Abstract
Autism spectrum disorders (ASDs) are heterogeneous complex neurodevelopmental pathologies defined by behavioral symptoms, but which have well-characterized genetic, immunological, and physiological comorbidities. Despite extensive research efforts, there are presently no agreed upon therapeutic approaches for either the core behaviors or the associated comorbidities. In particular, the known autoimmune disorders associated with autism are appealing targets for potential stem cell therapeutics. Of the various stem cell populations, fetal stem cells (FSCs) offer the potent immunoregulatory functions found in primordial mesenchymal stem cells, while exhibiting rapid expansion capacity and recognized plasticity. These properties enhance their potential for clinical use. Furthermore, FSCs are potent and implantable “biopharmacies” capable of delivering trophic signals to the host, which could influence brain development. This study investigated the safety and efficacy of FSC transplantations in treating children diagnosed with ASDs. Subjects were monitored at pre, and then 6 and 12 months following the transplantations, which consisted of two doses of intravenously and subcutaneously administered FSCs. The Autism Treatment Evaluation Checklist (ATEC) test and Aberrant Behavior Checklist (ABC) scores were performed. Laboratory examinations and clinical assessment of adverse effects were performed in order to evaluate treatment safety. No adverse events of significance were observed in ASD children treated with FSCs, including no transmitted infections or immunological complications. Statistically significant differences (p < 0.05) were shown on ATEC/ABC scores for the domains of speech, sociability, sensory, and overall health, as well as reductions in the total scores when compared to pretreatment values. We recognize that the use of FSCs remains controversial for the present. The results of this study, however, warrant additional investigations into the mechanisms of cell therapies for ASDs, while prompting the exploration of FSCs as “biopharmacies” capable of manufacturing the full array of cell-signaling chemistry. This manuscript is published as part of the International Association of Neurorestoratology (IANR) special issue of Cell Transplantation.
Introduction
Autism and autism spectrum disorders (ASDs) are neurodevelopmental pathologies affecting the brain and starting during the first 3 years of life (4). Autistic individuals seem to live in their own world; they are socially withdrawn and generally are unable to verbally express their emotions and needs. ASDs are characterized by a wide range of cognitive, emotional, and neurobehavioral abnormalities, social interaction and communication dysfunctions, restricted interests, and repetitive and stereotypic verbal and nonverbal behaviors (2,19).
The prevalence of children found to be affected by autism is rapidly increasing: a change that cannot be explained by mere data-gathering methods, nor is it likely due to changed diagnostic approaches (3). A recent study reports that 2% (3.23% of boys) of US school-aged children are now affected (5).
In spite of outstanding achievements in the study of pathogenesis of autism and other ASDs, including immunological and immunogenetic research resulting in the new trends of specific therapy, autism remains one of the unresolved issues of modern neurology. Currently available pharmaceuticals, that is, antidepressants and antipsychotics, only target specific limited symptoms, without resolving the basic underlying etiologies (17). Indeed, no effective drug therapies are currently available (9). In addition, several concerns on their usefulness have been raised, as their efficacy in treating ASDs is not definitively proven, and side effects are common (11). There is a lack of internationally standardized and defined approaches, although the therapies commonly used for ASDs involve educational, behavioral, sensory-integration, nutritional, hyperbaric oxygen, heavy metal detoxification, and immunological interventions (19). Recently, the potent immune regulator and vitamin D-binding protein, Gc macrophage-activating factor (GcMAF), has been used with positive results (6).
At present, new autism treatment approaches are being developed (23). One novel and promising intervention is stem cell therapy (24,26). Within the categories of available stem cells, fetal stem cells (FSCs) are presently being studied and used in animal models and clinical trials of neurological diseases (1). These cells are more specialized, compared to embryonic stem cells (ESCs), but, at the same time, have a higher proliferative potential and are effective in a number of illnesses, including those affecting the nervous system, without the need for human leukocyte antigen (HLA) matching (1). FSCs are easily obtainable and have higher proliferative and multiplication potential than adult bone marrow cells.
The aim of this research study was to examine FSC treatment safety and efficacy in autistic children. We studied clinical, neurological, and laboratory changes in autistic patients before and after FSC therapy.
Materials and Methods
Subjects
This is an open-label prospective study on the use of FSCs in ASD patients. The primary objective of this study was to assess and establish the safety of FSC transplantation (FSCT) in children with autism. A secondary objective was to evaluate the effectiveness of FSCT for reducing the core symptoms of ASDs. This study included patients from several nations, including Great Britain, Italy, Canada, Kuwait, UAE, Poland, Serbia, US, and Georgia (Sakartvelo).
