Abstract
Following central nervous system injury in mammals, failed axonal regeneration is closely related to dysneuria. Previous studies have shown that the obvious effects of apolipoprotein E (ApoE) on traumatic brain injury (TBI) were associated with an axonal mechanism. However, little information on the actions of ApoE and its isoforms on axonal regeneration following TBI was provided. In our study, the cerebral cortices of ApoE-deficient (ApoE-/-) and wild-type (ApoE
Introduction
After a mammalian central nervous system (CNS) injury, such as stroke, neurodegenerative disease and traumatic brain injury (TBI), the damage usually contributes to irreversible neurological impairment, which can involve coma, sensory disorder, paralysis, etc. Axonal regeneration failure is regarded as the main reason for persistent neurological dysfunction following a CNS injury 1 . Promoting axonal regeneration might be a potential treatment strategy for CNS injury. In contrast to the initial research on axonal regeneration, which supposed that injured axons failed to regenerate in the mammalian CNS, recent studies support that injured axons possess an intrinsic regeneration capability, which can be stimulated by certain regulating molecules 2,3 .
As a major apolipoprotein in the brain, apolipoprotein E (ApoE) mainly transports cholesterol and other lipids via ApoE membrane receptors 4 . Accumulating evidence shows an association between ApoE and TBI 5,6 . In animal experiments, ApoE considerably relieved impaired neurological function compared with conditions without it 7,8 . The three isoforms in humans, in contrast to those in animals, are ApoE2, ApoE3 and ApoE4 9 . Some clinical studies found that ApoE also significantly affected patients with TBI, and the prognosis of patients carrying the ApoE3 isoform was better than that of patients carrying the ApoE2/4 isoforms 10,11 . However, the protective mechanism of ApoE on TBI has not been fully revealed.
Accumulating studies indicate that the effect of ApoE on TBI is associated with the axons. In the peripheral nervous system (PNS), several studies have demonstrated that ApoE and its isoforms promote axonal extension of the dorsal root ganglia and axonal regeneration of sciatic and olfactory nerves 12 –14 . In the CNS, ApoE and its isoforms can also promote axonal extension of retinal ganglion cells and cortical neurons 15 –17 . However, the function of ApoE and its isoforms in axonal regeneration has not been fully elucidated. Just as axons are almost unavoidably damaged or are unable to cross an injury in TBI, here we speculate that ApoE and its isoforms can improve TBI prognosis by promoting axonal regeneration.
To simulate an environment similar to that of living neurons and better observe the axonal growth and regeneration, explants from cortical brain tissue were cultured, and an axonal transection model was established to imitate axonal injury in TBI. We attempted to explore the potential role of ApoE and its isoforms in axonal growth and regeneration.
Materials and Methods
Experimental Design and Cortical Explant Culture
All procedures were evaluated and approved by the Animal Care and Use Committee. A total of 61 neonatal mice (nApoE+/+ = 22, nApoE-/- = 39) were randomly assigned to the following experimental parts: Part 1: Model establishment: nApoE+/+ = 2; Part 2: Descriptive study: nApoE+/+ = 12, nApoE-/- = 15; Part 3: Mechanism study: nApoE+/+ = 8, nApoE-/- = 24. Usually, more than 10 cortical explants could be obtained for one brain cortex, and the best six mature explants would be adopted in our experiments. Based on this proportion, excluding the four neonatal mice used for astrocytes conditioned medium, a total 342 of cortical explants (57 pups) were used in our experiments.
A previously reported procedure of culturing cortical explants was applied to this study
18,19
, but the planting method was improved for obtaining an intact axon sample in this study. Briefly, cortical brain tissue was obtained from postnatal day 1
20
ApoE-deficient (ApoE
Axonal Transection and Sample Collection
After culture for 7 days in vitro, the distal axons in the cortical explants were transected by a scalpel under a microscope (40×; Fig. 1E). The distally transected axons were separately moved from the cortical explant into medium by a 0.22 mm diameter needle. All medium was transferred into a centrifuge tube and centrifuged (10,000 g) for 10 min at 4°C. The precipitate was taken as the RT-PCR/Western blot sample. For observing and detecting the regenerated axons, the distal axons were removed, and the rest of the cortical explant was placed into an incubator for next 48 h. Then, the regenerated axons that crossed the transecting line were collected by a second transection just as the sample that was collected for RT-PCR/Western blot.
Conditioned Medium (CM) of Astrocytes
Primary culture of astrocytes
Astrocytes were obtained from the ApoE-/- and ApoE
Preparation of the astrocyte CM
The ApoE-/- and ApoE
Incubation of explants with the astrocyte CM
After planting the explants, the astrocyte-ApoE
Human Recombinant ApoE Protein and JNK/ERK/p38 Inhibitors
Three human recombinant ApoE proteins, isoforms 2, 3 and 4 (PeproTech, Rocky Hill, NJ, USA), were separately added into Neurobasal/B27 to culture the explants of ApoE-/- mice after planting. The ApoE final concentration was 10 µg/ml in medium, which is similar to the concentration of ApoE in human cerebrospinal fluid (CSF) 22 –24 . In addition, after axonal transection, the explants of the ApoE-/- mice were continuously cultured in Neurobasal/B27 with human recombinant ApoE2/3/4 proteins for 24 h.
