Abstract
Tissue-specific stem cells found in adult tissues can participate in the repair process following injury. However, adult tissues, such as articular cartilage and intervertebral disc, have low regeneration capacity, whereas fetal tissues, such as articular cartilage, show high regeneration ability. The presence of fetal stem cells in fetal cartilaginous tissues and their involvement in the regeneration of fetal cartilage is unknown. The aim of the study was to assess the chondrogenic differentiation and the plasticity of fetal cartilaginous cells. We compared the TGF-β3-induced chondrogenic differentiation of human fetal cells isolated from spine and cartilage tissues to that of human bone marrow stromal cells (BMSC). Stem cell surface markers and adipogenic and osteogenic plasticity of the two fetal cell types were also assessed. TGF-β3 stimulation of fetal cells cultured in high cell density led to the production of aggrecan, type I and II collagens, and variable levels of type X collagen. Although fetal cells showed the same pattern of surface stem cell markers as BMSCs, both type of fetal cells had lower adipogenic and osteogenic differentiation capacity than BMSCs. Fetal cells from femoral head showed higher adipogenic differentiation than fetal cells from spine. These results show that fetal cells are already differentiated cells and may be a good compromise between stem cells and adult tissue cells for a cell-based therapy.
Introduction
Degeneration of cartilaginous tissues such as articular cartilage and intervertebral disc leads to disability, decreased quality of life, and higher medical costs. Cell-based therapies for cartilaginous tissue regeneration are an attractive prospective as they allow potential formation of a functional tissue. Autologous cell transplantation has first been investigated for immunocompatibility reasons. Autologous chondrocyte transplantation is clinically applied for full-thickness cartilage lesions and autologous disc cell transplantation has also been effected in clinical trials (6,7,28,33). Although these methods achieve good clinical results, limitations regarding the availability of the cells, their in vitro amplification, as well as their regeneration capacity can hamper the feasibility of these techniques (11,16,25).
The use of mesenchymal stem cells (MSC) opened new opportunities in treating different tissue such as nerve (46), bone (47), or for cartilaginous tissue regeneration (27,42,43). MSCs can be retrieved from bone marrow or adipose tissue with minimally invasive procedures and can be differentiated in vitro towards chondrogenic, osteogenic, or adipogenic phenotypes (2,18,44). Adult MSCs can only be differentiated into a limited number of phenotypes, whereas embryonic stem cells can virtually be differentiated into all phenotypes. Transition from embryonic to adult stage involves the proliferation and the specialization of embryonic cells under the control of morphogens. As cells become specialized, they lose their differentiation potential. The presence of tissue-specific stem cells in various adult tissues shows that unspecialized cells remain in adult tissues (36). In case of tissue injury, these cells can proliferate, differentiate, and regenerate the damaged tissue. However, the percentage of tissue-specific stem cells has been shown to decrease with age, concomitantly to regeneration potential of these tissues (9).
Fetal tissues, including articular cartilage, have high regeneration capacity, which seems inherent to fetal cells and not dependant on the local environment in the womb (26,31,38). Specific mechanisms involved in fetal cartilage regeneration are still largely unknown. Particularly, the presence of fetal stem cells in fetal cartilage and their involvement in the fetal regeneration process is unknown. Fetal stem cells have already been isolated from fetal tissues such as bone marrow, lung, liver, spleen, and umbilical cord (8,22,23). In addition, as fetal cells are in an intermediate stage of development between undifferentiated embryonic cells and specialized adult cells, questions remain about their degree of differentiation and their plasticity potential, that is to say that their capacity to transdifferentiate into multiple lineages. Fetal tissues offer very low immunogenic properties, which are closely dependent on the organ and gestational age (15). As for fetal tissues, fetal cells showed immunogenic properties, which can be partially explained by a low level of expression of HLA-I and the absence of HLA-II expression (1,17,20,29). As fetal cells offer low immunogenicity, good regeneration properties, and are readily cultured in vitro, they are good candidates to be used for a cell-based therapy (15,20,34,35).
Thus, the aim of this study was to assess the chondrogenic differentiation of fetal cells isolated from two fetal cartilaginous tissues, fetal femoral head, and fetal spine. Plasticity of these cells was assessed by analyzing their stem cell surface markers and their adipogenic and osteogenic differentiation potential.
