Abstract
The purpose of the study was to further scrutinize the potential of βB2-crystallin in supporting regeneration of injured retinal ganglion cell axons both in vitro and in vivo. Retinal explants obtained from animals after treatment either with lens injury (LI) alone or with combined LI 5 days or 3 days before or simultaneously with an optic nerve crush (ONC) were cultured for 96 h under regenerative conditions, and the regenerating axons were quantified and compared with untreated controls. These measurements were then repeated with LI replaced by intravitreal injections of γ-crystallin and β-crystallin at 5 days before ONC. Finally, bB2-crystallin-overexpressing transfected neural progenitor cells (bB2-crystallin-NPCs) in the eye were studied after crushing the optic nerve in vivo. Regeneration was monitored with the aid of immunoblotting of the retina and optic nerve both distal and proximal to the lesion site, and this was compared with controls that received injections of phosphate buffer only. LI performed 5 days or 3 days before ONC significantly promoted axonal outgrowth in vitro (p < 0.001), while LI performed alone before explantation did not. Intravitreal injections of β-crystallin and γ-crystallin mimicked the effects of LI and significantly increased axonal regeneration in culture at the same time intervals (p < 0.001). Western blot analysis revealed that crystallins were present in the proximal optic nerve stump at the lesion site in ONC, but were neither expressed in the undamaged distal optic nerve nor in uninjured tissue. bB2-crystallin-NPCs supported the regeneration of cut optic nerve axons within the distal optic nerve stump in vivo. The reported data suggest that bB2-crystallin-producing “cell factories” could be used to provide novel therapeutic drugs for central nervous system injuries.
Introduction
Retinal ganglion cells (RGCs) in adult mammals do not spontaneously regenerate axons after injury, which is presumably attributable to three main reasons: (A) the maturation-dependent decline of the intrinsic ability of the neurons to regenerate axons, (B) the growth-inhibiting environment surrounding the damaged neurons (4), and (C) insufficient support from target-derived neurotrophic factors (NTFs). However, adult RGCs can regenerate their axons under experimental conditions, implying that they do not lose their regenerative potential irreversibly. Several NTFs, for example, neurotropin-4/5, ciliary neurotrophic factor (CNTF), brain-derived neurotrophic factor (BDNF), and pigment epithelium-derived factor (PEDF), increase the intrinsic ability for axonal regeneration of the optic nerve in vivo and in vitro (7–9,32). Such experimental setups involve the intravitreal and/or neural administration of NTFs in vivo (33,43). In particular, neural progenitor cells (NPCs) exert relating NTFs, for example, BDNF, and other factors like oncomodulin and crystallins both in vitro and in vivo (1,3,5). Alternative approaches include the α-2 adrenergic receptor inhibitor brimonidine that promotes axonal regeneration by activating the phosphorylated Erk1/2 pathway via the tropomyosin receptor kinase B (17). The contribution of oncomodulin- expressing neutrophils also triggers axonal regeneration in the injured optic nerve of mice (24,46).
Another successful approach for inducing regeneration both in vivo and in vitro is lens injury (LI) after optic nerve crush (ONC) (14). The mechanisms underlying this growth-promoting effect may involve both inflammation-related (45) and noninflammatory (14,31) components, and the outcome of LI is clearly the long regrowth of axons. The beneficial growth-promoting inflammatory effect of LI has been attributed to the activation and infiltration of macrophages that produce oncomodulin (45) and to lens crystallins of the β/γ family, as demonstrated in both retinal explants and in vivo (12,14,31). β-Crystallin and γ-crystallin mimic the effects of LI and may act through activation of intrinsic CNTF (12).
Crystallins exhibit exceptional stability and comprise three major families (α-, β-, and γ-crystallins) that exhibit different degrees of homology to heat-shock proteins (HSPs) (19). Owing to their homology and relation to HSPs, they have been classified as stress proteins (19). Crystallins are abundant in the lens and were first considered to be lens-specific proteins but were rapidly recognized as neuronal and retinal proteins (29). While in lenticular tissue they are synergistically responsible for the maintenance of the lens transparency, their function in neurons is different—regulating various regenerative and degenerative conditions of the central nervous system (CNS), indicating their involvement in developmental injury and repair (29).
There are several lines of evidence that lens crystallins, which are released into the vitreous body following LI, mediate the growth-promoting effect of LI. Crystallins have been found to enhance axonal growth in animal models in vivo and in vitro (12,29,39). Crystallins are thought to transform RGCs into a robust regenerative state after injury, enabling them to regrow axons at higher growth rates (14,16). Crystallins are predominantly expressed in regenerating growth cones, indicating their crucial role in axonogenesis and growth cone formation (12,29). Moreover, regenerating retinal tissue can both secrete and internalize crystallins in vitro (29). These proteins may therefore be useful in novel drug-based therapeutic approaches for the treatment of optic nerve injury. However, to be effective, the drug has to reach the site of the lesion and be available when axonal repair is required.
One of the strategies available to test the effects of individual crystallins is the continuous en loco expression and release of crystallins from NPCs. Those NPCs activate matrix metalloproteinase-dependent proteolytic mechanisms and induce the proteolysis of chondroitin sulfate proteoglycans after intraneural administration supporting neurite growth (11,48). Moreover, NPCs migrate through the entire eye and maintain protective effects on diseased neurons (1,3,23). This approach suggests a high bioavailability at the lesion site. Neural stem cells and NPCs have been isolated from different parts of the embryonic and adult CNS of several mammalian species (18,22,35,36). NPCs can secrete several NTFs (1,5,23) and can be transfected to also produce any of the crystallins (18,27, 35,36). Beneficial effects of intravitreally injected βB2-crystallin-overexpressing NPCs (βB2-crystallin-NPCs) into injured RGCs and photoreceptors in rats were described recently (3). In a subsequent study, injected embryonic retinal progenitor cells were able to repair damaged optic nerves in mice (6).
The present study was undertaken to further scrutinize the regeneration-promoting effect of crystallins. For this purpose, we first examined under which time-dependent conditions crystallins exert their growth-promoting effects best, whether crystallins released by the injured lens or injected into the vitreous body are present at the injured optic nerve. We then injected NPCs and βB2-crystallin-NPCs into the vitreous after injuring the optic nerve to examine whether they stimulate axonal regrowth.
