs, transdifferentiation induced by somatic lineage-specific TFs is slow and inefficient. Some studies (31) have revealed that the efficiency of Ascl1/Ngn2-induced neuronal conversion of human fibroblasts could be enhanced by the combination of three small molecules that inhibit transforming growth factor-β, glycogen synthase kinase-3, and bone morphogenic protein (BMP) receptors. This study demonstrated direct conversion of fibroblasts to cardiac, neural, and endothelial tissue using transient expression of iPSC TFs followed by treatment with BMP4 (14). Other studies revealed that induced dopaminergic (iDA) cells were able to demonstrate spontaneous electrical activity organized in regular spikes consistent with pacemaker activity of brain dopaminergic neurons (4). Direct conversion of iDA cells from somatic cells might have a significant role in the understanding of critical processes of neuronal development, disease modeling, and cell replacement therapies (4).
The random nature of the iPSC reprogramming process may present “hidden” risks for iPSCs, such as residual epigenetic memory, inappropriately induced epigenetic changes, plus accumulation and selection of other subtle genetic and epigenetic abnormalities during the reprogramming process. Several small molecules that replace exogenous reprogramming transcription factors have been used as an alternative approach to address the above complications (15).
iPSC Reprogramming From Human Blood
The induction of iPSCs from human peripheral blood cells was first reported by Loh et al. in 2009 (35). After a 3-day injection of granulocyte colony-stimulating factor, the authors used retroviral vectors for gene delivery and infected the granulocyte colony into mobilized CD34+ blood cells isolated from a donor. This method is relatively labor intensive and places a major burden on the donor, thus making it difficult to be used as a conventional tool. In addition, retrovirus integration disrupts the endogenous genomic organization and might affect the disease modeling in vitro. Some recent studies revealed that human keratinocytes isolated from plucked hair can be reprogrammed to iPSCs (1). However, it is still unclear whether hair cells will be a good source for reprogramming because the quality and growth of the hair follicles are dependent on the age, genotype, and medical conditions of the human donors (3,29). Reprogramming iPSCs from human blood cells represents a better way of establishing iPSCs from donor cells because it requires little manipulation time in culture. Besides, peripheral blood is the original source of cells for immunotherapy, hematopoietic transplantation, and gene therapy (47). Induction of iPSCs from human blood cells is an attractive choice. This is not only advantageous of the large collections of umbilical cord blood stored in cord blood banks, but is also much less invasive (Fig. 1).
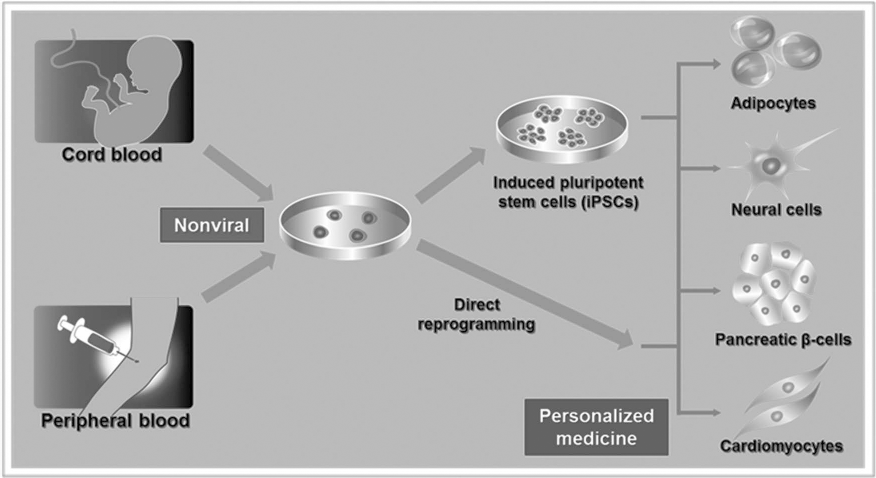
Strategies for iPSCs and direct reprogramming.