JNK/ERK/p38 inhibitors, which included SP600125, U0126, and SB203580 (Beyotime, Shanghai, China), were also used as interventional means. The final concentration of the three inhibitors was 10 μM, and they were dissolved in dimethyl sulfoxide (DMSO, Beyotime) 25,26 . In the control group, only DMSO was added without the inhibitors. After explant planting, the explants were separately treated by medium with the JNK, ERK and p38 inhibitors for 24 h. Subsequently, the media were replaced with fresh media without the inhibitors 27 .
Immunofluorescence
Coverslips that contained the explants were rinsed three times with phosphate-buffered saline (PBS). The coverslips were fixed with 4% paraformaldehyde at room temperature for 20 min, and 0.1% Triton X-100 was added to them for 1 h. Normal goat serum was utilized to dilute primary antibodies at the following dilutions: β-tubulin III antibody (1:1000, Sigma-Aldrich), MAP2 antibody (1:200, Boster, Wuhan, China), and ApoE antibody (1:100, Santa Cruz, Dallas, TX, USA). Coverslips were incubated with primary antibodies overnight at 4°C. FITC (green) and Alexa 555 (red) were diluted with PBS and used for secondary antibodies. Coverslips with secondary antibodies were incubated for 30 min at 37°C in a water bath. The nuclei were stained by 4’,6-diamidino-2-phenylindole (DAPI, Beyotime). The excess DAPI was washed off, and the coverslips were mounted with 50% glycerol. In addition, phalloidin (1:100, FITC labeled, SigmaAldrich) was used to dye F-actin in growth cones. A fluorescence microscope (NIKON TE2000-U, Tokyo, Japan) was used to capture all images.
RT-PCR
The samples for RT-PCR came from intact axons, from the axons that regenerated after the transection and from the residual explants. Total RNA was extracted by a MicroElute Total RNA Kit (Omega, Norcross, GA, USA) following the manufacturer’s protocol 28 . A PrimeScript RT reagent Kit (Takara, Tokyo, Japan) was used to reverse transcribe RNA to cDNA, and Premix Taq Version 2.0 (Takara) was used to amplify genes by PCR. We examined typical mRNA involved in axon regeneration, including H1f0 (GenBank ID: 14958), MAP2 (GenBank ID: 17756), GAPDH (GenBank ID: 14433) and ApoE (GenBank ID: 11816). The primer sequences (forward, reverse) and product lengths were as follows: H1f0, 5’-ggaccaccccaagtattcagac-3’, 5’-actaggcgcttgatggacaact-3’, 159 bp; MAP2, 5’-atcaggagacagggaggagaact-3’, 5’-aggggtagtaggtgtggaggtg-3’, 124 bp; GAPDH, 5’-gacatcaagaaggtggtgaagc-3’, 5’-gaaggtggaagagtgggagtt-3’, 117 bp; ApoE, 5’-aaccgcttctgggattacctg-3’, 5’-cgtcatagtgtcctccatcagtg-3’, 117 bp. The PCR conditions were 94°C for 30 s, 55°C for 30 s and 72°C for 1 min for a total of 35 cycles. The PCR products were separated in 4% agarose gels and scanned by a Gel Imager system (Bio-Rad, Hercules, CA, USA). ImageJ was used for image processing and analysis.
Western Blotting
After collection of the intact axons and the regenerated axons, Western blotting was performed as previously described 29,30 . Primary antibodies used in the western blot assay were ApoE (1:500, Santa Cruz), p-JNK (1:1000, Santa Cruz), p-ERK (1:1000, Santa Cruz), p-p38 (1:1000, Santa Cruz), and β-actin (1:2000, Santa Cruz). Then, they were incubated with the corresponding secondary antibodies at room temperature. The Western blot results were analyzed via ImageJ software.
Assessment of Axonal Growth and Regeneration
Axonal growth
In mature cortical explants, the maximum distance of axonal growth and the area of the axons were used to assess the axonal growth ability. The maximum distance of axons was determined by the maximum straight-line distance from the edge of the cortical brain tissue to the axonal terminal. The area of axons was determined by the total axonal coverage area in a cortical explant (see Fig. 4A, C).
Axonal regeneration
At 48 h after transection of the cortical explants, the maximum distance of regenerated axons was determined by the maximum straight-line distance from the transection line to the axonal terminal. The area of regenerated axons was determined by the total regenerated axonal coverage area that existed outside the transection line (see Fig. 4E, G). In every experimental group, 10 cortical explants were examined. The maximum distance and the area of axons/regenerated axons were measured via ImageJ software.
Statistical Analysis
Data are expressed as the mean±SD. The Statistical Package for the Social Sciences (SPSS, Armonk, NY, USA) software was used for data analysis. The data were evaluated by t-tests or analysis of variance (ANOVA), followed by S-N-K test for multiple comparisons. Values of p < 0.05 were considered statistically significant.
