Abstract
This study aimed to enhance the attachment of periodontal ligament stem cells (PDLSCs) onto the decellularized dental root surface using surface coating with fibronectin and/or calcium phosphate (CaP) and to evaluate the activity of PDLSCs attached to a coated dental root surface following tooth replantation. PDLSCs were isolated from five dogs, and the other dental roots were used as a scaffold for carrying PDLSCs and then assigned to one of four groups according to whether their surface was coated with CaP, fibronectin, CaP/fibronectin, or left uncoated (control). Fibronectin increased the adhesion of PDLSCs onto dental root surfaces compared to both the control and CaP-coated groups, and simultaneous surface coating with CaP and fibronectin significantly accelerated and increased PDLSC adhesion compared to the fibronectin-only group. On in vivo tooth replantation, functionally oriented periodontal new attachment was observed on the CaP/fibronectin-coated dental roots to which autologous PDLSCs had adhered, while in the control condition, dental root replantation was associated only with root resorption and ankylosis along the entire root length. CaP and fibronectin synergistically enhanced the attachment of PDLSCs onto dental root surfaces, and autologous PDLSCs could produce de novo periodontal new attachment in an experimental in vivo model.
Introduction
During the past decades, mesenchymal stem cells (MSCs) have received both scientific and clinical research attention due to their potential for self-renewal and differentiation into various other tissues (4,8,33). Although the pluripotency of MSCs is a subject of some debate (2), tissue stem cells (which can be observed and cultured from various postnatal tissues) are clearly able to differentiate into several specific types of cells to produce tissue that is the same as that from which they originated (2,17). Therefore, stem cells from various tissue types have been isolated and studied in many research fields, including several types of dental stem cell from dental pulp, dental papilla, and periodontal ligament (PDL) (2,17,18). These dental stem cells are expected to provide or aid the bona fide regeneration of dental pulp and PDL tissues.
In the field of regenerative medicine, clinical techniques based around the regenerative potential of PDL cells, known as “guided tissue regeneration,” have been used for periodontal regeneration over several decades (11,20,37). However, two major limitations have meant that clinically successful results have not been achieved in most clinical cases: (1) the minimal regeneration of specific tissue (ligament and cementum), which is caused by limited participation of intact PDL cells from the adjacent tissue to the defect area (22,41) and (2) the significantly unsteady attachment between the regenerated specific tissues (PDL and cementum) or between the regenerated tissue (cementum) and the dental root surface(3,22).
The PDL is a unique tissue that connects and adheres the alveolar bone and the tooth via a periodontal attachment, which comprises densely organized PDL fibers inserted into the cementum and dentin of dental roots (Sharpey's fibers). This adherence between these periodontal structures plays an important role in PDL function, as well as the characteristics of each structure. However, insertion of functionally oriented PDL fibers into the dental root surface is observed less in regenerated PDL tissues than in the pristine periodontal apparatus (22). To increase insertion of functionally oriented PDL fibers (attachment), cell-to-root surface interaction would be a key factor in the healing processes, which includes attachment, proliferation, differentiation, functional arrangement, and peripheral mineralization of collagen fibrils onto the dental root surfaces.
Various approaches using periodontal ligament stem cells (PDLSCs) have recently been applied for periodontal regeneration in research studies (18,27,28,32). Cultured PDLSCs were transplanted directly into the experimental periodontal defect with or without a biomaterial scaffold (32,42) or indirectly by applying collagen/ gelatin-containing stem cells to destroyed dental root surfaces (15,40). In addition, a multilayered stem cell sheet wrapped around the natural/engineered tooth surface has been replanted in an extracted socket in some recent studies (14,38). However, these studies did not reveal any evidence of cell attachment onto the dental root surface, which is the first step in the formation of a periodontal attachment. Therefore, whether the applied stem cells survive and participate in various cellular events or immediately disappear in the in vivo environment has yet to be established.
The present study focused on the attachment of PDLSCs onto the dental root surface, as a scaffold for carrying and maintaining the cells for in vivo transplantation. Cells adhere to the surface of other cells or tissue membranes via binding between cell adhesion molecules (CAM) or the extracellular matrix (ECM) (9,25,43). In the in vitro condition, fibronectin enhances cell attachment/ migration (19,23,43) and the control of divalent cations, such as calcium and magnesium, facilitates cell adhesion (12,30,43). The aims of this study were thus to enhance the attachment of PDLSCs onto decellularized dental root surface using surface coating with fibronectin and/ or calcium phosphate (CaP) and to evaluate the activity of autologous PDLSCs attached to a coated dental root surface following tooth replantation.
Materials and Methods
Animals
Five male Beagle dogs (approximately 15 months old and weighing 9 kg; OrientBio, Seongnam, Korea) that had been bred exclusively for biomedical research purposes were used. The animals possessed an intact dentition with a healthy periodontium. Animal selection and management, the surgical protocol, and preparation followed routines approved by the Institutional Animal Care and Use Committee of Yonsei Medical Center, Seoul, Korea.
