Abstract
The long-term effect of magnetically targeted neural stem cells in a rat focal cerebral ischemia model was investigated. In middle cerebral artery occlusion (MCAO) stroke model rats, ferumoxide-labeled human neural stem cells (hNSCs) were injected into the tail vein. MCAO rats were divided into three groups: ischemia only (IO), ischemia with NSC injection (IC), and ischemia with NSC injection and the use of magnet targeting (IM). Four weeks after MCAO and 3 weeks posttransplantation, a greater number of hNSCs were found in ischemic lesion sites in IM rat brain compared with IO and IC animals. In addition, differentiation of hNSCs into neurons or astrocytes and angiogenesis were markedly increased. In IM rats, infarct volume was considerably reduced, and function was significantly improved. The present study indicates that long-term use of magnetic fields may be a useful way to improve the efficacy of targeted migration of stem cells and functional deficits in stem cell-based therapy for ischemic brain injury.
Keywords
Introduction
Neural stem cell (NSC)-based cell therapy is a promising treatment for neurological diseases, such as stroke, spinal cord injury, Parkinson's disease, Huntington's disease, and amyotrophic lateral sclerosis (1–3,11,17,18). In animal models of stroke, intravenously transplanted human neural stem cells (hNSCs) migrated selectively to the brain lesions caused by ischemia and intracerebral hemorrhage, differentiated into neurons and astrocytes, and promoted angiogenesis and functional recovery in these animals (4–6, 8–10,13,14,25). However, delivery of NSCs via intravenous injection shows limited efficacy because only a small number of injected cells migrate to the target lesion sites (8,11–13,19,20). An efficient delivery of stem cells via an intravenous injection route was shown when rats were injected intravenously with ferumoxide-labeled NSCs, and an external magnet placed over the animal's brain increased the number of labeled cells in the brain (1,14). Ferumoxide, as superparamagnetic iron oxides (SPIOs), is a conventional magnetic resonance imaging (MRI) contrast agent and has been routinely used for clinical diagnoses. Efficient cellular internalization methods of SPIOs have been established such that the migration of injected NSCs can be monitored using MRI (16,18,26,27). Using this method, our previous study reported that hNSCs were labeled with ferumoxides in vitro and attracted to the site of an exterior magnetic field. Additionally, hNSCs labeled with ferumoxide, in combination with magnetic force, enhanced delivery of cells to target areas in focal cerebral ischemia and reduced brain infarct volume (17,24). For future clinical applications, it is necessary to investigate whether ferumoxide-incorporated hNSCs affect the efficacy of targeted delivery to the lesion site or differentiation of migrated hNSCs under long-term magnetism. However, the long-term effects of targeted, migrated hNSCs using a magnet in focal cerebral ischemia models have not been investigated.
In the present study, we first determined whether under long-term magnetism, ferumoxide-labeled hNSCs could be directed to a specific target area and differentiated in vivo in the ischemic hemisphere. Furthermore, in vivo studies were performed to determine the functional recovery effects of targeted delivery of ferumoxide-labeled hNSCs to the ischemic hemisphere in focal cerebral ischemia rat models.
Materials and Methods
Cell Culture
A stable, immortal female hNSC line, HB1.F3 (F3), was generated from primary fetal human brain cell cultures by infection with a recombinant replication incompetent retroviral vector pLNX.v-myc (12,13). F3 human NSCs express ATP-binding cassette subfamily G member 2 (ABCG2), nestin, and Musashi1, which are cell type-specific markers for NSCs and were grown in Dulbecco's modified Eagle's medium with high glucose (Sigma, St. Louis, MO, USA) supplemented with 5% fetal bovine serum (Sigma), 5% horse serum (Sigma), 100 U/ml penicillin, and 100 μg/ml streptomycin (Sigma).
Cell Labeling with Ferumoxide
As described in our previous study, F3 human NSCs (5 × 105 cell/ml) were incubated for 60 min at room temperature in culture media containing 25 μg/ml ferumoxide (Feridex IV; Advanced Magnetics, Cambridge, MA, USA) and 2 μg/ml poly-l-lysine (Sigma) prior to transplantation (27).
