Abstract
Bone marrow mononuclear cell (BM-MNC) intra-arterial transplantation improves recovery in experimental models of ischemic stroke through secretion of cytokines and growth factors (GFs), enhancing neoangiogenesis, and enhancing neuroplasticity. In this study, we tested whether BM-MNC transplantation in stroke patients induces changes in serum levels of cytokines and GFs. A phase I/II trial was conducted in middle cerebral artery (MCA) stroke patients with autologous intra-arterial BM-MNC transplantation between 5 and 9 days after stroke. Follow-up was done for up to 6 months. Eight cases and nine controls were included, and the serum levels of granulocyte-macrophage colony-stimulating factor (GM-CSF), platelet-derived growth factor-BB (PDGF-BB), β nerve growth factor (β-NGF), and matrix metalloproteinases 2 (MMP-2) and 9 (MMP-9) were measured before and 4, 8, and 90 days after transplantation. The correlation of these serum levels with dose of cells and clinical outcomes was studied. A total of 1.59 × 108 (±1.21 × 108) BM-MNCs were injected in cases; of them 3.38 × 106 (±2.33 × 106) were CD34+ cells. There was a positive correlation between total BM-MNCs injected and levels of GM-CSF and PDGF-BB at 90 days after transplantation (r = 0.929, p = 0.001 and r = 0.714, p = 0.047, respectively), and a negative correlation between total CD34+ cells injected and MMP-2 levels at 4 days after transplantation (r = −0.786, p = 0.036). Lower plasma levels of MMP-2 at 4 days and higher levels of PDGF-BB at 90 days were associated with better functional outcomes during follow-up (p = 0.019 and p = 0.037, respectively). When administered intra-arterially in subacute MCA stroke patients, BM-MNCs seem to induce changes in serum levels of GM-CSF, PDGF-BB, and MMP-2, even 3 months after transplantation, which could be associated with better functional outcomes. This manuscript is published as part of the International Association of Neurorestoratology (IANR) special issue of Cell Transplantation.
Introduction
Stroke is one the leading causes of morbidity and long-term disability in the world, with 15% to 30% of survivors being permanently disabled (22). However, no effective neuroprotective or neurorestorative therapies have been approved for ischemic stroke (8). Cell therapy is a potential new approach to improve neurological deficits after a stroke. Bone marrow mononuclear cells (BM-MNCs) have consistently demonstrated efficacy in animal stroke models in different laboratories and species(2,11,33). BM-MNCs have also the advantage of being rapidly isolated from bone marrow and permit autologous applications within hours from bone marrow aspiration, allowing for treatment of acute stroke patients. In the last years, some preliminary studies have been published, demonstrating the safety and feasibility of autologous bone marrow transplantation in stroke patients (10,14,18,24).
It has been reported that BM-MNCs can show neural markers in the rodent stroke model (13). However, in animal stroke models, the beneficial effects of BM-MNCs seem to be modulated by secretion of a broad spectrum of growth factors (GFs) and cytokines (33). Several biological effects, such as modulating microglia, reducing proinflammatory responses, and increasing neoangiogenesis, have been invoked (2,11,33). However, whether BM-MNC transplantation induces these biological effects in stroke patients is still not known.
Since we have previously shown safety and feasibility results of intra-arterial BM-MNC transplantation in stroke patients (18), we aimed to study the changes in cytokines related to neoangiogenesis, brain plasticity, and basement membrane degradation in subacute stroke patients included in our previous clinical trial.
Materials and Methods
Patients
Between September 2008 and March 2011, 20 patients (10 cases and 10 controls) with severe ischemic stroke in the middle cerebral artery (MCA) territory were prospectively enrolled in this single-blind (outcomes assessor) phase I/II controlled clinical trial. Criteria for inclusion were age between 18 and 80 years, an ischemic stroke in the MCA territory, a National Institute of Health stroke scale (NIHSS) score ≥8 at inclusion, and a treatment window within 5–9 days of stroke onset.
