Abstract
Cell-based angiogenic therapies offer potential for the repair of ischemic injuries, while avoiding several of the limitations associated with material-based growth factor delivery strategies. Evidence supports that applying MSCs as spheroids rather than dispersed cells can improve retention and enhance therapeutic effect through increased secretion of angiogenic factors due to hypoxia. However, while spheroid culture appears to modulate MSC behavior, there has been little investigation of how major culture parameters that affect cellular oxygen tension, such as external oxygenation and culture size, impact the angiogenic potential of spheroids. We cultured equal numbers of adipose-derived stem cells (ASCs) as spheroids containing 10,000 (10k) or 60,000 (60k) cells each, in 20% and 2% oxygen. VEGF secretion varied among the sample groups, with 10k, 2% O2 spheroids exhibiting the highest production. Spheroid-conditioned media was applied to HUVEC monolayers, and proliferation was assessed. Spheroids of either size in 2% oxygen induced comparable proliferation compared to a 2 ng/ml VEGF control sample, while spheroids in 20% oxygen induced less proliferation. Spheroids were also applied in coculture with HUVEC monolayers, and induction of migration through a Transwell membrane was evaluated. Sixty thousand, 2% O2 spheroids induced similar levels of migration as VEGF controls, while 10k, 2% O2 spheroids induced significantly more. Ten thousand, 20% spheroids performed no better than VEGF-free controls. We conclude that the therapeutic ability of ASC spheroids to stimulate angiogenesis in endothelial cells is affected by both culture size and oxygenation parameters, suggesting that, while ASC spheroids offer potential in the treatment of injured and ischemic tissues, careful consideration of culture size in respect to in vivo local oxygen tension will be necessary for optimal results.
Introduction
There is a clinical need for therapies that drive efficient vascular growth. Such treatments offer the potential to promote healing in ischemic injuries, such as infarcts and critical-size defects, and to facilitate rapid engraftment and survival of engineered tissue implants. The process of vascularization is largely governed by the activity of specific growth factors, such as vascular endothelial growth factor (VEGF), which must be available in distinct temporal and spatial profiles in order to successfully direct formation of a mature vascular network (33). However, delivery of angiogenic factors by bolus injection or release from synthetic vehicles can lead to ineffectual outcomes and unintended side effects. Key challenges to address include 1) nonphysiological release profile(s), 2) poor localization and/or retention, 3) difficulty delivering multiple agents, 4) lack of bioresponsiveness, and 5) immunogenicity of implanted materials or material breakdown products (3,5,10,25,38,52).
A promising alternative approach is to harness the innate capabilities of cells, applying them as living systems for growth factor production and delivery. Toward this goal, human mesenchymal stem cells (MSCs) have garnered increasing interest as primary candidates for cell-based angiogenic therapies (15,32,27,28,35). While it was initially assumed that the regenerative effects of MSCs would be exerted largely through differentiation and integration, recent studies have suggested that paracrine activity is the primary mechanism by which MSCs promote tissue healing and repair (23,26,31). Proteomic analyses have revealed that the secretome of cultured MSCs includes a large number of chemokines, anti-inflammatory cytokines, angiogenic factors, and growth factors (34,40,45). In addition, MSCs have been shown to secrete membranous vesicles (exosomes) that shuttle many of these same factors as well as bioactive matrix particles and miRNAs (4,37,53). Together, these factors have the potential to form a highly angiogenic milieu in the local MSC microenvironment. Coculture of MSCs with endothelial cells can induce spontaneous capillary-like endothelial arrangement, suggesting a promising strategy for tissue engineering (51). In MSC-treated animal injury models, elevated levels of angiogenic factors are reported to be the key effector of increased vascularization and enhanced healing (20,21,24,39). A recent study demonstrating that MSC-conditioned medium alone can be sufficient to enhance capillary formation and healing in the infarcted porcine heart further highlights the centrality of paracrine signaling in MSC-based therapies (48).