Results
Establishing the Axonal Transection Model
After the cortical explants grew to maturity, the cortical brain tissue was located in the center and contained numerous neurons, and the tissue was surrounded by radial axons (Fig. 1E). In addition, outgrowth of dendrites from neurons mixed with axons was observed. To obtain intact axon samples without dendrites or cell bodies for the subsequent experiments, the axons, dendrites and nuclei were separately stained by immunofluorescence (Fig. 1A, B, C). The merged results show the dendrites and nuclei existed only around the edge of the cortical brain tissue in mature explants (Fig. 1D). Therefore, to obtain intact axon samples, only the distal axons were transected and harvested (Fig. 1E). In addition, RT-PCR was further used to detect the purity of the axonal samples. The results showed that no dendrites or nuclei were mixed into the harvested axons (Fig. 1F). After harvesting the axons, the rest of cortical explant was placed into a cell incubator for further culture. Two days later, a limited number of regenerated axons was observed and used for the follow-up experiments (Fig. 1G).

Culturing cortical explants and establishing an axonal transection model.
ApoE mRNA and Protein Expression in Intact Axons and Regenerated Axons
Immunofluorescence staining, RT-PCR and western blotting were used to analyze the expression and localization of ApoE mRNA and protein in the ApoE

ApoE mRNA and protein expression in intact axons and regenerated axons of ApoE
ApoE Protein Expression in the Growth Cones of Intact Axons and Regenerated Axons