Autologous Dog PDLSC Culture
A slightly modified version of a previously reported protocol for human PDLSCs (34) was used to isolate and culture autologous dog PDLSCs (aD-PDLSCs). Maxillary molars were extracted gently after hemisection into two or four individual roots. The PDL tissues were separated from the root surface using a scalpel and minced into the smallest size possible. The minced PDL tissues were digested five times at 30-min intervals in α-minimum essential medium (α-MEM; Gibco, Invitrogen, Grand Island, NY, USA) containing 100 U/ml type I collagenase (Wako, Tokyo, Japan) and 2.5 U/ml dispase (Gibco) at 37°C. Single-cell suspensions were obtained by passing the mixture through a strainer with a pore size of 70 μm (Falcon; BD Labware, Franklin Lakes, NJ, USA), and 5 × 105 cells were seeded onto T75 cell-culture flasks (BD Biosciences, Franklin Lakes, NJ, USA) containing α-MEM supplemented with 15% fetal bovine serum (FBS; Gibco), 100 μM l-ascorbic acid 2-phosphate (Sigma-Aldrich, St. Louis, MO, USA), 2 μM l-glutamine (Gibco), 100 U/ml penicillin, and 100 μg/ml streptomycin (Gibco), and incubated at 37°C in an atmosphere containing 5% CO2. Single-cell colonies were observed after 3–7 days, and aD-PDLSCs at passage P0 were cultured. Cells at P3 or P4 were used for this study.
Characterization of Isolated aD-PDLSCs as MSCs
The basic characteristics of MSCs were determined using the following protocol.
Colony-Forming-Units Assay
Cells were plated at 1 × 103 cells/10 ml onto 100-mm culture dishes (BD Biosciences) with growth media, and the samples were fixed with 4% formaldehyde and then stained with crystal violet (Sigma-Aldrich). Cells were observed using light microscopy (CK40, Olympus Optical, Tokyo, Japan) at 14 days after seeding.
Osteogenic and Adipogenic Differentiation
Cells were seeded into six-well plates (BD Biosciences) at 1 × 105 cells/ well and cultured until they reached subconfluence. The culture medium for osteogenic differentiation comprised α-MEM (Gibco) containing 15% FBS (Gibco), 2 mM l-glutamine (Gibco), 100 mM l-ascorbic acid 2-phosphate, 10−8 M dexamethasone (Sigma-Aldrich), 2 mM β-glycerophosphate (Sigma-Aldrich), 55 mM 2-mercaptoethanol (AMRESCO, Solon, OH, USA), 1.8 mM KH2PO4 (Sigma-Aldrich), 100 U/ml penicillin (Gibco), and 100 mg/ml streptomycin (Gibco) and was refreshed at 3-day intervals. The culture medium for adipogenic differentiation comprised α-MEM containing 15% FBS, 2 mM l-glutamine, 100 mM l-ascorbic acid 2-phosphate, 500 mM isobutyl-methylxanthine (Sigma-Aldrich), 60 mM indomethacin (Sigma-Aldrich), 5 mM hydrocortisone (Sigma-Aldrich), 10 mM insulin (Sigma-Aldrich), 100 U/ml penicillin, and 100 mg/ml streptomycin. After 2 weeks (adipogenic differentiation) and 4 weeks (osteogenic differentiation) of induction, the cells were stained with Oil red O (Sigma-Aldrich) and Alizarin red (Sigma-Aldrich), respectively.
Fluorescence-Activated Cell Sorting Analysis (FACS)
aD-PDLSCs in T75 flasks were treated with trypsin-EDTA (Gibco). After the cells were harvested and transferred to a 1.7-ml tube (Oxygen, Union City, CA, USA), they were fixed by adding 4% paraformaldehyde (Santa Cruz Biotechnology, Dallas, TX, USA) for 15 min. The cells were then incubated for 1 h with 3% bovine serum albumin (Gibco) and then for a further 1 h with specific FITC- or PE-conjugated primary antibodies raised against CD14 (IM0645U; Beckman Coulter, Brea, CA, USA), CD34 (IM1870; Beckman Coulter), CD44 (555479; BD Pharmingen, San Jose, CA, USA), or CD90 (IM1839U; Beckman Coulter) with the concentration of 20 μl per 1 × 106 cells, washed with wash buffer (PBS; Gibco). After 45 min of incubation at room temperature, the cells were washed three times and then subjected to FACS using a flow cytometer (FACSCalibur; BD Biosciences).
Ectopic Transplantation
An in vivo transplantation assay was performed to evaluate the regenerative potential of aD-PDLSCs by ectopically implanting them into subcutaneous pockets on the dorsal region of immunodeficient mice (5-week-old, male, immunocompromised mice). aD-PDLSCs (P3), 6 × 106, were mixed with 80 mg of biphasic CaP (BCP; Biomatlante, Vigneux, France) particles. The cell mixtures were transplanted into four mice after incubation for 1.5 h; BCP particles alone were transplanted into the other four mice as a control experiment (n = 4 in each of the control and test groups). The animals were sacrificed after an 8-week healing period, and block sections were fixed in 4% neutral-buffered formalin (BBC Biochemical, Seattle, WA, USA). The specimens were decalcified with 5% EDTA (pH 7.2; Gibco) and then embedded in paraffin. The two central-most sections were cut at a thickness of 5 μm at 50-μm intervals and stained with hematoxylin and eosin (H&E; Sigma-Aldrich) and Masson's trichrome (Sigma-Aldrich).