Materials and Methods
Cell Isolation and Culture of Fetal Cells
Biopsies for cell isolation were obtained in accordance with the Ethics Committee of University Hospital in Lausanne (Ethical Protocol 51/01). Human fetal spinal column tissues (1 cm length, n = 4: FS12w, FS13w, FS14w, FS16w) and human fetal femurs (n = 3, FC14w, FC16w-a, FC16w-b) were obtained from fetuses after voluntary interruption of pregnancy at 12–16 weeks of gestation. Biopsies were first rinsed with 1% penicillin/streptomycin/PBS, cleaned from adjacent tissue, and minced in small pieces. Fetal spine units, composed of one intervertebral disc and the two adjacent vertebrae, were minced in small pieces with a scalpel and put into culture in DMEM (Gibco, Switzerland) supplemented with 10% fetal bovine serum (FBS) and 1% 100 mM l-glutamine (Gibco) (hereafter referred to as growth medium) at 37°C under 5% CO2. Femoral heads of fetal femurs were isolated, minced in small pieces with a scalpel, and put into culture in the same conditions as for fetal spine cells. For both tissues, cells migrated from the minced pieces, adhered to the plastic dishes, and began to proliferate. When primary cultures reached 80% confluency, cells were detached with trypsin/EDTA and plated in T75 flask in growth medium for further cell expansion (passage 1). When cells reached 80% confluency, cells were detached by trypsin/EDTA, mixed together to ensure consistency, and stored in frozen vials of cells at passage 1. Thus, a dedicated and consistent cell bank was established from each biopsy as described in De Buys Roessingh et al. (12) and Quintin et al. (35). For experiments, frozen vials were thawed and cells were expanded in monolayer and used for experiments at passage 4 unless otherwise stated. Human bone marrow stromal cells (BMSC) isolated from iliac crest bone marrow by plastic adherence selection (n = 2, aged 19 and 28 years) were kindly provided by Dr. Ivan Martin from Basel University. MSCs were cultured in MSC medium consisting of α-MEM (Gibco) supplemented with 10% FBS, 1% 100 mM l-glutamine, and freshly added 10 ng/ml fibroblast growth factor (FGF; Sigma, Switzerland). Cells were used in experiments between passages 3 and 6.
FACS Analysis of Surface Markers
Fetal cells at passage 0 were thawed and cultured in either growth medium or MSC medium at passage 1. At 80% confluency, cells were trypsinized and analyzed for the following surface markers: CD34, CD44, CD45-PE, CD73-PE, CD105-fluorescein, CD166-PE (all from R&D Systems, Minneapolis, MN, USA, except CD34 and CD73 from BD Bioscience Europe, Erembodegem, Belgium). First, cells were washed in ice-cold PBS and then incubated with primary antibody in PBS with 2% FBS for 30 min at 4°C. For CD34 and CD44 staining, bound antibodies were visualized by a 30-min incubation at 4°C with a secondary goat anti-mouse IgG-FITC labeled antibody (Invitrogen, Switzerland). After ice-cold PBS washing, cells were fixed with 4% PAF at 4°C for 15 min and analyzed with a CyAN ADP Analyser (DaKoCytomation, Switzerland) within 24 h. Controls were performed by substituting primary antibody by isotype-PE antibody for direct staining and by omitting primary antibody for indirect staining. Percentage of positive cells was calculated by subtracting control from sample with the Summit software (DaKoCytomation, Switzerland).
Chondrogenic Differentiation
Four hundred thousand fetal cells or BMSCs were centrifuged at 400 x g for 5 min to obtain cell pellets. Pellets were cultured in chondrogenic medium for 28 days with renewal of medium twice a week. Chondrogenic medium was made of DMEM supplemented with 1% of 100 mM l-glutamine, 1x insulin-transferrin-selenium (ITS), 10 mM sodium pyruvate, 40 μg/ml l-proline, 50 μg/ml ascorbic acid, and 10 ng/ml transforming growth factor-3 (TGF-β3). At the end of the culture period, pellets were fixed with 4% PAF, dehydrated through 70%, 80%, 94%, and 100% ethanol and xylol. Pellets were embedded in paraffin and 7-μm sections were cut.
Histology of Pellet
Deparafinized and rehydrated sections of pellets were stained for 15 min in Weigert's hematoxylin, stained for 10 min in Alcian Blue (pH 2.5) and molybdic acid for 10 min, and in Sirius Red for 5 min.
Immunohistochemistry of Pellet
Pellet sections were digested with hyaluronidase (Sigma, Switzerland) for aggrecan staining and with pepsin (Sigma) for type I, II, and X collagen stainings. After washing, sections were treated with 0.1% phenyl-hydrazine (Merck, Switzerland), washed, and blocked in PBS containing Triton X-100, FBS, and normal goat serum. Sections were then incubated with primary antibodies, aggrecan (1/2000; Biosource, Belgium), type I collagen (5 μg/ml; MP Biomedicals, Switzerland), type II collagen (1 μg/ml; Chemicon, Switzerland), and type X collagen (1/250; Sigma, Switzerland), then washed and incubated with goat anti-mouse biotinylated secondary antibody (Vector Laboratories, USA). After washing, sections were treated with Vectastain ABC kit and DAB revelation system (Vector Laboratories). Sections were counterstained with Fast Red, dehydrated, and mounted for microscopic observation.