Growth cones exist at the axonal tip and are regarded as the leader of axonal growth and regeneration. To investigate the role of ApoE in axonal regeneration, it was essential to determine whether ApoE is expressed in the growth cone. In cortical explants, the growth cones of intact axons and regenerated axons were both stained by F-actin and ApoE. The merged results showed that ApoE was present in the growth cones of intact axons and regenerated axons (Fig. 3A, B).

ApoE protein expression in intact axonal growth cones and regenerated axonal growth cones in ApoE
Axonal Growth and Regeneration were Attenuated in ApoE-/- mice and Improved by Exogenous ApoE
As mentioned in the experimental procedures, axonal growth and regeneration were assessed by the maximum distance and area of axons (Fig. 4A, C, E, G). The maximum distance and area of the axons in the ApoE-/- group and the ApoE-/-+CMApoE-/- group were significantly decreased compared with those in the ApoE
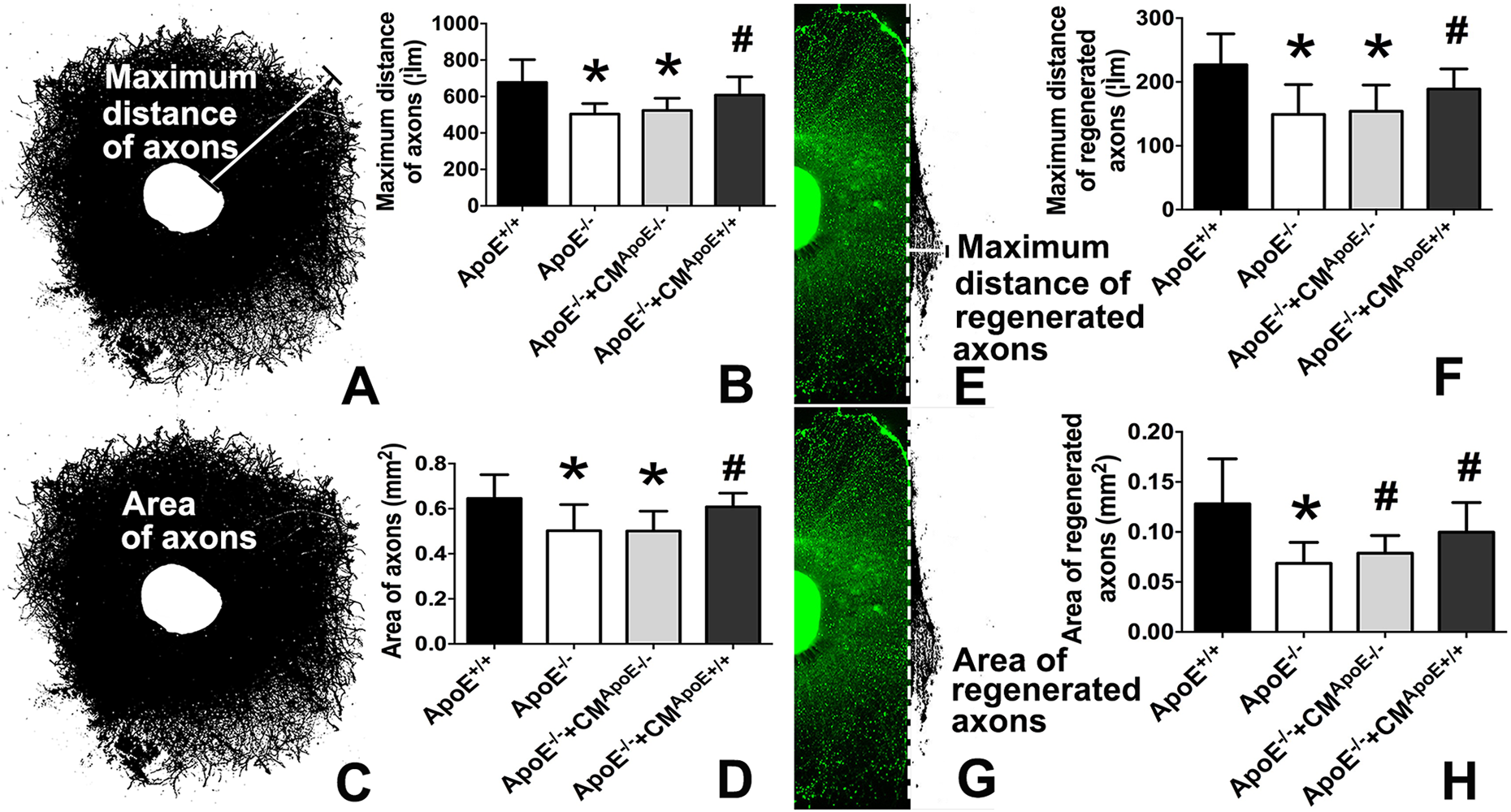
The effects of ApoE on axonal growth and regeneration. In intact axons:
ApoE Improved Axonal Growth and Regeneration via the JNK/ERK/p38 Pathway
In consideration of the ApoE effect on axonal growth and regeneration, the possible molecular mechanisms were explored. In intact axons and regenerated axons, the expression levels of p-JNK, p-ERK and p-p38 were obviously decreased in the ApoE-/- group compared with those in the ApoE

The expression levels of p-JNK, p-ERK and p-p38 were decreased in the ApoE-/- group. In intact axons and regenerated axons, the expression levels of p-JNK, p-ERK and p-p38 were significantly decreased in the ApoE-/- group compared with those in the ApoE