Animal Model and Transplantation Procedure
All experimental procedures were approved by the Animal Care and Use Committee of Seoul National University Hospital. Male Sprague–Dawley rats weighing 250–300 g (Orient, Seoul, South Korea) were used. Transient focal cerebral ischemia was induced using intraluminal thread occlusion of the left middle cerebral artery (MCAO) as described previously (15).
Three experimental groups were used: ischemia only (IO, n = 10), ischemia with cell injection (IC, n = 12), and ischemia with cell injection and magnet (IM, n = 11). IO rats received only MCAO, IC rats were injected with labeled F3 NSCs after MCAO without placement of magnet, and IM rats were hNSC-administered rats in which a neodymium magnet (5 × 10 × 2 mm, 0.32 T) was attached to the outside of the skull near the damaged left brain with a high-strength adhesive immediately after MCAO modeling. The magnet was removed 28 days after MCAO. Ferumoxide-labeled F3 cells [4 × 106 cells in 500 μl phosphate-buffered saline (PBS; Sigma)] were administered via tail vein injection 24 h post-MCAO.
Measurement of Iron Content
The iron content of rat brains was determined using inductively coupled plasma mass spectrometry [SPS-7000 (Ver. 2.03); Seiko Instruments, Tokyo, Japan].
Briefly, each contralateral and ipsilateral hemisphere was digested for 3 h at 60°C in a mixture (2.4 ml) of 35% hydrochloric acid (1.8 ml; Sigma) and 65% nitric acid (0.6 ml; Sigma). The mixture was diluted by adding sterile water to a volume of 5 ml, followed by filtering. The iron concentration was calculated using a standard curve obtained from ferrous chloride calibration standards containing 0, 1.25, 2.5, and 5 mg/L iron chloride (Sigma) in the above mixture. Iron content was confirmed by ferrozine-based spectrophotometric assays (7) using triplicate aciddigested samples. The average iron content per cell was calculated as the mean value divided by the tissue weight (kg) in each sample.
Histology and Immunohistochemistry
Rats were sacrificed 28 days post-MCAO. Briefly, animals were anesthetized and perfused through the heart with 100 ml cold saline (Sigma), followed by 100 ml 4% paraformaldehyde in 0.1 M phosphate buffer (Sigma). Brains were postfixed in the same fixative for 24 h, followed by immersion in 30% sucrose (Sigma) for 24 h, and sectioning on a cryostat (Leica CM 1900, Wetzlar, Germany) at 30 μm thickness. Five sections at 2-mm intervals were collected for Nissl staining. Briefly, the collected sections are gently mounted on gelatin-coated slides (Marienfeld GmbH & Co., Lauda-Koenigshofen, Germany) with a paintbrush, and then dried at room temperature overnight. The dried slides were dipped in 95% ethanol for 20 min and in chloroform (Merck, Darmstadt, Germany) solution for 10 min. They were dipped in a grades series of ethanol (95%, 100%; Merck) for 2 min each, exchanges of xylene (Sigma) for 5 min each, and then in a graded series of ethanol (100%, 95%, 95%) for 5 min each, after which slides were washed in distilled water (DW) for 5 min and then were dipped in 0.1% thionin solution (Sigma) at room temperature for 7 min. The stained slides were washed in DW for 2 min twice. The slides were dipped in 0.4% formalin acetic solution (Sigma) for 8 s, and then were washed with DW for 2 min. For dehydration, slides were dipped in a graded series of ethanol (95%, 95%) for 2 min, and were differentiated in 100% butanol (Sigma) for 2 min and then in cedarwood oil (Fisher Scientific, Inc.) for 5 min. The sections were cleared in xylene (5 min × 3). Finally, the sections were coverslipped. The area of the lesions was determined by Nissl staining, followed by image analysis (ImageJ; NIH, Bethesda, MD, USA).
