Abstract
The main challenge of cell transplantation as a treatment of myopathies is the large amount of tissue to treat. Intravascular delivery of cells may be an ideal route if proven to be effective and safe. Given the importance of nonhuman primates for preclinical research in transplantation, we tested the intra-arterial injection of β-galactosidase (β-Gal)-labeled myoblasts in macaques. Cells were injected into one of the femoral arteries in seven monkeys. Some muscle sites were damaged concomitantly in three monkeys. Various organs and muscles were sampled 1 h, 1 day, 12 days, 3 weeks, and 5 weeks after transplantation. Samples were analyzed by histology. Most β-Gal+ cells were observed in the capillaries and arterioles of muscles and other tissues of the leg homolateral to the cell injection. Groups of necrotic myofibers in the proximity of an arteriole plugged by a β-Gal+ embolus were interpreted as microinfarcts. Scarce β-Gal+ cells were observed in the lungs 1 h and 1 day posttransplantation. No β-Gal+ cells were observed in other organs or muscles. β-Gal+ myofibers were observed 12 days, 3 weeks, and 5 weeks after transplantation in muscles of the leg after the cell injection, in sites that were damaged at the time of cell injection. In conclusion, most intra-arterially injected myoblasts were retained in vessels of the leg homolateral to the cell injection site, and they fused with myofibers in regions in which there was a process of myofiber regeneration. This manuscript is published as part of the International Association of Neurorestoratology (IANR) special issue of Cell Transplantation.
Keywords
Introduction
Transplantation of cells competent to differentiate into myofibers (hereafter, myogenic cells) is a potential therapeutic tool for myopathies of genetic recessive origin. The principal property sought in these cells is the ability to fuse with myofibers of the recipient to form hybrid myofibers, that is, myofibers with myonuclei from recipient and donor origin and synthesizing proteins from both. Thus, hybrid myofibers can synthesize proteins whose genetic deficiency in the recipient causes a myopathy, like dystrophin in Duchenne muscular dystrophy (14,27,28,32).
The challenge of cell transplantation in myopathies is important given the volume of tissue to be treated; skeletal muscle contributes 40% to 50% of body weight(13). Two routes are possible for cell administration: intramuscular (IM) and intravascular. IM has been so far the most widely used for myogenic cell transplantation, but has complex technical challenges (33). The intravascular route could have potential advantages, the most obvious being that cells could be delivered to regions that cannot be easily reached by IM injections.
There are two possibilities for intravascular delivery: intravenous (IV) and intra-arterial (IA). The IV route has easier access, but there is the disadvantage that the cells first find a barrier in the capillary bed of the lungs, and those that pass this filter are indiscriminately distributed throughout the body. Conversely, IA injection could convey the cells to the required region. A few experiments in rodents showed far better results with the IA route than with the IV. Myoblasts engrafted into damaged muscles of rats after IA delivery (16), while IV delivery in mice was unsuccessful even after extensive muscle injury (17). IV delivery of muscle side population cells in mice led to <1% of hybrid myofibers versus 5–8% after IA delivery (1). Thus, the IA becomes the preferred route for the intravascular delivery of other myogenic cells in mice (22,35) and dogs (20,21).
The IA delivery of myogenic cells has never been tested in nonhuman primates, which are the most rigorous animal model in preclinical transplantation research. The close phylogenetic relationship between human and nonhuman primates provides the best anatomical, physiological, antigenic, and immunological similarities for preclinical transplantation research (9,10,19). Therefore, given our experience with myogenic cell transplantation in nonhuman primates, we wanted to see in this model whether:
the IA infusion of myogenic cells is feasible;
the injected cells remain in the skeletal muscles downstream from the injection;
the injected cells first pass the capillary filter and are retained in other organs;
there is any problem with the animal's health;
the injected cells are able to form hybrid myofibers.
Given our extensive experience with myoblast transplantation, we used these cells for this first approach. Furthermore, only myoblasts are proven so far to engraft in skeletal muscles of nonhuman primates (33) and, most importantly, in humans (14,27,28). We report here the observations made in seven monkeys, four of which were used for an early posttransplantation analysis and three for long-term analysis.