Recent success of episomal plasmid-based reprogramming of human blood cells (10,22) and a series of viral vector-based blood cell reprogramming (35,64) has significantly advanced iPSC technology. However, iPSCs have not been derived from Epstein–Barr virus-immortalized B-cell lines, an important source of genetic information of patients (and their family members) with various diseases, including a few inherited diseases (46,63). Blood cells represent a novel source of cells, and the use of blood cells replaces the need for skin biopsies and requires minimal maintenance in culture before reprogramming.
To reprogram blood cells is a very important step for the development of efficient ways of generating stem cells with patient-specific pluripotency.
Delivery System of iPSCs
There are several methods of inducing pluripotency in cells, including using viral vectors, nonviral vectors, small molecules accompanied by chemical treatment, and RNAs. Each method of producing iPSCs has its own advantages and disadvantages. Many studies have shown the use of viral-based vectors to deliver target genes into somatic cells that are reprogramming for enhanced lineage specification on viral vectors (16). There are many viral vectors, such as retroviruses, lentiviruses, and adenoviruses, that have been used to reprogram somatic cells to generate iPSCs (69). Some studies revealed that highly efficient gene transfer and long-term expression can be accomplished by viral vectors, but critical safety risks such as tumorigenicity due to insertional mutagenesis and uncontrolled gene expressions have been reported (43). Thus, nonviral delivery systems have been studied as another approach to reprogram cells and enhance their differentiation. Gonzalez et al. revealed that the delivery of plasmid DNA or RNA [such as, small interfering RNA (siRNA), microRNA (miRNA)] with electroporation or lipid-based NPs has been shown without vector and transgene integration into the host genome (17). In spite of the advantages of nonviral delivery systems, the delivery efficiency in nonviral vectors is generally less than that in viral vectors (20). Therefore, it is critical to improve the efficiency of nonviral delivery systems for reprogramming. In the following sections, we will focus on discussing the recent advances in nonviral delivery for iPSC generation and direct conversion; however, a brief introduction of viral methods will be given below in the A Viral Revolution of iPSCs section.
A Viral Revolution of iPSCs
Since the first successful establishment of human iPSCs from human skin dermal fibroblasts via viral transduction of the four Yamanaka factor genes in 2007, viral vectors such as lentiviruses, retroviruses, and adenoviruses have been achieved for gene delivery of these transcription factors to generate iPSCs. However, the use of viral vectors to integrate into the genome has some risks, including insertional mutagenesis and uncontrolled expression of exogenous factors. These risks significantly limit the utility of iPSCs in clinical settings. Therefore, for iPSC generation, several nonviral delivery vectors have recently been used as alternatives to viral vectors.
Nonviral/Nonintegrating Delivery of iPSCs
Nonviral delivery is a promising method for reprogramming to produce specific cell types from terminally differentiated somatic cells or undifferentiated stem cells. The genes of nonviral delivery, such as DNA (e.g., episomal, minicircle) or RNA (e.g., siRNA, miRNA) by electroporation or polymer/lipid-based NPs, proteins, or peptides, and gene-polymer/lipid complexes have the abilities to promote the differentiation of stem cells to specific lineages, and to reprogram somatic cells to pluripotent stem cells or other lineage cells. In 2008, Okita et al. (40) showed that pluripotency could indeed be achieved through nonintegrating viral methods. Strategies for non-viral reprogramming will be discussed below.
DNA Delivery
Episomal Vectors
The delivery of episomal plasmids can be transfected without viral packaging. First developed by Yu et al. (66), the episomal iPSC reprogramming vectors have proven successful in reprogramming many different somatic cell types. Subsequently, Yu et al. revealed that episomal plasmids were removed from cells even without drug selection. (66). Yu et al. revealed that a single transfection of three plasmids expressing the seven reprogramming factors led to generation of human iPSCs without transgene integration into the host genome in their reprogramming system. Generally, DNA vectors are more stable than viruses and can be refrozen through several usages, but the efficiency is very low (66).