Sections were used with Prussian blue staining to detect the presence of transplanted cells in the brain. Prussian blue staining was processed as follows: the sections were washed three times with PBS, incubated for 30 min with 5% potassium ferrocyanide (Sigma) in 5% hydrochloric acid (Sigma), rewashed, and then counterstained with nuclear fast red (Sigma). Prussian blue-stained slices were selected to determine the number of F3 NSCs that migrated into the rat brain. At least five fields of view (objective magnification: 100×) were selected randomly, photographed, and evaluated for Prussian blue staining using the image analyzer program (Qwin Pro, Leica).
We examined the systemic organs to detect any adverse reaction produced by the cells with a magnet. Systemic organs such as liver, spleen, kidney, lung, and heart of IO and IM animals were examined.
To detect transplanted hNSCs in the brain, sections were processed for immunofluorescence microscopy using anti-human nuclear matrix antigen (hNuA, mouse, 1:20; Millipore Biosciences, Temecula, CA, USA). To visualize colocalization of hNuA and cell type-specific markers [neuronal nuclear antigen (NeuN) for neurons or glial fibrillary acidic protein (GFAP) for astrocytes] in the same cell, double staining was employed. Briefly, sections were incubated with the hNuA antibody and cell type-specific antibodies directed against NeuN (1:100, rabbit; Millipore) to identify neurons, or anti-GFAP (1:1,000, rabbit; Millipore) antibody to identify astrocytes, followed by mixture of secondary antibodies [goat anti-mouse IgG Alexa Fluro 568 (1:200; Molecular Probes, Eugene, OR, USA) and goat anti-rabbit IgG Alexa Fluro 488 (1:200; Molecular Probes)]. To investigate if magnetically targeted hNSCs in the IM group enhance angiogenesis, hNSCs that migrated to brain lesions in both the IM and IC groups were stained with von Willebrand factor (vWF, angiogenic marker, rabbit, 1:200; Sigma), followed by secondary antibody goat antirabbit IgG Alexa Fluoro 488 (1:200; Molecular Probes). Cell nuclei were visualized using 4′,6-diamidino-2-phenylindole (DAPI; Sigma) staining. Immunofluorescent-stained sections were viewed using fluorescence microscopy (AH-3, Olympus, Tokyo, Japan).
Behavioral Tests
To determine whether the transplanted hNSCs can improve sensorimotor deficits, behavioral tests were compared (n = 9 for each group). Behavioral testing was performed by experienced testers blinded to the experimental group. The methods for each behavioral test are detailed below. Behavioral tests were performed in a blind fashion using modified neurological severity scores (mNSS). Briefly, the mNSS is a composite of motor, sensory, reflex, and balance tests (2). The scoring system was used with minor modifications, and neurological function was assessed on a scale of 0 to 18 (normal score = 0; maximal deficit score = 18). The limb-placing test was performed weekly, and it was used to assess forelimb and hindlimb responses to tactile and proprioceptive (forefoot, hind leg) stimulation (10,21). Briefly, the test consisted of four limb-placing tasks. The following scores were used to detect impairment of the forelimb and hindlimb: 0 points, the rat performed normally; 1 point, the rat performed with a delay of more than 2 s and/or incompletely; 2 points, the rat did not perform the task. Both sides of the body were tested. The maximum possible score achieved by highly damaged rats was 8.
Statistical Analyses
Statistical analysis was conducted by using GraphPad Prism version 6 for Windows (GraphPad Software, San Diego, CA, USA). Difference between two groups in terms of continuous variables was analyzed by using the Student's t test, and for multiple comparisons, one-way ANOVA was used with the Bonferroni correction. A value of p < 0.05 was considered statistically significant.
Results
Long-Term External Magnet-Enhanced Migration of Injected Ferumoxide-Labeled hNSCs in Focal Cerebral Ischemic Rat Brains
Twenty-eight days after injection of hNSCs in focal cerebral ischemic rats, Prussian blue staining of brain slices showed a higher number of Prussian blue-positive F3 NSCs in IM brains (MCAO + F3 NSCs + magnet) compared to IC brains (MCAO + F3 NSCs) (Fig. 1a). In addition, the area of Prussian blue staining was significantly larger in brain slices from IM rats compared to IC rats (2.1 ± 0.3 mm2 vs. 0.8 ± 0.6 mm2, p < 0.05) (Fig. 1b).