Materials and Methods
Animals
Six cynomolgus monkeys (Macaca fascicularis, two females and four males, 3 to 8 years old) and one rhesus monkey (Macaca mulatta, male, 5 years old) were used as receivers of myoblast transplantations. For transplantation and biopsies, the animals were kept under general anesthesia using isofluorane (1.5-2% in oxygen; Abbott Laboratories, Montreal, QC, Canada) after induction with IM ketamine (10 mg/kg; Bioniche, belleville, ON, Canada) and glycopyrrolate (0.05 mg/kg; Sandoz Canada, boucherville, QC, Canada). Buprenorphine (0.01 mg/kg b.i.d. for 3 days; Champion Alstoe, Whitby, ON, Canada) was given for postoperative analgesia. Euthanasia was done at the end of the experiments by IV administration of a pentobarbital sodium overdose (120 mg/kg; Euthanyl; Bimeda-MTC, Cambridge, ON, Canada) after anesthesia using IM ketamine (15 mg/kg). The Laval University Animal Care Committee authorized these procedures.
Cell Culture
We used frozen cells corresponding to two cell lines obtained in our laboratory from skeletal muscle samples of two additional female macaques (3 and 4 years old) and transduced with the LacZ (β-galactosidase) reporter gene as described (26). Only one cell line was injected per animal. The percentage of cluster of differentiation 56-positive (CD56+) cells (used as an indicator of the percentage of myoblasts in the cell lines) was analyzed in a sample of cells by flow cytometry. These cells were incubated with a phycoerythrin-coupled anti-CD56 antibody (1/40; Beckman Coulter, Fullerton, CA, USA) and analyzed in a Coulter Epics XL flow cytometer (Beckman Coulter). The percentage of CD56+ cells in the two cell lines was 91% and 99%, respectively (Fig. 1).
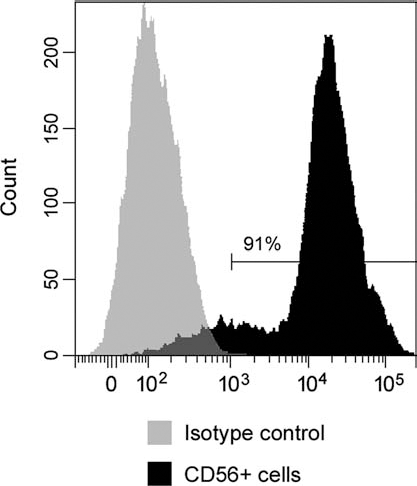
Flow cytometry analysis of myoblasts in cultured cells. Flow cytometry analysis performed in the cultured cells to determine the percentage of CD56+ cells (used as an indicator of the percentage of myoblasts).
After thawing, cells were proliferated in vitro during two passages in Molecular, Cellular, and Developmental Biology-120 (MCDB-120) culture medium (7) with 15% fetal bovine serum (FBS; Hyclone, Logan, UT, USA), 10 ng/ml of basic fibroblast growth factor (Feldan, St-Laurent, QC, Canada), 0.5 mg/ml of bovine serum albumin (Sigma, St. Louis, MO, USA), 0.39 μg/ml of dexamethasone (Sigma), and 5 μg/ml of human insulin (Sigma). For transplantation, cells were detached using 0.1% trypsin (Gibco, Grand Island, NY, USA) and washed three times with Hank's balanced salt solution (HBSS; Gibco). The final cell pellets were resuspended in HBSS and transplanted by IA cell infusion or IM injection as described below. A sample of cells was used in each case to verify the cell viability with a trypan blue exclusion method. For this, we added 0.1 ml of a 0.4% trypan blue (Sigma) solution to 1 ml of the cell suspension, which was loaded in a hemocytometer and examined immediately under an inverted microscope (Bausch & Lomb Place, Rochester, NY, USA).
IA Cell Infusion
The cells were resuspended in HBSS with 50 U/ml of heparin sodium (LEO Pharma, Thornhill, ON, Canada) and injected slowly in one femoral artery, at the region of the Scarpa's triangle, using disposable syringes with 27 to 30 gauge × 0.5-inch needles (Terumo, Somerset, NJ, USA). The amount of cells injected and other quantitative details of the IA cell infusion are shown in Table 1. In the first experiments, we began with an amount of cells in the range of 1 × 108. As we observed that these amounts of injected cells were well tolerated in the short term, we increased them for the long-term follow-ups (Table 1). We also increased the volume of cell suspension during the study (Table 1).