The study group consisted of 45 children with confirmed autism (39 boys and 6 girls) with ages ranging from 3 to 15 (mean = 6.94 ± 0.89). The patients were advised to not change their current therapies, that is, behavioral, dietary, and speech. All patients underwent transplantation of FSC suspension at the EmCell facility in Kyiv, Ukraine. Before entering the study, the children were examined by a pediatrician and a child neurologist and administered the Autism Diagnostic Interview-Revised version (ADI-R), the Childhood Autism Rating Scale (CARS), and the Autism Diagnostic Observation Schedule-Generic (ADOS) to document the diagnoses of autism.
Additionally, all included patients met the Diagnostic and Statistical Manual of Mental DisordersIV Revised criteria for autism (DSM-IV-TR) (2). Further, children were screened with the Aberrant Behavior Checklist (ABC). Potential subjects were excluded if they had any of the following: a neurological or comorbid psychiatric disorder, epilepsy, history of liver, renal, or endocrine disorders, or current infection of any origin. Mental retardation without autism, including pervasive developmental disorder-not otherwise specified (PDD-NOS), was considered exclusion criteria. Children diagnosed with Asperger's syndrome, fragile X syndrome, or tuberous sclerosis were also excluded from the study. IQ testing was not performed but was frequently available from the pediatric records. None of the subjects in the study group were receiving antipsychotic or stimulant medications.
All children underwent general and neurologic examination, routine blood tests, and immunology testing. Neurological evaluation and laboratory tests were performed before FSCT as well as 6 and 12 months after transplantation. All patients underwent immunological testing [B-lymphocytes: CD19+, T-lymphocytes: CD3+, T-helpers: CD4+, T-suppressors: CD8+, natural killer (NK): CD16+] before FSCT and 6 and 12 months after it. This test was performed at EmCell laboratory (Kyiv, Ukraine) by cytofluorimetry method (wavelength 464–480 mm, using cytofluorimeter FASTER 2010, Beckton Dickinson, North Ryde, NSW, Australia). Leukocyte and lymphocyte counts were measured by impedancemetry method (measurement range: 0–100 × 109/L), with hematologic analyzer MYTHIC 18 (Orphee Medical, Plan-les-Ouates, Switzerland), at EmCell laboratory. Before the treatment, all of the patients' parents, or their legal representatives, signed the informed consent for blood tests, FSCT, and further study and evaluation of the potential comorbidity in autistic patients. This study is a registered scientific research protocol under N 0113U004140 in the State Agency of Science, Innovations and Informatization of Ukraine of the Ukrainian Institute for Research and Development and Economic Information. Extensive informed consent was administered in the native languages of the parents. While some of the patients did pay for their treatment in this study, this was not criteria for their inclusion. Patients who were willing to pay were excluded if comorbidity was present or they met the exclusion criteria, or they failed the inclusion criteria.
Stem Cell Procedure: Donor Screening and Sterility Processing
We used suspensions containing stem cells harvested from 5- to 9-week-old human fetuses following voluntarily elective pregnancy terminations (legally available in the Ukraine). One donor source was utilized for each individual sample, that is, cells from different donors were never combined. Fetal material was harvested in the operatory pursuant to aseptic surgical requirements. Gestational age was accomplished with abdominal or transvaginal ultrasound and by examination of the harvested material. All donors had extensive health histories and physical examinations documented in the medical records. Tissue was collected upon written informed consent of the maternal donor from liver and brain nervous tissues of 5- to 9-week gestational fetuses aborted for family planning purposes.
Infectious Disease Screening
In order to assure safety, both women donors and the hematopoietic stem cells (HSCs) from each fetal liver suspension were tested for bacterial, fungal, and viral infections and parasites such as human immunodeficiency virus 1 (HIV 1) and 2, hepatitis B virus (HBV), HCV, syphilis (Treponema pallidum), toxoplasmosis (Toxoplasma gondii), rubella, cytomegalovirus (CMV), herpes simplex virus 1 (HSV 1) and 2, Epstein–Barr virus (EBV), Mycoplasma genitalium, Ureaplasma urealyticum, Ureaplasma parvum, and Chlamydia trachomatis.