Prussian blue staining to detect presence of labeled cells. (a) Prussian blue-stained hNSCs from ischemia only (IO), ischemia with NSC injection (IC), and ischemia with NSC injection and the use of magnet targeting (IM) groups. High-magnification images of area outlined. Scale bar: 50 μm. (b) Absolute areas of Prussian blue-stained cells in the brain of MCAO-treated rats were analyzed using an image analyzer (*p < 0.05). (c) Iron content of rat brains. ipsil., ipsilateral hemisphere; contra., contralateral hemisphere (n = 3). ns, nonsignificant, *p < 0.05, **p < 0.005.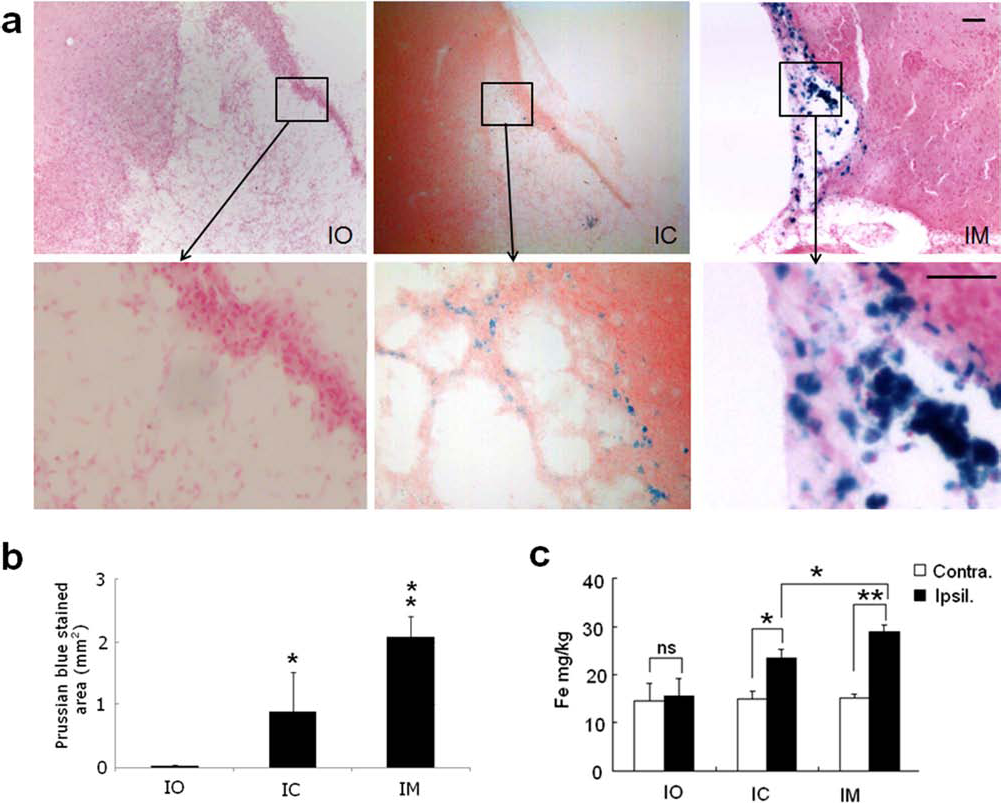
In the IO (MCAO alone) group, there was no significant difference between the iron content of the contralateral (14.4 ± 3.5 mg/kg) and ipsilateral (15.5 ± 3.6 mg/kg) hemispheres of the brain. However, the iron content of the ipsilateral hemisphere was higher than the contralateral hemisphere iron content in both IC (23.3 ± 1.8 vs. 14.7 ± 1.6 mg/kg, p < 0.05) and IM (30.1 ± 6.1 vs. 14.8 ± 0.4 mg/kg, p < 0.05) brain slices. Comparison of iron concentration in the ipsilateral hemisphere between the IC and IM groups showed that there was a significantly higher iron concentration in the IM group (p < 0.05) (Fig. 1c).