Materials and Methods
Animals and Drugs
All experiments were conducted in accordance with the Association for Research in Vision and Ophthalmology Statement on the Use of Animals in Ophthalmic and Vision Research. Sprague–Dawley rats (in total: n = 80) from our own breeding were housed in a standard animal room under a 12-h light/dark cycle with food and water provided ad libitum. The ethics committee (Regional government of North-Rheine/Westfalia) approved this study (Permission No.: 84-02.04.2011.A127). Surgical procedures were performed unilaterally on the left eye of female rats weighting 180–250 g, under general anesthesia induced by a mixture of ketamine and xylazine (both at 2 mg/kg body weight; Ceva-Sanofi, Düsseldorf, Germany) administered intraperitoneally. Gentamicin (Gentamytrex; Dr. Mann Pharma, Berlin, Germany) was applied topically after each surgical intervention. Tables 1 and 2 summarize the experimental setups of the in vitro and in vivo experiments.
Experimental Setup—In Vitro Experiments

IHC, immunohistochemistry; LI, lens injury; ONC, optic nerve crush; n/a, not applicable.
Experimental Setup—In Vivo Experiments
IHC, immunohistochemistry; ONC, optic nerve crush; n/a, not applicable.
Isolation of Purified Lens γ-Crystallin and β-Crystallin
Purified lens crystallins were obtained by extraction and separation using high-pressure liquid chromatography (HPLC) (12). Preliminary tests with fractionated lenses were performed to analyze the effects of lens crystallins on axonal growth in vitro. γ-Crystallin and β-crystallin dissolved in PBS (each 1 μg/μl), corresponding to fractions 3 and 4 from HPLC eluates, were used for intravitreal injections.
Synthesizing of Recombinant βB2-Crystallin
To synthesize recombinant βB2-crystallin (rβB2-crystallin) for intravitreal injections, the RNA was extracted from rat lenses using the RNAeasy Kit (Qiagen, Hilden, Germany) and transcribed into cDNA using a cDNA kit (Qiagen).
The 5′ end of each primer (forward: GGGCTCGAG ATGGCCTCAGACCACCAGAC; reverse: CCCGGAT CCTTAGCTGGAGGGGTGGAAGG) was modified to contain BamHI or HindIII restriction endonuclease sites to facilitate directional cloning in a pQE32 vector (Qiagen). Two additional bases were added between BamHI and the ATG to connect the HIS-Tag in the frame to rβB2-crystallin. The PCR product (of approximately 650 bp) was purified and ligated into pQE32 and transformed into competent C600/pREP4 cells. Cultures of these cells were grown at 37°C in LB broth containing 25 μg/ml kanamycin sulfate (Fluka Chemica AG, Buchs, Switzerland) and 50 μg/ml ampicillin sodium salt (Sigma-Aldrich, St. Louis, MO, USA) until an optical density of 0.4–0.5 was reached at 600 nm, when protein expression was induced by the addition of IPTG in a final concentration of 1 mM (Promega, Madison, WI, USA). The cells were grown overnight, and the protein was purified with TALON according to the protocols provided by the manufacturer (Clontech, Mountain View, CA, USA). The identity and purity of βB2-crystallin were verified by sodium dodecyl sulfate (SDS)–polyacrylamide gel (Sigma-Aldrich) electrophoresis and Western blotting. The synthesized βB2-crystallin was dissolved in PBS (180 μg/μl; Sigma-Aldrich) for intravitreal injections.
Lens Injury and Intravitreal Crystallin Injection
LI was performed (n = 3 for each group) by retracting the globe to expose its posterior aspect and fixed without using sutures. A glass capillary (Hilgenberg GmbH, Malsfeld, Germany) with a tip diameter of 20 μm was inserted through the sclera and retina at 2 mm from the optic nerve. The tip of the needle was bent at a 90° angle and inserted into the eye perpendicular to the sclera so as to intentionally puncture the lens surface. Ten microliters of PBS was slowly injected into the lens to promote its dissolution (15). The presence of LI was confirmed by direct visualization immediately after surgery and by monitoring the slow opacification that developed over the following few days.
To enable the intravitreal injection of larger volumes, 10 μl of anterior chamber fluid was removed by penetrating the cornea with a glass capillary (Hilgenberg GmbH, Malsfeld, Germany). Ten microliters of PBS containing γ-crystallin, β-crystallin, or rβB2-crystallin was injected slowly into the vitreous body (n = 3 for each group). An untreated group served as control (n = 3). Care was taken to avoid damage to the lens and uncontrolled dissemination of lens crystallins. Table 1 provides details of the experimental approaches in the different groups.
ONC and In Vitro Experiments
ONC was performed simultaneously with LI, at 3 days or 5 days (n = 3 each for axonal quantification, the 5-day group also for Western blotting) after LI, or 5 days after the intravitreal injection of γ-crystallin, β-crystallin (n = 3 each for axonal quantification and Western blotting), and rβB2-crystallin (n = 3 for Western blotting). The effects in noninjured tissue were analyzed by performing LI or rβB2-crystallin injection without ONC in the sham control groups (n = 3 each for axonal quantification and Western blotting). As control served untreated animals (for axonal quantification) or the contralateral retina and optic nerve (for Western blotting) (each n = 3). For ONC briefly, under general anesthesia a 1.0- to 1.5-cm-long incision was made in the skin above the right orbit of female Sprague–Dawley rats weighing 180–250 g. The optic nerve was surgically exposed inside the orbit under microscopic illumination. The meninges were opened longitudinally, and the optic nerve was crushed 4 mm behind the eye for 10 s using a jeweler's forceps (Geuder AG, Heidelberg, Germany), while avoiding injury to the central ophthalmic artery. A clear indentation mark at the lesion site confirmed the presence of effective nerve injury induced by ONC. The vascular integrity of the retina was verified by funduscopic examination. Rats were allowed to survive for 24 h after ONC.
Rats were sacrificed under a CO2 atmosphere. The eyecups were removed and placed in Hank's balanced salt solution (HBSS; Krackeler Scientific, Albany, NY, USA). Under sterile conditions, the retina was isolated, flat mounted on a nitrocellulose filter (Sartorius, Göttingen, Germany), divided into eight wedge-shaped pieces, and placed with the ganglion cell layer on a substrate of polylysine (20 mg/ml; Sigma-Aldrich, München, Germany) and laminin (20 mg/ml; Roche, Mannheim, Germany). Three milliliters of serum-free, S4 growth medium (PromoCell, Heidelberg, Germany) containing 1% gentamicin was added to the cultures, which were maintained for the following 5 days at 37°C in a humidified atmosphere containing 5% CO2. The number of outgrowing axons after 96 h in vitro was determined with the aid of an inverted phase-contrast microscope (Axiovert 135; Carl Zeiss, Jena, Germany) at a magnification of 200-fold. Axons that were present up to an imaginary line 200 μm from the explant margin were counted. When fasciculation was present, a single bundle was counted as one axon in all groups. Each experimental group comprised three eyes, corresponding to 24 slices.
