Abstract
As a refractory fibrosis disease, intrauterine adhesions (IUAs) is defined as fibrosis of the physiological endometrium. Although hysteroscopic adhesiolysis is widely recommended as an effective treatment, prognosis and recurrence remain poor in severe cases. Recently, stem cell therapy has been promoted as a promising treatment for IUAs. The ability of human amniotic epithelial cells (hAECs), emerging as a new candidate for stem cell therapy, to treat IUAs has not been demonstrated. To study the potential effects of hAECs on IUAs, we created an IUA rat model using mechanical injury and injected cultured primary hAECs into the rats’ uteri. Next, we observed the morphological structure of endometrial thickness and glands using hematoxylin and eosin staining, and we detected extracellular-matrix collagen deposition using Masson staining. In addition, we performed immunohistochemical staining and reverse-transcription polymerase chain reaction (RT-PCR) to investigate potential fibrosis molecules and angiogenesis factors 7 d after hAECs transplantation. Finally, we detected estrogen receptor (ER) and growth factors via RT-PCR to verify the molecular mechanism underlying cell therapy. In the IUA rat models, endometrial thickness and endometrial glands proliferated and collagen deposition decreased significantly after hAEC transplantation. We found that during the recovery of injured endometrium, the crucial fibrosis marker transforming growth factor-β (TGF-β) was regulated and angiogenesis occurred in the endometrial tissue with the up-regulation of vascular endothelial growth factor. Furthermore, hAECs were shown to promote ER expression in the endometrium and regulate the inflammatory reaction in the uterine microenvironment. In conclusion, these results demonstrated that hAEC transplantation could inhibit the progression of fibrosis and promote proliferation and angiogenesis in IUA rat models. The current study suggests hAECs as a novel stem cell candidate in the treatment of severe IUA.
Keywords
Introduction
Intrauterine adhesions (IUAs), also known as Asherman’s syndrome, are characterized as fibrous strings connecting the opposite walls of the uterus and partially or completely obstructing the endometrial cavity. As a result, the fibrosed endometrium is usually avascular and unresponsive to hormonal stimulation 1,2 . Any damage to the basal layer of the endometrium can lead to IUAs. Patients with IUAs are associated with hypomenorrhea, amenorrhea, abnormal chronic pain, infertility, and recurrent miscarriages 3 –5 . Though hysteroscopic adhesiolysis is widely recommended for diagnosis and treatment, the prognosis remains poor in severe IUA cases and the recurrence rate is still high (up to 20.0% to 62.5%) 6 . In order to reduce the high recurrence rate, numerous antiadhesion treatments are applied, such as hormonal treatment 7 , insertion of an intrauterine device (IUD) 8 , or barrier gels 9 . But no consistent result has been delineated yet. Nowadays, regenerative engineering based on stem cell therapy offers a new treatment for the fibrotic endometrium. The existence of endometrial stem/progenitor cells in the endometrial basal layer is the theoretical foundation of this application of stem cell therapy 10 .
It is universally accepted that stem cells are unique lines of cells characterized by self-renewal and differentiation. Over the last few decades, stem cells from different sources—such as bone marrow-derived mesenchymal stem cells (BM-MSCs) 11 –13 , menstrual blood-derived mesenchymal stromal cells (mbMSCs) 14 , adipose-derived mesenchymal stromal cells (ASCs) 15 , and oral mucosal epithelial-cell sheets 16 —have been collected and applied in animal models and even in human clinical trials. BM-MSCs 17 –19 and mbMSCs 14 have been autologously engrafted into patients with severe refractory IUAs. Though the number of patients in those clinical trials was limited, the thickness of the endometrium was significantly increased and the prognosis was promising. However, the use of stem cells in clinical practice still presents many difficulties. For example, isolation of these candidate cells is unavailable. Recruitment and collection of BM-MSCs and ASCs is an invasive and expensive procedure. The mbMSCs are rare in menstrual blood and require more attention to aseptic concept. These limitations encouraged us to explore a novel candidate of stem cell that is not only easy to obtain but also effective.
