Abstract
For the majority of CNS malignancies, radiotherapy provides the best option for forestalling tumor growth, but is frequently associated with debilitating and progressive cognitive dysfunction. Despite the recognition of this serious side effect, satisfactory long-term solutions are not currently available and have prompted our efforts to explore the potential therapeutic efficacy of cranial stem cell transplants. We have demonstrated that intrahippocampal transplantation of human neural stem cells (hNSCs) can provide long-lasting cognitive benefits using an athymic rat model subjected to cranial irradiation. To explore the possible mechanisms underlying the capability of engrafted cells to ameliorate radiation-induced cognitive dysfunction we analyzed the expression patterns of the behaviorally induced activity-regulated cytoskeleton-associated protein (Arc) in the hippocampus at 1 and 8 months postgrafting. While immunohistochemical analyses revealed a small fraction (4.5%) of surviving hNSCs in the irradiated brain that did not express neuronal or astroglial makers, hNSC transplantation impacted the irradiated microenvironment of the host brain by promoting the expression of Arc at both time points. Arc is known to play key roles in the neuronal mechanisms underlying long-term synaptic plasticity and memory and provides a reliable marker for detecting neurons that are actively engaged in spatial and contextual information processing associated with memory consolidation. Cranial irradiation significantly reduced the number of pyramidal (CA1) and granule neurons (DG) expressing behaviorally induced Arc at 1 and 8 months postirradiation. Transplantation of hNSCs restored the expression of plasticity-related Arc in the host brain to control levels. These findings suggest that hNSC transplantation promotes the long-term recovery of host hippocampal neurons and indicates that one mechanism promoting the preservation of cognition after irradiation involves trophic support from engrafted cells.
Keywords
Introduction
Clinicians have recognized the beneficial effects of cranial irradiation used to control tumor growth in the brain, but have long realized that such treatments come at a cost, as radiotherapy frequently leads to progressive and long-lasting declines in cognition that can severely impact quality of life (1, 13, 30). Furthermore, as cancer treatments have advanced, increased numbers of long-term survivors of intracranial tumors are forced to cope with a range of neurocognitive sequelae in the relative absence of any satisfactory interventional recourse. To address this growing problem, we have used intrahippocampal transplantation of human neural stem cells (hNSCs) to ameliorate radiation-induced cognitive impairment ranging from 1 to 8 months posttransplantation in rodents (4-6, 8), thereby providing evidence that such a strategy may one day provide relief to those suffering from the side effects of cranial radiotherapy.
While the mechanisms underlying radiation-induced cognitive impairment are incompletely understood, evidence suggests that persistent changes to the microenvironment of the irradiated brain involving oxidative stress and inflammation (15, 16, 33, 42) can adversely impact neural stem/progenitor cell (NSC) proliferation, differentiation, and the structure of immature and mature neurons (33-35, 42). Recent findings from our laboratory have shown that cranial irradiation alters mature neuronal architecture (dendrites and spines) and modulates proteins involved in synaptic function in the hippocampus (34). The hippocampus is a brain region critical for the acquisition (learning), consolidation, and retrieval of declarative memories [for review see (15, 41)]. These processes modulate the strength and efficacy of synaptic signaling (i.e., synaptic plasticity), which in turn involves gene expression (14). Gene expression induced during learning produces proteins that alter the composition of networks and provides a mechanism for translating synaptic plasticity into changes in synaptic strength (memory). The activity-regulated cytoskeletal (Arc) gene encodes a protein that is critical for memory formation and synaptic plasticity (20-22). Arc is rapidly activated by robust patterned synaptic activity related to memory [reviewed in (18)], and reducing Arc expression impairs memory retention and long-term potentiation (19). Arc expression has been utilized extensively to map neuronal networks that underlie information processing and plasticity (22, 39, 40). Further, we have reported that the transcription and translation of Arc are significantly affected by cranial irradiation (10, 38). Given the specificity and the well-characterized dynamics of behaviorally induced Arc expression and its critical role in synaptic plasticity and memory, we monitored the expression of Arc in this study to determine whether grafted hNSCs could restore functional circuits within the irradiated hippocampus at a long-term time point (8 months) postirradiation. The present findings provide insight into the potential mechanisms underlying the restoration of cognitive function by stem cell transplantation into the irradiated brain.