Western Blotting
After intravitreal injection of β-crystallin and γ-crystallin and rβB2-crystallin and ONC, the eyes were enucleated, and the retina and the optic nerve (or, if lesioned, the respective proximal and distal parts of the optic nerve) were isolated, embedded, and frozen in liquid nitrogen. Probes were subsequently homogenized in SDS sample buffer containing 130 mM Tris-HCl (Carl Roth GmbH + Co. KG, Karslruhe, Germany), 10% w/v SDS, 10% mercaptophenol, 20% glycerol, and 0.06% w/v bromophenol blue (each Sigma-Aldrich). After sonicating and heating the samples, the protein concentration was determined using Bradford reagents (Bio-Rad, München, Germany). Fifty micrograms of protein from each sample were fractionated on 8%, 10%, or 12% SDS– polyacrylamide gels (depending on the examined protein) with a protein marker (Bio-Rad, Hercules, CA, USA). After electrophoresis, the proteins were transferred onto a nitrocellulose membrane (Whatman; GE Healthcare Europe GmbH, Freiburg, Germany). The blots were incubated in blocking solution containing 5% fat-free dry milk (Carl Roth GmbH + Co. KG) and 0.1% Tween-20 PBS (Sigma-Aldrich) for 1 h, followed by incubation overnight at 4°C with polyclonal anti-rabbit γ-crystallin and anti-goat β-crystallin (Santa Cruz Biotechnology, Santa Cruz, CA, USA) used at dilutions of 1:700 and 1:1,000, respectively, in samples following LI. Polyclonal anti-goat βB2- crystallin (Santa Cruz Biotechnology) was used at a dilution of 1:700 for samples after rβB2-crystallin injection. The application control antibody anti-calnexin (Sigma-Aldrich) was used at a dilution of 1:10,000. The membrane was then incubated with the secondary antibody conjugated with horseradish peroxidase (Sigma-Aldrich) in blocking solution for 1 h at room temperature (RT). Antibodies were detected by enhanced chemiluminescence (Amersham, Rockville, UK), with the relative densities of the protein spots analyzed using Alpha Ease (Alpha-Ease FC software 4.0; Alpha Innotech Cooperation, San Leandro, CA, USA). The protein density of a fixed area was determined for each spot after subtracting the specific background density in the surrounding region. The spot density was correlated and corrected with the relative density of the particular application control. The spot density of the proximal part of the optic nerve after LI performed 5 days before ONC and the spot density of the retina after intravitreal rβB2-crystallin injection were defined as the respective reference marks, and the relative values were calculated.
Preparation and Transfection of NPCs
To retrieve and culture NPCs, embryos were removed from pregnant Sprague–Dawley rats (n = 5), under lethal anesthesia by cesarean section at days E13.5 and E15, and were stored in sterile HBSS (PAA Laboratories, Pasching, Germany) with 1% penicillin streptomycin (Sigma-Aldrich, Hamburg, Germany) on ice until dissection. The brains were removed and mechanically homogenized, and the homogenate was treated with 0.1% trypsin (Sigma-Aldrich, St. Louis, MO, USA) in HBSS to obtain single cells. The suspension was transferred into a culture medium comprising Dulbecco's modified Eagle's medium and Ham's nutrient mixture (DMEM-F12; PAA Laboratories) supplemented with 1% PenStrep, 1% N2 supplement (PAA Laboratories), 10 ng/μl recombinant human basic fibroblast growth factor (R&D Systems, Minneapolis, MN, USA), and 10 μl of insulin (2.5 mg/ml; PromoCell, Heidelberg, Germany). The suspension was gently triturated, and 4.5 × 106 cells were plated at a density of 200,000 cells/ml on uncoated Advanced TC petri dishes (Greiner Bio-One, Frickenhausen, Germany) to inhibit further differentiation. The cells were incubated at 37°C in a humidified atmosphere of 5% CO2. All cell-harvesting steps were performed under sterile conditions.
For preparation of βB2-crystallin-NPCs, rat βB2-crystallin was cloned into pIRES-ACGFP (Clontech, Palo Alto, CA, USA) so that rat βB2-crystallin and green fluorescent protein (GFP) could be translated from a single bicistronic mRNA. The transfection protocol used was very similar to that described by the manufacturer and produced high transfection efficacies. NPCs were transfected by mechanically dissociating cultured cells that had clustered into neurospheres into single cells. The cells were centrifuged for 5 min at 400 × g (4°C). The culture medium was removed, and about 5 × 106 cells were resuspended in 100 μl of the Amaxa Rat NSC Nucleofector Kit (Lonza Cologne, Köln, Germany). The vector (comprising 3.5 μg of DNA) was added, and the cells and DNA were gently mixed. The cells were then electroporated using the Nucleofector device (Lonza Cologne). The efficiency of transfection was monitored based on GFP fluorescence. A transfection efficiency of 56.4 ± 10.9% was evaluated and was analogous to other studies (25,36). Cells were used for a maximum of 2 weeks after preparation. After this procedure was complete the βB2-crystallin-NPCs were placed into the culture medium and cultured for up to 7 days. Cultured NPCs were examined by immunohistochemistry (further details are provided below) for selected proteins: nestin (mouse, monoclonal, dilution 1:100; AbD Serotec, Puchheim, Germany) was used to reveal the retained stem cell-like features, and βB2-crystallin (goat, polyclonal, 1:100; Santa Cruz Biotechnology) was administered to examine its expression characteristics. The secondary antibodies used were Cy2 (goat, anti-mouse, dilution 1:200; Jackson ImmunoResearch Europe, Suffolk, UK) and TRITC (donkey, anti-goat, dilution 1:100; Jackson ImmunoResearch Europe). Transfected βB2-crystallin-NPCs were prepared for injection by first washing the cell suspensions in HBSS. Cell numbers were then counted using the trypan blue (Gibco, Life Technologies GmbH, Darmstadt, Germany) exclusion method resulting in 67.4 ± 4.3% viability of cells. The cells were then transferred to the transplantation medium (i.e., HBSS). Surgery and cell injection were performed within 1 h of cell preparation.