Human amniotic epithelial cells (hAECs), derived from pluripotent epiblast at day 8, are isolated from discarded placenta tissue
20
. Evidence shows that hAECs can express typical stem cell markers such as octamer-binding transcription factor 4, stage-specific embryonic antigen 3, and NANOG and have the ability to differentiate into ectoderm, endoderm, and mesoderm lineage cells
In this study, we created an IUA rat model and transplanted hAECs into rat uteri to assess whether the candidate cells could reverse the fibrosis and regenerate the endometrium. These results could guide future clinical trials and therapy for severe IUA patients.
Materials and Methods
Isolation and Culture of hAECs
Fresh amniotic membranes were collected from healthy mothers after cesarean deliveries with written and informed consent. The procedure was approved by the Institutional Ethics Committee of the Second Affiliated Hospital of Zhejiang University of Medical School, Hangzhou, China. All donors were tested and were negative for HIV-I and hepatitis A, B, C, and D. The amnions were peeled from the collected placentas, and blood and mucus carefully washed away with sterile saline solution carefully. Amniotic debris was dissociated by using 0.25% trypsin/EDTA for 30 min at 37°C in a water bath, then the sample suspension centrifuged for 10 min at 300 g. The cell pellet was then suspended in the complete culture medium (Dulbecco’s Modified Eagle’s medium/F12), added with 10% fetal bovine serum, 2 mM glutamine, 1% antibiotic-antimycotic (all from Thermo Fisher Scientific, Waltham, MA, USA), and incubated in a 5% CO2 atmosphere at 37°C. Cobble-shaped cells were observed under a light microscope (Nikon, Tokyo, Japan). Flow cytometry (FCM) was used to identify the specific immunophenotype of hAECs. The first-generation hAECs were selected for the transplantation. hAECs were transfected, 48 h before transplantation, by lentivirus parceled with green fluorescence protein (GFP) and the green fluorescence intensity observed over 70%.
Flow Cytometry
Cell surface antigens of hAECs were analyzed by FCM. Cells were harvested and stained with primary labeled antibodies according to the technical data sheet: cluster of differentiation 34 (CD34); CD324; CD45; human leukocyte antigen-DR (HLA-DR); stage specific embryonic antigen-4 (SSEA-4) (all from Thermo Fisher Scientific) at 4°C for 30 min. For FCM analysis, we used Kaluza Analysis Software version 1.5a (Beckman Coulter, Brea, CA, USA).
Animals