Preparation of Decellularized Dental Roots
The maxillary premolars were extracted, and the separate dental roots were used as a decellularized scaffold in this study (Fig. 1). Cementum was not removed in order to mimic the clinical situation (16), and excess soft tissue tags were removed gently from the cervical area of the extracted dental roots, for which a root canal treatment was performed; the dental pulp was extirpated using barbed broaches (MANI, Tokyo, Japan) and Ni-Ti rotary instruments (Pathfile, Dentsply Maillefer, Ballaigues, Switzerland), and the roots were left empty. All experimental dental roots were sterilized with 12% ethylene oxide gas (Steri-Gas; 3M, St. Paul, MN, USA) at 54°C for 6 h, and then degassed for 12 h to remove any particles of ethylene oxide. The dental roots were demineralized, and their organic matrix was exposed by treatment with 5% EDTA solution (pH 7.4) (7) and then washed out vigorously with phosphate-buffered saline (PBS) for 5 min.

Schematic diagrams and clinical photographs of the in vivo and ex vivo surgical protocols. (A) Periodontal ligament (PDL) stem cells (PDLSCs) were isolated from canine maxillary molars, and their characterization as mesenchymal stem cells (MSCs) was confirmed. (B) The effect of dental root surface treatment on PDLSC adhesion onto decellularized dental root (C) was evaluated by dividing the samples into four groups according to the treatment type: control (no surface treatment), calcium phosphate (CaP) coating, fibronectin coating, and simultaneous CaP/fibronectin coating. (C) After decellularization, PDLSCs were cultured on CaP/fibronectin-coated dental roots with regular rolling during the first 6 h to ensure an even distribution of cell adhesion. (D) After 60 h of ex vivo PDLSC culture on extracted/decellularized dental roots, dental roots with PDLSCs were replanted following widening of recipient extraction socket.
Evaluation of the Effect of Dental Root Surface Treatment on aD-PDLSC Attachment
Thirty-six decellularized dental roots were included, with nine being assigned to each of the following four experimental groups with different types of surface treatment: control, fibronectin coating, CaP coating, and CaP/ fibronectin coating (Fig. 1). Fibronectin coating was achieved by soaking the prepared decellularized dental roots overnight in fibronectin solution from bovine plasma (F1141; Sigma-Aldrich) at a concentration of 20 μg/ml at 4°C, and then washing them with PBS. Coating with CaP was also performed by soaking the roots overnight in CaP solution, which was fabricated by pulverizing BCP particles and dissolving the powder in distilled water at a concentration of 100 mg/ml, with magnetic stirring for 12 h at room temperature. For simultaneous surface treatment, the decellularized roots were coated first with CaP and then with fibronectin.
The aD-PDLSCs were suspended at a density of 1.2 × 107 cells/ml, added to six-well plates with a prepared experimental dental root, and then incubated in an α-MEM/FBS culture medium at 37°C in an atmosphere containing 5% CO2 for a maximum of 3 days. Scanning electron microscopy (SEM) was employed to evaluate the growth and degree of attachment of aD-PDLSCs on the fibronectin-, CaP-, and CaP/fibrin-treated dental root surfaces. The samples were fixed at 1, 2, and 3 days after cell culture onto the dental root surface (n = 3 at each time point and for each of the four groups, yielding 12 sample types). After fixation using Karnovsky's glutaraldehyde (Sigma-Aldrich) for SEM, the samples were observed with a scanning electron microscope (S-300N; Hitachi, Japan).