Unilateral ONC and Intravitreal Treatment In Vivo
The primary degeneration of RGCs was initiated by performing unilateral ONC followed by the intravitreal injection of βB2-crystallin-NPCs (n = 4). After making a skin incision near the superior orbital rim, ONC was performed according to the “micro crush lesion” technique (38), which was modified by the time used for ligation with a suture (8.0 Ethicon; Johnson & Johnson, St-Stevens-Woluwe, Belgium), that was 20 s instead of 60 s described in the original paper. A total of approximately 300,000 living cells in 10-μl volume of βB2-crystallin-NPCs were injected through the sclera with a pooled glass capillary (diameter about 20 μm), with care being taken to avoid damage to the lens. Controls were injected intravitreally with 10 μl of nontransfected NPCs (n = 4) or rβB2-crystallin protein (corresponding to 180 μg/μl) (n = 4). After subcloning into pQE32 (Qiagen), rat βB2-crystallin was expressed and purified; bacterial cultures were then centrifuged (1,500 × g for 20 min) and subsequently resuspended in buffer 1 (pH 8.0) containing 8 M urea, 50 mM NaH2PO4, 15 mM imidazole (Carl Roth GmbH + Co. KG), 10 mM Tris-HCl, and 100 mM NaCl (each Carl Roth GmbH + Co. KG). After cloning, cell lysates were centrifuged at 20,000 × g for 30 min. The supernatant was purified using immobilized metal affinity chromatography (Clontech); elution was performed using buffer 1 modified by supplementation with 100 mM imidazole (pH 6.0). The proteins were dialyzed in aqua dest. provided by a water purification system (EMD Millipore Corporation, Billerica, MA, USA). Owing to the decreased effects of intravitreally injected rβB2-crystallin in the case of ONC after 4 weeks (unpublished data from our laboratory), we performed an additional intravitreal injection of rβB2-crystallin in the 8-week group after 4 weeks. For sham control groups (see above), 10 μl of sterile phosphate-buffered saline (PBS) was injected after ONC (n = 4). The experimental protocols are listed in Table 2.
Immunohistochemistry and Western Blotting
The effects of rβB2-crystallin, nontransfected NPCs, and βB2-crystallin-NPCs on axonal degeneration and regeneration were investigated by sacrificing rats using CO2 inhalation (n = 4 from each group). The eyeballs containing the optic nerve were explanted, frozen separately in Tissue-Tek (Sakura Finetek Europe, Alphen aan den Rijn, The Netherlands), and then stored at −80°C until further use. For Western blotting to detect retinal βB2-crystallin expression, the tissue was prepared as mentioned above. As application control antibody β-actin (Sigma-Aldrich) was used at a dilution of 1:10,000. Sections were cut along the visual axis with a cryostat (10 μm thick; CM 1500, Leica, Bremen, Germany) and mounted onto glass slides (Engelbrecht, Edermünde, Germany). Primary antibodies for neurofilament-200 (NF-200) (mouse, monoclonal, dilution 1:400; Sigma-Aldrich), β-III-tubulin (mouse, monoclonal, dilution 1:500; Covance Inc., Munich, Germany), and GAP-43 (rabbit, polyclonal, dilution 1:200; Millipore, Bredford, MA, USA) were used to detect axonal degeneration and regeneration. The secondary antibody used was Cy2 (goat, anti-mouse, dilution 1:200; Jackson ImmunoResearch Europe; goat anti-rabbit, dilution 1:400; Sigma-Aldrich) and Cy3 (goat, anti-mouse, dilution 1:200; Jackson ImmunoResearch Europe). Slides were fixed in methanol for 10 min at −20°C. After rinsing the cells with PBS, they were incubated with blocking solution containing 10% goat serum (Sigma-Aldrich) for 2 h at RT; the primary antibodies were then applied. After washing, cells were incubated with secondary antibodies (1 h at RT) and then washed with PBS. Antifade mounting medium was used to cover the cells (Mowiol, Hoechst, Frankfurt, Germany). The slides were viewed using the appropriate filter on a microscope with an epifluorescence viewing capability (Axiophot; Carl Zeiss Meditec, Oberkochen, Germany). Negative controls comprised sections processed without the addition of the primary antibodies and by using preimmune sera. Control and experimental sections were stained simultaneously to reduce possible variations in immunohistochemical staining characteristics.
Data Evaluation
All data regarding the number of axons per retinal explant are presented as mean ± SD values. Data were analyzed statistically using the two-independent-samples test (Statistica version 7; StatSoft, Tulsa, OK, USA) to identify whether or not they conformed to a Gaussian distribution, and then processed using either the independent-samples t-test (for a Gaussian distribution) or the Mann–Whitney U-test (for a non-Gaussian distribution). The remaining quantitative data were analyzed statistically using two-way analysis of variance (ANOVA; Statistica version 7) or, if the distribution was not Gaussian, the Kruskal–Wallis H-test. Post hoc analyses using the Tukey HSD test were performed to identify possible differences among the experimental groups. Optic nerve sections of in vivo experiments were photographed with the aid of an Axiophot microscope (Carl Zeiss) equipped with AxioVision software (Carl Zeiss). NF-200-, GAP-43-, and β-III-tubulin-positive axons were counted to evaluate the number of axons within the regenerating optic nerve. All counts were performed at defined, distal distances from the crush site (e.g., distal 1: 500 μm, distal 2: 1,000 μm) after 4 weeks. Additional quantification was carried out with NF-200-positive axons after 8 weeks. The viability of cells was indicated by an intact nucleus provided by DAPI staining. In total, 15 sections from four optic nerves in each group were evaluated. One-way ANOVA was used to compare both groups overall, followed by a Student's t-test involving standard errors of the means to compare selected groups at the respective distances from the crush site. The estimated local p values were adjusted for multiple comparisons by Holm–Bonferroni (Project R, Version 3.0; Foundation for Statistical Computing, Vienna, Austria, http://www.r-project.org). A value of p < 0.05 was judged as statistically significant.
Results
Effect on Axonal Growth
To analyze whether the time of exposure to lenticular factors before ONC influences axonal growth in vitro, LI was performed 5 days or 3 days before or simultaneously with ONC (each n = 3). Retinas were explanted 24 h after ONC. An untreated group (n = 3) and a group with only LI (n = 3) served as the controls. Axonal growth was significantly (p < 0.001) greater for LI performed 3 days or 5 days before ONC than for LI alone, LI performed simultaneously with ONC and untreated controls. The increased axonal growth after LI performed 5 days before ONC compared to LI performed 3 days before ONC showed no significant difference. LI performed at the same time with ONC or LI alone had no growth-promoting effect (Fig. 1A), which suggests that some lead time for the exposure to lenticular factors is required for the neuron to enter a robust regenerative state. Moreover, LI seems to exert greater effects on injured tissue than on noninjured tissue (i.e., LI without ONC).