Despite their enormous potential in laboratory studies, inconsistent or mixed results have prevented MSC therapies from making the clinical transition. A major reason for this is low retention and survival of MSCs in single cell transplantations due to apoptosis or washout (2,14,19,36,46,49). To improve their performance, 3D culture of MSC spheroids has been investigated. Spheroid cultures maintain cell–cell and cell–matrix interactions, promoting cell retention and survival (22). Transplanted spheroids also retain matrix-associated growth factors, providing immediate protective effects to the MSCs as well as paracrine signals to surrounding endogenous cells (7). Furthermore, spheroid culture is reported to contribute to elevated secretion of many of the angiogenic factors produced by MSCs (1,6,9,11,12,57). This is commonly attributed to activation of hypoxia-inducible factor (HIF)-1α and HIF-2α due to increased hypoxic stress within 3D cultures (8,50,54). However, while hypoxic stimulation of angiogenic behaviors is desirable, hypoxia that is too sudden or severe can alternatively lead to loss of cell function and cell death (13,56). Thus, care must be taken to ensure that an optimal level of local oxygen concentration is maintained in 3D MSC cultures in order for beneficial behaviors to manifest.
Of the parameters that affect cellular oxygen tension in a 3D culture, culture size and local external oxygen concentration are the largest contributors. However, to date there has been little evaluation of how changes in these conditions affect the proangiogenic properties of MSC spheroids. We have previously shown that differences in culture diameter and external oxygen concentration result in variations in HIF activity and VEGF secretion in adipose-derived stem cell (ASCs) spheroids (43). The current work was undertaken to investigate how these results translated to angiogenic stimulation in endothelial cells, specifically addressing two key questions: 1) Do ASC spheroids function as well as cell-free administration of VEGF in stimulating angiogenic behaviors in endothelial cells? and 2) How do variations in ASC spheroid size and external culture oxygenation beneficially or detrimentally affect their angiogenic potential in terms of endothelial cell behavior? Answers to these questions would provide insights into approaches for optimizing culture geometry in respect to in vivo local oxygen tension when applying MSCs for the treatment of injured or ischemic tissues.
Materials and Methods
ASC and Human Umbilical Vein Endothelial Cell (HUVEC) Culture
Passage 1 (P1) ASCs and low-serum medium (MesenPRO RS™) were purchased from a supplier (Invitrogen, Carlsbad, CA, USA). Cells were cultured as monolayers in 75-cm2 vented tissue culture flasks (BD Biosciences, Franklin Lakes, NJ, USA) at 37°C, 5% CO2, and 95% air in a well-humidified environment (hereafter referred to as, 20% oxygen conditions). Upon reaching 80-90% confluence, the cells were trypsinized (Corning Life Sciences, Manassas, VA, USA) and counted by hemacytometer (Hausser Scientific, Horsham, PA, USA) then used for study or reseeded for continued expansion. In order to ensure consistent cell behavior, cells between P2 and P5 were used in all studies, according to the supplier's recommendations.
P1 HUVECs (Millipore, Billerica, MA, USA) were cultured as monolayers in a high-growth expansion medium containing VEGF (EndoGRO™; EMD Millipore, Billerica, MA, USA) in 20% oxygen. At 80-90% confluence, the cells were trypsinized (EMD Millipore) and counted, then used for study or reseeded for culture expansion. In certain experiments, a low-serum, VEGF-free medium (hereafter, LS medium; EMD Millipore) was used as a negative control, and LS medium supplemented with 2 ng/ml human recombinant VEGF (VEGF165, Invitrogen; hereafter, VEGF medium) was used as a positive control. To ensure consistent cell behavior, HUVECs between P2 and P5 were used for study.
ASC Spheroid Formation
ASC spheroids were formed in a manner previously detailed (43). Cells in a freshly detached dispersion were counted by hemacytometer, then resuspended in MesenPRO RS™ medium at a density of 500,000 cells/ml. Medium volumes containing 10,000 (10k) or 60,000 (60k) cells were added to 0.8 ml, siliconized, screw-cap microcentrifuge tubes (VWR, Radnor, PA, USA), and the tubes were spun at 450 x g for 2 min. The screw caps were then loosened slightly to allow for gas transfer, and the tubes were placed in an incubator at 20% oxygen overnight. Spontaneous self-assembly of 3D, spherical aggregates (spheroids) occurred after 18-24 h. The spheroids were then carefully collected using a micropipettor (Eppendorf, Hamburg, Germany) with a wide-mouth tip to prevent shearing and used for study.