Quantitative Details of the IA Cell Infusion in Each Monkey of the Study

The cell viability was verified with a trypan blue exclusion test before transplantation.
Rhesus macaque; the other six are cynomolgus.
IM Cell Transplantation
A muscle site of about 1 cm3 was grafted IM with the same cells used for IA infusion in the monkeys of the long-term follow-up. This was a control for cell engraftment and acute rejection. Cell transplantation was done percutaneously using a matrix of about 100 parallel equidistant injections per cm2 perpendicular to the muscle surface. We used a 250-μl Hamilton syringe attached to a PB600-1 repetitive device (Hamilton, Reno, NV, USA) (29) with 27 gauge × 0.5-inch needles (Terumo), delivering 5 μl of cell suspension in each injection. Approximately 20 × 106 cells were delivered per site. An OpSite sterile transparent dressing with a 5-mm grid (Smith & Nephew, Hull, UK) was adhered to the skin to control the pattern of needle penetrations. Each grafted site was identified by placing two points of Prolene® 5.0 (Ethicon, Inc., Somerville, NJ, USA) on the muscle surface, through small skin incisions, at ~5 mm from each side of the cell-grafted region.
Muscle Damage
This was performed in the three monkeys of the long-term follow-up. The damage was carried out during the same procedure of the IA infusion of cells. It was done in muscle regions of about 1 cm3, both in the quadratus femoris and gastrocnemius of the leg homolateral to the injection. Muscle damage consisted of 100 parallel percutaneous IM penetrations with a 27-gauge needle of 0.5-inch length, forming a matrix of 1 cm2 (similar to IM cell transplantation). As for IM transplantation, we used an OpSite transparent dressing to control the pattern of needle penetrations, and each site was identified by placing two points of Prolene® 5.0.
Immunosuppression
Monkeys in the long-term follow-up were immunosuppressed with an IM formulation of tacrolimus (a generous gift from Astellas Pharma, Inc., Osaka, Japan), beginning before transplantation and maintained until the end of the experiment. Tacrolimus was injected IM once a day. We began tacrolimus administration using 0.5 mg/kg/day, and then we adjusted the dosage to target tacrolimus blood levels of >40 μg/L. Tacrolimus blood levels were quantified in blood samples with an IMx tacrolimus II kit for microparticle enzyme immunoassay (Abbott, Wiesbaden, Germany). Dexamethasone (Omega, Montreal, QC, Canada) was injected IM once a day at 0.25 mg/kg/day to stimulate the appetite of monkeys, which is decreased during the first days of tacrolimus administration.
Sampling
Monkeys were submitted to euthanasia 1 h (n = 1), 1 day (n = 3) and 12 days (n = 1), and 3 (n = 1) and 5 weeks (n = 1) posttransplantation. The monkey euthanized at 12 days was planned for a follow-up of 5 weeks but developed severe thrombocytopenia, and the veterinary service decided to perform euthanasia at that time. This was one of the two monkeys that received the highest volume of cell suspension (10 ml), which was in heparin solution. Thus, it was a possible case of heparin-induced thrombocytopenia (8). This circumstance will be taken into account in future experiments. Given the number of monkeys available for the long-term experiments in this study, this animal could not be replaced.
Samples were taken from skeletal muscles of the homolateral and contralateral (in reference to the IA injection) posterior limbs and in the arms. Other tissues of the limb homolateral to the IA injection (sole skin, sciatic nerve, and lymph node) were sampled in one monkey 1 day posttransplantation. Several organs were also sampled 1 h (lungs) and 1 day posttransplantation (lungs, heart, brain, cerebellum, gut, liver, spleen, kidneys; n = 3). The tissue samples were mounted in Shandon™ Cryomatrix™ embedding medium (Thermo Scientific, Kalamazoo, MI, USA), snap frozen in liquid nitrogen, and stored at −80°C until sections of 10 to 15 μm were made in a cryostat (Thermo Scientific) at −25°C.