Materials and Methods
Animals and Cranial Irradiation Procedure
All animal procedures described are in accordance with NIH and approved by Institutional IACUC committee. Immunodeficient male athymic nude (ATN) rats (strain 0N01, Cr:NIH-rnu, X50 colony; NCI Frederick National Laboratory, Frederick, MD, USA) were maintained in sterile housing conditions (20°C ± 1°C; 70% ± 10% humidity; 12 h:12 h light and dark cycle) and had free access to sterilized diet and water. A total of 24 young (2 months old) ATN rats were divided in three experimental groups: 0 Gy (no irradiation), sham surgery (Cont-Sham, n=8), 10 Gy head-irradiated sham surgery (IRR-Sham, n = 8), and 10 Gy irradiated receiving hNSC grafting (IRR+hNSC, n = 8). An additional cohort of unirradiated control animals also received hNSC transplantation (Cont + hNSC, n=8) to assess the effects of stem cells in the uninjured hippocampus at 8 months postsurgery. For the irradiation procedures, animals were anesthetized, eyes and body were lead shielded and were exposed to cranial γ-irradiation (10 Gy) using a 137Cs irradiator (Mark I; J. L. Shepard, Glendale, CA, USA) at a dose rate of 2.07 Gy/min, as described in detail previously (4).
Transplantation Surgery
The use of hNSC (ENStem-A cell line; EMD Millipore, Billerica, MA, USA) was approved by the Institutional Human Stem Cell Research Oversight Committee (hSCRO). The hNSCs were maintained as a monolayer in T25 flasks (Corning, Fisher Scientific, Pittsburgh, PA, USA) in neural expansion media (EMD-Millipore), expressed stem cells markers (Sox2, nestin), and displayed multilineage potential, as described previously (7). For transplantation and identification of engrafted cells in the host brain, hNSCs (passages 5-9) were labeled in vitro with BrdU (5-bromo-2′-deoxyuridine, 4 μm, labeling index 90%; Sigma-Aldrich, St. Louis, MO, USA) as previously described (5). Two days postirradiation, rats received bilateral, intrahippocampal transplantation of hNSCs as described in detail previously (5). A total of 4.0 × 105 live hNSCs (1 μl/site in neural expansion media) were transplanted in four distinct hippocampal sites per hemisphere using defined streotaxic coordinates (4). Groups designated as sham surgery (Cont-Sham and IRR-Sham) received sterile vehicle (neural expansion media) at the same stereotaxic coordinates.
Exploration of Novelty for Arc Induction
To induce the expression of Arc, rats were subjected to novelty by allowing them to explore freely in an open arena (8 × 3 × 10 cm high) containing two toy objects for a single 5-min session. Rats were then returned to their holding cages until sacrificed 30 min later. A separate cohort of animals (n=4 in each group) did not undergo exploration and served as “caged controls ” to determine the basal expression of Arc.
Extraction of Brains and Immunohistochemistry
Animals were decapitated following deep anesthesia, and brains were rapidly (60-120 s) extracted and frozen in isopentane as described previously (40). Brains were cryosectioned at a thickness of 20 μm and collected on microscopic slides; each slide contained hemicoronal brain sections from each of the experimental conditions (40). Sections were selected from the medial portion of the dorsal hippocampus (from 3.2 to 4.0 mm posterior to bregma) and stained for neurons (NeuN monoclonal antibody clone A60, 1:500; Chemicon, EMD-Millipore), BrdU (mouse monoclonal antibody clone BMC9318, 1:20; Roche Applied Science, Indianapolis, IN, USA), Arc protein (1:1,000; antibody kindly provided by Dr. P. F. Worley laboratory, Johns Hopkins University), and astrocyte markers (GFAP, S100(β) after postfixation with 4% paraformaldehyde as described previously (5). Similar procedures were followed for detection of activated microglia (OX6; mouse monoclonal, 1:200; BD Pharmingen, San Diego, CA) and oligodendrocytes (APC-CC1, mouse monoclonal, 1:100; Abcam, Cambridge, MA, USA). For the present study, 8-month tissues were derived from a cohort of animals subjected to cognitive testing following irradiation and transplantation with hNSCs (8).