Minicircle Plasmid Vectors
Minicircle plasmid vectors are one type of episomal DNA vector produced as a circular expression cassette devoid of any bacterial plasmid DNA backbone. Recently, the delivery of nonviral minicircle DNA vectors was used for iPSC generation. The delivery system is based on supercoiled DNA molecules and is primarily composed of a eukaryotic expression cassette without bacterial DNA sequences (26). The reprogramming efficiency is the same as typically reported for viral delivery, but the minicircle vectors were introduced into human iPSCs by electroporation (26). Since reprogramming requires a continuous dose of minicircle DNA, the other groups later chose to use lipofectamin for subsequent delivery of the minicircle DNAs, which may have caused lower delivery efficiency.
Transposons
Transposons are mobile genetic elements with the capacity to “jump” to new target DNA. Originally discovered by McClintock, they are present in DNA genomes of species from all kingdoms (37). Transposons can be utilized as another efficient and safe tool for iPSC generation. Woltjen et al. showed the first successful application of the nonviral transposon and transposase system to reprogram embryonic fibroblasts into iPSCs (62), although others have used another type of transposon system for iPSC reprogramming of fibroblasts (30). Other disadvantages, such as variable transfection efficiency, have been mentioned in previous work.
RNA Delivery
Warren et al. first described a new methodology: using synthetic mRNA for the efficient generation of iPSCs without compromising genomic integrity (60). Other groups also reported using miRNA delivery for direct reprogramming (25). These studies identified a combination of miRNAs, namely miRNAs 1, 133, 208, and 499, that promote direct conversion of fibroblasts into cardiomyocyte-like cells in vitro (25). Some other studies also supported evidence of the principle that transfection of synthetic and modified RNA can result in cellular reprogramming to a pluripotent state and specialized cell types, but they have low and variable transfection efficiency (60).
Nanoparticle-Mediated Delivery
NP-mediated gene transfection has been used to improve the efficiency of iPSC reprogramming. For example, calcium phosphate-based nanoparticles were applied to deliver plasmid encodings of the Yamanaka factors into human umbilical cord mesenchymal stem cells. Moreover, the delivery system of polymer-based gene carriers has been thought to be potentially safer than viral-mediated delivery (58). The polymer complexes can protect DNA from nuclease degradation. In addition, these nanoparticles are small enough to enter the cell by endocytosis. However, cytotoxicity and nonbiodegradability might be harmful in long-term safety considerations for certain polymer-based carriers. Polyurethanes (PUs) are used as biomaterials in tissue engineering and in hydrogels because of their biocompatibility and physiochemical properties (19,53,59). Cationic polyurethane short branch polyethylenimine (PU-PEI) has a higher transfection efficiency with lower cytotoxicity (23,34). Thus, it is important to find out whether such nonviral vectors will be helpful in facilitating in vivo gene delivery in intractable malignant cancers, such as glioblastoma. Currently, the delivery efficiency of nonviral methods still needs to be improved for clinical applications. Combining the use of bioinspired strategies and biomaterial libraries to establish functional biomaterial development is expected to facilitate the establishment of nonviral delivery systems.
Nanomedicine
It is already well established that reprogramming somatic cells to iPSCs can be achieved by the delivery of a combination of the Yamanaka factors. In the past two decades, retroviral and lentiviral vectors have been widely utilized to express these four Yamanaka factors separately to acquire reprogrammed iPSCs. Unfortunately, despite the efficiency and reproducibility, these methods are time consuming and labor intensive. Most importantly, these approaches are with potentially harmful viruses overexpressing potent oncogenes, such as c-Myc. Therefore, scientists in the past few years have been focusing on generating iPSCs using nonviral methods, in particular, employing nanoparticulated vectors (Fig. 2). In the following sections, we will review some of the most up-to-date nanoparticulated delivery system-based applications on iPSC research and summarize these compelling works in Table 1.

Recent nonviral cell reprogramming techniques using nanotechnology-based vectors.