Tooth Replantation with Cultured aD-PDLSCs on the CaP/Fibronectin-Coated Surface
Unilateral mandibular premolars in five dogs were included as the experimental group, which was subjected to the following procedures. On the first day of the experimental replantation process, the bilateral mandibular second and fourth premolars were extracted gently after hemisection into two individual roots. Root canal treatment and sterilization were performed as described above, and the crown portion of the tooth was removed. On the second day, simultaneous CaP/fibronectin surface treatments were performed over a 24-h period, and then aD-PDLSCs were cultured with the prepared experimental roots for 60 h. To ensure an even distribution of aD-PDLSCs on the entire surface of the dental roots, they were rolled regularly during the first 6 h of culture. On the fifth day, the prepared experimental roots were replanted after removal of blood clots from the extraction socket and preparation of the socket by slight removal of the socket wall (Fig. 1). A full mucoperiosteal flap was raised and advanced with a periosteal releasing incision and sutured using a monofilament nylon suture material (Monosyn; B. Braun, Melsungen, Germany). The contralateral mandibular premolars were used as the control condition and were treated using the same protocol as for the experimental group, with the exception of cell application. The animals received an intramuscular administration of a broad-spectrum antibiotic (20 mg/kg cefazoline sodium; Yuhan, Seoul, Korea) and daily topical application of a 0.2% chlorhexidine solution (Hexamedine; Bukwang Pharm Co., Seoul, Korea) for 14 days. The suture materials were removed 7 days after the surgery. The animals were sacrificed at 8 weeks postsurgery using an overdose of pentobarbital sodium (90–120 mg/kg, IV; Entobar; Hanlim Pharm Co., Seoul, Korea). Block sections, including the experimental sites, were fixed with 10% neutral-buffered formalin for 10 days, and then microcomputed tomography (micro-CT; Skyscan 1072; Skyscan, Aartselaar, Belgium) images of these block specimens were obtained at a resolution of 35 μm (using 100 kV and 100 μA). The scanned micro-CT images were processed in DICOM format and three-dimensionally reconstructed with PC-based software (On-Demand3D; Cybermed, Seoul, Korea). Eight to 12 serial sagittal tomographic sections were chosen at 300-μm intervals for each experimental tooth, and the proportions of the PDL space and ankylosis relative to the entire length of the root surface were determined. Histology processing was performed by the aforementioned procedures described for the ectopic transplantation assay. Twelve coronal histologic sections were acquired from the most buccal to lingual surfaces of experimental roots. Ten slides of sections were stained with H&E and Masson's trichrome, and another two slides were used for immunohistochemical staining of PDL markers (collagen III and periostin). Five H&E-stained slides were used for histomorphometric analysis of the proportions of PDL-like tissue with/without root resorption and ankylosis with root resorption relative to the entire length of the root surface. Anticollagen III (diluted 1:500; ab23445; Abcam, Cambridge MA, USA), and antiperiostin (diluted 1:200; ab14041; Abcam) antibodies were used, and a commercially available kit (SuperPicture polymer detection kit; Zymed/Invitrogen, Carlsbad, CA, USA) was used to detect the antibodies, in accordance with the manufacturer's protocol.
Statistical Analyses
The statistical analysis was performed using a standard software program (SPSS 15.0; SPSS, Chicago, IL, USA). Paired t-tests were used to evaluate the results from radiographic and histomorphometric analyses, with p < 0.05 used as the cutoff for statistical significance. Measured data are presented as mean ± SD values based on the animal means.
Results
Characterization of the aD-PDLSCs
Single-cell suspensions generated from dog PDL formed adherent clonogenic cell clusters of fibroblast-like cells after 7–14 days, which confirmed the capacity of putative stem cells in dog PDL. Long spindle-like fibroblastic cells were observed at 4–7 days after cell plating, and 76–114 single colonies were generated from 1 × 103 single cells (Fig. 2A, B). Although there was a tendency for the number of single colonies produced by cells to decrease as the number of passages increased, P4 cells also exhibited a high density of single colonies on culture plates. Differentiation assays demonstrated Alizarin red-positive mineral deposition and Oil red O-positive cells in the cell-seeded plates with osteogenic and adipogenic differentiation media, respectively, which showed evidence of multipotential differentiation of aD-PDLSCs (Fig. 2C, D). FACSs revealed that aD-PDLSCs were negative for hematopoietic markers (CD14 and CD34) but positive for MSC markers (CD44 and CD90) (Fig. 2E). The capacity of aD-PDLSCs to regenerate various tissues was observed in the in vivo ectopic transplantation model (Fig. 2F–K). Unidirectional PDL-like tissue and cementum-like mineralized tissue formation was observed on the surfaces of the biomaterial. Sharpey's fibers inserting into cementum-like tissue and cementoblasts scattered between inserting fibers were observed in the experimental transplanted area. Comparison with the control transplanted group (connective tissue capsulation by parallel-arranged transplanted particles, without any mineralized tissue formation) further confirmed the multipotency of the aD-PDLSCs in the in vivo model.

Characterization of PDLSCs as MSCs. (A, B) Colony-forming units assay revealed colony formation at 14 days after cell plating (stained with crystal violet). Although the colony formation decreased with increasing passage number [number of colonies were significantly decreased between passage 1 (P1) and P4; p = 0.02], the sizes of the colonies were similar from P1 to P4. Adipogenic (C) and osteogenic (D) differentiation were induced after 4 and 2 weeks, respectively. (E) Fluorescence-activated cell sorting analysis revealed positive findings for mesenchymal stem cell markers (CD44 and CD90; 97.97% and 51.30%, respectively), but negative findings for hematopoietic markers (CD14 and CD34; 0.05% in both). (F–K) Histology views of transplanted biphasic CaP (BCP) with/without PDLSCs revealed highly mineralized tissue (arrows) and PDL-like tissue formation by PDLSCs: (F) BCP alone, (G) BCP with PDLSCs [hematoxylin and eosin (H&E)]. While sites that received BCP alone exhibited grafted particles encapsulated by parallel-arranged connective tissue fibers (F, H), experimental sites that received BCP with PDLSCs exhibited unidirectionally arranged, PDL-like fibers inserting into the grafted particles (I), and cementum formation (J, K). (K) High-magnification view of the red box shown in panel (J), revealing Sharpey's fibers (arrowheads) inserted into cementum and cementoblasts between the fibers (H to K; Masson's trichrome stain). B, bone; C, cementum; PDL, periodontal ligament; PDLSC, periodontal ligament stem cell. Scale bar: 100 μm in (C), (D), and (H–K); 1 mm in (F) and (G).