For the analyses of Arc protein expression and activated microglia, Z-stack images (200× magnification; 1 μ optical thickness per plane; eight planes) of the hippocampus were acquired using a Zeiss Apotome microscope (Carl Zeiss Microscopy GmbH, Jena, Germany); offline analyses were performed using Zeiss AxioVision software. The percentage of Arc immunoreactive neurons from the entire dentate gyrus (DG) enclosed blade and the CA1 subfield (two counting frame/area/slide) was assessed using two stained slides from the dorsal hippocampus as described in detail previously (37, 40). The density of major histocompatibility complex, class II (MHC-II; OX6+) immunoreactive cells in the hippocampus was measured blindly to the experimental group, as described in detail previously (37, 40).
Fluorescence-Based Stereology
For the assessment of the yield of hNSC transplant-derived cells at 8 months postsurgery, fluorescence stereological quantification was carried out. Every 10th section through the entire hippocampus was processed for BrdU immunostaining (1:20, mouse monoclonal, anti-BrdU IgG1, formalin grade; Roche Applied Science). Fluorescence color development was facilitated by anti-mouse FITC conjugate (1:200, donkey IgG; Jackson ImmunoResearch, PA, USA) and counterstained with DAPI blue nuclear stain (Sigma-Aldrich). Stereological assessment was conducted using a Zeiss epifluorescence microscope (Carl Zeiss Microscopy) equipped with a MBF monochrome digital camera, 100× (oil-immersion, 1.30NA; Zeiss) objective lens, three-axis motorized stage, and an optical fractionator probe (Stereo Investigator, v10.0; MBF Biosciences, Williston, VT, USA). Systemic random sampling (SRS) and image stack analysis modules were used to acquire a batch of images through anterioposterior planes of the hippocampus. The acquired images were uploaded on MBF Workstation (MBF Biosciences), and the yield of surviving hNSCs was quantified by counting BrdU+ nuclei (green) using the optical fractionator probe and SRS according to unbiased stereological principles. Sampling parameters (grid and counting frame size) were empirically determined to achieve low coefficients of error (Gunderson's CE, <0.06 ± 0.003, n = 12) for each hippocampus.
Analyses and Statistics
For the analysis of activated microglia measured by OX6+ cells/area, the treatment groups (Cont-Sham, IRR-Sham, and IRR+hNSC) were the independent variable (factors) and the number of OX6+ cells/area was the dependent variable. For the Arc analysis, the caged animals and the behavioral groups from each treatment (Cont-Sham, IRR-Sham, Cont + hNSC, IRR + hNSC) were the independent variable, and the percentage of Arc+ neurons/total neurons counted was the dependent variable. The nature of the dependent variable for both OX6 and Arc measurements (level of measurement) was continuous while the nature of the independent variable (treatment) was categorical. Thus, an ANOVA test for each region of interest (DG and CA1) was performed using GraphPad Prism software (v5.0c; San Diego, CA, USA). When an overall ANOVA was significant, Bonferroni's multiple comparisons test was used. For the analyses of activated microglia, Z-stack images (200× magnification; 1 μ optical thickness per plane; eight planes) of the hippocampus were acquired using a Zeiss Apotome microscope (Carl Zeiss Microscopy); offline analyses were performed using Zeiss AxioVision software. For the Arc and OX6 analyses, GraphPad Prism software (v5.0c) was used to perform ANOVA tests for each region of interest. The percentage of Arc-positive neurons and activated microglia was calculated as previously described in detail (9, 38-40).