Female Sprague-Dawley rats (220 to 250 g; 8 weeks old) were purchased from the Animal Center of the Chinese Academy of Sciences, Shanghai, China. All animals were kept in a pathogen-free environment and fed ad libitum. The procedures for care and use of animals were approved by the Ethics Committee of the Zhejiang University and all applicable institutional and governmental regulations concerning the ethical use of animals were followed.
Model Building and hAEC Transplantation
The IUA model was constructed while all rats were in estrus, as implied by the characterization of their vaginal cytology. Vaginal smears were collected with a cotton swab dipped in sterile saline solution at 9:00 to 10:00 AM every day. Under a light microscope (Nikon) at 100× total magnification, classic keratinized, needle-like cells were observed, indicating that the rats were in estrus. All rats were randomly assigned to four groups, including blank group (
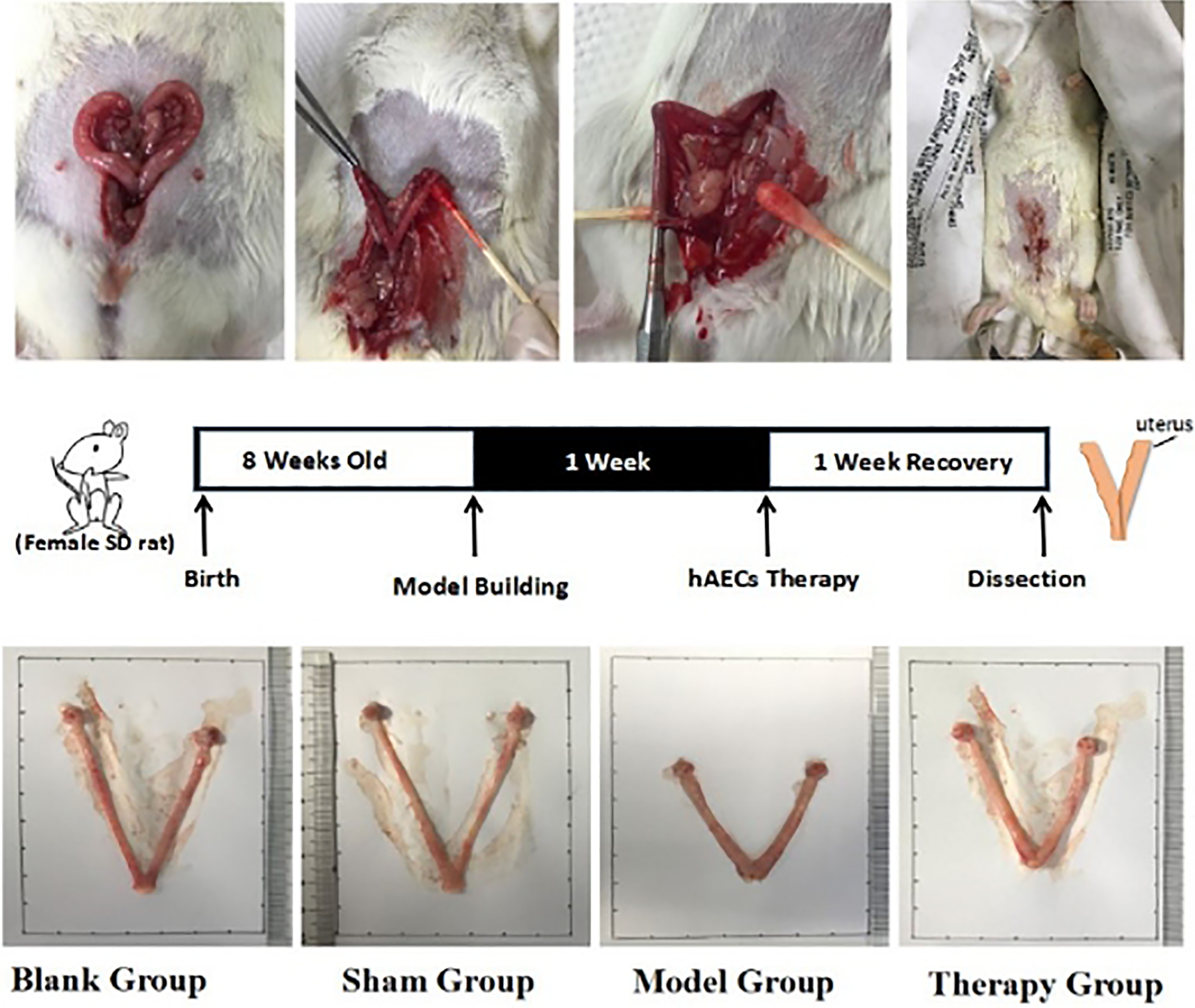
Model building and hAEC transplantation. Female SD rats were used to build IUA models by mechanism curettage until it was rough in texture. In the therapy group, a suspension of GFP-labeled hAEC suspension was transplanted intrauterinely into the bilateral uterine horns 7 d after surgery, while rats in the model group were injected with sterile saline solution only. Uteri were collected after 7 d’ recovery for assessment. GFP, green flourescence protein; hAEC, human amniotic epithelial cell; IUA, intrauterine adhesion; SD, Sprague-Dawley.
Histological Examination
The morphologic structure of endometrial thickness and glands was examined via hematoxylin and eosin (H&E) staining. Collagen deposition was detected via Masson staining. Briefly, the uteri excised from rats were fixed with 4% paraformaldehyde solution immediately, then embedded in paraffin, sliced into 5-µm thick sections, and stained with H&E and Masson stains (Beyotime Institute of Biotechnology, Shanghai, China) according to a standard protocol. Briefly, after deparaffinization and rehydration, tissue slices were stained with hematoxylin solution for 5 min, soak in 1% acid ethanol (1% HCl in 70% ethanol) 5 times and then rinsed in distilled water. Then the slices were stained with eosin solution for 3 min, dehydrated with graded alcohol and clearing in xylene. Microscopic examination performed and the number of endometrial glands, endometrial thickness were analysed by Image-Pro Plus 6.0 software. According to the routine Masson staining protocol, after deparaffinization and rehydration, reagent A, B, C, D and E were stained in turn to assess collagen deposition. The expression levels were evaluated by integrated optical density (IOD) of the positive cells. IOD/area was the average cumulative optical density of the positive staining area determined by Image-Pro Plus 6.0 software.