No Prussian blue-positive NSCs were found in other organs including liver, spleen, kidney, lung, and heart in IO and IM animals.
Magnetically Targeted hNSCs Induce Long-Term Infarct Volume and Functional Recovery
Control rats exhibited significant functional recovery over the course of the 28 days following MCAO, as assessed by several measurements, including mNSS and limb-placing tests.
mNSS results showed that functional deficits in the IM group were significantly reduced compared with the control IO group 21 days after MCAO (IO vs. IM on day 21: 6.1 vs. 5.2, p < 0.05). Twenty-eight days post-MCAO, the IM group had significantly reduced mNSS results compared to the IO and IC groups (IO vs. IM: 5.7 vs. 4.7, p < 0.05; IC vs. IM; 5.5 vs. 4.7, p < 0.05) (Fig. 2a).
Behavioral functional tests and infarct size after MCAO. (a) In the mNSS test, rats from the IM group had significantly reduced functional deficits compared to IO rats and both IO and IC rats on days 21 and 28, respectively. (b) No significant differences between the groups were observed in the limb-placing test. IO, rat with MCAO (n = 9); IC, MCAO rats transplanted with F3 human NSCs (n = 9); IM, MCAO rats transplanted with F3 hNSCs in the presence of a magnet on the left side of the brain (n = 9) (*p < 0.05). (c) Infarct size of rat brains 28 days after MCAO. Infarct volume measurements were performed on Nissl-stained coronal sections. Infarct volumes were measured using image analysis. Infarct volumes in the IM group were significantly reduced compared with that in IO and IC rats. IO (n = 10), IC (n = 12), IM (n = 11), *p < 0.05, **p < 0.01.
In limb-placing tests, the result in the IM group was slightly reduced compared to those in IC and IO groups on days 14 to 21 following MCAO. However, no significant difference was found among the groups on days 1 to 21 after MCAO (Fig. 2b).
In order to examine the effects of magnetically targeted hNSCs on MCAO lesions, rats were sacrificed 28 days post-MCAO, and infarct volumes were analyzed in brain sections processed for Nissl staining. There was a minor difference in infarct volume between IO and IC brains (221.9 ± 34.48 vs. 188.2 ± 22.27 mm3). However, the average lesion size in IM brains (152.4 ± 19.7 mm3) was significantly smaller than lesions observed in either IO or IC brains (p = 0.001 and 0.04, respectively) (Fig. 2c).
Magnetically Targeted hNSCs Enhance Angiogenesis
The migrated hNSCs were positive for hNuA staining, a specific marker for human cellular nuclei. Differentiated hNSCs were also positive for NeuN, a neuronal-specific marker, and GFAP, an astrocyte-specific marker. Twenty-eight days after MCAO, no difference was noted in the differentiation pattern into neurons or astrocytes from magnetically targeted or lesion-targeted hNSCs (Fig. 3a). Thus, we conclude that the presence of the magnetic field did not affect long-term differentiation of hNSCs into neurons or astrocytes. However, the number of cells positive for vWF, an endothelial cell-specific protein, in the IM group was significantly increased compared to the IC group 28 days after MCAO (Fig. 3b). This observation indicates that the IM group significantly promotes angiogenesis.