The Up-to-Date Nanoparticulated Delivery System-Based Applications on iPSC Research and a Summary of These Compelling Works

Liposomes
Park et al. employed liposomal magnetofection (LMF) for iPSC generation (44). Briefly, ternary complexes of cationic lipids self-assembled with plasmid DNA (pCX-OKS-2A and pCX-cMyc) associated with superparamagnetic iron oxide nanoparticles were prepared. Park and coworkers performed gene transfection by applying a magnetic field to concentrate CombiMag-DNA lipoplexes onto MEF cells. An integration-free LMF-iPSC line was obtained, and it displayed pluripotency both in vitro and in vivo, including teratoma formation and chimeric mouse production. Moreover, the previous studies have shown that “CombiMag-DNA lipoplexes” for the transfection of MEF cells was safe, proven by lactate dehydrogenase activity assay and transmission electron microscopy. Therefore, LMF might be an effective and safe procedure for nonviral, integration-free reprogramming of iPSCs for future clinical applications, once the transfection efficiency is further improved.
Polymeric Nanoparticles
In spite of safety concerns, viral delivery is still the most efficient method for the generation of iPSCs. Transfection is a common process used for delivery of nucleic acids into cells. As mentioned above, PLGA nanoparticles are both biodegradable and biocompatible and can be a good cargo-loading carrier for targeted drug delivery. However, the low electrostatic interaction between PLGA and nucleic acid hinders the use of PLGA nanoparticles on DNA delivery or gene therapy. On the other hand, the positively charged amine groups of branch-chained polyethyleneimine (bPEI) contribute to high cationic-charge-density of the polymer and make bPEI an ideal carrier for nucleic acid. Seo and coworkers developed PLGA/bPEI NPs in transfection and virus production (52). The addition of positively charged bPEI can interact with the negatively charged nucleic acid and increase the absorption of nucleic acid onto the PLGA/bPEI nanoparticles. In addition, bPEI can further interact with the negatively charged cell membrane and facilitate nucleic acid delivery by rupturing the endosome before it reaches the lysosome through the socalled proton sponge mechanism. From Seo's study, PLGA/bPEI-DNA (w/w ratio 6:3:1) NPs demonstrated a better transfection efficiency compared to liposome–DNA complexes. Moreover, the culture supernatants of HEK293FT cells transfected with PLGA/bPEI-DNA of viral reprogramming factors (Oct4, Sox2, Klf4, or c-Myc) produced iPSC colonies from mouse embryonic fibroblasts in a more efficient manner compared to the liposome–DNA (52). However, although these results suggest that PLGA/bPEI-DNA nanoparticles have advantages in DNA delivery in various applications, such as in reprogramming or direct conversion, the use of viral DNA is still questionable and with major clinical issues. Therefore, a nonviral reprogramming system utilizing PLGA/bPEI with small molecules, such as miRNAs, may be beneficial in future iPSC research.
Numerous nonviral methods, including protein, mRNA, miRNA, and small molecule transduction, were established to prevent genomic integration and generate stem cell-like cells from mouse and human fibroblasts. Sohn et al. created microRNA-loaded (miR 302 family and 367) NPs using the polyketal polymer PK3 (PK3-miR) and delivered them to mouse bone marrow-derived hematopoietic cells. Interestingly, in just 8 days, the formation of colonies was found with considerable induction of the pluripotency markers: Oct4, Sox2, and Nanog. In addition, pluripotency markers, such as SSEA-1, were positively stained after the colonies were transferred to feeder layers. Their reprogramming results indicated that a polyketal-microRNA delivery platform might be used to produce assorted reprogrammed cells without long-term genetic manipulation (54).
Proteins, especially TFs, can modulate the gene expression of the host cells leading to complete transformation of the parental phenotype (28). Kahn et al. used cationic amphiphiles as the nonviral vehicle for protein delivery and reprogramming of iPSCs. After optimization, the cationic bolaamphiphile–protein complex ratio was set at 7:1 (w/w), and the incubation period was set for 3 h with human fibroblasts in the reprogramming procedure. The reprogrammed cells displayed the characteristics of embryonic stem cells, such as the expression of pluripotent markers, formation of teratoma in SCID mice, and capability of differentiation into a specific lineage (i.e., neuronal differentiation). Moreover, the produced iPSCs by cationic bolaamphiphiles–protein showed a similar property to human ESCs or iPSCs generated by traditional viral transduction.