Considering that the axonal growth and regeneration were attenuated and the level of p-JNK/ERK/p38 was decreased in the ApoE-/- group, the JNK/ERK/p38 inhibitors were used to further explore whether ApoE influenced axonal growth and regeneration by a p-JNK/ERK/p38-mediated mechanism. The results showed that the JNK/ERK/p38 inhibitors significantly suppressed the axonal growth and regeneration (Fig. 6).
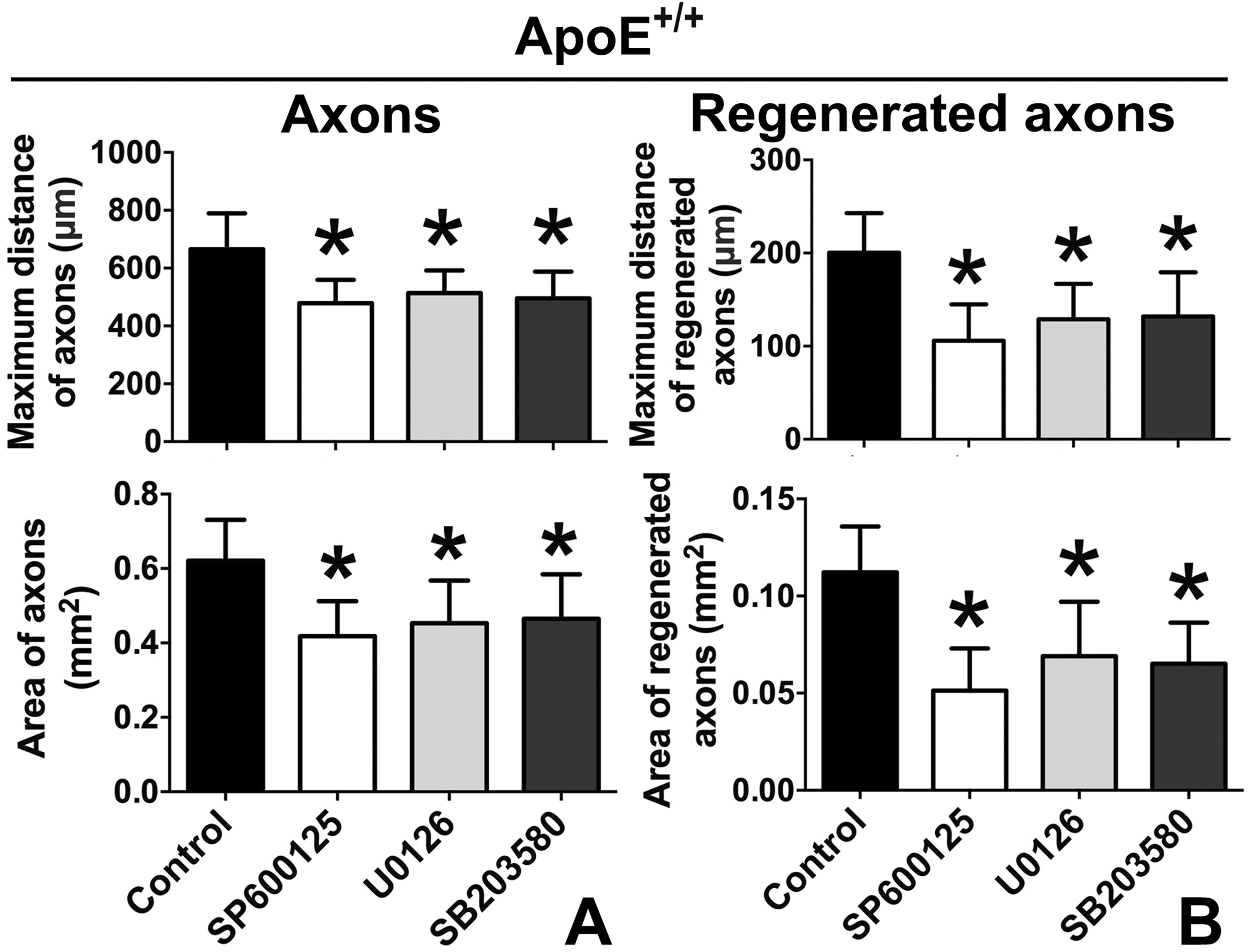
The JNK/ERK/p38 inhibitors attenuated the axonal growth and regeneration in the ApoE
Human Recombinant ApoE4 Negatively and ApoE3 Positively Influenced Axonal Growth and Regeneration
In contrast to those in mice, three isoforms of ApoE exist in humans. Explants from ApoE-/- mice were cultured, and human recombinant ApoE2, ApoE3 or ApoE4 was separately added into the culture medium to achieve the same concentration as ApoE in human CSF. The “maximum distance” and “area” were still used to quantify axonal growth and regeneration. In intact axons, the maximum distance was improved by ApoE2 and significantly improved by ApoE3, but only ApoE3 significantly improved the area (Fig. 7A). In regenerated axons, the maximum distance and area were significantly improved by ApoE3 (Fig. 7B). In contrast, ApoE4 did not improve the maximum distance or area in intact axons or in regenerated axons (Fig. 7A, B).
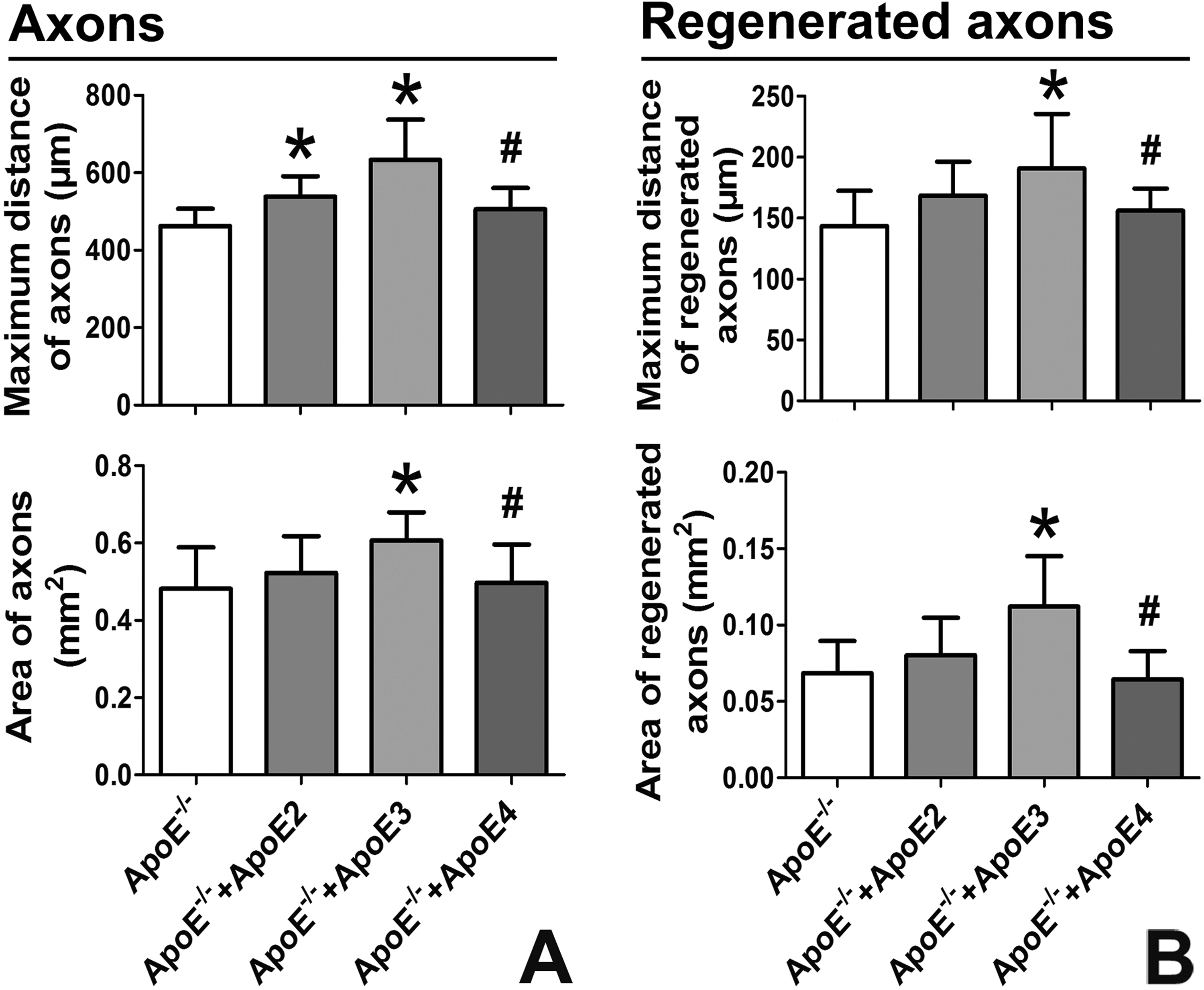
ApoE3 rather than ApoE4 improved the axonal growth and regeneration.