Bacteriological and fungal testing utilized standard screening performed by the Central Health and Disease Control Department of the Health Ministry of Ukraine, Kiev, Ukraine. After thawing in a sterile vessel, sterility testing was performed via inoculation in the following nutrient media: Hottinger sugar broth, thioglycollate medium, Saburo medium (PAA Laboratories, Pashing, Austria). In the laminar flow hood, cell suspension in saline solution was inoculated (with a dropper) into duplicate test tubes containing 10 ml of each medium. If no growth was observed after 14 days of inoculation in all media, a sterility certificate was issued for the sample. If bacterial growth was apparent in at least one test tube, the sample was discarded and properly destroyed.
Virological testing utilized enzyme-linked systems Diaprom Med (Kiev, Ukraine), Multiskan EX microtiter plate reader (Thermo LabSystems). ELISA and EIA (Diaprom Med, Ukraine) are used for HIV (1 and 2). screening. If antibodies were positive, both cell suspension and controls intended for long-term storage were discarded and destroyed pursuant to biohazardous waste requirements (direct high-temperature incineration). Testing and certification of suitability for transplantation was additionally performed by the Virological and AIDS Laboratory of the Central Health and Disease Control Department of the Health Ministry of Ukraine, Kiev, Ukraine.
Cell Suspension Isolation
One suspension was made of HSCs derived from fetal liver, while another was prepared from fetal brain nervous stem cells isolated using microscopic operating techniques by trained specialists (the initial cell viability was 85–95%). Fetal tissue was then placed into sterile transport medium made of Hank's solution (PAA Laboratories, Pashing, Austria) and penicillin–streptomycin (100 U–100 mg/ml) (Sigma Chemical Company, St. Louis, MO, USA). Tissues were aseptically separated and homogenized in the Hank's antibiotic solution. Stem cell suspensions were then filtered using individual 100-μm filters (BD, Italy).
Stem Cell Expansion and Colony-Forming Activity
Culturing was performed in 24-well plates (Greiner Bio-one, Germany) in an atmosphere of 5% CO2, air, at 100% humidity, while being maintained at 37°C. Cells were kept in the following semisolid medium: agar 33% (Difco, BD, Italy), glutamine 4.0 mM/L (Sigma Chemical Company), penicillin–streptomycin (100 U + 100 mg/ml) (Sigma Chemical Company), granulocyte/macrophage colony-stimulating factor 100 U/ml (Sigma Chemical Company), interleukin (IL)-3 100 U/ml (Behringwerke, Germany), stem cell factor (SCF) 50 ng/ml (R&D Systems, Minneapolis, MN, USA), and erythropoietin 10 U/ml (Epoetin b; Boehringer Manheim Pharmaceuticals, Germany). The medium was prepared on Dulbecco's modified Eagle's medium (DMEM) (PAA Laboratories). Cells were expanded for 14 days. Colony (CFU-A, CFU-GM, CFU-GEMM) number and type calculation was performed on the 14th day of culturing. Progenitor cells and stem cells of fetal neural tissue were characterized by culturing them in vitro. Neurospheres from fetal neural cells were formed using this method, and their number and histological characteristics pointed to their functional potency as neuroprogenitor cells.
Cryopreservation of expanded cell suspensions was performed in the controlled-rate freezer chamber (IceCube 14-S, Sy-lab Cryobiology, Austria) pursuant to the preselected programs. Suspensions containing HSCs from fetal liver and brain (nervous) stem cells were then stored in liquid nitrogen at −196°C in the EmCell cryobank. Dimethyl sulfoxide (DMSO, Sigma Chemical Company) 10% diluted in DMEM was used as cryoprotector.
Transplantation Methodology
Stem cell transplantation of suspensions containing cryopreserved FSCs was preceded by premedication of the subject via intravenous slow infusion of diphenhydramine (Darnitsa, Ukraine) 10 mg and prednisone (Darnitsa, Ukraine) 15 mg on day 1 and diphenhydramine (Darnitsa, Ukraine) 10 mg on day 2.