Montserrat and coworkers generated bona fide hiPSCs from human fibroblasts via poly-β-amino esters as the transfection vector for the delivery of a single CAG-driven polycistronic plasmid expressing Oct4, Sox2, Klf4, c-Myc, and a GFP reporter gene (OSKMG) (39). The use of poly-β-amino esters achieved remarkably enhanced transfection efficiency, compared to the conventional transfection reagents. Unfortunately, in spite of the successful reprogramming capability and efficiency of polycistronic plasmid carrying poly-β-amino esters, integrated transgenes were found in the clones of hiPSCs. Therefore, further studies are needed to evaluate the feasibility of using poly-β-amino ester-plasmid as a nonviral vector in the near future.
Cao et al. used calcium phosphate NPs for the generation of virus-free iPSCs from human umbilical cord mesenchymal stem cells by codelivery of the four plasmids (Oct4, Sox2, Klf4, and c-Myc) (5). The reprogramming efficiency was comparable to traditional viral transduction (about 0.049%). In addition, the iPSCs expressed pluripotency markers: OCT4, SSEA-3, SSEA-4, Nanog, and TRA-1-81. Similar to conventional viral approaches, the iPSCs were capable of differentiating into all three germ layers in vitro. Moreover, subcutaneous injection of the iPSCs into immunocompromised mice brought about the formation of teratomas containing a variety of tissues from all three germ layers. Thus, the codelivery of the four Yamanaka factors through plasmid-encapsulated calcium phosphate nanoparticles was able to produce virus-free iPSCs. This is vital for future regenerative medicine research in the clinical aspect.
Mesoporous Silica Nanoparticles
Chen et al. developed a nonviral gene delivery system using MSNs to quickly produce iPSC-derived definitive-lineage cells. MSNs with positive, neutral, and negative surface charges were generated and compared for their relative cell uptake property. Chen et al. further demonstrated that MSNs could present the practicability and efficiency of FITC-conjugated MSNs (FMSNs) for labeling of iPSCs. Interestingly, iPSCs were able to take up MSNs via energy-dependent endocytosis in the order of MSNs+ > MSNs (neutral) > MSNs- without cytotoxicity. In addition, the pluripotent status of the tested iPSCs remained the same. Moreover, while positively charged FMSNs were delivered with hepatocyte nuclear factor 3β (HNF3β) plasmid DNA (pDNA), the treated iPSCs showed significantly enhanced definitive endoderm formation and rapidly differentiated into hepatocyte-like cells with mature functions in 2 weeks. Chen's results demonstrated the advantages of FMSNs as ideal vectors for stem cell labeling and as a safe gene delivery system that can facilitate the generation of hepatocyte-like cells from iPSCs (9).
Patient-Specific iPSCs for Personalized Medicine
Recently iPSC technology has successfully generated hiPSCs by forced expression of TFs in somatic cells, thus providing new hope for regenerative medicine and disease modeling in vitro (7,51). Several studies have been done to generate iPSCs from patients with serial diseases (12). Some of the studies report that patient-specific iPSCs have been differentiated into neural crest precursors, motor neurons, and mature hepatocytes (13). These previous data revealed that hiPSCs could be used to model the specific pathogenesis of a genetically inherited disease, to perform cell replacement therapy, and to search for candidate drugs. In recent decades, several studies have focused on hiPSCs. iPSCs can propagate infinitely and maintain the ability to differentiate into many different cell types of the human body (27). Moreover, the high consistency, purity, and expandability of hiPSCs can be utilized for drug screening, toxicity testing, and the development of personalized medicine for degenerative diseases (51). Furthermore, direct reprogramming can be used for autologous transplantation without the risk of rejection and allows the creation of patient-specific pluripotent stem cells, avoiding the ethical concerns raised by using ESCs, and are considered candidates for cell therapy. Recently, some studies have developed the use of disease-specific hiPSCs from neurodegenerative disorder patients for treatment phenotypes. These patient-specific iPSCs and their differentiated progeny have offered models for particular individualized disease phenotypes. They can be used to understand disease mechanisms, such as neurodegenerative diseases, mitochondrial diseases, and retinal diseases, and investigate the pathogenesis of disease-causing mutations (50).