Immunological staining of rat brains to evaluate differentiation of injected hNSCs and effects on angiogenesis 28 days after MCAO. (a) Immunological staining of rat brains to evaluate differentiation of injected hNSCs 28 days after MCAO. The markers used were as follows: nucleus (DAPI, blue), human nuclear matrix antigen (hNuclei, red), astrocytes (GFAP, green), and neurons (NeuN, green). (b) Top: vWF-stained cells (green) in the brain lesion area from IC and IM groups. Scale bar: 200 μm. Bottom: absolute areas of vWF-stained cells in the brain from MCAO rats were analyzed using an image analyzer. vWF-stained cells in IM rats were significantly greater than IC (*p < 0.05).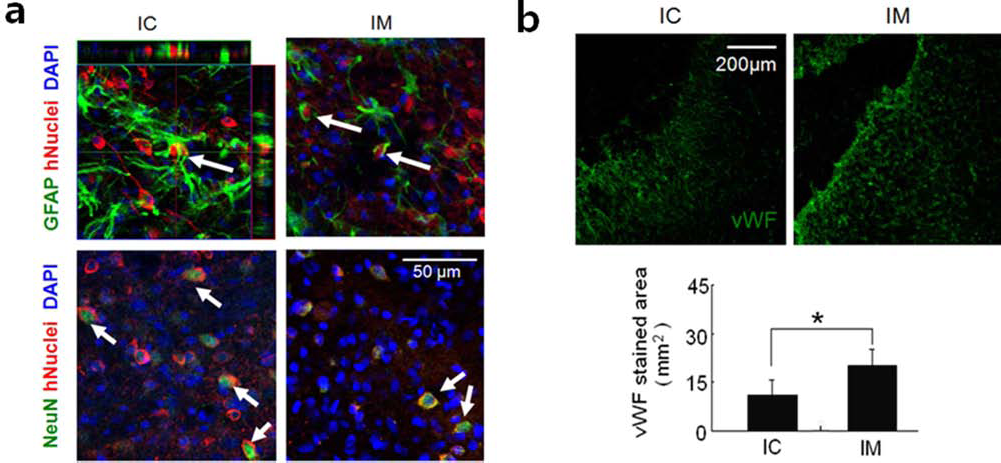
Discussion
The aim of this study was to investigate the long-term effects of targeted ferumoxide-labeled hNSCs using a magnetic field in focal cerebral ischemia model rat brain. In this study, we observed enhanced migration and differentiation of ferumoxide-labeled hNSCs to the ischemic lesion site for the period up to 3 weeks posttransplantation and 4 weeks after MCAO. In addition, functional deficits of the ischemic animals improved, and angiogenesis in the lesion sites was enhanced via magnetic targeting of intravenously injected hNSCs. This is the first study to report that long-term magnetically targeted stem-based cell therapy provides enhanced migration and differentiation of grafted hNSCs and increased angiogenesis in focal cerebral ischemia.
Our previous study showed that ferumoxide-labeled hNSCs in cell culture or in MCAO animals (using a magnetic field to enhance delivery of the labeled NSCs to the region of interest) reduced brain infarct volume after 7 days (17). However, before clinical trials are undertaken, it is necessary to determine the long-term effect of magnetically targeting cells in focal cerebral ischemia.
Our results show that significantly more intravenously injected ferumoxide-labeled hNSCs migrated toward the ischemic hemisphere using a magnetic field. These results correspond well with an earlier study reporting that there were more intravenously injected SPIO nanoparticle (SPION)-labeled rat mesenchymal stem cells present in the upper retinal hemisphere when a magnet was placed within the orbit 1 month after cell injection compared with the nonmagnet group (22). Similarly, 3 weeks after cell injection, superparamagnetic microsphere-labeled rat cardiosphere cells were visibly attracted toward the magnet and accumulated around the ischemic zone in myocardial infarction (25) and similarly in a rat model of heart ischemia/reperfusion (27). hNSCs are well known to show a high capacity of tumor and pathology tropism as reported by Schmidt (23) and Kim and deVellis (11). hNSCs' migration potential is as good as embryonic stem cells (ESCs) or induced pluripotent stem cells (iPSCs). We suggest the magnet targeting can also be utilized for the cell types derived from ESCs or iPSCs. It is known that the ESCs or iPSCs are not transplanted directly into target animals, but specific cell types developed from the ESCs or iPSCs are utilized for transplantation.