Histological Techniques
Sections were stained routinely with hematoxylin (Laboratoire Mat, Quebec, QC, Canada) and eosin (Fisher Scientific, Hampton, NH, USA) (H&E). For histochemical detection of β-galactosidase (β-Gal), sections were fixed 3 min in 0.25% glutaraldehyde (Sigma), rinsed with phosphate-buffered saline (PBS; Sigma), incubated 24 h at room temperature in a solution containing 0.4 mM X-gal (5-bromo-4-chloro-3-indolyl-b-d-galactopyranoside) (Boehringer Mannheim, Vienna, Austria) from a 20-mg/ml stock in dimethylformamide, 1 mM magnesium chloride, 3 mM potassium ferrocyanide (all from J. T. Baker Chemicals, Phillipsburg, NJ, USA), and 3 mM potassium ferricyanide (Sigma) in PBS, and mounted in 1:1 glycerin jelly (Sigma). The membrane attack complex (MAC) of the complement system was immunodetected to identify potential myofiber necrosis (5). MAC was detected with a mouse anti-human/baboon C5b-9 monoclonal antibody (1/100; Abcam, Cambridge, MA, USA), followed by incubation with a biotinylated goat anti-mouse IgG antibody (1/150; Dako, Copenhagen, Denmark) and 30-min incubation with streptavidin-Cy3 (1/700; Sigma). Antibodies and streptavidin were diluted in PBS, pH 7.4, with 1% FbS. Incubations were at room temperature. As positive controls, we used muscle samples of monkeys with myofiber damage in which we previously detected MAC+ myofibers. The tissue sections were analyzed using an Axiophot microscope with epifluorescence and bright field optics (Zeiss, Oberkochen, Germany), and pictures were taken with a Canon A650 IS digital camera (Canon, Tokyo, Japan).
Results
The results are divided into two groups, according to the time after transplantation. First, the immediate period (1 h and 1 day posttransplantation) was designed to observe where the IA-injected cells were retained. Within this period, we follow the sequence of the vascular filters encountered by the injected cells, that is, (a) the leg homolateral to the injection, (b) the lungs, and (c) the rest of the body. Second, the intermediate period was designed to observe whether the injected cells gave rise to hybrid myofibers.
Immediate Postinjection Period (1 H or 1 Day After IA Injection)
Leg Homolateral to the IA Injection
β-Gal+ cells were observed in skeletal muscle samples taken in the leg homolateral to the IA injection (Fig. 2). These β-Gal+ cells were seen either as isolated cells or in clumps of varying sizes. β-Gal+ cell clumps were observed in arterioles (Fig. 2A, B). Isolated β-Gal+ cells were observed in the endomysium, in close contact with myofibers, a location that corresponds with capillaries (Fig. 2C, D).

Cross-sections of skeletal muscles of the leg homolateral to the IA injection of β-galactosidase (β-Gal)-labeled myoblasts. Sections are stained with H&E, for histochemical detection of β-Gal (blue-greenish stain) and for fluorescent immunodetection of the MAC of complement. (A–D) Gastrocnemius, 1 h posttransplantation. (A) An accumulation of β-Gal+ cells is indicated between arrowheads and an isolated β-Gal+ cell with an arrow. As seen in the serial section stained with H&E (B), the accumulation of β-Gal+ cells is located in an arteriole (between arrowheads). The region in which the section is tangential to the wall of the vessel is shown at higher magnification in the inset to show the closely packed and circumferentially arranged nuclei of smooth muscle cells, typical of arterioles. The isolated β-Gal+ cell indicated with an arrow in (A) is shown at higher magnification in (C) (arrowheads). The contour of the vessel (a semilongitudinal section) can be observed by diffraction in (C). The arrow indicates what seems to be an accumulation of small blood cells probably upstream to the plugged myoblast (they resemble erythrocytes, albeit they seem to be smaller than expected according to the scale). (D) A β-Gal+ cell (arrow) in the endomysium in the angle of three myofibers, in this case in a transversal section of the vessel. (E) A group of MAC+ myofibers (between arrowheads) in a tibialis anterior. The serial section stained with H&E (F) corresponds to the region in the square in (E). Asterisks indicate two myofibers in which MAC labeling indicates intracellular complement activation and thus sarcolemmal permeabilization. Close to this region, there is a perimysial vessel in which the lumen is occupied by an eosinophilic body with nuclei (F) (arrow, higher magnification in the inset). This eosinophilic element is β-Gal+ in a serial section (G) (arrow), confirming that is an embolus of IA injected cells. The embolus is MAC+, thus necrotic (E) (arrow). The arrowhead in (G) shows a smaller accumulation of β-Gal+ cells into a smaller arteriole (F). Scale bars: 10 μm (C and D), 100 μm (A, B, F and G), and 500 μm (E).