Results
Yield of hNSC Transplant-Derived Cells and Differentiation
At 8 months posttransplantation, analysis of brain sections using BrdU immunofluorescence staining revealed the survival of a fraction of hNSC transplant-derived cells in the host hippocampus (Fig. 1). Transplanted cells were found to be located predominately near the corpus callosum (CC) and partly extended in the CA1 regions of hippocampus (see below). Stereological quantification using an optical fractionator (Fig. 1) revealed that ~16,000 (16,242 ± 4,412, n=4) of transplant-derived cells (4.5% of total; 4.51 ± 1.22, n=4) survived at 8 months posttransplantation; that is approximately five times and three times less compared to 1 month and 4 months posttransplantation, respectively (5, 6) (Fig. 1). Thus, 16,000 new cells were added to each hippocampus at 8 months posttransplantation.
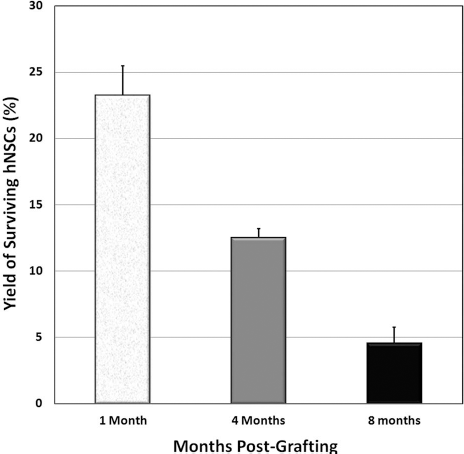
The yield of hNSC transplant-derived cells at 1, 4, and 8 months postsurgery. Transplant-derived cells were tracked by BrdU immunofluorescence staining and quantified using fluorescence stereology and an optical fractionator by counting BrdU-positive nuclei. Data are represented at mean ± SEM of four individual observations.
To further determine the phenotypic fate of hNSC transplant-derived cells at 8 months postsurgery, we carried out dual immunofluorescence staining for BrdU and neuronal (NeuN), astrocytic (GFAP, S100β), and oligodendrocyte (APC-CC1) markers. However, differentiation at 8 months posttransplantation was not evident for neuronal and/or astroglial markers. Nonetheless, 1% (1.07 ± 0.66, n=7) of cells surviving at 8 months expressed nestin, suggesting that a small percentage of the transplanted cells remained multipotent.
Inflammatory Signatures and Transplant Location at 1 and 8 Months Posttransplantation
Qualitative analyses of activated microglia (OX6+ cells per mm2 area) revealed the presence of MHC-II+ cells in Cont-Sham, IRR Sham, and IRR+hNSC groups at each postirradiation time (Fig. 2A-F). At 1 month postsurgery, there was a significant increase in the number of OX6+ cells in the hippocampus (DG, DH, CA1, and CA3 subfields) (Fig. 2A, C, E) of IRR+hNSC transplanted group compared to the Cont-Sham and IRR-Sham [F(2, 18) = 6.36, p = 0.008] (Fig. 2G). In contrast, at the longer postirradiation time (8 months), irradiated animals (IRR-Sham) showed higher OX6+ immunoreactive cells (Fig. 2B, D, F) compared to control (Cont-Sham) and hNSC-transplanted (IRR+hNSC) animals [F(2, 18) = 3.2, p = 0.06] (Fig. 2H). Interestingly, the location of transplanted hNSCs and host OX6+ cells were found to be in close proximity at 8 months (Fig. 3A-D). Moreover, close examination of transplant-derived cells (BrdU+) and OX6 revealed interaction of activated microglia with human cells (Fig. 3C-D, arrows). Extended processes of activated microglia were in close proximity with BrdU+ human cells (Fig. 3E, F, arrows).

Representative fluorescent micrographs of OX6+ microglia (MHC-II, red and nucleus, DAPI, blue) in sham surgery control (Cont-Sham), irradiated (IRR-Sham), and irradiated with hNSC transplantation (IRR + hNSC) at 1 month (A, C, E) and 8 months (B, D, F) postsurgery. Quantification of OX6 in the hippocampus (DG, DH, CA1, and CA3) indicated significant upregulation (*p < 0.008) in the transplanted group (IRR + hNSC) at 1 month postsurgery (D). The trend was reversed (higher OX6 in IRR sham) at 8 months postsurgery group (H), albeit not significant (p < 0.06) at this time point. Data are represented as mean ± SEM of four individual observations. Scale bar: 50 μm (A-F).