PEG Encapsulation of ASC Spheroids
For secretion studies, ASC spheroids of the same size were pooled. In order to prevent substrate attachment or spheroid fusion, the spheroids were immobilized in a poly(ethylene glycol) (PEG) hydrogel matrix, as previously described (43). PEG (10 kDa; Sigma-Aldrich, St. Louis, MO, USA) was functionalized to PEG-dimethacrylate (PEGDM) with methacrylic anhydride (Sigma-Aldrich) by microwave methacrylation and then purified. A 10% (wt) photo-crosslinking solution of PEGDM was prepared by dissolving PEGDM in Hank's balanced salt solution (HBSS; Corning Life Sciences) and adding Irgacure 2959 (Ciba, Basel, Switzerland) to 0.025% (wt) (42). This solution was sterilized by passage through a 0.22-μm filter (VWR) prior to use. Spheroids were encapsulated in a two-step procedure. First, 18 μl of photoactive PEGDM solution was added on top of the plunger of a vertical, decapitated 1-ml syringe, and cross-linking was initiated by 8 min of exposure under a UV lamp (365 nm, ~7 mW/ cm2; UVP, Upland, CA, USA). Then pooled spheroids were placed on top of this layer, and a second 18-μl volume of photoactive PEGDM solution was added. Hydrogel cross-linking was completed by another 8-min UV exposure, ensuring full encapsulation of spheroids.
Growth Factor Secretion From ASCs
Following spheroid encapsulation, the hydrogels were rinsed once in HBSS and each placed in 1 ml of MesenPRO RS™ medium in a 24-well plate (GBO, Monroe, NC, USA). Samples were prepared such that each hydrogel contained an equivalent number of cells. Hydrogels contained either four 60k spheroids or 24 10k spheroids. All hydrogels were placed in 20% oxygen for 24 h. After addition of fresh media, a group of 10k and a group of 60k spheroids were each placed in an incubator at 20% oxygen, while a second group of each size was placed in an incubator held at 2% oxygen (Napco series 8000 programmable incubator; Thermo Fisher, Waltham, MA, USA). Four samples were prepared for each group. Every 24 h, the medium from each sample was collected and frozen, and fresh medium was added. For samples cultured in 2% O2, media were preequilibrated to 2% O2 prior to use, with an overnight hypoxic incubation period being sufficient (41). Secretion of VEGF from HUVEC monolayers cultured in 20% or 2% oxygen was also evaluated.
Growth factor concentrations in media samples were analyzed by ELISAs for the respective factor. ELISAs for VEGF, transforming growth factor-β (TGF-β1), hepatocyte growth factor (HGF), and epidermal growth factor (EGF) were purchased from Invitrogen. ELISAs for fibro-blast growth factor-2 (FGF-2), platelet-derived growth factor (PDGF-BB), and stromal cell-derived factor-1α (SDF-1α) were purchased from Abcam (Cambridge, MA, USA). ELISAs for bone morphogenetic protein-2 (BMP-2) and angiopoietin-2 (Ang-2) were purchased from R&D Systems (Minneapolis, MN, USA). Assays were performed in accordance with the manufactures' recommendations. The ELISAs used are listed in Table 1 along with the lower detectable protein concentration of each assay.
List of ELISAs Used

HUVEC Proliferation Assay
The effects of ASC-secreted factors on endothelial cell proliferation were evaluated by a conditioned media swap study. Hydrogels containing 10k or 60k ASC spheroids were prepared in the same manner as described, and groups were placed in 20% or 2% oxygen in LS medium. Twenty hours after spheroids were assigned to different oxygen conditions, HUVECs were detached from a flask, counted, and then added into the wells of six-well plates (GBO) at 50,000 cells per well. EndoGRO™ medium was added to a total volume of 1.5 ml, and the cells were incubated at 20% oxygen conditions for 4 h to allow for attachment. After this time, the HUVECs were rinsed once with HBSS, then 500 μl of LS medium was added to each well. Each well then received 1 ml of ASC-conditioned medium transferred from one of the four ASC spheroid groups (10k or 60k at 20% or 2% oxygen). Control wells received 1 ml of LS medium or 1 ml of VEGF medium. The concentration of VEGF in positive control medium (2 ng/ml) was selected based on the highest expected average VEGF secretion from ASC spheroids as determined by ELISA results. ASC hydrogels were given 1 ml of fresh LS medium and returned to their respective incubator. Preequilibrated LS media was used for ASCs in 2% oxygen. HUVEC monolayers were returned to incubation at 20% oxygen. Media swaps were performed in this manner every 24 h. Four replicate samples were prepared for each group.