Adipogenic Differentiation
Cells were cultured in growth or MSC medium for fetal cells and MSC medium only for MSCs. At confluency, cells were treated with growth medium with ITS, 1 μM dexamethasone, 0.1 mM indomethacin, and 0.5 mM isobutylmethylxanthine. Media was changed twice a week during 21 days. Then cells were washed with PBS, fixed in 4% PAF, and stained with Oil Red and counterstained with Mayer hematoxylin.
Osteogenic Differentiation
Cells were cultured in growth or MSC medium for fetal cells and in MSC medium only for MSCs until confluency. Cells were then cultured in growth medium with 10 mM phosphoglycerol, 200 mg/ml ascorbic acid, and 100 nM dexamethasone for 28 days. At the end of the culture period, cells were washed with PBS, fixed in 4% PAF, washed with distilled water, and stained with 2% silver nitrate, washed with water, and then with sodium thiosulfate.
Results
Chondrogenic Differentiation
Stimulation of BMSCs, fetal spine cells, and femoral head cells by chondrogenic media led to the production of sulfate-rich matrix as shown by the positive alcian blue staining (Fig. 1). Aggrecan was largely produced by BMSCs, fetal spine cells, and femoral head cells (Fig. 1b). BMSCs produced type II collagen in a specific region of the pellet, whereas fetal spine and femoral head cells produced type II collagen homogenously throughout the whole pellet (Fig. 1c). However, two fetal spine cell donors (FS12w and FS13w) produced very low level or no type II collagen at all. BMSCs produced type I collagen in the whole pellet but a slightly more intense staining was observed in the type II collagen-positive region (Fig. 1d). Type I collagen was strongly produced by three donors of fetal spine cells (FS12w, FS13w, FS15w) but the fourth donor (FS14w) showed a lower production of type I collagen. All three donors of fetal femoral head cells produced type I collagen. In both types of fetal cells, a more intense staining for type I collagen was observed at the periphery of the pellets. Type X collagen was not detected in the BMSC pellets. Fetal spine cell donors were heterogenous for type X collagen production. Type X collagen was detected in the extracellular matrix at a low level in two fetal spine donors (FS12w, FS14w) and it was only detected at the cellular level in the the other fetal spine cell donors (FS13w, FS15w). On the other hand, fetal femoral head cells showed a high level of type X collagen expression localized at the cellular and matrix levels. Presence of enlarged chondrocytes was observed in fetal spine and femoral head pellets.

Chondrogenic differentiation of BMSCs, fetal spine cells, and fetal femoral head cells. (a) Alcian blue-Weigert-Siruis Red, (b) aggrecan, (c) type II collagen, (d) type I collagen, and (e) type X collagen stainings. When heterogenous results were obtained among fetal spine or femoral head cells, lower and higher staining can be seen. Scale bars: 200 μm.
Expression of Surface Markers by Fetal Spine and Femoral Head Cells at Passage 1
BMSCs cultured in MSC medium were positive for CD44 (99.2%), CD73 (98.8%; 99.0%), CD105 (76.9%; 93.7%), and CD166 (99.4%) and negative for CD34 (2.9%; 5.1%), CD45 (0.2%; 0.4%) (Fig. 2, Table 1). Fetal spine and femoral head cells cultured in growth medium showed the same expression pattern and were positive for CD44 (95.0 ± 2.8; 97.8 ± 0.3), CD73 (98.8 ± 0.8; 98.8 ± 0.2), CD105 (79.1 ± 13.7; 86.7 ± 3.8), and CD166 (99.1 ± 0.3; 98.9 ± 0.9) and negative for CD34 (3.9 ± 0.9; 3.9 ± 0.3) and CD45 (2.8 ± 1.2; 1.7 ± 1.5). When cultured in MSC media, both type of fetal cells had the same level of expression of surface markers as that of in growth media (Table 1).

Representative FACS analysis of CD34, CD44, CD45, CD73, CD105, and CD166 expression of (a) BMSCs, (b) fetal spine cells at passage 1, and (c) fetal femoral head cells at passage 1. Negative control: gray curve, sample: white curve, positive cells: dashed curve.