Representative confocal fluorescent micrographs of location of transplanted cells and its close proximity with activated microglia. Intrahippocampal location of transplanted hNSC (green, BrdU+ and blue, DAPI+ nucleus) is shown at 5× and 40× magnification (A, B). Transplanted hNSCs and host OX6+ microglia (red) were found in the close vicinity at 8 months postsurgery (arrows, C and D). The activated microglia extended process to interact with transplanted hNSCs (arrows, E, F). Scale bar: 100 μm (A-B), 50 μm (C-D), and 5 μm (E-F).
hNSC Transplantation Restores the Endogenous Expression of the Plasticity-Related Arc in the Irradiated Hippocampus
To establish if irradiation and hNSC transplantation affect spatial information processing from a cellular perspective, we analyzed the proportion of neurons expressing the plasticity-related immediate early gene product Arc in the DG and CA1 areas of the hippocampus 30 min after exploration of novelty (Figs. 4 and 5). Animals that did not undergo exploration served as “caged controls. ” At 1 month postsurgery, in the IRR + hNSC group, the percentage of granule cells expressing behaviorally induced Arc was significantly elevated in all the experimental groups compared to the caged controls except the IRR-Sham animals [F(2, 24) = 16.8, p < 0.0001] (Fig. 4). Similar significant trends were observed in the CA1 subfield (Fig. 5). The percentage of pyramidal neurons expressing Arc in the CA1 was significantly elevated compared to caged control animals in all treatment groups except for the IRR-Sham animals that showed decline in Arc expression [F(4, 20)=20.10, p < 0.0001] (Fig. 5). Notably, in the IRR-Sham animals the percentage of pyramidal neurons expressing Arc was not different from that of the cage control animals and was significantly reduced compared to the Cont-Sham animals (post hoc test, p < 0.001) (Fig. 5). Most importantly, at 8 months posttransplantation, the proportion of Arc-expressing neurons in both the DG and CA1 areas were elevated after novel exploration in all treatment groups except for the irradiated animals [F(3, 14) = 7.26, p < 0.004 for the DG (Fig. 4) and F(3, 14) = 29.8, p < 0.0001 for the CA1 (Fig. 5)]. No significant differences were found across the caged control among the different groups (data not shown).

Representative fluorescent micrographs of Arc protein expression (red) 30 min after memory test in the DG neurons (blue) of the hippocampus. Memory test induced a significant increase in Arc+ neurons compared to caged control animals for both 1 and 8 months postgrafting in all treatment groups except the irradiated animals (*p < 0.0001 vs. caged). Scale bar: 200 μm.

Representative fluorescent micrographs of Arc protein expression (red) 30 min after memory test in the CA1 pyramidal neurons (blue) of the hippocampus. Thirty minutes after behavioral exploration in the memory test the percentage of Arc+ pyramidal neurons was significantly higher than the cage control in all the treatment except irradiated animals (*p < 0.0001, vs. caged) both 1 and 8 months postirradiation and was significantly reduced compared to sham control animals (+p < 0.001, vs. Cont-Sham). Scale bar: 50 μm.
The percentage of Arc-expressing neurons in the CA1 and DG areas after exploration was similar to that previously reported after similar exploration paradigms (10, 38-40) and following the Morris water maze (11). These data further demonstrate that irradiation affects neuronal function as indicated by the decline in behaviorally induced Arc expression. Importantly, hNSC transplantation was able to restore neuronal function at the cellular levels (Figs. 4 and 5). Treatment with hNSC prevented the alteration in Arc expression induced by irradiation and restored neuronal plasticity. Notably, the hNSC transplantation did not alter behaviorally induced Arc expression in control animals.