Evaluation of aD-PDLSC Attachment Onto the Decellularized Dental Root Surface According to Surface Treatment Type (Fig. 3)
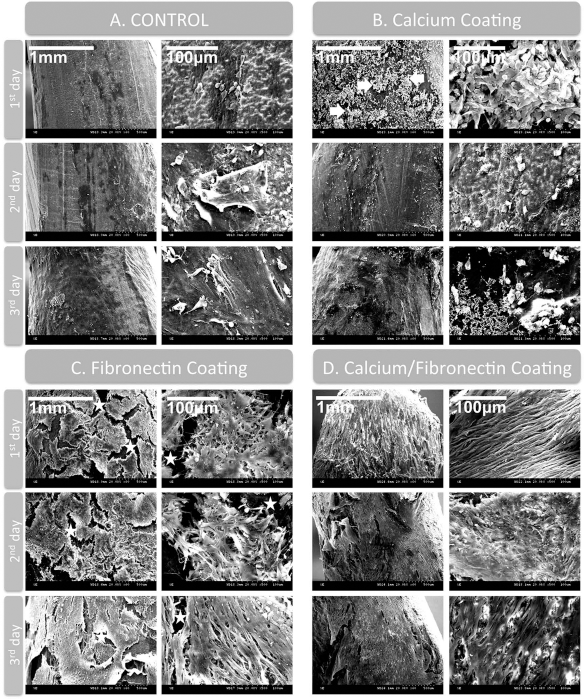
Scanning electron microscopic views of dental root surfaces treated with various experimental coating techniques [left and right columns in the same group: original magnifications of 60× (scale bar: 1 mm) and 500× (100 μm), respectively]. Limited PDLSC attachment was observed throughout the observation period (3 days) in the control (A) and CaP-coated (B) samples, despite CaP crystal formation on the CaP-coated dental root surface. Fibronectin-coated surfaces (C) exhibited significantly increased PDLSC attachment compared to the control and CaP-coated samples. A sheet-like PDLSC mass formed on the root surface from day 1 of observation, but denuded root surface was exposed at crevice-like fissures (asterisks). These fissures still remained, and margins of PDLSC mass were detached on the third day of observation, despite the increased cell mass. Surfaces coated with both CaP and fibronectin (D) exhibited a significantly increased cell mass compared to the fibronectin-coated sites. Consolidation of cell–cell interactions by calcium ions may have stabilized the cell mass formation onto the dental root surface; a sheet-like PDLSC mass was observed without any fissures from day 1 of observation, which suggests that the PDLSC mass that adheres to the root surface can be well maintained by coating the surface with CaP and fibronectin.
Decellularized dental root surfaces without any coating (control group) exhibited limited cell attachment throughout the 3-day observation period. Most of the surfaces were exposed, and a few cultured cells could be observed in a limited area but with a spheroidal shape rather than a spindle-like appearance. After 3 days of cell culture, some cells had produced cellular processes that attached to the root surface, but there were no more attached cells than after the first day of cell culture.
In the CaP-coated group, most of the dental root surface was covered with small particles of CaP crystals, and a few spheroidal-shaped cells could be also observed on day 1 of the experimental period. However, some of these CaP crystals had disappeared by day 2, and few cells were observed throughout the remainder of the observation period, with no increase in cell number.
In the fibronectin-coated group, there was a marked increase in cell attachments in a sheet-like state by day 1 of the observation period. High-magnification views demonstrated the presence of several layers of cells that had produced several cellular processes that spread multidirectionally and appeared to be contacting/adhering with other cells in either the same or other layers. By the second day, these cellular processes were stretched more widely, and the network of sheet-like cell mass was larger. By day 3, the cellular processes of the sheet-like cell mass stretched unidirectionally and formed a regularly patterned fabric-like structure. While crevice-like fissures could be observed on day 1, and the root surface was exposed at these fissures, these fissures reduced over time. Most of the root surface was covered with a sheetlike cell mass, but the fissures still remained by day 3.
Samples coated simultaneously with CaP and fibronectin exhibited significantly increased cell attachments compared to all of the other groups. At day 1 of observation, a sheet-like cell mass could be observed with no fissures or cellular detachment, and a fabric-like structure with unidirectionally stretching cellular processes was evident. Detachment of some layers of the sheet-like cell mass was observed in a limited area by days 2 and 3, but the dental root surfaces were not exposed because only partial layers appeared to be peeling off in these areas. Based on these in vitro results, root surfaces for in vivo tooth replantation were coated by CaP and fibronectin simultaneously.
In Vivo Tooth Replantation with aD-PDLSCs
In micro-CT images, most of the root surface (66.32 ± 10.05%) was in direct contact with the alveolar bone at control sites replanted with dental root only, whereas the experimental sites containing replanted dental roots with aD-PDLSC attachment exhibited a persistent (but reduced) PDL space (60.93 ± 5.15%), despite the presence of direct bony contact along a portion (39.07 ± 5.15%) of the same dental root (Table 1 and Fig. 4). In control sites, root resorption was found simultaneously with the bony ankylosis more often and extensively than in experimental sites (p < 0.01).