On day 1, we used HSCs from fetal liver containing an average of 1.6 ml of cell suspension with a nucleated cell count >30 × 106/ml per transplantation. All cells used were cryopreserved and thawed the same way. Cell suspension in cryo-test tubes (Changzhou Wideroad Int., Changzhou, China) were left for 5–7 s in order to allow evaporation of liquid nitrogen from the surface of cryo-test tubes. Each cryo-test tube was disinfected using a special agent (ethyl alcohol 70%). Then, for the thaw process, they were supported in water balneum (37°C) in a vertical position with the help of forceps. The cap of the cryo-test tube was above the water level, but the cryo-test tube with the cell suspension was sunk in water. The process of thawing was checked visually; after 45–60 s each cryo-test tube was removed with forceps from the support to detect the state of the liquid phase. If the liquid phase was not observed, cryo-test tubes were sunk in water as required. Cells were then centrifuged and washed twice in saline solution. Cells were resuspended in saline solution before transplantation. Five-milliliter disposable syringes with 0.7 × 40-mm needle (BD Discardit™ II, Fraga, Spain) were filled with the cell suspension. The cell suspension was administered via drip-feed IV (Hemoplast, Belgorod-Dniester, Ukraine) to the patient along with 200 ml of saline solution (Yuriya-Pharm, Kyiv, Ukraine), by means of a blood transfusion system (Hemoplast, Odessa, Ukraine), with 1.2 × 38-mm needle and infusion volume of 22 cm3.
On day 2, we administered neuroprogenitor cells from fetal brain into the subcutaneous abdominal adipose tissues. The cell suspension volume (in saline solution) averaged 2.12 ± 0.49 ml with a nucleated neuroprogenitor cell count >8.70 × 106/ml per transplantation. Cluster of differentiation 34-positive (CD34+) cells were counted by using flow cytofluorometry (Beckton Dickinson, Australia) with fluorescent-tagged antibodies (Santa Cruz Biotechnology, Santa Cruz, CA, USA).
Statistics
Statistical analysis of the data was performed with Statistica v.6.0 (StatSoft, Inc., Tulsa, OK, USA), including calculation of averages and standard deviation (SD). Bonferroni multiple comparison correction test and Student's t test were used to determine statistical significance of the results (p < 0.05 was considered statistically significant).
Results
Early posttransplantation effects were reported in 78% of children: 26% of these children became calmer; eye contact was improved in 9%, while 29% had better appetite, and 23% had an improved affect. No adverse events were observed in ASD children treated with FSCs. No side effects were observed from the steroid and antihistamine combination, and these initial changes likely represent some immunological benefit of the medications. No transmittable diseases were noted during the 12-month follow-up. We analyzed the immunology findings of autistic children before FSCT and at 6 and 12 months after it.
According to Table 1, within 6 months after the treatment, B-lymphocyte (CD19+) count reduced significantly. This was felt to be indicative of improved cell-mediated immunity in children, which was confirmed by the significant increase of CD3+ and CD4+ counts 12 months after the treatment. Again, no side effects were observed among the autism patients during this period of observation.
Immunological Findings

Immunology findings of autistic children before FSCT, and at 6 and 12 months after are reported. Within 6 months after the stem cell treatment, B-lymphocyte [cluster of differentiation 19 (CD19+)] count was reduced significantly; this result indicates a cell-mediated immunity improvement in children after FSCT, which is confirmed by the significant increase in CD3+ and CD4+ counts 12 months after the treatment. Values are expressed as mean ± SD. Reference ranges refer to the normal healthy conditions based on the average population of patients and healthy controls examined at our clinic.
p < 0.05 between baseline and results before FSCT.
p < 0.05 between results before and 6 months after FSCT.
p < 0.05 between results before and 12 months after FSCT.
There was a marked reduction in the mean score on the ATEC scale reflected by lowering of the average rates in all subtests of the scale (Fig. 1). A statistically significant reduction in the overall score on the ATEC scale was observed in the patients 12 months after treatment (59 ± 9.5) compared to before treatment (83 ± 5.5; p < 0.05). Also, a significant decrease in the mean score according to the sociability subscale was defined in the patients at 12 months (11.5 ± 21) after treatment compared to before treatment (19.8 ± 1.9; p < 0.05); cognitive subscale results were 15.0 ± 2.5 at 6 months posttransplantation and 8.0 ± 2.0 post 12 months, compared to 21.4 ± 1.5 before treatment (p < 0.05). Clinically significant improvement in the behavior of subscale data revealed a 12-month posttreatment score of 9.3 ± 2.1 compared to a pretreatment score of (24.5 ± 2.9; p < 0.05).