Discussion
Here we show the long-term benefits of engrafted cells on host brain activity following cranial irradiation. Studies were prompted to further delineate the mechanisms underlying the beneficial effects of stem cell transplantation on cognition at early (1 month) and long-term (8 months) postirradiation that we have previously reported (3-6). Recent work has shown that hNSC grafting improved behavioral performance on a novel place recognition task administered 8 months after irradiation and transplantation surgery. Tissues derived from these cohorts were used here to substantiate further the potential mechanisms underlying the beneficial effects of cranial stem cell transplantation. These studies are driven by the need to improve quality of life in brain cancer survivors, as the adverse effects of radiation treatment compromise cognition and quality of life, while remaining a serious unmet medical need (1, 13, 30). While it remains uncertain precisely how irradiation impacts specific subpopulations of cells within the CNS to disrupt cognition, the present results demonstrate the capability of hNSC transplantation to restore host neuronal activity at a protracted treatment interval (i.e., 8 months).
Past work from our group has characterized the beneficial effects of pluripotent and multipotent human stem cells on cognition following irradiation and elucidated the yields and differentiated phenotypes associated with those effects at 1, 4, and 8 months following treatment (4-6, 8). In the present study, we analyzed the functional consequences of engrafted cells in the brain from a cellular perspective at an extended time (8 months) after irradiation and transplantation surgery. We were able to demonstrate that engrafted cells impact host hippocampal neurons to improve activity patterns (i.e., Arc expression) known to be associated with synaptic plasticity and memory. These data demonstrate that engrafted cells impart persistent and beneficial effects to host neurons of the irradiated brain.
Unbiased stereology conducted to quantify engrafted cell survival over this timeframe showed that at 1, 4, and 8 months following irradiation, the number of engrafted cells was 23.2%, 12.5%, and 4.5%, respectively (Fig. 1). Since the yield of engrafted cells fell over 80% during the course of 8 months, these data suggest that the positive effects of stem cell transplantation in the irradiated brain are not entirely due to engrafted cell survival.
To explore the potential mechanisms that might influence cognitive processing in the irradiated brain, we quantified the number of activated microglia present 1 or 8 months following irradiation in our control and transplanted cohorts. At 1 month, the numbers of activated microglia (OX6+) were significantly higher for animals receiving stem cells compared to those that did not (Fig. 2G). Past work has shown that NSCs can express immune molecules that may facilitate their interaction with microglia and/or reactive astrocytes to promote repair under conditions of inflammation or other CNS injury (24, 25). The mild inflammatory signature at 1 month may aid homing of the microglia to sites of stem cell engraftment where they may promote CNS repair through removal of dead cell debris. The presence of microglia has also been shown to regulate the number of functional synapses (26), where the secretion of microvesicles and/or exosomes laden with an array of neuromodulatory proteins may serve to restore neuronal activity through the stimulation of synaptic transmission (43). Interestingly, the longer-term inflammatory signature observed at 8 months reverses, where the number of activated microglia trended lower in irradiated animals receiving stem cell grafts compared to those that did not (Fig. 2H). The earlier increase in microglia immunoreactivity may also suggest an initial rejection response that decreases over time. Recent evidence indicates that activation of the macrophage lineage is a polarized process leading to a potentially neurotoxic M1 “classical activation ” or potentially neuroprotective M2 “alternative activation ” (28, 31). We recently demonstrated that microglia display a neuroprotective (M2) phenotype when involved in remodeling and repair 3 months after cranial irradiation (10). Because activation of microglia is a temporally regulated process dependent on the injury and treatment (9, 10), future studies will be needed to investigate the consequences of immune activation in the irradiated brain following transplantation.
The activated microglia were found distributed primarily throughout the septotemporal axis of the CC dorsal to the CA1 and CA3 subfields of the hippocampus. Interestingly, this region corresponded to the location where the majority of engrafted cells were found (Fig. 3C-F). Further confocal analysis of all cohorts confirmed that the primary site of engrafted cell survival 8 months after treatment was the CC (Fig. 3C-F). The regional proximity of these cells supports the idea that activated microglia homing to sites of engrafted cells may initially facilitate the repair of the irradiated host brain by promoting synaptic activity and maintaining circuit integrity.