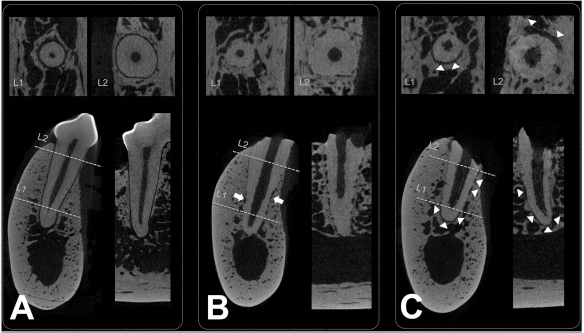
Microcomputed tomography views of experimentally replanted dental roots and the adjacent natural tooth. (A) The adjacent natural tooth: APDL space is evident between the alveolar bone and the tooth structure in all tomography planes (sagittal, lower left; coronal, lower right; axial, upper). L1 and L2 refer to the level of axial view (L1, apical one third; L2, coronal one third). (B) Control replanted dental root without PDLSCs: bony ankylosis can be observed along the entire length of the tooth surface. Simultaneously, extensive root resorption is found at the middle level (arrows). (C) A persistent PDL space could be observed in the experimental sites at which PDLSCs had adhered onto the CaP/fibronectin-coated surface of the dental root (arrowheads), although root resorption and ankylosis were evident in other areas of the same root surfaces.
Results of Morphometric Measurements in Radiographic and Histologic Analyses

Significantly different from control sites that received decellularized dental root only; aD-PDLSC(-)/p < 0.01. aD-PDLSC, autologous dog periodontal ligament stem cell.
Histologic analysis revealed a functionally oriented PDL-like tissue layer (periodontal attachment) between the alveolar bone and replanted dental root surface only at experimental sites where aD-PDLSCs had been ex vivo cultured on CaP/fibronectin-coated root surface (Figs. 5 and 6); control sites without any cell application exhibited irregularly resorbed root surfaces that were in direct contact with the alveolar bone, without any mediation of a PDL or PDL-like tissue layer (Fig. 5). Highly magnified views revealed that the PDL-like tissue included basic structures of PDL: obliquely/perpendicularly arranged collagen fibers, Sharpey's fiber insertion into both alveolar bone and newly formed cementum, and cells (Fig. 6); however, despite this, the width of the PDL space was significantly reduced. In addition, experimental sites demonstrated two types of PDL-like tissue. Most of the root surfaces contacting PDL-like tissue maintained their original shape, without any irregular border indicative of root resorption (Fig. 6B). Perpendicularly/obliquely arranged PDL collagen fibers were inserted into the newly formed cementum that had been deposited onto the old cementum. In contrast, some other parts of the PDL-like tissues remained as an extension of the above-mentioned PDL-like tissue despite resorption of the contacting root surface and having been encircled by newly formed bone (Fig. 6D). In the latter tissue type, there was a purple-colored layer including collagen fibers and cells maintained between both sides of mineralized alveolar bone. PDL-like tissue, contacting bone marrow instead of mineralized tissue, also maintained its own structures, even though several multinuclear cells emerged at the outermost surface (Fig. 6C). Immunohistochemistry with PDL-specific markers (collagen III and periostin) also revealed a dark-stained specific layer adjacent to the dental root surface and inserting collagen fibers (Fig. 6C, D). However, the PDL-like tissue was narrower and had fewer inserting Sharpey's fibers compared to the natural PDL in the adjacent tooth that had not received any experimental procedures.
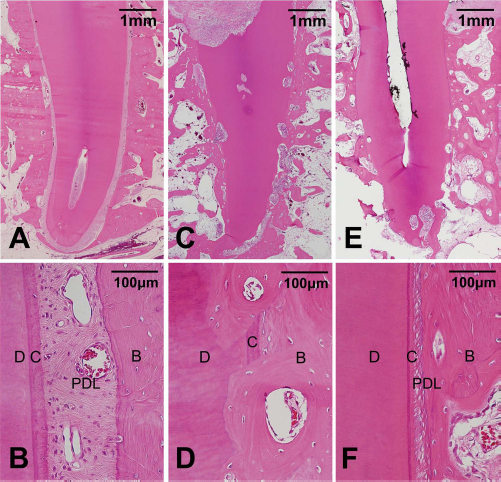
Histology photomicrographs of natural tooth (A, B) and replanted dental roots with/without PDLSCs (C–F). Histologic views of natural tooth demonstrate pristine PDL between cementum and alveolar bone (A, B). The histologic healing pattern differed significantly between sites that received dental roots with and without PDLSCs. Root resorption and ankylosis were observed at control sites along the entire root surface without any mediation of periodontal attachment (C, D). However, PDL-like tissue (PDL in F) comprising functionally oriented connective tissue fibers could be observed on most of the dental root surface at both low (E) and high (F) magnifications at experimental sites with PDLSCs. High-magnification views show direct contact between the alveolar bone and the dental root surface (D) and PDLSC-derived PDL-like tissue (F). B, bone; D, root dentin; C, cementum; PDL, PDL or PDL-like tissue. (H&E; scale bar: 1 mm in A, C, and E; 100 μm in B, D, and F).