Immunohistochemistry
The transverse paraffinized uterine sections were deparaffinized, rehydrated, and then incubated in 5% bovine serum albumin (Beyotime) for 30 min at 37°C to block the nonspecific antibody. Then the samples were incubated with the following primary antibodies: collagen 1 (1:100 dilution), vascular endothelial growth factor (VEGF; 1:100 dilution), transforming growth factor-β (TGF-β; 1:100 dilution), and estrogen receptor (ER; 1:100 dilution, all from Abcam, Cambridge, UK) at 4°C overnight. And then the sections were incubated with the corresponding secondary antibody and the reaction was stopped with 3,3-diaminobenzidine. The results were imaged under a light microscope (Nikon). Under 40 × 10 magnification of the field of vision, five fields were randomly selected to calculate the positive-area percentage of the whole selected area.
RNA Isolation and Polymerase Chain Reactions
Rats were euthanized 2, 12, and 24 h, and 1 wk, respectively, after transplantation of GFP-labeled cells. Total RNA samples were collected from uterine horns using TRIzol Reagent (Omega, Thermo Scientific, Waltham, MA, USA) as per the manufacturer’s instructions. The purity and concentration of all RNA were analyzed and the nucleotide to protein ratios (A260:A280) ranged from 1.8 to 2.1, within the acceptable boundaries. Complementary DNA (cDNA) was synthesized using a cDNA Reverse Transcription Kit (Toyobo, Shanghai, China), allowing the quantitative conversion of up to 2 mg of total RNA to cDNA. Amplification and quantification of gene expression were executed by reverse-transcription polymerase chain reaction (RT-PCR) using gene-specific primers and SYBR Green enzyme (Bio-Rad, Hercules, CA, USA) as follows: 35 cycles of 95°C for 5 min and then 60°C for 30 s. The expression level of β-actin was used as the internal control. Primer sequences are presented in supplemental Table S1.
Statistical Analysis
Statistical analysis was performed using GraphPad Prism version 7 (GraphPad Software, San Diego, CA, USA). Student’s
Results
Characterization of hAECs
Under light microscope, the hAECs appeared as cobble-shaped cells, a typical appearance for epithelial cells (Fig. 2A). After labeling with GFP, the green fluorescence intensity could be observed over 70% before the hAECs were transplanted into rat uteri (Fig. 2B). FCM was used to identify immunophenotypic characterization of hAECs. Cells applied in this study were positive for epithelial marker CD324 and pluripotent marker SSEA-4, negative for hematopoietic lineage marker CD45, endothelial marker CD34, and immunogenicity identification marker HLA-DR (Fig. 2C).

Characterization of hAECs. (A) hAECs were cobble-shaped under a light microscope; (B) cells were transfected with green fluorescence protein under fluorescence microscope; (C) specific hAECs with special immunophenotypic characterization (CD324, SSEA-4, HLA-DR, CD45, CD3) were identified using flow cytometry. CD, cluster of differentiation; hAEC, human amniotic epithelial cell; HLA-DR, human leukocyte antigen-DR; SSEA-4, stage specific embryonic antigen-4.