In the present study, we found that infarct volume in the IM group was significantly decreased compared to IC rats 28 days after MCAO, similar to our previous study 7 days after MCAO (17). The results of the present study agree with results from a previous study that reported on transient MCAO; magnetic field treatment of endothelial progenitor cells (EPCs) labeled with SPIONs significantly reduced atrophy volume compared with groups without magnetic field treatment. However, the EPCs were injected 90 min after transient MCAO rather than 24 h after MCAO, and the magnet was placed on the skull of the ischemic hemisphere for 2 h rather than 28 days (26).
Furthermore, the present study shows that the rats in the IM group had significantly reduced functional deficits compared to rats in the IC and IO groups. These findings are nearly identical to results from earlier studies showing that neurobehavioral deficiencies were markedly reduced in SPION-labeled EPCs-treated mice 28 days after transient MCAO (26). Hindlimb motor function of injected feridexlabeled bone marrow stromal cell group using a magnet demonstrated significant improvement compared with the nonmagnet group 6 weeks after a rat spinal cord injury (26). Therefore, we suggest that the marked improvement in neurological functional deficits 28 days after MCAO was induced by an increased number of intravenously injected ferumoxide-labeled hNSCs using magnetic targeting to the ischemic area.
The replacement of functional neurons using stem cell grafts possibly contributes to the behavioral improvement in the stroke-damaged brain (2). In the present study, we found improvement in functional deficits and infarct areas in ischemic rats following intravenous transplantation of ferumoxide-labeled hNSCs combined with magnetic targeting. As for a follow-up study, we investigated if the injected hNSCs differentiate into neurons or astrocytes under long-term magnetism. Earlier studies showed in vitro magnetic fields did not affect differentiation of rat mesenchymal stem cells (28) and in hNSCs (17). In these studies, in vivo differentiation of stem cells in animal model under long-term magnetism was not investigated. The results of the present study show that magnetically targeting hNSCs did not affect differentiation of hNSCs into neurons or astrocytes compared with IC rats under nonmagnetism conditions. These results indicate that the long-term magnetism does not affect differentiation status of transplanted hNSCs.
In a previous study, intravenously administered hNSCs were found to reduce deficits in rats by inducing angiogenesis (29). Brain transplantation of F3 hNSCs and F3 NSCs overexpressing vascular endothelial growth factor in mouse hemorrhage stroke model provided functional recovery and renewed angiogenesis of host brain. Transplantation of F3 hNSCs induced a three- to fourfold increase in vWF-positive microvessels compared with control PBS-injected brain (9). These results suggest a possible application of NSCs as a therapeutic agent for stroke to induce functional recovery and also to improve angiogenesis, in addition to cell replacement (2). In the present study, we found that angiogenesis in the damaged brain region of the IM group was significantly higher compared with that in the IC group, which is in agreement with previous studies (2,29,30). We suggest that the reduction in functional deficits is a result of the stimulation of angiogenesis, which is a consequence of dramatic accumulation of magnetically targeted hNSCs in the lesion area.
The present study has some limitations as well. First, the therapeutic benefits were obtained after long-term magnetism with targeted delivery of ferumoxide-labeled hNSCs, without comparing the effects of long-term and short-term magnetism. In the future, it will be necessary to obtain the time period that is sufficient to magnetically target injected cells to the region of interest. Second, the F3 stably immortalized hNSC line might be restricted in clinical therapeutic applications because it bears the v-myc oncogene. Despite these limitations, the present study demonstrates a novel method for enhancing the targeted migration of intravenously administered stem cells to the area of interest to promote functional recovery.
In summary, we show that the magnetically targeted ferumoxide-labeled hNSCs in MCAO animals enhanced delivery of labeled hNSCs to the area of interest, resulting in reduced brain infarct volume and improved function. Our results suggest that the magnetic targeting method enhances the therapeutic effects of stem cell-based cell therapy and is promising for future clinical applications.
Footnotes
Acknowledgments
This work was supported by the grants from Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education, Science and Technology (2012 NRF-2012R1A5A2051384) and the Korea Health technology R&D Project, Ministry of Health & Welfare, Republic of Korea. (HI12C03810000). The authors declare no conflict of interests.