Axonal growth-promoting effects after lens injury in vitro. (A) Quantification of axonal growth in vitro for lens injury (LI) performed simultaneously with optic nerve crush (LI + ONC), or 3 days before ONC (3 days LI + ONC) or 5 days before ONC (5 days LI + ONC) if samples remained untreated (controls) or received only LI. Outgrowth was significantly increased (*p < 0.05) for 3 days and 5 days LI + ONC compared to control, LI, and LI + ONC samples, and did not differ among the latter three conditions. (B) Axonal regeneration of control samples, samples after 5 days LI + ONC, and intravitreal injection of γ-crystallin and β-crystallin, showing that axonal outgrowth was significantly increased (**p < 0.01) for both γ-crystallin and β-crystallin compared to controls and 5 days LI + ONC samples. (C) Western blot analyses of lenses. Lysates of lenses were prepared and probed for γ-crystallin and β-crystallin. (D-F) γ-Crystallin and β-crystallin facilitates axon outgrowth from retinal explants. (D) Untreated control compared to intravitreal injection of (E) γ-crystallin or (F) β-crystallin ONC. Statistically significant differences: *p < 0.05, **p < 0.01 Scale bars: 50 μm (D-F).
To determine whether crystallins are the main factors triggering the growth-promoting effect of LI, purified crystallin fractions of γ-crystallin and β-crystallin were injected intravitreally without injuring the lens at 5 days before ONC (each n = 3). An untreated sham group served as the corresponding control (Fig. 1B). Western blot analysis revealed the presence of γ-crystallin and β-crystallin (Fig. 1C). Intravitreal injections of purified γ-crystallin and β-crystallin fully mimicked the effect of LI and significantly (p < 0.01) enhanced axonal growth by 20-fold compared to sham animals. Whether the effects of crystallin do not depend exclusively on inflammatory responses was tested by measuring neurogenic effects in retinal slices cultured in vitro (Fig. 1D–F). A dense growth of axons was found in vitro after the injection γ-crystallin and β-crystallin (Fig. 1E, F), which differed significantly (p < 0.01) from the growth in untreated controls (Fig. 1D). The growth-promoting effects of intravitreal γ-crystallin and β-crystallin injections even exceeded those of LI performed 5 days before ONC, while the axonal growth did not differ significantly between γ-crystallin and β-crystallin injections (Fig. 1B).
Presence of Crystallins Inside Injured Axons
To examine whether crystallins are expressed at the site of a lesion Western blotting of the retina and optic nerve (proximal and distal to the crush site if lesioned) was performed after LI (n = 3). The levels of crystallins were compared between injured (with ONC), untreated (contralateral to ONC), and noninjured (sham control without ONC) cases. γ-Crystallin was markedly upregulated in the retina and the proximal part of the optic nerve, while it was absent and not detectable in the distal part of the optic nerve. It was also absent in both the retina and optic nerve samples of the contralateral parts and with LI alone (i.e., without ONC) (Fig. 2A). The relative density analysis confirmed these results (Fig. 2B). Comparable results were obtained for β-crystallin (Fig. 2C), showing sparse β-crystallin in the contralateral retina and no crystallin in noninjured samples or distal to the optic nerve in ONC cases, as confirmed by relative density analysis (Fig. 2D). These data show that LI-released crystallins are expressed at the site of lesion in the proximal optic nerve stump.

Expression of γ-crystallin and β-crystallin in retina and optic nerve after ONC in vivo. Western blot analysis for γ-crystallin and β-crystallin of the proximal (prox ON) and distal parts (dist ON) of the nerve, respectively, as well as of the retina after LI and performed optic nerve crush (LI + ONC). The contralateral ON and contralateral retina to ONC served as untreated control (CL to ONC). LI alone (i.e., without ONC) for the retina and the nerve for (A) γ-crystallin and (C) β-crystallin served as sham control (sham LI – ONC), including the application control with calnexin, and the corresponding relative densities of (B) γ-crystallin and for (D) β-crystallin. The percentage densities are given relative to the density of the proximal nerve for 5 days LI + ONC. Statistically significant differences: *p < 0.05.
To further determine whether recombinant βB2-crystallin is also expressed at the site of the lesion, rβB2-crystallin was injected intravitreally (n = 3). Western blotting for βB2-crystallin of the proximal and distal part of the optic nerve and the retina was subsequently performed, with the results compared with those for a sham group in which rβB2-crystallin was injected into the vitreous body, but without ONC. Confirming the results after LI, βB2-crystallin was strikingly upregulated in the retina and the proximal part of the optic nerve, while it was scarce in the contralateral retina and absent in the contralateral optic nerve. In the sham group, βB2-crystallin was found in the retina, while no βB2-crystallin was detectable in noninjured optic nerves (Fig. 3A). The spot density measurements confirmed these results (Fig. 3B).

Expression of βB2-crystallin in retina and optic nerve after optic nerve crush in vivo. Western blot analysis for βB2-crystallin of the ipsilateral retina and the ipsilateral proximal optic nerve (prox ON) after intravitreal injection of recombinant βB2-crystallin (rβB2-crystallin) and performed optic nerve crush (IL + ONC). Contralateral retina and contralateral optic nerve to ONC serves as untreated control (CL to ONC). Intravitreal injection of rβB2-crystallin for ON and retina without ONC served as sham control (sham – ONC). (A) Loading control with calnexin and (B) corresponding relative plot densities. Statistically significant differences: *p < 0.05.
Effects of rβB2-Crystallin and βB2-Crystallin-NPCs on Regeneration In Vivo
Cultured NPCs were examined so as to reveal their expression characteristics in vitro (Fig. 4). NPCs in culture retain their stem-cell feature of expressing nestin (Fig. 4A–C), which remains after stable transfection (Fig. 4D), and they also show a stable expression of several NTFs (not shown), including βB2-crystallin (Fig. 4E–H). We previously showed the continuing expression of several NTFs (e.g., βB2-crystallin) by βB2-crystallin-overexpressing NPCs after intravitreal administration in vivo (3).