Personalized medicine is an emerging medical practice that proposes the customization of health care for the prevention, diagnosis, and treatment of disease. Importantly, personalized medicine can cater to individual patients based on the patient's unique genetic profile (33). In recent years, quite a few studies have been concentrating on hiPSCs due to their ability to propagate infinitely while still maintaining the ability to differentiate into a variety of cells of the human body (27). The purity, high consistency, and expandability of hiPSCs will greatly help drug screening, toxicity testing, and the development of personalized medicine for the treatment of degenerative diseases (51). Patient-specific iPSCs and their differentiated progeny can afford the individual models for particular individualized disease phenotypes that are useful in understanding mechanisms in the diseases and investigating the pathogenesis of disease-causing mutations (50).
The integration of nanomaterials and biology has influenced modern nanomedicine, especially toward personalized medicine (41). However, there is still a gap between nanotechnology and iPSCs. Although NP-based drug delivery systems have been widely studied, not much attention has been given to the application of NPs in patient-specific iPSCs or patient-specific drug screening. Notwithstanding, due to the nanoscale structure and large surface area, we consider that NPs can be a potential tool in both reprogramming of disease-specific iPSCs and the follow-up drug screening process performed on the obtained disease-specific iPSCs. For instance, MSNs have high surface area and exceptional biocompatibility that could provide a large amount of drug loading, which makes MSNs a suitable platform for efficient drug screening. The manner in which NPs interact with iPSCs is still not fully understood. Formerly, Chen and coworkers confirmed that a plasmid-carrying MSN system (pHNF3β-FMSN+) was able to enhance iPSC differentiation toward hepatocyte-like lineage with mature liver function, and this doubled delivery of pHNF3β-FMSN+ further improving mRNA expression levels of liver-specific genes (9). In addition, MSNs with various surface charges (negative, neutral, or positive charge) could be efficiently internalized by iPSCs without causing cytotoxicity. Accordingly, MSNs could be a possible candidate for biomolecule delivery and serve as a good vector for stem cell labeling, gene delivery, as well as a potential drug carrier for inducing patient-specific differentiation and subsequent personalized therapeutics.
Conclusions
In this article, we have provided an account of studies to show that adult stem cells can differentiate into some or all major specialized cell types of the body. Progress in research and clinical applications using adult stem cells is still being made, but there are still several unsolved problems, such as (i) inaccessibility (bone marrow-derived mesenchymal stem cells) and (ii) differentiation efficiency (e.g., differentiation into neural progenitor cells or pancreatic progenitor cells). Recent progress in iPSC research opened up an era of stem cell biology. These iPSCs are derived from somatic tissues and are reprogrammed from exogenous genes. They are pluripotent and able to differentiate into tridermal lineages. iPSC research has been able to avoid several disadvantages of ESCs (e.g., ethical issues and invasive operation procedures) as well as adult stem cells (multipotency and inaccessibility in some kind of adult stem cells). Nevertheless, the incidence of in vivo teratoma formation has been a major unresolved problem for hiPSC-based transplantation (8). Research on patient-specific hiPSCs provides one new strategy in personalized medicine. Patient-specific iPSCs and NP-based medicine have become very important components of personalized medicine. Moreover, the genetic information available through hiPSC disease phenotypes could be used to predict disease progression and clinical prognosis. Researchers have also developed many successful NP-based drug delivery systems as a treatment for several neurodegenerative diseases. Therefore, the utility of hiPSC models in combination with nanoparticle medicine could help elucidate further pathological mechanism and progression and ultimately be applied to personalized therapeutic development.
Footnotes
Acknowledgments
The authors declare no conflicts of interest.