Given the possibility of focal muscle ischemia due to blood flow interruption in the vessels plugged by the injected cells, we searched for myofiber necrosis. There was no histological evidence of myofiber necrosis in H&E-stained sections (Fig 2F). However, MAC immunodetection revealed myofibers with intracellular complement deposition at 1 day posttransplantation (Fig 2E). MAC+ myofibers were in dispersed or compact groups. In most cases, we observed intravascular β-Gal+ emboli in the perimysium close to the focal groups of necrotic myofibers (Fig. 2F, G), indicating that occlusion of a small vessel might explain the focal myofiber necrosis. These emboli were MAC+ (Fig 2E), indicating that they were necrotic. Following this observation, we analyzed the cell suspension after the IA transplantation in the following two monkeys by microscopic observation in a hemocytometer with trypan blue staining. We observed some cell aggregates, which in several cases were trypan blue+ (thus necrotic).
We observed β-Gal+ cells in the samples of nonmuscular tissues taken in the leg homolateral to the IA injection, that is, the sole skin (Fig. 3A–C), sciatic nerve (Fig. 3D, E) and lymph nodes (not shown). These β-Gal+ cells were also seen either as isolated cells or in small clumps of varying size.

β-Gal+ cells in nonmuscular tissues after IA injection. The tissues were sampled 1 day (A–E) and 1 h (F, G) after the IA injection. Sections are stained with H&E and for histochemical detection of β-Gal. (A) A section of sole skin in the limb homolateral to the IA injection (a: stratum corneum, b: epidermis, c: dermis). The region in the square is shown at higher magnification in (B) (arrowheads indicate sweat glands). (C) A serial section shows two small clusters of β-Gal+ cells (arrows, they indicate the same approximate locations in B). (D) A section of the sciatic nerve (between arrowheads). The region in the square corresponds to the serial section in (E), stained for β-Gal detection. Arrows indicate β-Gal+ cells, isolated or in small clusters. (F) β-Gal+ cells in the lung (arrow). Owing to sampling without fixation, the lung is collapsed, and the alveolar structure is not seen in the serial section (G), in which cavities correspond to vessels (asterisks). The arrow in (G) indicates the location of the β-Gal+ cells. Scale bars: 100 μm.
Lungs
We observed some β-Gal+ cells in the lungs at 1 h and 1 day posttransplantation (Fig. 3F, G). They were mostly isolated and less frequently seen as small accumulations. Accumulations of β-Gal+ cells were never as large as described above for the leg and were never in arterioles. β-Gal+ cells were more dispersed than in the leg homolateral to the IA injection.
Rest of the Body
No β-Gal+ cells were observed in the samples of the other organs or in the samples of skeletal muscles of the other limbs.
Intermediate Postinjection Period
There were β-Gal+ myofibers in skeletal muscle samples of the leg homolateral to the IA cell injection in the three monkeys during this follow-up (Fig. 4A–C). β-Gal+ myofibers were observed only in the sites of muscle damage. The positive controls (sites of IM transplantation) confirmed the engraftment capacity of the myoblasts used for IA injection (Fig 4D), according to our standard results of IM myoblast transplantation in monkeys (31). Good control of acute rejection was confirmed by the absence of lymphocyte accumulations around the hybrid myofibers (Fig 4C), according to our criteria for the diagnosis of acute rejection in this context (25).