ApoE3 Improved Axonal Growth and Regeneration via the JNK/ERK/p38 Pathway
In view of the distinctive effects of ApoE3 and ApoE4 on axonal growth and regeneration in the cortical explants, the relationship between ApoE3/ApoE4 and the p-JNK/ERK/p38 pathway was further investigated. Compared with ApoE3, p-JNK and p-ERK were obviously reduced by ApoE4 in intact axons. However, in the regenerated axons, only p-JNK was significantly decreased by ApoE4 (Fig. 8).
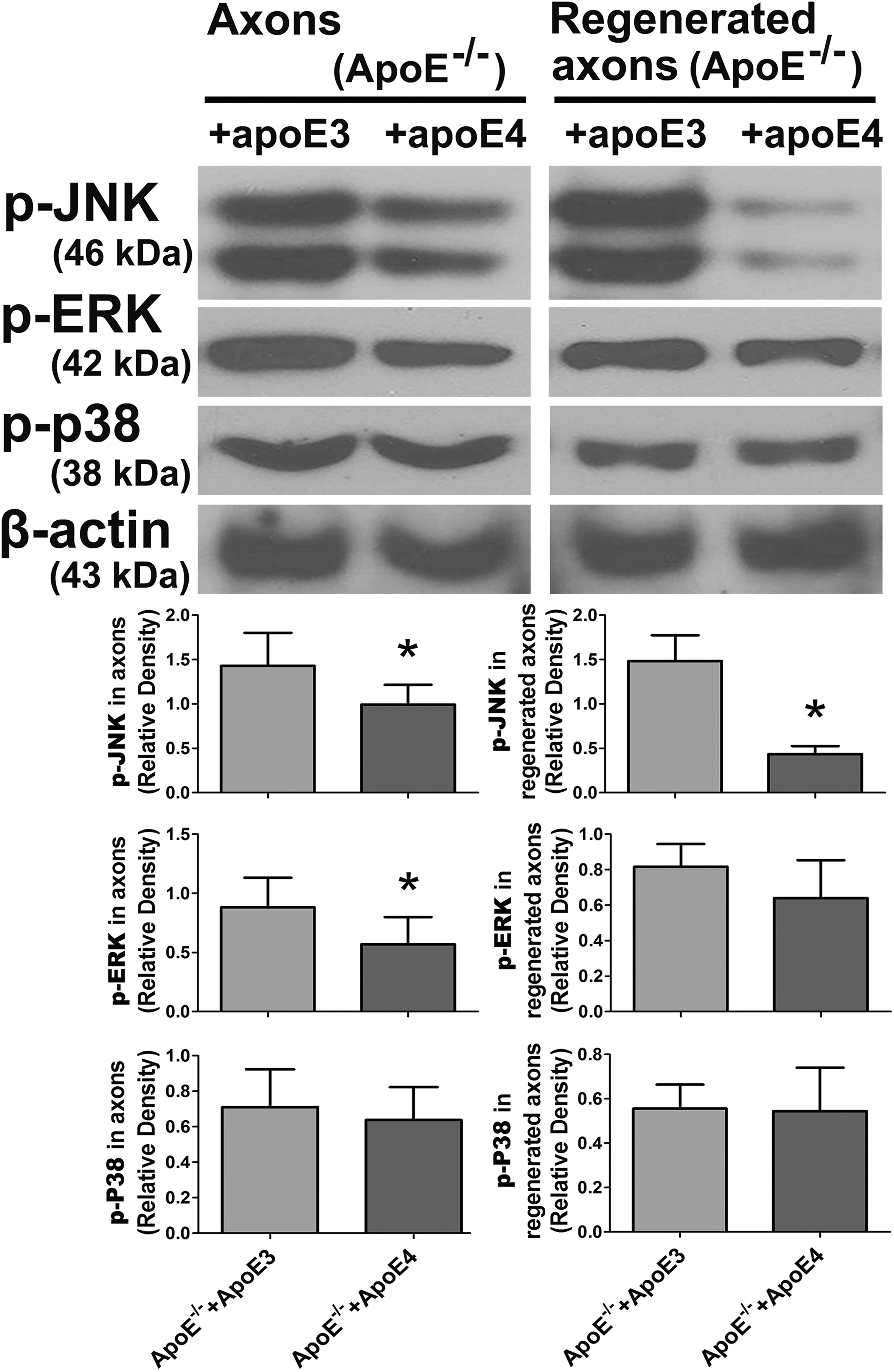
ApoE4 significantly decreased the expression of p-JNK in regenerated axons. In intact axons, the expression levels of p-JNK and p-ERK were obviously decreased in the ApoE-/-+ApoE4 group compared with the ApoE-/-+ApoE3 group. However, in the regenerated axons, only the expression of p-JNK was significantly decreased in the ApoE-/-+ApoE4 group compared with that in the ApoE-/-+ApoE3 group (*p < 0.05 vs. ApoE-/-+ApoE3).