Patients with lacunar or hemorrhagic stroke were excluded. Other exclusion criteria were pregnancy, childbearing potential, history of present or previous malignant disease during the last 5 years, life-threatening illness, stroke of hematological cause, significant previous disability (prestroke modified Rankin Scale score ≥3), and severe comorbidity (severe hepatic or renal dysfunction) that would prevent follow-up.
This study was done in accordance with the Declaration of Helsinki and the Good Clinical Practice Guidelines and approved by the regional ethics committee and the Spanish Regulatory Agency. Written informed consent was obtained from each patient or their representatives. The trial was registered with clinicaltrials.gov (trial identification number NCT00761982). The patients did not pay for their inclusion or treatment in this study.
Procedures
The transplantation procedure was done in the BM-MNC-treated group between 5 and 9 days after stroke onset. Once the patient was in the operating room and under local analgesia [1% lidocaine without epinephrine (B. Braun Medical, Barcelona, Spain)] and mild sedation [midazolam (Laboratorios Normon, Madrid, Spain) 2–3 mg intravenous], 50 ml of bone marrow was obtained aseptically by puncture in the posterior iliac crest. Each 5-ml bone marrow aspiration was deposited into a transfer bag (Baxter, Fenwal division, Mont Saint Guibert, Belgium) with 10 ml of acid–citrate–dextrose (ACD) solution (Grifols, Barcelona, Spain) and 1 ml of preservative-free heparin (Laboratorios Farmacéuticos ROVI, S.A., Madrid, Spain).
The BM-MNCs were isolated by density gradient centrifugation over Ficoll-Hypaque solution (Fresenius KabiNorge AS, Oslo, Norway). After three washes, the BM-MNCs were filtered and resuspended in a mean volume of 7.5 ml (±2.12) of 0.9% sodium chloride [in phosphate-buffered saline (PBS); Grifols] supplemented with 0.1% heparin for infusion into the patient.
Aliquots of the final BM-MNC product were obtained for automated cell counting (Sysmex xp 5000, Norderstedt, Germany) and flow cytometry. Cells were stained with anti-cluster of differentiation 34 (CD34) MoAb conjugated to fluorescein isothiocyanate (FITC) according to the manufacturer's instructions (Becton Dickinson Pharmingen™; San Diego, CA, USA). Flow cytometer analyses were performed on a FACSCantoTM® (BD Biosciences, San Jose, CA, USA). Data were analyzed with BD CellQuest™ Pro software (BD Biosciences). Then, number of total CD34+ cells was calculated by multiplying for total volume (ml).
On the same day and 3–4 h after bone marrow harvest, a cerebral angiography (Philips Integris H5000, Best, Netherlands) (3000 U heparin) was done in the neuro-interventional room. Then, a microcatheter (Excelsior SL-10 Stryker Neurovascular, Fremont, CA, USA) was placed in the M1 segment of the infarct-related MCA, and BM-MNCs were transplanted at low pressure over approximately 10 min. No bone marrow aspiration or sham injection was performed in the control group.
Clinical and functional evaluations were performed the day after transplantation and 1, 3, and 6 months after the stroke. Neurological deficit was measured with the NIHSS (3). Disability and dependence in activities of daily living were measured with the modified Rankin Scale (31) and Barthel index (5), respectively. Neurologists assessing the patients during follow-up were unaware of the treatment allocation.
Of the patients included in the trial, blood samples were obtained from 17 patients that were included in this study. Three patients from the original study were excluded as we could not obtain every blood sample in the prespecified time points to analyze the temporal curve of the different cytokines and GFs. In eight active cases and nine controls, blood samples were drawn before and 4, 8, and 90 days after transplantation (or the day after inclusion for control patients) for determination of matrix metalloproteinases 2 (MMP-2) and 9 (MMP-9), granulocyte-macrophage colony-stimulating factor (GM-CSF), platelet-derived growth factor-BB (PDGF-BB), and β nerve growth factor (β-NGF) serum levels by means of enzyme-linked immunosorbent assay (ELISA) (Aushon Biosystem, Cultek, Billerica, MA, USA; ref: 84739, 84991, and 84794; R&D Systems, Minneapolis, MN, USA; ref: DY256; respectively).
