Abstract
Dental pulp stem cell (DPSC) subsets mobilized by granulocyte-colony-stimulating factor (G-CSF) are safe and efficacious for complete pulp regeneration. The supply of autologous pulp tissue, however, is very limited in the aged. Therefore, alternative sources of mesenchymal stem/progenitor cells (MSCs) are needed for the cell therapy. In this study, DPSCs, bone marrow (BM), and adipose tissue (AD)-derived stem cells of the same individual dog were isolated using G-CSF-induced mobilization (MDPSCs, MBMSCs, and MADSCs). The positive rates of CXCR4 and G-CSFR in MDPSCs were similar to MADSCs and were significantly higher than those in MBMSCs. Trophic effects of MDPSCs on angiogenesis, neurite extension, migration, and anti-apoptosis were higher than those of MBMSCs and MADSCs. Pulp-like loose connective tissues were regenerated in all three MSC transplantations. Significantly higher volume of regenerated pulp and higher density of vascularization and innervation were observed in response to MDPSCs compared to MBMSC and MADSC transplantation. Collagenous matrix containing dentin sialophosphoprotein (DSPP)-positive odontoblast-like cells was the highest in MBMSCs and significantly higher in MADSCs compared to MDPSCs. MBMSCs and MADSCs, therefore, have potential for pulp regeneration, although the volume of regenerated pulp tissue, angiogenesis, and reinnervation, were less. Thus, in conclusion, an alternative cell source for dental pulp/dentin regeneration are stem cells from BM and AD tissue.
Keywords
Introduction
Dental pulp tissue has a critical role in homeostasis of teeth. Maintenance of the function of pulp tissue is critical for longevity of teeth and improved quality of life. Thus, an unmet need exists for novel clinical applications of pulp regenerative therapy for endodontic treatment for functional restoration of dental pulp.
Recently, we have demonstrated that autologous transplantation of pulp cluster of differentiation 31 negative (CD31-) side population (SP) cells or CD105+ cells with stromal cell-derived factor (SDF)-1 induce complete pulp regeneration in a root canal after pulpectomy in dogs (6,22). It was revealed that subfractions of dental pulp stem cells (DPSCs), such as CD31- SP cells and CD105+ cells, have a greater effect on angiogenesis/ vasculogenesis and neurogenesis after transplantation both in mouse hindlimb ischemia and rat brain ischemia compared with colony-derived DPSCs (10,11,23,34). The efficacy of autologous transplantation of CD31- SP cells and CD105+ cells into canine pulpectomized root canal is also much higher than colony-derived DPSC transplantation. In addition, transplantation of these DPSC sub-fractions with SDF-1 resulted in much higher volume of regenerated pulp tissue compared with transplantation of DPSC subfractions only, due to homing of CXCR4+ stem cells by the SDF-1–CXCR4 axis during pulp regeneration (6,13,22).
Since safety of these CD31- SP cells and CD105+ cells isolated by flow cytometry has not been established and SDF-1 was not approved for clinical use, we recently devised a novel method involving the use of granulocytecolony-stimulating factor (G-CSF)-induced mobilization for isolation of DPSC subsets (24). DPSCs mobilized by G-CSF (termed MDPSCs) have a similar phenotype to the CD105+ cells with high angiogenic/ vasculogenic, neurogenic, and regenerative potential in a mouse hindlimb ischemia model and subcutaneous ectopic tooth transplantation model (20). In addition, an alternative to SDF-1 for pulp regeneration, G-CSF, which has a migratory effect on stem cells (30), was selected to establish pulp regeneration therapy into a clinical application (7). G-CSF has an effect on migration, antiapoptosis, proliferation, and immunosuppression as demonstrated in vitro and in vivo, and the combinatorial therapy of G-CSF and MDPSCs resulted in higher regenerative potential compared to G-CSF only or MDPSCs only after transplantation in a canine pulpectomized model (7).
However, supply of pulp tissue is very limited with age, and alternative sources of mesenchymal stem cell/ progenitor cells (MSCs) need to be identified for clinical application of cell therapy for pulp regeneration. Complete pulp regeneration has already been demonstrated by transplantation of CD31- SP cells from bone marrow and adipose tissue with SDF-1 in pulpectomized teeth, and its quality was similar to dental pulp CD31- SP cell transplantation (12).
Thus, in this investigation, canine subsets of DPSCs, bone marrow-derived stem cells (BMSCs), and adipose-derived stem cells (ADSCs) in the dog were isolated by G-CSF-induced mobilization. The stem cell properties including trophic effects and the regenerative potential in the pulpectomized teeth were compared systematically.
Materials and Methods
This study was approved by the ethics committee and the Animal Care and Use Committee of Aichi-gakuin University and the National Center for Geriatrics and Gerontology, Research Institute. All experiments were conducted using the strict guidelines of DNA Safety Programs.
Cell Culture and Cell Isolation
Dental pulp cells were separated from pulp tissues of 8- to 10-month-old female beagle dogs (n = 5) (Kitayama Labes, Ina, Japan) that had been extracted from upper canine teeth as described previously (7). Primary adipose cells were separated from the abdominal subcutaneous adipose tissue, and bone marrow cells were collected by bone marrow puncture from the sternum of the same dog, respectively.