To pursue the foregoing ideas further, we investigated the potential impact of engrafted cells on host brain function through an assessment of the molecular distribution of the plasticity-related Arc protein in the host neurons of the hippocampus 1 or 8 months after irradiation and transplantation. Exposure to environmental enrichment and/or novelty leads to gene expression changes known to alter the composition of neuronal networks, thereby providing mechanisms for translating synaptic plasticity into changes in synaptic strength that impact learning and memory (32). While several immediate early genes such as c-fos and zif268 have been found to influence memory retention (2, 22), Arc expression has been shown to correlate both temporally and spatially with the inducing stimuli (23). Furthermore, lower basal Arc expression is associated with memory impairments in Alzheimer's patients (17) and genetic disruption of Arc expression leads to impairments in long-term memory (19, 36). The capability of Arc to rapidly respond to behavioral experiences (37) to maintain long-term potentiation and spatial memory consolidation (19) provides convincing evidence for Arc's role as a molecular determinant of memory.
Analysis of behaviorally induced Arc expression patterns in granule cell neurons of the DG and the pyramidal neurons of the CA1 in controls show that Arc expression was elevated only when animals were engaged in novel exploration, thus confirming that Arc can be rapidly induced by neuronal activity associated with new behavioral experiences. However, in irradiated animals, the percentage of neurons expressing Arc following novel exploration was not significantly different than caged controls. Notably, after cranial irradiation animals receiving stem cell grafts had increased numbers of Arc+ neurons at both 1 and 8 months postirradiation similar to that of the nonirradiated controls (Figs. 4 and 5). Although the percentage of Arc-expressing neurons in the DG is relatively low (i.e., <4%, Fig. 4), maintaining a small fraction of Arc activity during a behavioral experience has been shown to be critical for proper hippocampal function (22, 24). This is also consistent with electrophysiological recordings showing sparse activity in the DG during behavior and with the concept of sparse distributed coding that suggests only small subsets of neurons are required for optimal information encoding and processing (27, 29). Thus, a modest reduction in the number of granule cells expressing Arc in the DG may be sufficient to disrupt the tightly regulated sparse coding important for memory. Arc expression in pyramidal neurons of the CA1 was significantly higher in controls and in irradiated animals receiving stem cells when compared to either cage controls or, importantly, irradiated animals (Fig. 5). These findings clearly demonstrate the long-term and beneficial effects of stem cell grafting in the irradiated brain, where the presence of engrafted cells promotes the activity of host neurons by enhancing the expression of Arc in both granule cells (DG) and pyramidal neurons (CA1) of the hippocampus and thereby preserving cognition (8).
In summary, we have quantified engrafted cell survival 8 months following transplantation and irradiation and found that the majority of the surviving cells were regionally colocalized to activated microglia in the CC. Our past work has shown that irradiation disrupts Arc expression in the DG and CA areas (10, 38) and our present findings suggest that stem cell grafting may help overcome these disruptions throughout multiple hippocampal subfields. We have also recently reported that engrafted cells expressed Arc and NeuN when analyzed 1 month after transplantation, suggesting that engrafted cells possess the capability to functionally integrate into hippocampal circuits (5). Engrafted cells positive for these same markers were not observed at 8 months posttreatment suggesting that the majority of the long-term beneficial effects of stem cell grafting in the irradiated brain are due to trophic support that facilitates Arc expression in host neurons. The idea that the predominant beneficial effects of stem cell transplantation are derived from trophic support has been found in the majority of related studies using a variety of animal-based injury models (12). In this light, the secretion of neurotrophic factors from engrafted cells may nurture the host microenvironment to facilitate Arc expression over protracted postirradiation intervals, thereby promoting cognitive improvement.
Footnotes
Acknowledgments
This work was supported by the NIH-NINDS R01 NS074388581 (C.L.L.), NIH-NCI R01 CA133216 (S.R.), NIH Shared Instrumentation Grant OD010420 (Dr. Frank M. LaFerla), and California Institute for Regenerative Medicine (CIRM) training grant TG-01152 (M.M.A.). We are thankful to Mary Lan for excellent technical assistance. The authors declare no conflicts of interest.