High-magnification histology photomicrographs of CaP/fibronectin-coated dental roots with PDLSCs and the adjacent natural tooth. (A) Histology of the PDL of a natural tooth adjacent to an experimental site; dense PDL (Sharpey's) fibers can be seen inserting into both sides of the dental root and the alveolar bone (H&E, scale bar: 100 μm). (B) Functionally oriented PDL-like tissue at the experimental sites shows perpendicularly/obliquely arranged collagen fibers on the root surface and their insertion into both the root surface and alveolar bone (B), despite the tissue being narrower than the normal PDL shown in (A). PDLSC-derived PDL-like tissue was maintained in the area contacting bone marrow tissue (C) and even between bone tissues in some areas showing root resorption and ankylosis (D). PDL-like tissue was inserted into newly formed cementum that was deposited onto old cementum (B, E); newly formed cementum can be distinguished clearly in both normal light microscopy (B) and depolarized light microscopy (E). Putative PDLSCs can be observed in PDL-like tissue (F), and immunohistochemical staining against PDL-specific markers (collagen III and periostin) revealed PDL fibers (white arrowheads) inserting into dental root (G; collagen III), and confirmed the presence of a specific tissue layer on the dental root surface (H; periostin) in PDL-like tissue at the experimental sites. B, bone; BM, bone marrow tissue; C, cementum; D, root dentin; OC, old cementum; NC, newly formed cementum; PDL, PDL/PDL-like tissue. (A: H&E, scale bar: 100 μm; B to D: H&E; E: depolarized view/ Masson's trichrome; F: Masson's trichrome; G: collagen III; and H: periostin; scale bar: 50 μm in B to H).
Discussion
Over the past decade, many researchers have developed and studied bioengineered teeth by applying two types of developmental approach: whole-tooth regeneration using a concept of tooth development (1,29), and partial-tooth regeneration using a tooth-shaped scaffold and stem cells (36). Although bioengineered teeth produced using tooth germ exhibit complete regeneration of individual dental structures, there remain many biologic and technical hurdles before they can be applied clinically. However, the partial-regeneration approach, which has focused on the periodontal attachment between preexisting alveolar bone and the tooth scaffold, is expected to be an obtainable goal for clinical application, since it often requires only a small modification of the conventional treatment procedure. Sonoyama et al. bioengineered a functional tooth using a tooth-shaped ceramic scaffold and MSCs from the dental root apical papilla and provided evidence of dentin–PDL complex regeneration (36). However, there are several limitations with this system, including low mechanical strength, additional requirement of prosthetic treatment, and an unclear prognosis regarding resorption of the engineered scaffold.
Recent studies have tested the efficacy of natural teeth as a carrying scaffold for stem cells in bioengineering tooth development. Zhou et al. (44) replanted avulsed teeth wrapped in PDLSC sheets, and demonstrated reduction of dental root resorption compared to sites replanted with dental root only. Zhao et al. (42) transplanted PDLSC sheet fragments directly into the extraction socket before tooth replantation and showed evidence of periodontal regeneration. However, these studies applied a PDLSC mass in replanted extraction sockets without any adhesion onto the dental root. Since most of the cellular metabolism is based on cellular adhesion onto a substrate or other cells, reproducible roles of separately applied cell masses cannot be guaranteed for clinical application.
Dangaria et al. (7) indicated that the microtopography of natural teeth is an important feature for the application of PDLSCs in bioengineered tooth development. They demonstrated that a rough surface topography increased ECM signaling pathways related to the focal adhesion of cells and promoted the attachment of PDLSCs onto natural dental root surfaces. In the present study, a small number of aD-PDLSCs were attached to the rough surface of EDTA-treated dental roots. Despite evidence of aD-PDLSC attachment, decreased cell attachment was observed in the present study compared to the results of Dangaria et al. (7). This discrepancy may be attributable to interspecies differences in the dental composition or microstructure—Dangaria et al. used mice, while we used dogs as experimental subjects. However, the fibronectin surface coating resulted in significantly increased aDPDLSC attachment, and even a sheet-like cell mass was formed on the dental root surface. The addition of CaP simultaneously with fibronectin resulted in accelerated and significantly increased formation of a sheet-like cell mass that adhered onto the root surface. Given the finding of minimal cell attachment on dental root surfaces coated with CaP alone, this synergism might arise from acceleration or stabilization of fibronectin-induced aD-PDLSC attachment by CaP, rather than the sum of independent effects of both coating substances.