After 24 h, HUVEC proliferation was quantified via crystal violet cell staining assay. Monolayers were rinsed once with HBSS and then 400 μl of a 0.1% crystal violet (Alfa Aesar, Ward Hill, MA, USA) solution was added to each well. After 10 min, all wells were thoroughly rinsed with deionized water and decanted. Stain was released by addition of 300 μl of 10% acetic acid (Ricca Chemical Company, Arlington, TX, USA) and orbital shaking for 10 min. A 100-μl sample was collected from each well, and the optical density for each sample was determined at 560 nm using a plate reader (Synergy 2; Biotek, Winooski, VT, USA). A schematic of the workflow for proliferation studies is shown in Figure 1.

Work flow of HUVEC proliferation assay.
HUVEC Migration Assay
The effects of ASC-secreted factors on endothelial cell migration were evaluated by a Transwell migration study. ASC spheroids of 10k and 60k sizes were prepared as described, but not encapsulated. After overnight self-assembly, the spheroids were collected and pooled into 24-well plates in 500 μl of LS medium and placed in either 2% or 20% oxygen conditions. Wells contained three 60k spheroids or 18 10k spheroids. After 6 h, HUVECs were detached from a flask, counted, and resuspended in LS medium at a density of 1 × 106 cells/ml. Transwell membrane inserts (Cell Biolabs, San Diego, CA) were then placed into the wells of the 24-well plates above the ASC spheroids. As controls, inserts were also added to wells containing only 500 μl LS medium and wells containing only 500 μl VEGF medium. Two hundred fifty microliters (2.5 × 105 cells) of dispersed HUVECs was added to the upper chamber of the inserts according to the manufacturer's instructions, and the plates were returned to 2% or 20% oxygen conditions for 18 h. Determination of cell migration was performed following the assay manufacturer's instructions. Four replicate samples were performed per group. A schematic of the workflow for migration studies is shown in Figure 2.

Work flow of HUVEC Transwell migration assay.
Data Analysis
Growth factor secretion results are reported as average nanograms of protein per milliliter of sample ± standard deviation. HUVEC proliferation and migration results have been normalized to the negative control group (LS medium alone). Results are reported as the average percent difference between the sample and negative control ± percent error of the sample mean. Statistical significance of differences between experimental groups was determined by Student's t-test assuming unequal variance. False discovery rate (FDR) corrections were employed to account for multiple comparisons with the FDR set at 0.05 for these calculations. Significance of differences between sample means was determined by comparing p values to modified critical values determined by FDR. Statistically significant differences among samples are indicated in the results.
Results
Encapsulated ASCs
During overnight aggregation, pelleted ASCs spontaneously assembled into spheroids. Figure 3 shows representative images of ASC spheroids at the time of encapsulation. Spheroids appeared spherical or slightly oblong in shape. Morphologically, the spheroid surfaces were smooth, with the boundary between cells being difficult to distinguish. Spheroid size was dependent upon the number of cells seeded, with spheroid sizes within each group being highly consistent. Sixty thousand and 10k spheroids exhibited average diameters of 877 ± 20 μm and 462 ± 15 μm, respectively. All spheroids were fully encapsulated within the PEG hydrogels, which were approximately 6 mm in diameter.
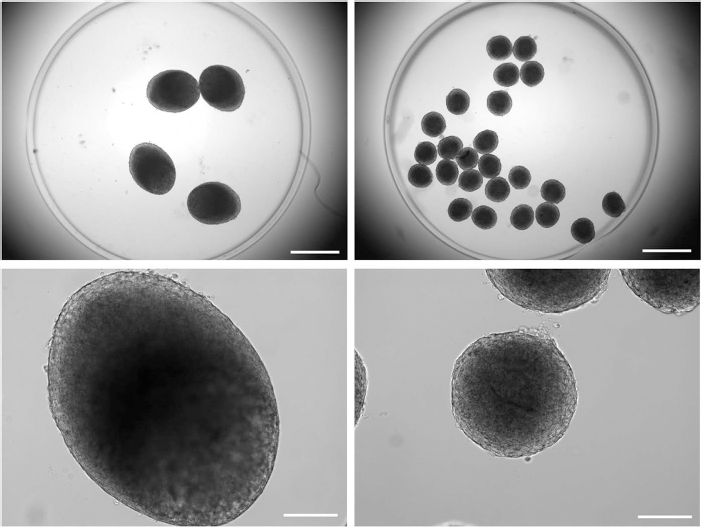
The 60k (left) and 10k (right) spheroids immediately following encapsulation. Magnification: (top) 20x, scale bar: 1 mm; (bottom) 100x, scale bar: 200 μm.