Percentage of Positive Cells for CD34, CD44, CD45, CD73, CD105, and CD166 Expression of BMSCs (n = 2), Fetal Spine Cells (n = 4) at Passage 1 in Growth Media and MSC Media, and Fetal Femoral Head Cells (n = 3) at Passage 1 in Growth Media and in MSC Media

Adipogenic Differentiation of Fetal Cells
Fetal cells were cultured in growth or MSC media and then treated with adipogenic media. BMSCs showed a high number of adipose vacuole-positive cells, whereas fetal spine cells in growth or BMSC media showed little production of small lipidic vacuoles (Fig. 3Aa, b, d). Fetal femoral head cells showed more cells positive for lipidic vacuoles and the size of lipidic vacuoles was much larger than in fetal spine cells (Fig. 3Aa, c, e). For both types of fetal cells, no difference in the adipogenic differentiation was observed between growth and BMSC media conditions. Similar results were obtained when fetal cells were cultured in DMEM up to passage 4 and then treated with adipogenic media (Fig. 3Af, g).
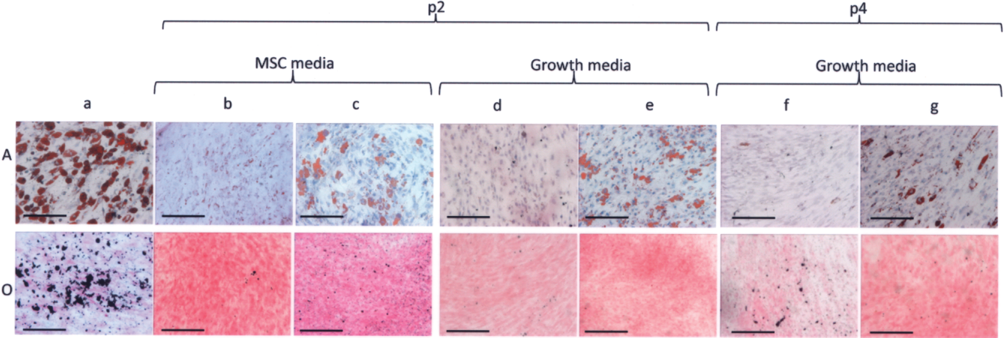
Adipogenic (A) and osteogenic (O) differentiation of (a) BMSCs, (b) fetal spine cells at passage 2 in MSC media, (c) fetal femoral head cells at passage 2 in MSC media, (d) fetal spine cells at passage 2 in growth media, (e) fetal femoral head cells at passage 2 in growth media, (f) fetal spine cells at passage 4 in growth media, and g) fetal femoral head cells at passage 4 in growth media. Scale bars: 200 μm.
Osteogenic Differentiation
Fetal cells were cultured in MSC media and then treated with osteogenic media. Fetal spine and femoral head cells produced very low levels of calcium salts compared with BMSCs (Fig. 3Oa–e). No difference was observed between fetal spine and femoral head cells. Similar results were obtained when fetal cells were cultured in DMEM up to passage 4 and then treated with osteogenic media (Fig. 3Of, g).
Discussion
In this study, we first examined the TGF-β3-induced chondrogenic potential of cell populations isolated from fetal spine and fetal femoral head. As fetal cells were isolated from cartilaginous tissues, it was expected that they show high chondrogenic property. Alcian blue and type II collagen staining indicated chondrogenic differentiation for BMSCs and fetal cells. However, type II collagen production by BMSCs was spatially restricted whereas it was homogenous in type II-positive fetal cell pellets. BMSC populations may be heterogenous for type II collagen production or more sensitive to local mechanical and/or physicochemical conditions than fetal cells. Unexpectedly, type X collagen was not detected in BMSC pellets as previously described. Chondrogenic stimulation of MSCs is usually achieved by TGF-β1 or -β3 and dexamethasone stimulation in high cell mass culture (4,21,24,32,45). Although now debated, in this model, type X collagen is sequentially induced after induction of type II collagen (4,30,32). It is possible that, in our model, TGF-β3 stimulation was less potent than in combination with dexamethasone to induce chondrogenic differentiation of BMSCs. On the other hand, fetal spine and femoral head cells may have been more responsive to chondrogenic stimulation as they are likely to be more differentiated than BMSCs. Longer term chondrogenic culture and type X expression time course could give additional information on the development of a hypertrophic phenotype. Fetal femoral head cells expressed high level of type X collagen, which strongly suggests that chondrogenic medium induces hypertrophic chondrocyte formation and raises the risk of in vivo endochondral ossification. This result could be explained by the fact that cells from the secondary ossification center of the fetal femoral head may have been isolated together with cells from the articular cartilage.