Cell adhesion is the first and essential step for normal cellular metabolism for cell survival and is mediated by CAM (10,43). It is known that CAM provides two-way targets and functions: cells adhere to a substrate or adjacent cells, and CAM engages mechanical attachment to both targets or intra-/intercellular signaling (13). A previous study has already demonstrated that impaired integrin function results in cells detaching from substrates and could trigger their apoptosis (9). The metabolism of pluripotent stem cells is also sensitive to both cell–cell and cell–substrate interactions via various CAM, with regard to both cell survival and self-renewal (24–26,39). Recent reviews have found that cell–ECM or cell–cell adhesion via various CAM makes the stimuli to intracellular signaling pathways and regulates the activities of growth factor receptors that play important roles in basic cellular metabolism and functions (43). An increase/stabilization of the attached sheet-like cell mass by the CaP surface coating observed in the present study may be attributable to consolidation of cell–cell interactions. Many types of CAM in cell–cell interactions have the characteristic of calcium-dependent activity (12,30,43), and thus a CaP coating on the root surfaces could provide calcium ions for these cell–cell interactions, which could strengthen the intercellular relationship between aD-PDLSCs attached to the root surface via fibronectin. Resultantly, both CaP and fibronectin could affect the acceleration of aD-PDLSC mass formation and be expected to survive transplanted in the in vivo environment.
The histology pattern observed in the present study differed significantly between experimental sites at which dental roots were replanted with aD-PDLSCs and control sites at which decellularized teeth were replanted. aD-PDLSC-derived functionally oriented periodontal new attachment was clearly evident between alveolar bone and the replanted root surface, with Sharpey's fibers inserting into both sides around experimental dental roots. Even in some areas that showed root resorption and bony ankylosis despite the application of aD-PDLSCs, the extensions of this PDL-like tissue layer maintained their own properties comprising PDL collagen fibers and replanted cells between newly formed bone tissues. It appears that the PDL-like tissue resisted overwhelming osteogenic induction by the surrounding tissues, which is a normal physiologic function of the PDL to maintain its space against the alveolar bone. These might have been caused by the self-renewal potential of the replanted aD-PDLSCs that were attached to the dental roots, given the results of the control group, in which ankylosis was observed along the entire dental root surface without any mediation of PDL/PDL-like tissue. Periodontal new attachment formation by aD-PDLSCs is in agreement with previous studies showing de novo formation of cementum and PDL by transplanted dental stem cells at ectopic experimental sites (21,31,35). While the previous studies demonstrated that the transplanted stem cells themselves had directly differentiated into cementum or PDL individually in ectopic models, in the present tooth replantation model, there was a functionally combined complex of periodontal attachment observed on the dental root surface to which aD-PDLSCs had been attached ex vivo.
The present study has demonstrated the possibilities of the in vivo application of ex vivo cultured stem cells in periodontal regeneration, as well as the limitations of partial regeneration of PDL and root resorption in some areas. The findings suggest that the application of these stem cells in clinical scenarios could result in enhancement of periodontal regeneration in cases of tooth avulsion and that this technique could be used during intentional tooth replantation or transplantation. Both of these clinical situations were mirrored in the present experimental protocols. However, the present results reveal the limitations of partial root resorption and ankylosis; these limitations could be explained by two factors. First, the loss of physiologic structure by the tooth socket (bundle bone for the PDL) that occurred after tooth extraction was followed by a vigorous healing/ remodeling phase (5,6). Not all of the attached aDPDLSCs on the root surface survived during this in vivo environment, and those that did survive failed to regenerate the original features of the PDL. Second, the cells that adhered onto root surfaces may have been distributed unevenly during the pretreatment cell application. To overcome this issue, the experimental protocols included regularly rolling the roots during the initial 6 h so that cultured cells could become evenly attached onto the root surfaces. However, during the later pretreatment period, the roots were cultured in a static state to enable the adhered cells to stabilize; this could have affected the formation of the cell mass on the upper- or floor-side surface. In the interests of attaining complete periodontal regeneration in the clinical setting, future studies should include advanced ex vivo periodontal tissue-engineering techniques focusing on PDLSC attachment and cell mass formation on the root surface.
Cell adhesion is the first critical step for the survival and correct functioning of mesenchymal stem cells in clinical applications, which prompted the present study to focus on increasing both the attachment of aD-PDLSCs onto the dental root surface by coating it with fibronectin and CaP and the in vivo periodontal regeneration by aD-PDLSCs carried by a decellularized natural tooth. The presence of CaP and fibronectin accelerated and increased the adhesion of aD-PDLSCs onto the decellularized dental root surface in vitro, and functionally oriented PDL-like tissue (de novo periodontal new attachment formation) was demonstrated in vivo in the tooth replantation model following ex vivo aD-PDLSC culturing with a CaP/fibronectin coating. The present results suggest that coating the root surface simultaneously with CaP and fibronectin may synergistically enhance the adhesion of PDLSCs onto the natural tooth surface, and ex vivo cultured autogenous PDLSCs carried by the CaP/fibronectin-coated natural tooth can produce de novo periodontal regeneration. These results demonstrate the possibilities of stem cell therapy in periodontal regeneration and indicate decellularized teeth to be an ideal biomaterial scaffold for bioengineered teeth.
Footnotes
Acknowledgments
This research was supported by the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT, and Future Planning (grant numbers: 2012M3A9B2052521 and 2012R1A2A4A01007124). The authors declare no conflicts of interest.