Growth Factor Secretion From ASC Spheroids
Encapsulated ASC spheroids were incubated in 20% or 2% oxygen, and daily media samples were assayed to quantify secretion of a number of growth factors. No detectable secretion of Ang-2, BMP-2, EGF, FGF-2, HGF, PDGF-BB, SDF-1α, or TGF-β1 was observed in any sample. VEGF secretion from ASC spheroids was detected and is shown in Figure 4. In spheroids of both sizes, significantly more VEGF was secreted in 2% oxygen conditions than in 20% oxygen. In 2% oxygen, secretion increased from day 1 to a peak at day 2 or 3 and then gradually declined through the remaining days of the study. For 60k spheroids in 2% oxygen, daily VEGF production peaked at 2.37 ± 0.17 ng on day 2, which was 1.69-fold higher than secretion from spheroids of the same size in 20% oxygen. For 10k spheroids in 2% oxygen, these differences were more pronounced, with a peak of 2.76 ± 0.39 ng secreted on day 3, which was 2.28-fold higher than secretion in 20% oxygen. Calculation of cumulative, 5-day secretion confirmed that significantly more total VEGF was produced by spheroids of either size in 2% oxygen than in 20% oxygen. Cumulative secretion from 10k spheroids in 2% oxygen was 9.52 ± 0.86 ng VEGF, which was significantly higher than the cumulative 8.35 ± 0.37 ng VEGF secreted from 60k spheroids in 2% oxygen. Cumulative VEGF secretion from 10k and 60k spheroids in 20% oxygen did not significantly differ.

VEGF secretion from 60k and 10k spheroids in 20% and 2% O2 (top), and total VEGF secretion from spheroids of over 5 days (bottom). *Significant difference between oxygen concentrations. †Significant difference between the spheroid sizes.
ASC-Conditioned Media and HUVEC Proliferation
Monolayers of HUVECs were grown in medium that had been conditioned with 10k or 60k ASC spheroids in 20% or 2% oxygen, or in LS medium as a negative control or VEGF medium as a positive control. After 3 days, proliferation of each culture was assessed. HUVEC proliferation results for each test group are quantified in Figure 5. VEGF media induced significantly more proliferation than LS media. Compared to LS media, media conditioned with 10k or 60k ASC spheroids in 20% oxygen or 60k spheroids in 2% oxygen exhibited no significant difference in effect on proliferation. Media conditioned with 10k spheroids in 2% oxygen resulted in 14.6 ± 4.6% more HUVEC proliferation, respectively, compared to LS medium. Media conditioned with 60k or 10k spheroids in 2% oxygen did not perform significantly different than VEGF media. Media conditioned with 10k spheroids in 20% oxygen induced significantly less HUVEC proliferation than VEGF media.

HUVEC proliferation in conditioned and control media. *Significance compared to LS. †Significance between oxygen conditions.
HUVEC Migration in Transwell Coculture with ASC Spheroids
HUVEC monolayers were grown on the apical surface of porous, Transwell inserts through which the cells were capable of migrating basolaterally in response to chemotaxic stimuli. Figure 6A shows the relative migratory responses of HUVECs in LS media or VEGF media (4.5 ng/ml) that had been added to the apical, basolateral, or both chambers. The selected concentration of VEGF was comparable to that found in common endothelial growth media formulations and ensured that a clear chemotaxic effect would be observed. Compared to the condition in which both chambers received only LS medium, addition of VEGF to the apical chamber did not result in significantly more basolateral migration of the HUVECs. Addition of VEGF to both chambers resulted in a slight (1.5-fold), though significant, increase in HUVEC migration, which may have represented a general (nondirected) increase in HUVEC motility. Addition of VEGF to the basolateral chamber alone resulted in a noticeable, 3.1-fold increase in HUVEC migration to the basolateral membrane surface. Because autologous VEGF production by HUVECs could also affect their migration, HUVECs were cultured in 20% and 2% oxygen for 5 days, and media samples were assayed for VEGF. Neither condition resulted in detectable VEGF production.