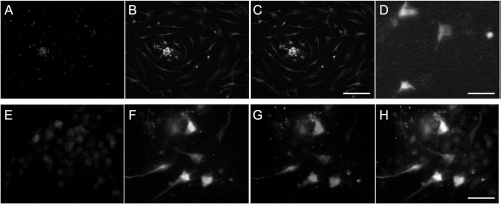
Expression of βB2-crystallin in cultured NPCs in vitro. Immunohistochemistry characterization of cultured neural progenitor cells (NPCs) derived from cultured rat fetal neural tissue. (A–C) Cultured NPCs have stem cell features (using nestin, B) and were found as single cells and in cell clusters called neurospheres. (C) Nestin-DAPI merged image. (D) GFP-positive cells reveal transfection efficiencies. (E–H) Transfected NPCs stained with nestin (F) and βB2-crystallin (G) to show expression characteristics in culture. (H) Nestin-βB2-cyrstallin-DAPI merged image. In these images, the transfection rate is not representative and is about 25%. DAPI (A, E) was used to stain cell nuclei. Scale bars: 50 μm (A–C) and 20 μm (D–H).
To analyze whether rβB2-crystallin or βB2-crystallin-NPCs prevent RGC axon degeneration and promote RGC axon regeneration in vivo, we evaluated their effects in the rat ONC model (each n = 4). Dynamic remodeling of neuro-filaments is necessary for axonal maintenance and regeneration. Recent reports provide lines of evidence that NF-200 is an indicator for axonal damage and regrowth (40). Changes in axonal NF-200 staining patterns indicate swellings and irregular granulation of axons (2). Also, NF-200 shows the capacity of nerve fibers to regrow into injury sites within the CNS (10). Therefore, we performed NF-200 staining to quantify axons at 4 and 8 weeks after ONC (37) (Fig. 5). Injection of rβB2-crystallin significantly increased the number of NF-200-positive axons after 4 weeks [proximal: 100 ± 6.08%, p > 0.05; distance 1: 83.36 ± 4.81%, p > 0.05; distance 2: 72.8 ± 6.9%, p < 0.05) (Fig. 5AB) and after 8 weeks (proximal: 100 ± 9.67%, p > 0.05 (Fig. 5M–O, P–R, AC); distance 1: 89.64 ± 19.82%, p < 0.05 (Fig. 5M–O, S–U, AC); distance 2: 77.03 ± 12.20%, p < 0.05 (Fig. 5V–X, AC)] distal to the crush site compared to controls (after 4 weeks, proximal: 100 ± 13.25%; distance 1: 77.27 ± 19.22%; distance 2: 57.95 ± 12.13%; after 8 weeks, proximal: 100 ± 20.86%; distance 1: 54.44 ± 13.14%; distance 2: 36.29 ± 7.61%) (Fig. 5AB, AC). These data indicate that the application of rβB2-crystallin results in elongation of axons within the injured rat optic nerve.
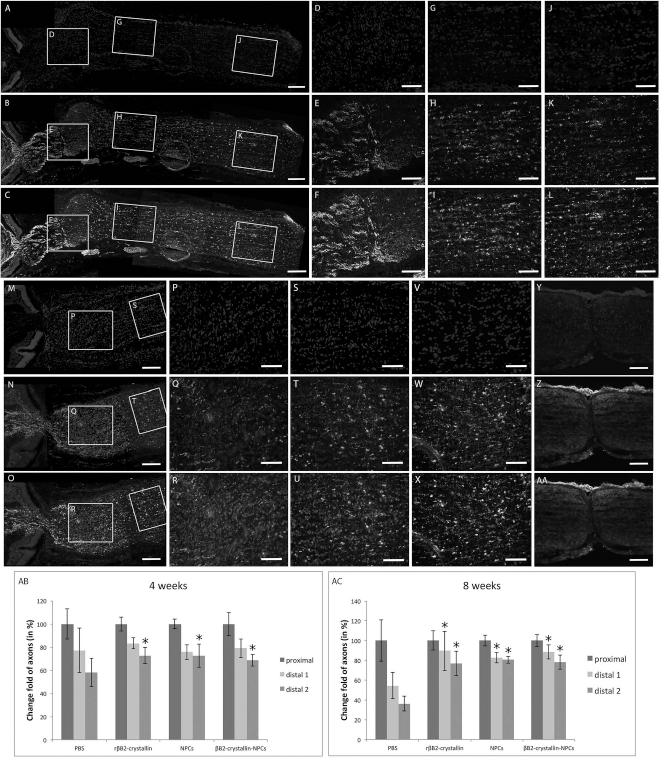
Intravitreal application of rβB2-crystallin and βB2-crystallin-NPCs prevents axonal degeneration after optic nerve crush in vivo. Effects of intravitreal injected (A–L) βB2-crystallin-NPCs and (M–AA) rβB2-crystallin in the model of crushed optic nerve (ONC) after 8 weeks. Slides of proximal part (A–C, D–F), distal part 1 (A–C, G–I), and distal part 2 (A–C, J–L) based on lesion site of ONC after intra-vitreal injection of βB2-crystallin-NPCs are shown. (B, E, H, K) Axon fibers were stained with NF-200. (A, D, G, J) DAPI was used to stain cell nuclei. (C, F, I, L) NF-200-DAPI merged images. Also proximal part (M–O, P–R, Y–AA), distal part 1 (M–O, S–U), and distal part 2 (V–X) of the optic nerve after intravitreal injection of rβB2-crystallin are shown. (N, Q, T, W, Z) Axon fibers were stained with NF-200. (M, P, S, V, Y) DAPI was used to stain cell nuclei. (O, R, U, X, AA) NF-200-DAPI merged images. Scale bars: 100 μm (A–C, M–O, Y–AA) and 50 μm (D–L, P–X). Relative changes in axonal fibers proximal and distal to the crush site after intravitreal injection of PBS, rβB2-crystallin, nontransfected NPCs, or βB2-crystallin-NPCs after (AB) 4 weeks and (AC) 8 weeks. Statistically significant differences: *p < 0.05.