In addition, of the three human ApoE isoforms, only ApoE4 obviously inhibited axonal growth and regeneration. The level of p-JNK/ERK/p38 was also downregulated by ApoE4, but ApoE3 showed an opposite effect, more so than that of ApoE2. Therefore, whether ApoE3 assisted axonal growth and regeneration via p-JNK/ERK/p38 was investigated by adding JNK/ERK/p38 inhibitors. The inhibitors markedly attenuated the positive effects of ApoE3 on axonal growth and regeneration (Fig. 9A, B).
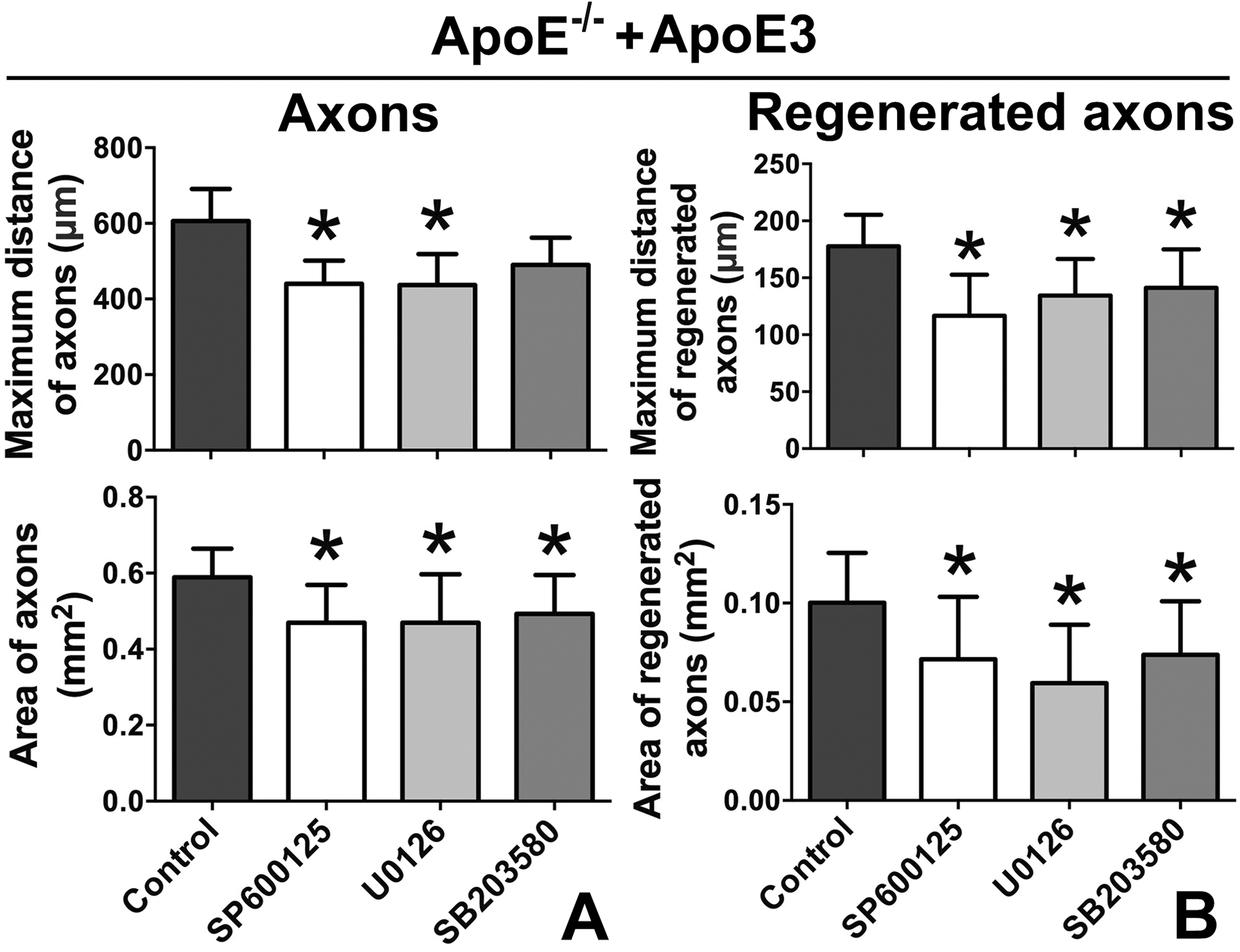
Inhibiting the JNK/ERK/p38 pathway attenuated the positive effects of ApoE3 on axonal growth and regeneration.
Discussion
In this study, we discovered that ApoE not only exists in regenerated axons, but also plays a role in promoting axonal regeneration in the mammalian CNS. In addition, human recombinant ApoE4 negatively influenced axonal regeneration and ApoE3 did so positively, and these effects were mediated by the JNK/ERK/p38 pathway.