(A) Migration data of HUVECs with 4.5 ng/ml VEGF added to the apical (A), basolateral (B), or both chambers. *Significance compared to A:LS/B:LS. (B) Migration data of HUVECs with ASC-conditioned, LS, and VEGF control media (2 ng/ml). *Significance compared with LS. †Significance between oxygen conditions. ‡Significance compared to VEGF. (C) Representative images of stained, migratory HUVECs on the basolateral surface of the different treatment chambers. Cells were stained with crystal violet, which was then released in solution and colorimetrically quantified.
Coculture chambers were also prepared in which HUVEC monolayers were grown on the apical insert surface, and 10k or 60k ASC spheroids were placed in the basolateral chamber. Preparations with ASCs were then cultured in 20% or 2% oxygen, and HUVEC migration was assessed. These results are quantified in Figure 6B, and representative images of the stained migratory cells for each condition are shown in Figure 6C. Compared to preparations in which the basolateral chamber received only LS medium, treatment of the basolateral chamber with VEGF medium, 10k or 60k ASC spheroids in 2% oxygen, and 60k spheroids in 20% oxygen resulted in significantly more HUVEC migration. The fold increases were 1.5, 2.4, 1.8, and 1.5, respectively. Addition of 10k spheroids in 20% oxygen did not significantly alter HUVEC migration compared to the LS treatment. The amount of migration observed in HUVECs treated with 60k spheroids in 2% oxygen did not significantly differ from that observed in VEGF control samples. However, 10k spheroids in 2% oxygen performed significantly better than the VEGF control, inducing 38.3% more HUVEC migration. Levels of migration induced by 10k spheroids in 2% oxygen were also significantly higher than levels of migration induced by 60k spheroids in 2% oxygen.
Discussion and Conclusions
To improve their clinical efficacy, a better understanding of how culture parameters that affect cellular oxygen tension impact the angiogenic potential of MSC spheroids is needed. We have previously shown that variations in external oxygen concentration and spheroid size can cause differences in HIF-1 activity and VEGF secretion in ASC spheroids (43). The present study investigated the resultant angiogenic behaviors of endothelial cells exposed to ASC spheroids or spheroid-conditioned media when spheroid size and culture oxygenation were varied. To our knowledge, this is the first study to evaluate how size and oxygen parameters combine to impact the effectiveness of ASC spheroids in driving prevascular behaviors in endothelial cells. Our studies demonstrate that for an equivalent number of cells, variations in spheroid geometry and culture oxygenation can result in differences in the ability of ASC spheroids to promote angiogenic behaviors in endothelial cells. While ASCs had the potential to surpass comparable cell-free, growth factor administration, they could also perform more poorly, depending upon the culture parameters. These findings illustrate that the effectiveness of 3D ASC therapies in promoting vascularization will be heavily dependent upon choosing appropriate culture geometry to fit the oxygen status of the target implant site.
In agreement with numerous previous studies, ASCs cultured in low external oxygen conditions exhibited an increase in VEGF secretion compared to those cultured in standard oxygen conditions (8,17,43,47). This supports the suggestion that hypoxic preconditioning can prime ASCs to more immediately drive vascularization upon implantation (16,17,44). In addition, as previously observed, a size-dependent difference in secretion levels was seen, with 10k spheroids producing more VEGF than 60k spheroids. Differences in secretion levels from ASC spheroids due to spheroid size have not been widely investigated, though size-dependent differences in degree and homogeneity of chondrogenic differentiation of MSC spheroids have been reported (29). Necrosis of cells within the center of large spheroids has also been reported, suggesting an explanation for the superior per-cell production from 10k spheroids (43,55). Regional differences in cell behavior, including potential phenotypic commitment and corresponding changes in gene expression, could help explain the lack of detectable production of other angiogenic factors that have been commonly reported, such as EGF, PDGF, FGF-2, and HGF.