Muscle engraftment of β-Gal-labeled myoblasts. The gastrocnemius homolateral to the cell injection, 5 weeks after transplantation, is shown in (A–C). This site was damaged at the moment of transplantation. (A) A cross-section of the muscle stained for β-Gal detection shows several β-Gal+ myofibers (3.2% of the surface in this section was β-Gal+). The region in the square in (A) is shown at a higher magnification in (B), with a serial section stained with H&E (C) (β-Gal+ myofibers are colocalized with numbers) to illustrate the muscle structure and the absence of lymphocyte accumulations typical of acute rejection. A small β-Gal+ element is located in the endomysium (B) (arrow), and in the serial section stained with H&E seems to coincide with a small vessel (C) (arrow). (D) The muscle site in which the same myoblasts were transplanted intramuscularly, as a control of their engraftment capacity. Scale bars: 1 mm (A, D) and 50 μm (B, C).
Discussion
We confirmed that myoblasts can be delivered by IA infusion to the limbs of primates and that they can participate in myofiber regeneration, giving rise to hybrid myofibers in these limbs. There was only one previous study dedicated to the IA transplantation of myoblasts, using the L6 cell line in rats (16). The significance of our study lies in two facts. First, in the use of nonhuman primates, as mentioned in the introduction. Second, in the use of cells obtained and cultured similarly to those used in our last clinical trials (26) (except for the retroviral labeling with LacZ).
Most of the IA-injected cells were retained in different tissues of the leg homolateral to the injection, that is, in the first vascular filter encountered. Some injected cells passed through the vascular bed of the leg and were retained in the second vascular filter, that is, the lungs. We did not observe β-Gal+ cells in other organs. Indeed, if some cells escaped the barrier of the lungs, their number should be low, and their dispersion throughout the whole body would make it difficult to find them by histology. These results agree with the IA delivery of L6 myoblasts in rats, in which most injected cells were found in muscles downstream to the IA injection, very few in the lungs (only in one of 12 animals), and none in the liver and spleen (16). IA injection of CD34+ stem cell antigen-positive (Sca-1+) cells in mice also resulted in their retention in muscles of the injected side, being absent from the lungs or other organs or muscles out of the injected limb (35). In contrast, this was different from the IA delivery of mesoangioblasts in mice; many mesoangioblasts passed through the vascular filter of the limbs and lungs and were captured in the liver and spleen (6).
The fact that the injected cells occluded small vessels (principally arterioles) implies the possibility of focal muscle ischemia. In effect, 1 day posttransplantation, we observed necrotic myofibers focally grouped near a small vessel occluded by a β-Gal+ embolus, corresponding to microinfarcts. The fact that these emboli were necrotic indicates that either the cells did not survive in these emboli or the emboli were formed by the aggregates of necrotic cells that we observed in the cell suspension. Small vessel occlusion could also be caused by myotubes present in the cell suspension. Arteriole occlusion should be avoided by preventing the passage of myotubes and cell aggregates into the circulation. It would be useful to investigate whether microinfarcts are produced by other myogenic cells administered IA.
The few myoblasts trapped in the lungs did not cause evident problems to the animal's health. Since capillary plugging is the usual mechanism of leukocyte migration in the lungs during inflammation (23), we can assume that plugging of a certain number of capillaries in the lungs is not harmful.
Since β-Gal+ myofibers were observed only in the muscle sites that were damaged, it is clear that muscle damage was necessary for the engraftment of the IA-injected myoblasts. Indeed, muscle regeneration is a prerequisite for the incorporation of muscle precursor cells into myofibers (30). It is logical that β-Gal+ myofibers in these sites were fewer than in the sites of IM transplantation; the dispersion of the IA-injected cells throughout the leg results in a low density of β-Gal+ myoblasts in the muscle. We have established in macaques that the engraftment is proportional to the amount of cells injected in a given volume of muscle (31). The sites of IM transplantation in this study were injected with about 20 × 106 cells per cm3, which is over the optimum amount of 10 × 106 cells per cm3 (31). The engraftment of the IA-injected cells was similar to that obtained by IM transplantation of about 10,000 to 100,000 cells per cm3 of muscle (31). Therefore, perhaps the engraftment could be improved if the number of IA-injected cells is increased in future experiments. Another aspect to consider in future experiments is how this engraftment can be produced with myofiber damage similar to that occurring spontaneously in muscular dystrophies. Mechanical damage in this study may have helped myoblast extravasation by damaging the vessels, and it is pertinent to see what would happen without vessel injury. We have preliminary evidence that this could happen, though perhaps less efficiently than with mechanical damage. In any case, the extravasation of myoblasts could be enhanced by inducing the secretion of enzymes that degrade the extracellular matrix, such as metalloproteinases (4,12,15). In fact, plugging in capillaries is a physiological mechanism of neutrophil extravasation in inflamed tissues, and this seems to require proteolytic breakdown of the basement membrane by secretion of elastase and matrix metalloproteinases (23).