To provide a sustainable production and release of βB2-crystallin, NPCs producing the protein were injected into the vitreous body in ONC eyes. Four weeks later, the number of NF-200-positive axons that had crossed the lesion site and grown into the distal part of the optic nerve was similar to that in the rβB2-crystallin-protein group (Fig. 5AB). After 4 weeks, the loss of NF-200-positive axons at the longest distance from the site of injury was significantly lower in the βB2-crystallin-NPCs group (proximal: 100 ± 9.98%, p > 0.05; distance 1: 79.02 ± 6.57%, p > 0.05; distance 2: 68.78 ± 5.13%, p < 0.05) compared to control (Fig. 5AB). After 8 weeks, there were more NF-200-positive axons remaining in the βB2-crystallin-NPC group than in controls irrespective of the distance from the crush site [proximal: 100 ± 6.16%, p > 0.05 (Fig. 5A–C, D–F, AC); distance 1: 88.48 ± 6.99%, p < 0.05 (Fig. 5A–C, G–I, AC); distance 2: 78.01 ± 7.02%, p < 0.05 (Fig. 5A–C, J–L, AC)]. Although a slight increase in NF-200-positive axons was observed in distance 1, in general no significant difference was evaluated between nontransfected NPCs (proximal: 100 ± 4.34%, p > 0.05; distance 1: 75.67 ± 8.1%, p > 0.05; distance 2: 72.6 ± 10%, p > 0.05) compared to βB2-crystallin-NPC after 4 weeks (Fig. 5AB). After 8 weeks increased NF-200-positive axons were also found in the βB2-crystallin-NPC group compared to nontransfected NPCs (proximal: 100 ± 5.35%, p > 0.05; distance 1: 82.64 ± 8.1%, p > 0.05; distance 2: 80.56 ± 3.42%, p > 0.05) in distance 1 without statistical significance (Fig. 5AC).
In addition, we performed β-III-tubulin and GAP-43 staining to show the relation of regenerating axons within the crushed optic nerve after application of βB2-crystallin-NPCs compared to PBS-, rβB2-crystallin, and NPCs group. Counts of GAP-43 and β-III-positive axons revealed increased numbers of axons in the experimental groups (Fig. 6).

Intravitreal application of rβB2-crystallin and βB2-crystallin-NPCs promote axonal regeneration after optic nerve crush in vivo. Effects of intravitreal injected (A–H) βB2-crystallin-NPCs in the model of crushed optic nerve (ONC) after 4 weeks. Area of ONC in different magnifications (A–D: 10×; E–H: 20×) is shown. Axons were stained with β-III-tubulin (Cy2) (B, F) and GAP-43 (Cy3) (C, G), while cell nuclei were counterstained with DAPI (A, E). (D, H) β-III-tubulin-GAP-43-DAPI merged images. Distal part (I) of the optic nerve after intravitreal injection of βB2-crystallin-NPCs shows GAP-43-positive axons. Scale bars: 100 μm (A–D) and 50 μm (E–I). Relative changes in GAP-43-positive axons in relation to β-III-tubulin-stained axons proximal and distal to the crush site after intravitreal injection of PBS, rβB2-crystallin, nontransfected NPCs, or βB2-crystallin-NPCs were quantified after 4 weeks (J). Western blot analysis to reveal expression levels of βB2-crystallin within the PBS group, the nontransfected NPCs group, and the βB2-crystallin-NPCs group, including the application control with β-actin (K). The percentage densities are given relative to the density of the control group (L). Statistically significant differences: *p < 0.05.
An increase in GAP-43-positive axons was seen in the rβB2-crystallin group (proximal: 100 ± 7.6%, p < 0.05; distance 1: 58 ± 2.7%, p < 0.05; distance 2: 46.3 ± 2.3%, p < 0.05) compared to control (proximal: 100 ± 1.5%; distance 1: 0 ± 0%; distance 2: 0 ± 0%), that showed no regenerating axons (Fig. 6J). Application of nontransfected NPCs results in increase in GAP-43-positive axons at all sites of optic nerve (proximal: 100 ± 8.7%, p < 0.05; distance 1: 72.9 ± 3.5%, p < 0.05; distance 2: 78.3 ± 3.4%, p < 0.05) compared to control and the βB2-crystallin-NPCs group (Fig. 6J). In distance 1, a significant increase in GAP-43 axons was found in the rβB2-crystallin group (proximal: 100 ± 9.4%, p > 0.05; distance 1: 81.6 ± 4.1%, p < 0.05; distance 2: 79.9 ± 3.6%, p > 0.05) compared to nontransfected NPCs (Fig. 6J).
Finally, we performed Western blotting to reveal the retinal expression level of βB2-crystallin in the different experimental groups after 4 weeks (Fig. 6K). Relative optical density revealed increased levels of βB2-crystallin in the rβB2-crystallin group (234.2 ± 14.8%, p < 0.05), in the nontransfected NPCs group (392.6 ± 29.8%, p < 0.05), and the βB2-crystallin-NPCs group (488.6 ± 29.7%, p < 0.05) compared to control. A significant difference was found between both rβB2-crystallin and nontransfected NPCs (p < 0.05) and βB2-crystallin-NPCs and nontransfected NPCs p < 0.05) (Fig. 6L).
These data show that βB2-crystallin-NPCs significantly reduce the degree of axonal degeneration and trigger axonal regeneration in the rat optic nerve, which further implies that rβB2-crystallin and βB2-crystallin-NPCs expression improves the neuron-intrinsic capability of rat RGCs to override the growth-inhibiting environment in vivo and empowers adult mammalian neurons to reextend lengthy axons.
Discussion
The principal findings of this study are as follows:
LI promotes axonal growth of retinal explants in vitro if it is performed before ONC.
LI exerts its growth-promoting effect mainly in injured tissue, while showing no effects in noninjured tissue (i.e., LI without ONC).
Crystallins of the β and γ superfamily are the main factors involved in the triggering of axonal regeneration by LI.
Crystallins are expressed at the lesion site to the proximal part of the ON both after LI and the intra-vitreal injection of rβB2-crystallin, while they are not expressed beyond the scar in the distal part of the optic nerve or in noninjured tissue.
Recombinant βB2-crystallin and NPCs producing βB2-crystallin prevent axonal degeneration and trigger axonal regeneration in the injured optic nerve in vivo.
Adult mammalian RGCs lack the ability to spontaneously regenerate axons after injury (4). Although they fail to regenerate axons under their usual inhospitable environmental conditions, RGCs may form growth cones and regenerate axons under certain experimental conditions in vivo and in vitro (7,8,17,24,32,46,47). It is well documented that LI exerts strong neuroprotective and axon growthpromoting effects on axotomized RGCs (13,16,20,45). Those effects have been related to the activation of macrophages, microglia, astrocytes, and Müller cells, which themselves mediate growth-facilitating factors (20,26,31).