Endometrial Morphological Structure and ECM Deposition
Seven d after therapy, uteri were collected from all rat groups (blank, sham, model, and therapy groups) and H&E and Masson staining were performed to ensure the model was successful and to assess collagen deposition in endometrium. There was no statistically significant difference in either the morphological structure or the collagen deposition between the blank group and the sham group. Unlike the blank group, the endometrium of the model group was damaged; H&E staining showed that their thickness and number of glands were significantly decreased (

Morphologic structure of endometrium. (A) H&E staining shows the number of endometrial glands and endometrial thickness (the black arrows refer to endometrial glands). (B) Masson staining indicates the degree of endometrial fibrosis. Images are shown at 4× magnification (n.s.: no statistical significance;*
Effects of hAEC Therapy on Fibrosis Regulation
Messenger ribonucleic acid (mRNA) levels of fibrosis in rat endometria indicated that levels of TGF-β family members and collagen 1 were altered prominently after injury and therapy. The profibrosis molecules of TGF-β1 and TGF-β2 were apparent in the model group versus the blank group (

Effects of hAEC therapy on fibrosis regulation. (A) RT-PCR results showed that in mRNA level, profibrosis molecules TGF-β1, TGF-β2, and collagen 1 decreased after cell therapy, while antifibrosis marker TGF-β3 increased in the therapy group compared with the model group. (B) Immunohistochemistry results demonstrated the protein levels of TGF-β, α-SMA, PDGF, and collagen 1 expression. Mean area percentage
Effects of hAEC Therapy on Angiogenesis and Endometrium Proliferation
In order to explore the effects of hAEC therapy on angiogenesis and endometrium proliferation, we detected the expressions of VEGF, mammalian target of rapamycin (MTOR), Rho-associated protein kinase (ROCK), and microenvironment-related molecules such as insulin-like growth factor (IGF) and interleukin-4 (IL-4). In the model group, new vascular formation was inhibited and VEGF1 and VEGF2 expressions were significantly lower than in the blank group (

Effects of hAEC therapy on endometrial angiogenesis and proliferation. (A) RT-PCR results showed that at the mRNA level, VEGF1 and VEGF2 increased after cell therapy, MTOR was upregulated, and ROCK was downregulated compared with the model group. IGF and IL-4 regulation reflected the change to the endometrial microenvironment. (B, C) Immunohistochemistry results demonstrated the protein levels of VEGF and ER expression. Mean area percentage
Engraftment of GFP-Labeled hAECs in Rat Endometrium
At 2, 12, and 24 h and 1 wk after transplantation, rats were sacrificed sequentially. Uteri were collected to assess whether the injected hAECs had migrated to the endometrium and how long they were sustained there. The GFP-labeled hAECs were observed in the endometrium under fluorescence microscope (Fig. 6A). Accumulation of GFP RNA indicated the presence of hAECs. Results showed that transplanted hAECs first appeared 2 h posttransplantation and were sustained there for at least 1 wk (Fig. 6B).
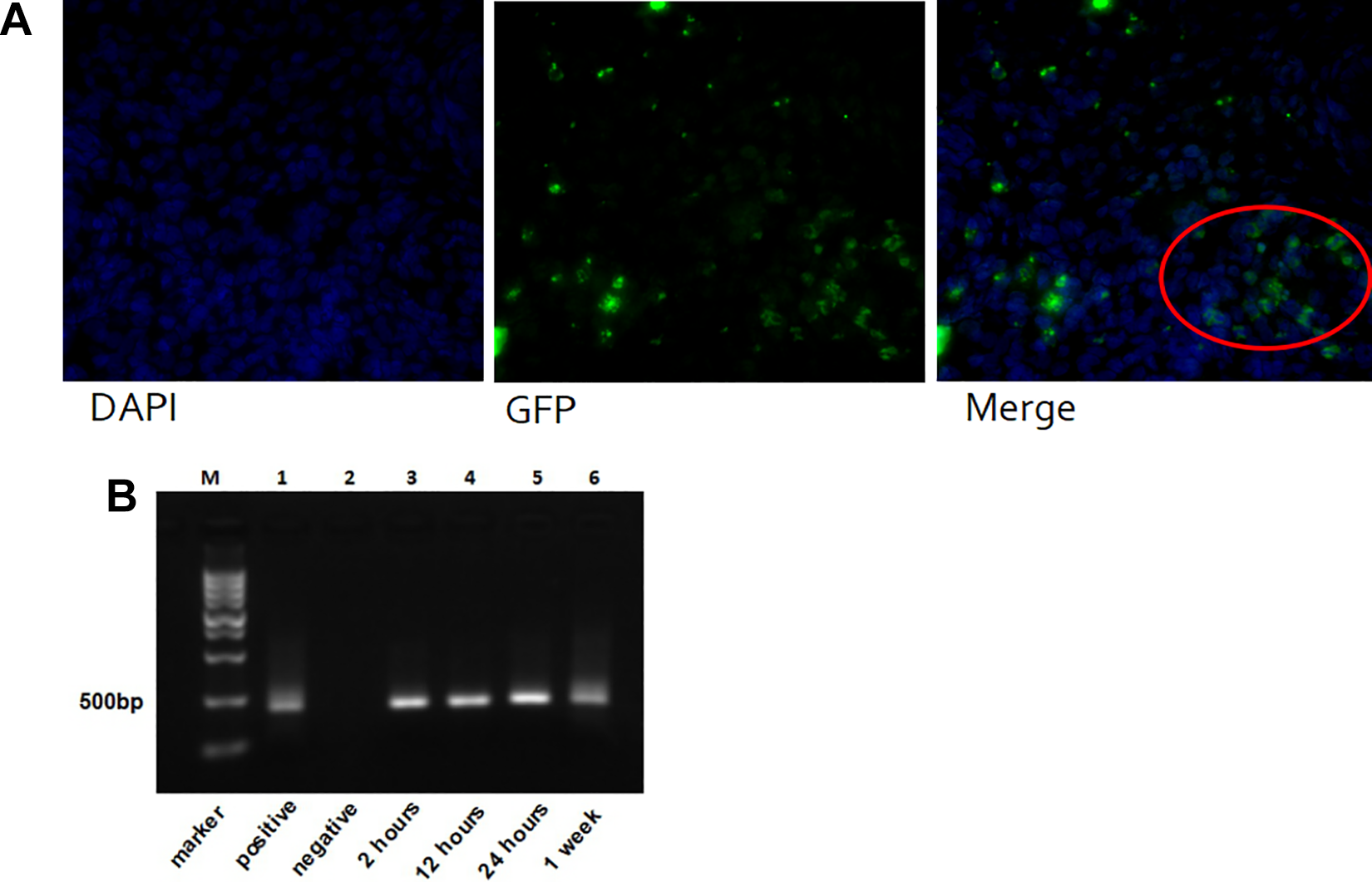