Statistical Analysis
Spearman's rank correlation coefficient was used to assess the association between MMP-2, MMP-9, GM-CSF, PDGF-BB, and β-NGF serum levels and the number of BM-MNCs and cluster of differentiation 34-positive (CD34+) cells injected intra-arterially. Also, serum levels were correlated with neurological status on follow-up (NIHSS, modified Rankin, and Barthel index scores). Finally, analysis of the PDGF-BB, MMP-2, and MMP-9 serum levels at each individual time point was done by repeated measures ANOVA analyses with Bonferroni correction for multiple comparisons, to study changes in their temporal profiles similar to our previously reported results on β-NGF and GM-CSF (18). All statistical analyses were performed using the SPSS software package version 17.0 for Windows (IBM, Armonk, NY, USA). Differences were considered to be statistically significant when two-tailed values were p < 0.05.
Results
Of the 17 patients included, 47.1% were men, and the mean age was 68.1 years (±12.9). Baseline characteristics and vascular risk factors were similar in both groups (Table 1). All had a severe MCA stroke at inclusion with a mean NIHSS score of 15.6. Mean time from stroke onset to BM-MNC transplantation was 6.4 days (±1.3). A mean of 1.59 × 108 BM-MNCs (±1.21 × 108) were intraarterially injected into the MCA. Of them, 3.38 × 106 (±2.33) were CD34+ cells. By weight, we injected a mean of 2.17 × 106 BM-MNCs per kilogram and 47,363 CD34+ cells per kilogram.
Baseline Characteristics

Data are number (%) or mean (SD). NIHSS, National Institutes of Health stroke scale; BM-MNC, bone marrow mononuclear cell.
At day 4 after BM-MNC transplantation, we found significantly lower serum levels of MMP-2 in cases versus controls (188.20 ± 23.23 ng/ml vs. 258.86 ± 55.31 ng/ml, p = 0.006), with no significant differences at day 8 (193.80 ± 38.91 vs. 234.47 ± 68.37, p = 0.173) or day 90 (255.69 ± 59.94 vs. 277.29 ± 11.74, p = 0.654). Similar levels of MMP-9 at baseline or during follow-up were observed between the two groups (Fig. 1). There was also a negative correlation between levels of MMP-2 at day 4 after transplantation and number of CD34+ cells injected (r = −0.667, p = 0.071) and CD34+ per kilogram injected (r = −0.786, p = 0.036) (Fig. 2).
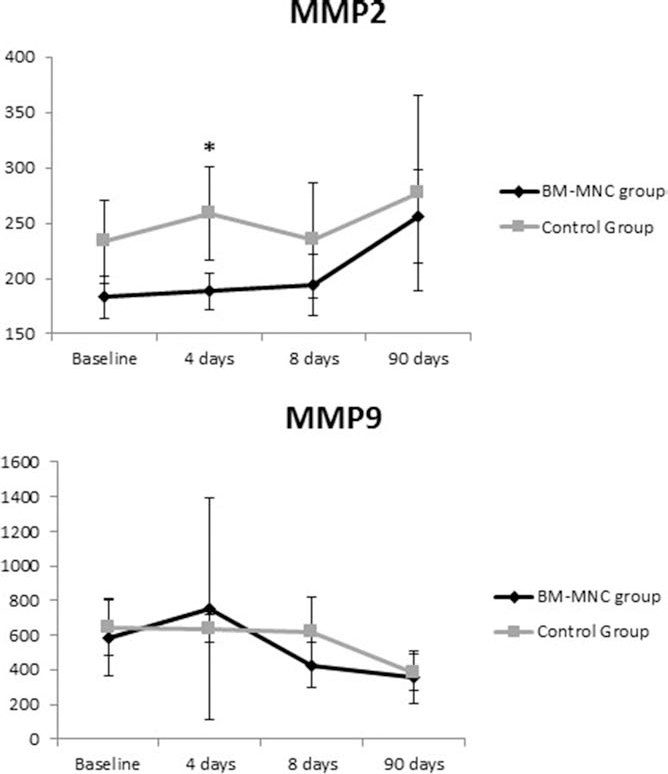
MMP-2 and MMP-9 levels before and after transplantation. Values are expressed as mean ± SD ng/ml. *p < 0.01. BM-MNC group indicates bone marrow mononuclear cell transplantation group.