Among those factors, astrocyte-derived CNTF is regarded as a key contributor mediating the beneficial effects of LI. However, growth-promoting effects of LI have also been seen in the absence of macrophages (34). Previous studies revealed the intravitreal release of crystallins mediating the growth-promoting effects after LI. An improved axonal growth has been linked with crystallins in different studies performed in vitro and in vivo (12,29,39).
In order to further elucidate how LI promotes axonal growth, the period between LI and ONC was varied in order to further elucidate how LI promotes axonal growth. LI performed 3–5 days before ONC enhanced axonal outgrowth significantly, while LI performed simultaneously with ONC failed to promote axonal growth. This suggests that RGCs require several days of exposure to lens-derived factors before they enter a robust regenerative state to enable axonal regrowth. This time span may be explained by lens-derived factors exerting their growth-promoting effect indirectly through the activation of processes that take several days. Indirect effects of LI include activation of several cell types in the retina with upregulation of activated cell-derived factors such as CNTF (12). Furthermore, any therapeutic drug must reach the lesion site before it can exert its protective effects. The release of lens-derived factors from the lens into the vitreous body and the penetration to the retina takes several days. However, the lens-derived factors that mediate enhanced axonal growth still need to be considered.
Recent data strongly suggest that crystallins mediate the growth-promoting effect of LI. Crystallin isoforms have previously been shown to enhance axonal growth in animal models in vitro and in vivo (12,29), and the present findings are in accordance with this. Intravitreal injections of purified γ-crystallin and β-crystallin fractions enhanced axonal growth significantly and independently of the presence of previous LI, supporting the hypothesis that crystallins mediate the growth-promoting effect of LI. The exact mechanism for these neuronal survival and neurite growth-promoting effects of crystallins remains to be explained. The effects of crystallins have been seen to imitate the key regulatory effects constituting the growth-promoting cascade of LI inducing both CNTF expression and macrophage activation, thereby imitating the effects of LI (12). Alternatively, crystallins may act as ligands inhibiting or downregulating apoptotic receptors such as FAS/APO-1. Crystallins are very stable proteins due to their characteristic “Greek key motif” structure, comprising four antiparallel strands, which is also believed to be responsible for their molecular chaperoning function (19,28,30). Their homology and close relationship to HSPs have led them to be regarded as stress proteins. Indeed, crystallins are regulated under both neuroregenerative and neurodegenerative conditions (29,41,42). Crystallins may act as stress proteins that switch RGCs into a robust regenerative state that acts to protect these cells against further injury. Future studies should analyze the mechanisms underlying this growth-promoting effect.
LI exerts its effects in injured tissue, but not in noninjured tissue, since there were no LI-induced effects when ONC was not also performed. This may be due to the mediating crystallins only being able to penetrate injured tissue. Moreover, the tissue must be a target of the applied drug. We assumed that the availability of lens-derived factors is greater in injured tissue or that injured tissue is more susceptible and responds better to crystallins. We therefore performed Western blot experiments to analyze the lens-derived crystallins in the injured and noninjured retina, and optic nerve experiments as described above. Our results are the first to clearly show that crystallin expression is increased in the retina and proximal part of the optic nerve after both lens injury and injection with purified rβB2-crystallin, but not in noninjured tissue, which may be explained by uptake out of the vitreous and anterograde axonal transport of γ-crystallin and β-crystallin to the lesion site. However, noninjured tissue lacked crystallin expression. Such a mechanism would require the internalization of crystallins, and investigations of uptake have yielded data supporting the transfer of βB2-crystallin into the cytoplasm (29). Neither γ-crystallin nor β-crystallin penetrate noninjured tissue, which possibly explains why LI alone (i.e., without ONC) did not enhance axonal growth, with this also not occurring in the presence of ONC beyond the lesion site of the optic nerve. The observation that crystallins are not expressed distal to the scar leads to the assumption that they cannot reach the glial scar and may explain previous observations that mature RGCs fail to regenerate injured axons beyond the site of a lesion (33). Our data are the first indicating that crystallins are transported anterogradely into injured tissue where they exert their growth-promoting effect.
Our data confirm former results suggesting that crystallins stabilize growth cone formation and are mediators of axogenesis (29). The results of our in vitro experiments also confirm that the growth cone formation and axogenesis of cut axons was comparable with previous γ-crystallin and β-crystallin in vivo injections (12).
The exact mechanism underlying the effects of NPCs in the damaged optic nerve is not fully understood. The beneficial effects may be realized by the release of specific growth factors such as CNTF or other factors known to delay the death of RGCs and photoreceptors (3,12). Besides CNTF, NTFs contain a cocktail of proteins including BDNF, which may prolong the survival of RGCs in vitro and promote the survival of injured adult neurons (21,32,33). It has been suggested that oncomodulin acts as an important signaling molecule between macrophages and neurons to promote axonal regeneration within inflammatory conditions (45). More recent studies confirmed that oncomodulin mediates inflammation and influences axonal regeneration, adding that neutrophils rather than macrophages are the cell type responsible (20,24,44,46). In addition to inflammatory molecules, we previously observed that the overexpression of βB2-crystallin by modified βB2-crystallin-NPCs influences cell survival within the retina and lasts for up to 4 weeks in vivo (3). In the present study, we extended the observations of beneficial effects in the model of ONC mediated by βB2-crystallin-overexpressing NPCs for up to 8 weeks. From the effects observed by administering rβB2-crystallin to injured axons we conclude that βB2-crystallin maintains these proactive effects.
Conclusions
In summary, we have demonstrated that crystallins—and in particular βB2-crystallin—are the main lenticular components mediating the neuroprotective and axon growth-promoting effects of LI. Furthermore, crystallins are transported to the site of the lesion in injured tissue to exert their growth-promoting effect, while exerting no effect in noninjured tissue. This leads to βB2-crystallin-producing NPCs increasing the probability of axonal growth in the injured optic nerve. These observations show that NPCs may be used as local growth-promoting “cell factories” for assuring a sustained and constant delivery of crystallins into the area of a lesion. Further NTFs may be introduced using this procedure to ensure no viral influences.
Footnotes
Acknowledgments
The authors thank M. Wissing and M. Langkamp-Flock for their skillful technical assistance. The authors thank English Science Editing (ESE) for native linguistic editing of the manuscript. Supported by a DFG grant to S.T. (Th 386/18), by IZKF grants to M.R.R.B. and S.T. (THa3/002/09), and DFG-Excellence Cluster “Cells in Motion, CiM”, area C.4. The authors declare no conflicts of interest.