Engraftment of GFP-labeled hAECs in rats’ endometrium. (A) GFP-labeled hAECs were observed in the endometrium under a fluorescence microscope. Cells in red circle refer to hAECs in the endometrium (DAPI: 4′,6-diamidino-2-phenylindole). (B) Electrophoresis image shows that hAECs were first found 2 h after transplantation and were sustained for at least 1 wk after therapy. DAPI, diamidino phenylindole; GFP, green flourescence protein; hAEC, human amniotic epithelial cell.
Discussion
Regenerative engineering and stem cell therapy have gradually become a novel and heated treatment for endometrial damage and fibrosis in recent years, due to the high recurrence rate for severe IUAs 31 . In the present study, we built an IUA model in female SD rats by mechanical curettage in order to simulate the pathomechanism in IUA patients. The hAECs as an original stem cell source were transplanted intrauterinely into model rats and the curative effect was prominent. The results of the present study show that (i) the transplantation of hAECs promoted endometrial thickness, increased the number of endometrial glands, and limited the area of fibrosis; (ii) after hAECs therapy, expression of profibrosis molecule TGF-β was inhibited and collagen deposition decreased; (iii) hAECs treatment facilitated endometrial angiogenesis and proliferation, as well as the expression of ER. IGF and IL-4 participated in the regulation of the endometrial microenvironment; (iv) transplanted hAECs were sustained in the endometrium for at least 1 wk. These findings could guide future clinical trials and treatment.
Promising data from preclinical studies have demonstrated the significant therapeutic properties of hAECs as candidate stem cells in regenerative medicine to treat fibrotic disease 32 . Cargnoni et al. built a lung fibrosis mouse model induced by bleomycin and then injected a conditioned medium that was generated from cells separated from human amniotic membrane 33 . Pro-fibrosis molecules such as TGF-β, macrophage inflammatory protein-1 alpha, and monocyte chemoattractant protein-1 were downregulated, suggesting that amniotic products could play a crucial antifibrotic role 33 . The researchers performed a subsequent study of hAECs’ antifibrotic effects on liver fibrosis in a rat model. The hAECs exerted a strong effect on myofibroblast precursors, inhibiting their proliferation specifically by suppressing αvβ6 integrin expression, with a resultant decrease in TGF-β activation 34 . To our knowledge, the current study is the first study to examine the potential effect of hAECs on intrauterine fibrotic disease.
Across different fields of fibrosis diseases, experts agree that myofibroblasts are the key cells responsible for the formation of fibrosis and the development of scar tissue 35 . Once the fibrotic reaction is triggered, myofibroblasts become highly active and express α-SMA, promoting a remarkable increase in collagen deposition 36,37 . In the present study, myofibroblasts were seen to be synthetically active after the injury of endometrium. The α-SMA was upregulated significantly and collagen 1 was altered prominently. They were downregulated remarkably after hAEC transplantation, which indicated that hAECs could inhibit the activity of myofibroblasts and prevent progression of fibrosis.