Correlation between number of CD34+ cells per kilogram injected and MMP-2 serum levels at day 4 after transplantation. Values of CD34+ cells are expressed as millions of cells per kilogram injected, and MMP-2 values are expressed in ng/ml.
Although there were no significant differences in the levels of GM-CSF and PDGF-BB between BM-MNC-treated patients and controls, a strong correlation was detected between serum levels of GM-CSF at day 90 after transplantation and the total number of BM-MNCs injected (r = 0.929, p = 0.001) and BM-MNC per kilogram injected (r = 0.929, p = 0.003) (Fig. 3). Also, PDGF-BB levels at days 8 and 90 after intra-arterial injection were positively correlated with the number of transplanted BM-MNCs (r = 0.714, p = 0.047 and r = 0.900, p = 0.037, respectively).

Correlation between number of CM-MNCs injected and GM-CSF serum levels at day 90 after transplantation. BM-MNC indicates bone marrow mononuclear cell; GM-CSF, granulocyte-macrophage colony-stimulating factor. Values of CM-MNCs are expressed as millions of cells injected, and GM-CSF values are expressed in pg/ml.
When evaluating the neurological outcomes, we found that higher levels of PDGF-BB were associated with better functional status, measured with the Barthel index at 90 days (r = 0.900, p = 0.037; data not shown).
Also, lower levels of MMP-2 at day 4 were correlated with lower neurological deficit (NIHSS at day 30) (r = 0.775, p = 0.041). There was better functional status measured with the Rankin scale at days 90 (r = 0.792, p = 0.019) and 180 (r = 0.776, p = 0.024) and with the Barthel index at day 180 from transplantation (r = −0.690, p = 0.058). No correlation was detected between MMP-9 and β-NGF levels and neurological outcomes (data not shown).
Discussion
Until now, neuroprotective and neurorestorative drugs targeting single steps in the cascade of cerebral ischemia have failed to improve neurological deficits. This is probably related to stroke complexity, with necrosis, apoptosis, inflammation, and remodeling occurring as a continuum. Cell therapy, such as transplantation of BM-MNCs, might target simultaneously several of these processes by releasing different factors, inducing neuroprotection, brain remodeling, and modulating the postischemic inflammatory response (2,4,11,29,33). BM-MNCs include a population of mesenchymal (MSC) and hematopoietic progenitor stem cells (HPCs), which secrete cytokines and GFs, which are involved in the repair processes that are enhanced under hypoxic conditions (29). It has been speculated that the cytokines produced by BM-MNCs play a more important role than their differentiation and proliferation (15). However, whether these mechanisms occur in stroke patients after cell therapy remains unclear.
In a previous paper we described an increase of β-NGF serum levels after intra-arterial BM-MNC transplantation, a neurotrophic factor that contributes to brain plasticity after stroke (18). However, until now, there is little evidence documented on the biological effects of BM-MNC transplantation in stroke patients.
First, we analyzed markers of basement membrane degradation after stroke. MMPs are known to have a dual role by acutely disrupting the blood–brain barrier (BBB) and chronically promoting angiogenesis. MMPs degrade extracellular matrix and basal lamina proteins leading to BBB leakage, edema, and leukocyte infiltration, increasing further brain injury in the first days after stroke (19). MMP-9 is associated with acute BBB opening, whereas MMP-2 levels are increased in the subacute stroke phase and associated with delayed BBB opening (19). After BM-MNC injection, there was a significant decrease in MMP-2 levels in BM-MNC-treated patients in the first 4 days, but not in later time points, that were correlated with lower neurological deficit and better functional status during follow-up (up to 6 months). Interestingly, it has been found that early inhibition of MMPs after stroke reduces ischemic volume without compromising the beneficial effects of matrix plasticity that promotes BBB restoration and angiogenesis in the long term (32).