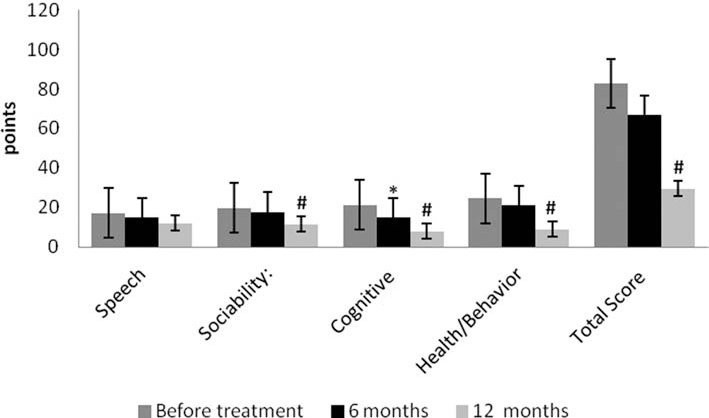
Dynamics on a scale of ATEC in autistic patients before FSCT and 6 and 12 month later. *p < 0.05 between baseline and results at 6 months. #p < 0.05 between baseline and results at 12 months. Patients: children with confirmed autism (39 boys and 6 girls).
According to the ABC scale, a significant decrease in the mean score was apparent in the patients after both 6 and 12 months (63 ± 1.6 and 54 ± 2.9, respectively) compared to before treatment (80 ± 2; p < 0.05) (Fig. 2).
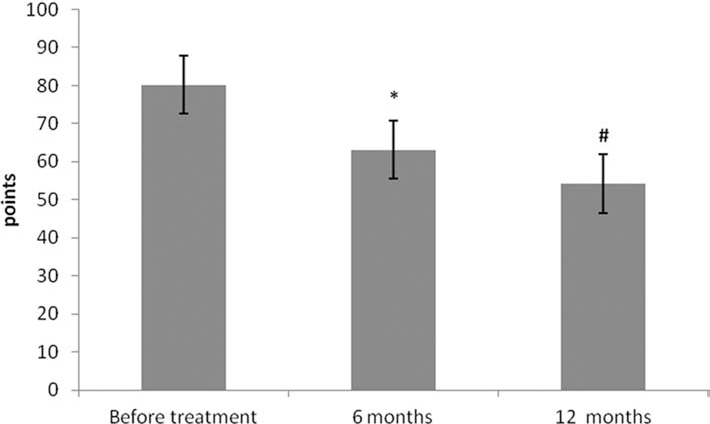
Dynamics on a scale of ABC in autistic patients before FSCT and 6 and 12 months later. *p < 0.05 between baseline and results at 6 months. #p < 0.05 between baseline and results at 12 months. Patients: children with confirmed autism (39 boys and 6 girls).
Discussion
In this study, we evaluated the safety and potentially beneficial effects of FSCT in autistic children. ASDs are generally considered lifelong and incurable disorders (19,9). However, the immunological literature points to some encouraging possibilities for immunotherapies (6,13). Cell-based therapy offers a novel approach for ASD management (25,20). Recently, it has been demonstrated that autologous bone marrow-derived mononuclear cells (BMMNCs), delivered intrathecally into the L4–L5 spinal area of a 14-year-old boy with severe autism, resulted in an improved quality of life (20). The boy showed improvements from 1 week after cell transplantation until the 1-year follow-up. These improvements ranged from increased spontaneous eye contact and included better attention and improved social interaction (20). The CARS rating fell to the nonautistic range after treatment. Similarly, in a study conducted from the same research group, BMMNCs were able to improve autistic behaviors in 32 autistic patients (21).
Among cell populations, FSCs offer a new treatment modality for ASDs (24,27). The rationale for the use of stem cells in ASDs, in part, comes from the paracrine and immunomodulatory effects of stem cells (26,29). It has been demonstrated that ASDs are associated with immune changes and overproduction of proinflammatory cytokines [i.e., interleukin (IL)-1β] (30). Stem cells are able to synthesize and release several significant bioactive molecules (cytokines, chemokines, and growth factors) (29), which, in turn, are able to suppress the proinflammatory immune skewing of the ASD-associated immune system (6,13,31). Stem cells are further known to inhibit apoptosis and stimulate recruitment, retention, mitosis, and differentiation of tissue-residing native stem cells (26). It has been demonstrated that HSC, as well as human umbilical cord mesenchymal stem cell, transplantations are able to affect the levels of circulating inflammatory cytokines (15,35). The mechanism of action exerted by these stem cells could also be applicable to FSCs.
Among presently available stem cell populations, FSCs are more primitive and have greater potentiality than their adult counterparts (1). They are more specialized than ESCs, which is desirable for specialized paracrine activity. They show higher proliferative and expansion potential and lower immunogenicity than other stem cell types (8). FSCs are more rapidly, easily, and efficiently reprogrammed to pluripotency than neonatal and adult cells (12). Their rapid proliferation, stable karyotype, and low immunogenicity render them useful candidates for cell transplantation (1). In addition, unlike ESCs, they have greatly reduced potential to form in vivo teratomas (27). Ethical issues are also of less controversy than ECSs, as FSCs are obtained from tissues that would otherwise be discarded.