What Is the Mechanism of Cell Capture in the Tissues?
Previous studies of IA delivery with other cells in mice interpreted that their capture in the capillaries of the muscles is due to specific molecular interactions between the grafted cells and the vessels, similar to those occurring during leukocyte extravasation at sites of inflammation (18). These studies hypothesized that the engraftment of IA-delivered myogenic cells in muscles involves margination, rolling, attachment, and extravasation (18).
The problem of this interpretation is that margination and rolling of leukocytes do not occur in capillaries. They occur predominantly in postcapillaries and venules, where the lumen is wide enough to allow leukocyte margination without stopping the flow of erythrocytes (essential for margination and rolling of leukocytes), and the flow rate is low enough to facilitate these mechanisms (24). Capillaries are too small to permit that, since their lumen is smaller than an erythrocyte (34). The transit of leukocytes in capillaries requires deformation of cells (with a diameter of ~6.5 μm for an average lymphocyte and ~8 μm for neutrophils) into a compressed shape fitting into the lumen of capillaries (5 to 6 μm) (24). For this reason, leukocyte entrapment in capillaries is not uncommon (24). This occurs frequently during inflammation, even if permanent plugging is supposed to need molecular interactions between the endothelium and leukocytes (24). Cell size and deformability condition the capillary arrest of cells (3). Neutrophils and monocytes are trapped more frequently than lymphocytes, which are smaller, through 5-μm pores (3). Since the diameter of human myoblasts is larger than that of leukocytes (11), their passage through capillaries should be ever more challenging. Therefore, retention of myoblasts in capillaries must be attributed to mechanical entrapment (Fig. 5). The presence of β-Gal+ myoblasts in the lungs suggests that some of them passed the vascular bed of the leg. This may be achieved by passing through arteriovenous anastomoses, which are larger than capillaries.

Capture of the IA-injected myoblasts in capillaries. The drawing depicts only essential elements for illustration purposes: the vascular endothelium (yellow), the injected myoblasts (blue), and the erythrocytes (red). Red arrows indicate the direction of blood flow. A terminal arteriole is represented (vertical vessel) with two capillaries (horizontal vessels A and B). Capillary (A) shows the normal flow or erythrocytes. In capillary (B), the plugged myoblast produced a flow arrest with accumulation of erythrocytes (as probably observed in Fig. 2C). The illustration was inspired by Figure 7 of Schmid–Schonbein (24), which shows examples of leukocyte plugging at the bifurcation of terminal arterioles and inside capillaries and was drawn by Dr. Daniel Skuk.
It is not up to us to analyze other cells that were delivered IA in mouse and dog experiments (2,20–22). However, it is pertinent to ask whether their homing in skeletal muscles is produced by margination, rolling, specific attachment, and extravasation in postcapillaries and venules or whether they are mechanically trapped in capillaries. This does not exclude that specific interactions with the endothelium could be important to produce extravasation.
Footnotes
Acknowledgments
This work was supported by a grant of the Jesse's Journey Foundation for Gene and Cell Therapy of Canada to Dr. Daniel Skuk. The authors wish to express their gratitude to Marlyne Goulet for excellent technical work in cell culture, histology and follow-up of immunosuppression in monkeys and to Steve Brochu for his technical assistance during monkey procedures. Authors' contribution: D.S. is responsible of the design, surgery, analysis, interpretation, and manuscript drafting. J.P.T. is the director of the research team; he read and approved the manuscript. The authors declare no conflict of interest.