As we previously described, a higher number of CD34+ cells injected may induce better functional outcomes measured by the Barthel index (18). Cells expressing CD34 (CD34+ cells) are normally found in the bone marrow as HPCs and are probably recruited by inflammatory signals originating in the ischemic brain. HPCs reduce cerebral postischemic inflammation, attenuate peripheral immune activation, and mediate neuroprotection after ischemic stroke in animal models (25). In relation to these findings, in this study we determined that a higher number of CD34+ cells correlated with lower levels of MMP-2. Therefore, we may speculate that CD34+ cells could play an anti-inflammatory role in stroke by decreasing MMP-2 levels.
Second, we analyzed cytokines related to neoangiogenesis and brain plasticity in stroke, such as GM-CSF, PDGF-BB, and β-NGF. Induction of angiogenesis stimulates endogenous recovery mechanisms after stroke, including neuronal plasticity, synaptogenesis, and long-term migration of neuroblasts (7,30). It has been described that a proangiogenic status is associated with better functional outcomes in stroke patients (20). GM-CSF enhances angiogenesis and ameliorates cerebral damage after stroke, and PDGF-BB improves angiogenesis and regeneration of damaged axons after stroke and promotes neural stem cell migration (9, 16,28). Also, β-NGF reduces apoptotic neuronal death and contributes to brain plasticity after stroke (6,23). It has been found that BM-MNCs produce PDGF under hypoxic conditions and that it contributes to functional improvement in the infarcted heart, inducing angiogenesis and inhibiting apoptosis (29).
Surprisingly, we found a strong correlation between the number of BM-MNCs transplanted and the levels of GM-CSF and PDGF-BB after 3 months. Also, higher levels of PDGF-BB at 3 months were associated with better functional outcomes, suggesting a beneficial effect of BM-MNC transplantation.
However, BM-MNCs seem to survive only for a few days after transplantation, even in humans (1,2,12). Despite this short survival, previous studies have observed a substantial increase in axonal sprouting in the subventricular zone and migration and survival of neuroblasts even 4 months after treatment with bone marrow mesenchymal stem cells in stroke animals (17,26). Enhancement of oligodendrocyte progenitor cells after bone marrow cell therapy has been proposed as a mechanism that contributes to improvement of neurological outcomes in the long term as a response to cell therapy in the acute stroke phase (17). Also, some cytokines, such as granulocyte-colony-stimulating factor (G-CSF), are known to mobilize endogenous bone marrow stem cells in patients with subacute ischemic stroke (27). We then hypothesized that BM-MNC transplantation may activate other restorative mechanisms in the subacute-chronic phase of stroke, such as activation of neural stem cells or mobilization of endogenous bone marrow stem cells that could explain these cytokine changes after 90 days of transplantation.
Despite promising preclinical data, administration of single cytokines failed to improve stroke outcomes (21). Our data strengthens the notion that stem cells play a role as a “factory” of trophic factors and cytokines, making this therapy more likely to improve neurological deficits, as they could target different symptoms of brain ischemia.
There are several limitations in this study. First is the small sample size included in this study, making it possible that some of the findings could be by chance. However, the strong correlations detected and similar findings previously described in stroke animal models make this possibility very unlikely.
Another limitation is that with this study design approach we cannot prove a relationship of causality between the levels of cytokines and GFs and functional recovery. However, some of our findings and those of others point toward an interesting relationship, as we have found clear correlations between the levels of MMP-2 and PDGF-BB and better functional status in the long term after therapy, supporting this hypothesis. Also, changes in levels of MMP-2, GM-CSF, and PDGF-BB, as those detected in this study, have already been correlated with better functional status in stroke (9,26,28,32).
Whether these changes in serum cytokines and MMPs improve neurological outcomes is still to be proven. Further work is, therefore, needed to determine the efficacy of BM-MNC transplantation in stroke patients.
In conclusion, intra-arterial injection of BM-MNCs in subacute stroke patients, seem to induce changes in serum levels of GM-CSF, PDGF-BB, and MMP-2, even 3 months after transplantation, which could be associated with better functional outcomes.
Footnotes
Acknowledgments
This study was partially funded by Junta de Andalucía grant TCRM 0001/2006. The authors declare no conflicts of interest.