The cortical explant culture was chosen as our experimental model based on the following considerations. First, the in vitro cortical explant culture allowed axonal growth and regeneration without obstructions, which are usually composed of glial scars in vivo 2,3 . This study examined the intrinsic capacity of axonal regeneration, and the cortical explant model allowed axons to freely grow and regenerate without barriers, which was suitable for our research goals. Second, the morphological structure of axons could be effectively exhibited in this model. As mentioned in Fig. 1, entire axons were radially distributed around the cortical brain tissue in the mature explants, making it convenient for us to observe the axonal growth and regeneration. In addition, because the axons gathered around the cortical brain tissue, the axonal transection and recovery were easy; for example, precisely transecting the axons to observe the regeneration and collecting enough axonal samples for RT-PCR and western blotting, etc., were easy using this model. Finally, the explant culture provided a similar environment to that in vivo for neurons. In primary neuron cultures, the neurons are isolated and cultured away from glial cells. However, in explant cultures, a piece of cortical brain tissue that contains neurons is cultured, so the surrounding environment of the neurons better simulates that of the brain in vivo.
Previous studies have suggested a correlation between ApoE and TBI 5,6 . One study has also shown that ApoE exists in the cell bodies and axons of mouse primary cortical neurons in vitro 31 , and the subsequent research also showed that ApoE was increased in injured neurons in vivo 21,32 . These previous results suggested that ApoE might participate in axonal growth, but whether it was involved in axonal regeneration was unknown. Our research provides supporting evidence for this possibility. After the regenerated axons grew from the site of axonal transection, ApoE was observed in the regenerated axons by immunofluorescence. To further validate the role of ApoE in axonal regeneration, the growth cone, located at the tip of axon, was also analyzed in this study. Growth cones play a vital role in guiding axonal growth and regeneration 33,34 , and there are a large number of mRNA and proteins in a tiny growth cone that actively take part in many pathological and physiological processes in the CNS 35,36 . Our results showed that ApoE exists not only in the growth cones of intact axons but also in the growth cones of regenerated axons, which indicates that ApoE might participate in axonal regeneration as well as the corresponding new growth cone formation.
Although axons are an important component of neurons, most of the proteins are synthesized in the neuronal soma then slowly transported to distal axons through the transport system within the axons. In contrast, a small amount of proteins are rapidly transcribed and translated in the axon itself 37 –39 . After axonal injury, proteins that can be timely and locally synthesized may be more essential for axonal repair or regeneration 39,40 . Therefore, whether ApoE mRNA was present in axons was further analyzed in our research. The results showed that ApoE mRNA exists in regenerated axons, which implied that injured axons could synthesize ApoE protein, and that it may even be used for axonal regeneration. However, it was completely possible that the intact axons and regenerated axons had taken up exogenous ApoE that was produced by glial cells. Indeed, considering the nature of the cortical explant culture, our study cannot differentiate these two possibilities, but our data demonstrate the importance of ApoE to axonal growth and regeneration.
Further, of the three human ApoE isoforms, ApoE3 and ApoE4 distinctively affected axonal growth. In the mouse PNS, Comley et al. found that ApoE4 obviously negatively affected peripheral nerve regeneration and neuromuscular junction re-innervation
41
. However, the specific influence of the ApoE isoforms on axonal regeneration in the CNS has not been well studied. In our axonal transection model of cortical explants, the results supported that ApoE3 significantly improved axonal growth and regeneration, but the effect of ApoE4 was opposite. The above results might partly reveal an unfavorable prognosis for TBI patients with the ApoE4 genotype with regard to injured axons. To address this phenomenon, we examined the possibly related signaling molecules. As an essential signaling pathway protein, the MAPK protein family has demonstrated involvement in axonal regeneration by a series of experiments
42
. In a study by Korwek et al., the expression of JNK and ERK was not significantly different between ApoE-deficient and wild-type mice. However, the phosphorylated ratio of JNK to ERK was significantly attenuated
43
. This result demonstrated an association between ApoE and MAPK. Therefore, in our study, p-JNK, p-ERK and p-p38, which belong to the MAPK family, were detected in the regenerated axons of ApoE-/- and ApoE
There were two obvious limitations in our study. First, the previous studies did not pay enough attention to the relationship between ApoE and axonal regeneration, so our research was mainly concentrated on descriptive studies, as the mechanistic research on ApoE was relatively insufficient. Furthermore, we focused only on the MAPK7 family, but we cannot rule out the possibility that ApoE influences axonal growth and regeneration via other signaling proteins. Second, axonal growth and regeneration are extremely complicated pathophysiological processes, not only because of the axon itself but also its surrounding environment. In our research, only the intrinsic capacity of growth and regeneration with ApoE were assessed by the axonal transection model in vitro. The effects of external factors on axonal growth and regeneration were not explored in this study. Overall, we merely discovered a partial role of ApoE, and the relationship of ApoE to axonal growth and regeneration warrants further investigation.
Conclusions
ApoE-/- and ApoE
Footnotes
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Statement of Human and Animal Rights
All procedures performed in studies involving animals (No human in this study) were approved by the Institutional Animal Care and Use Committee (IACUC) of Sichuan Provincial People’s Hospital.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by grants from the National Natural Science Foundation of China (No. 81601031, 2017).