In the body, angiogenesis proceeds when endothelial cells in the wall of an existing vessel are stimulated to digest the basement membrane, proliferate, and migrate toward the source of local, angiogenic, and paracrine factors. The most potent stimulating factor is VEGF, while PDGF, FGF-2, and HGF are also known to induce early angiogenic behaviors. Therefore, increased angiogenic factor production from ASCs should be expected to correlate with improved angiogenic induction of endothelial cells. However, using cells to condition media or in coculture can lead to nutrient depletion and competition among cell types, which could detrimentally affect endothelial cell function. Aware of these competing effects, and knowing that VEGF production from ASC spheroids can vary depending upon culture conditions, we evaluated HUVEC proliferation and directed migration in response to various ASC spheroid cultures to determine how effective spheroids were in promoting angiogenesis. For comparison, we also applied VEGF-free medium and a positive VEGF control medium selected to simulate a simple delivery device that maintains a constant VEGF concentration. The concentration of the VEGF control was intended to closely match the average concentration secreted by the ASC spheroids, which maximally produced VEGF (10k, 2% O2). For all other study groups, VEGF control medium would be expected to contain more VEGF than the study sample. In addition, in coculture studies, VEGF from the control medium would be instantaneously available to the cells at the expected concentration, while VEGF provided by ASCs would gradually ramp up to the expected concentration as secretion accumulated. Figure 7 shows the estimated VEGF concentration in the media of the various sample groups.

Concentrations of VEGF secreted from 10k (A) and 60k (B) spheroids in 20% and 2% oxygen over 3 days with the concentration of VEGF in positive control medium (dashed line) indicated for comparison. (C) Estimated concentrations of VEGF in media swaps added to HUVEC cultures for proliferation control and sample groups. (D) Estimated initial and final concentrations of VEGF in coculture media for migration control and sample groups.
We found that both 10k and 60k ASC spheroids cultured in 2% oxygen induced HUVEC proliferation equally as well as VEGF control medium, while spheroids in 20% induced significantly less proliferation, though still more than VEGF-free medium. Additionally, 10k spheroids induced greater proliferation than 60k spheroids, confirming that spheroid size affects angiogenic potential. Furthermore, this outcome highlights that simply increasing spheroid size to increase HIF activation does not guarantee improved therapeutic potential. These results are consistent with the VEGF concentrations estimated to be in each sample. While the VEGF levels in 2% oxygen-conditioned media samples and the VEGF control were comparable, the ASC-conditioned media should be more nutrient depleted due to ASC metabolic requirements.
In HUVEC migration studies, 60k ASC spheroids in 2% oxygen promoted migration equally as well as VEGF control medium, while 10k, 2% oxygen spheroids promoted superior migration. The 60k and 10k spheroids in 20% oxygen did not induce migration any better than VEGF-free medium. This could suggest that the low levels of VEGF secretion were not sufficient to counteract nutrient competition exerted by the ASCs. Competition for nutrients in cocultures has been reported in other cell systems, with the potential to reduce expected cell behaviors (18,30). While 10k, 2% oxygen samples did achieve a higher final level of VEGF, this concentration represents cumulative secretion over 24 h and was not instantaneously available, as in VEGF control samples. Therefore, the median VEGF concentration in the conditioned media samples may represent a better direct comparison to the control. In this regard, 10k, 2% oxygen spheroids appeared to outperform the positive VEGF control. This suggests that factors in addition to VEGF may have contributed to promoting HUVEC migration. These factors were not identified as any of the additional cytokines tested for in this study, though they could represent such growth factors contained in exosomes, which may not have been detectable by ELISA (4,53). This potential for superior performance, even if incompletely understood, emphasizes the potential of cell-based therapies and underscores the need for more comprehensive investigation of the environmental stimuli and intrinsic pathways that regulate their behavior.
Taken together, our results address the two key questions initially posed. 1) Under particular, favorable culture conditions, ASC spheroids can better stimulate angiogenic behaviors in endothelial cells than cell-free growth factor administration. However, in alternate, though not abnormal, arrangements or culture conditions, the same number of ASCs can induce a much weaker angiogenic response. 2) Low external oxygen tension is important to increase the angiogenic potential of ASC spheroids, though larger spheroids (which would be expected to experience more severe hypoxia) are not overall as effective as smaller spheroids in inducing preangiogenic endothelial behaviors. These results highlight a challenge of cell-based systems: that they behave differently given their surrounding environmental conditions. There is therefore a need to better understand how cells respond to different in vivo microenvironments in order to predict their behavior and maximize their functionality. We conclude that no single ASC spheroid size will fit all applications, and strict consideration of an optimal tissue geometry based on the local oxygenation will produce optimal therapeutic outcomes.
Footnotes
Acknowledgments
We offer thanks to the laboratory of Dr. Melissa Moss of the University of South Carolina, Columbia, Chemical Engineering Department, for shared use of research space and instrumentation. This research was funded through a grant from the National Institutes of Health (R03 AR062816). The authors declare no conflicts of interest.