Induction of hypertrophy may also be due to the use of specific in vitro culture conditions (high cell density, nonphysiological growth factor concentration), and ossification of these cells should be tested in vivo using undifferentiated and differentiated cells. On the other hand, fetal spine cells showed very little production of type X collagen. However, cellular positive staining could be observed in two fetal spine cell donors (FS13w, FS15w). These cells may be at an earlier stage where type X collagen has not yet been exported into the extracellular compartment, which would indicate that these cells are also directed toward endochondral ossification. Only the FS14w cells showed very low level of type X collagen. This difference may be due to the age of the donor or to a specific cell population. The identification of the cell types present in the fetal spinal cell population would help to answer this question. Unfortunately, specific markers for identification of these cell populations are not available. The potential clinical use of fetal spine cells will not require in vitro differentiation and the use of growth factor-driven differentiation, possibly alleviating the risk of type X collagen production and endochondral ossification.
In a second part, we examined the plasticity of fetal cells. Stem cell surface marker expression, and adipogenic and osteogenic differentiation by fetal cells were analyzed and compared to those of BMSCs. BMSCs can be characterized by their ability to self-renew and to differentiate into multiple lineages. A panel of surface markers has been developed to isolate and characterize BMSCs, notably the expression of CD73, CD105, and CD166 and the absence of CD34 and CD45. However, the presence of specific combinations of surface markers is not enough to predict differentiation capacity and differentiation protocols are required (14,19). Fetal cells exhibited similar expression of the studied surface markers as BMSCs. However, fetal cells had lower adipogenic and osteogenic potential than BMSCs. Culture of MSCs requires specific culture conditions, such as the presence of FGF, to preserve their multilineage differentiation property whereas fetal cells are routinely cultured in growth media, which does not contain any growth factor (5,37,41). Thus, fetal cells were also cultured in MSC media to see if the presence of FGF had an effect on fetal cell differentiation potential. No effect of FGF on adipogenic nor for osteogenic differentiation was observed, confirming the observed low plasticity of fetal cells cultured in growth media.
Although additional stem cell surface markers can be used to refine fetal cell population characterization, these results strengthen the fact that surface marker characterization is not enough to predict multilineage differentiation potential of cells. In the same way, in't Anker et al. showed that isolated fetal MSCs from bone marrow, liver, lung, and spleen exhibited similar surface marker expressions but different levels of adipogenic and osteogenic differentiation (22). We also noticed that the two sources of fetal cells had different differentiation potential. Fetal femoral head cells showed a higher adipogenic differentiation potential than fetal spine cells. Cui et al. reported the presence of MSC-like cells in fetal articular chondrocyte population (10). These MSC-like cells could be differentiated into adipose, osteogenic, and neural cells. However, the percentage of differentiated cells was low, likely indicating that most of the cell population did not show plasticity properties. A very interesting series of papers demonstrated that adult articular chondrocyte showed plastic property, and that plasticity increased with passage and time in culture, that is to say with dedifferentiation level (3,13,39,40). However, we did not observe an increase plasticity property of fetal femoral head cells with from passage 2 to 4. We did not do later passages for these cells because for potential clinical application, these cells would not normally be used after passage 4, but it cannot be excluded that higher passages of fetal femoral head cells would show higher osteogenic and adipogenic differentiation properties.
Further studies, such as clonal selection, are needed to determine if differentiation properties are due to MSC-like cells or to subpopulation of fetal femoral head cells with plastic property. Interestingly, very low levels of adipogenic differentiation were seen with fetal spine cells. These cells have been isolated from fetal spine tissues and are likely to be a mixed population of fibroblastic and chondrocytic cells from fetal intervertebral disc annulus fibrosus, endplate, and vertebral body. This difference in plasticity level between spine and femoral head cells may be related to the origin of the cells. Although fetal spine cells may contain fibroblastic and chondrocytic cells from the annulus fibrosus, endplates, and vertebral body, a homogenous cell population for all surface markers was seen during the FACS analysis of the fetal spine cells.
In conclusion, chondrogenic stimulation by TGF-β3 of fetal cells isolated from fetal spine and femoral head led to the expression of aggrecan and type II collagen but also of type I and type X collagens. Although fetal cells exhibited a similar expression of stem cell markers as BMSCs, they showed low plasticity under adipogenic and osteogenic stimulation. As fetal spine cells showed lower plasticity and lower endochondral marker expression than fetal femoral head cells, it would be worth assessing their chondrogenic and regeneration potential in vivo.
Footnotes
Acknowledgments
This project was supported by the AO Research Fund 04–533, the Inter-institutional Center for Translational Biomechanics EPFL-CHUV-DAL, and the Inverni foundation.