It is widely known that the TGF-β family, as multifunctional signaling molecules, is involved in organism development, inflammation regulation, immunoregulation, neoplasm promotion and suppression, fibrosis development, and healing 38 . In the formation and progression of fibrotic disease, TGF-β is considered as a pivotal mediator and critical molecular driver 39 . One study showed that TGF-β1 and its two receptors play key roles in uncontrolled fibrotic response 40 . By contrast, TGF-β3 is reported to have antifibrotic potential to inhibit collagen synthesis and suppress scarring of the skin 41,42 . Another study reported that the expression of TGF-β1 in mRNA and protein was significantly upregulated in both animals and patients with IUAs. In addition, the expression level of TGF-β1 was apparently higher in severe patients than in those with mild or moderate IUAs 43 . In the current study, we saw that the profibrosis molecules TGF-β1 and TGF-β2 were remarkably inhibited after hAEC transplantation in comparison with the IUA model group, whereas the antifibrotic molecule TGF-β3 increased significantly in the therapy group. These results revealed that the potential antifibrotic function of hAECs could inhibit the TGF-β-mediated fibrotic progression in the endometrium.
During a woman’s reproductive years, human endometrium, known as a dynamic remodeling tissue, undergoes more than 400 cycles of regeneration, differentiation, and shedding. Estrogen induces endometrial cell proliferation through upregulating synthesis and secretion of growth factors, such as epidermal growth factor, TGF-α, and IGF 44 . In Chen et al.’s study, they found that vessels closed and hypoxic conditions changed in the endometrial tissues of IUA patients 45 . On the other hand, after conventional therapy, VEGF expression increased and angiogenesis occurred in the endometrium, which implied that angiogenesis in the endometrium may promote endometrial repair 45 . The present study demonstrates that after hAEC transplantation, the expression of VEGF and ER increased, which signifies angiogenesis and proliferation in the injured endometrium. Upregulation of IGF and IL-4 observed in the therapy group suggested that hAECs could regulate the microenvironment of the injured endometrium and promote recovery from fibrosis through paracrine growth factors.
In conclusion, the current study demonstrated that transplantation of hAECs could inhibit the progression of endometrial fibrosis and promote endometrial angiogenesis and regeneration in rats with IUAs. The regulation of IGF and IL-4 suggested that hAECs might exert their effect by secreting paracrine factors in the endometrial microenvironment. Their advantages of easy accessibility and antifibrotic properties make hAECs an ideal candidate to guide further clinical stem cell treatment strategies for severe IUA patients. The underlying molecular mechanism and long-term efficacy of hAECs need further exploration in the future.
Supplemental Material
Supplemental Material, Table_1 - Therapeutic Effect of Human Amniotic Epithelial Cells in Rat Models of Intrauterine Adhesions
Supplemental Material, Table_1 for Therapeutic Effect of Human Amniotic Epithelial Cells in Rat Models of Intrauterine Adhesions by Xuechai Bai, Jia Liu, Weixin Yuan, Yang Liu, Wei Li, Siyu Cao, Luyang Yu and Liang Wang in Cell Transplantation
Footnotes
Author Note
Joint first authors: Xuechai Bai, Jia Liu.
Author Contributions
XCB, JL, and YL carried out animal model establishment; WXY participated in FCM assays and analysis, immunohistochemistry, and polymerase chain reactions; JL, WL, and SYC participated in data acquisition and performed the statistical analysis. XCB and JL participated in data analysis and manuscript revision. XCB, LJ, YYL, and LW designed and conceived the study and drafted the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Tissue collection and analysis was approved by the review boards of the Second Affiliated Hospital of Zhejiang University of Medical School. All animal experiments were carried out with ethical approval from the Laboratory Animal Care and the Use Committee of Zhejiang University.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants to LW from the National Natural Science Foundation of China (Grant No.81472422).
Statement of Human and Animal Rights
Animals used in this study were treated according to the Laboratory Animal Care and the Use Committee of Zhejiang University. Human subjects were approved by the Institutional Ethics Committee of the Second Affiliated Hospital of Zhejiang University of Medical School.
Statement of Informed Consent
The human subjects used in this article were informed and consent was obtained from them.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