The FSCTs described in this article appear safe and well tolerated, both immunologically and neurodevelopmentally for the children. No immediate or long-term side effects were observed during the 1-year follow-up period studied. Equally, no apparent immunologically adverse reactions were noted during the time of these evaluations. Given the young age of our autistic patients, intravenous and subcutaneous routes of administration were chosen to be less invasive than intrathecal transplantation. It has already been demonstrated that intravenous injection is a safe and efficacious route for cell therapy in autistic children (14). As discussed below, FSCs could exert their function through paracrine and immunomodulatory effects. It has been already demonstrated that after systemic intravenous injection, stem cells show a high degree of chemotaxis based on proinflammatory cytokines (16). In addition, the clinical trial protocols currently performed have been using this route more effectively (www.clinicaltrials.gov).
Indeed, substantial clinical benefits from the intravenous delivery of stem cells have been observed (7). In a preclinical experimental model of neurodegeneration, this route of stem cell administration was verified as safe and efficacious (28).
Furthermore, transplanted stem cells are able to migrate into the CNS without loss of their immunological important regulatory capacity (22). They are able to move from the blood into the brain or the spinal cord through the blood– brain barrier (BBB) (10), where they can exert their therapeutic influences. Interestingly, it has been demonstrated that autistic patients show an increase in the permeability of the BBB (33). This endovascular dysfunction may further represent a therapeutic target of FSCs, since they have the potential to restore proper BBB characteristics (26). Subcutaneous administration was envisaged as a methodology to allow protracted stem cell survival of the neuroprogenitor cell transplantation. In the subcutaneous adipose tissue, the fetal neuroprogenitor cells can replicate and migrate into the surrounding vessels where they may traffic to the CNS. They can also release their related biopharmaceutical paracrine activity for extended periods of time. These compounds are likely transported via the lymph into the central vascular circulation where they may influence the CNS. Intravenous, as well as subcutaneous, administration of FSCs permits stem cells to circulate in the bloodstream, where disease-associated chemical signals could assist in the homing of the FSCs to damaged sites (32).
As discussed, a likely mechanism of FSCs is exerted through their paracrine effects. Among stem cells, they demonstrate an enhanced trophic influence on host tissues (27). They are able to secrete diffusible neurotrophic and several growth factors, which could influence the autistic central nervous system (8). It has been hypothesized that in ASDs, cellular transplantation is able to restore the damaged specialized neural systems by neuroprotection, neural circuit reconstruction, neural plasticity, neurogenesis, and immunomodulation (21).
FSCs do not cause rejection or graft-versus-host responses (1,18), which allows for their successful acceptance and functioning in the body of the recipient. This important fact could be explained by immaturity or inactivity of the fetal immune system. Definite expression of HLA-II molecules (which would be associated with rejection mechanisms) normally begins around the 14th week of gestation (34). Prior to the 10th week of gestation, this expression is either very weak or absent. Indeed, in the cell suspensions utilized, HLA-II-expressing cells are either absent or present in a very low quantity, such as leucocytes, vascular endothelium, and dendritic cells (34). Thus, FSCT requires no antirejection medication.
The ultimate fate of the transplanted FSCs is uncertain. They are a few possibilities: 1) remain as FSCs and persist in that state within the recipient, 2) mature into adult cells expressing HLA-II antigens, and either a) form a stable chimeric relationship with the recipient or b) are rejected by the host's immune system. In the presence of an intact host immune system, a graft-versus-host reaction is extremely unlikely and has not been observed by the EmCell team in the past 20 years.
Conclusions
This pilot study is a preliminary demonstration of the safety and efficacy of FSCs in autism. Our research has shown that FSCT, irrespective of ASD severity, favorably influences the course of children's development and their immunological markers. The procedure of cell transplantation is simple and minimally invasive. In order to further enhance stem cell effects, we recommend combined therapies where necessary: special diets, sensory integration, and behavioral therapy. How FSCs work in ASDs is incompletely understood and needs to be more fully investigated. These future research studies are urgently needed and larger randomized, placebo-controlled trials are needed to further characterize potential FSC-associated improvements in ASDs.
Footnotes
Acknowledgments
The authors N.S., M.K., O.I., I.M., and M.D. are EmCell employees. Authors declare no other conflicts of interest.