Freshly extracted, upper canine teeth after making a longitudinal cut, bone marrow, and adipose tissues were soaked in Hank's balanced salt solution (Life Technologies, Carlsbad, CA, USA). Dental pulp tissues separated from the teeth were minced into pieces and enzymatically digested in 0.04 mg/ml Liberase (Roche Diagnostics, Rotkreuz, Switzerland) for 30 min at 37°C. Adipose tissues were also minced into pieces and enzymatically digested in 0.04 mg/ml Liberase for 30 min at 37°C. Red blood cells of bone marrow were lysed with IOTest3 lysing solution (Beckman Coulter, Fullerton, CA, USA). The isolated pulp cells were plated at 2–6 × 104 cells/ml in a T25 flask (Asahi Technoglass, Funabashi, Japan) in Dulbecco's modified Eagle's medium (DMEM; Sigma-Aldrich, St. Louis, MO, USA) supplemented with 10% autologous canine serum (autoserum). Bone marrow cells and adipose cells were plated at 2–5 × 104 cells/ml and 2–8 × 104 cells/ml on a 35-mm collagen type I-coated dish (Asahi Technoglass) in DMEM supplemented with 10% autoserum. They were detached by incubation with TrypLE™ Select (Life Technologies) prior to 70% confluence.
We isolated DPSCs, BMSCs, and ADSCs by a novel method utilizing G-CSF (NEUTROGIN®; Chugai Pharmaceutical Co., Ltd., Tokyo, Japan)-induced stem cell mobilization (20,24). Costar Transwell® (a permeable support with an 8.0-mm polycarbonate membrane 6.5-mm insert; Corning, Lowell, MA, USA), used as the upper chamber, was inserted into 24-well tissue culture plates (BD Biosciences, Franklin Lakes, NJ, USA), which were used as the lower chamber. Colony-derived DPSCs, BMSCs, and ADSCs (2 × 104 cells/100 μl DMEM) at the second passage of culture were added to the upper chambers, and 390 μl of DMEM supplemented with 10% autoserum and G-CSF (final concentration: 100 ng/ml) was added to the lower chambers. After 48-h incubation, the medium was changed with DMEM supplemented with 10% autoserum without G-CSF. The cells that had transmigrated were enumerated, and isolation efficiency was calculated. Isolated cells were termed MDPSCs (DPSCs mobilized by G-CSF), MBMSCs (BMSCs mobilized by G-CSF), and MADSCs (ADSCs mobilized by G-CSF), respectively. Once the cells reached 60–70% confluence, they were detached by incubation with TrypLE™ Select and subcultured in 35-mm dishes (BD Biosciences) and further subcultured into cell culture flasks (25 cm2 and further 75 cm2; Asahi Technoglass) at 0.6 × 104 cells/cm2 in DMEM supplemented with 10% autoserum.
Canine bone marrow CD31- SP cells were isolated as described previously (9,12). Primary bone marrow cells were labeled with Hoechst 33342 (5 μg/ml; Sigma-Aldrich). Then the cells were preincubated with anti-Fc g III/II receptor antibody (1:10; Miltenyi Biotec, Bergisch Gladbach, Germany) for 20 min at 4°C. The cells were further incubated with the mouse IgG1 negative control [phycoerythrin (PE); 1:10; MCA928PE; AbD serotec, Oxford, UK] or anti-CD31 antibody (PE; 1:10; JC70A; Dako, Glostrup, Demark) for 60 min at 4°C. Cell sorting was performed using a FACSAria II (BD Biosciences). Bone marrow CD31- SP cells were plated into 35-mm collagen type I-coated dishes in DMEM supplemented with 10% fetal bovine serum (FBS; Lot #10D416; Cell Culture Bioscience, Nichirei Biosciences Inc., Tokyo, Japan) to maintain the cells. Once the cells reached 60–70% confluence, they were detached by incubation with TrypLE™ Select and subcultured. In some experiments, the cell lines were used. Human umbilical vein endothelial cells, (HUVECs; 7F3415, female; Lonza, Valais, Switzerland) were cultured in endothelial basal medium (EBM)-2 supplemented with growth factors and FBS as provided in the EGM™-2 BulletKit™ (Lonza). Human neuroblastoma cell line (TGW cells; 0618, male; Japanese Collection of Research Bioresources Cell Bank; JCRB, Osaka, Japan) were cultured in DMEM supplemented with 10% FBS.
Flow Cytometric Analysis
The phenotype of MDPSCs, MBMSCs, and MADSCs were characterized by flow cytometry (FACS Canto II; BD Biosciences) at the sixth passage of culture after immunolabeling with antigen cell surface markers. The cells were preincubated with anti-Fc g III/II receptor antibody (1:10; Miltenyi Biotec) for 20 min at 4°C. Then, they were immunolabeled for 60 min at 4°C with an anti-rabbit IgG negative control (Alexa Fluor 488; 1:10; #4340; Cell Signaling, Danvers, MA, USA), a mouse IgG2a negative control [fluorescein isothiocyanate (FITC); 1:10; sc-2856; Santa Cruz Biotechnology, Dallas, TX, USA], a mouse IgG2b negative control (Alexa Fluor 488; 1:10; MPC-11; Biolegend, San Diego, CA, USA), and the antibodies against CD105 (Alexa Fluor 488; 1:10; orb10285; biorbyt, Cambridge, UK), chemokine C-X-C motif receptor 4 (CXCR4; FITC; 1:10; 12G5; R&D Systems, Inc., Minneapolis, MN, USA), and G-CSFR (Alexa Fluor 488; 1:10; S1390; Abcam, Cambridge, UK).
Cell Proliferation and Migration Assay
To measure proliferation of MDPSCs, MBMSCs, and MADSCs compared with colony-derived DPSCs, BMSCs, and ADSCs, these cells at the sixth passage of culture at 103 cells per well in a 96-well plate were cultured in DMEM supplemented with 10% autoserum. Ten microliters of Cell Counting Kit-8 (Dojindo Laboratories, Kumamoto, Japan) was added to the 96-well plate, and the cell numbers were measured using a spectrophotometer (Appliskan™; Thermo Electron Corp., Vantaa, Finland) at 450 nm absorbance at 2, 12, 24, 36, 48, and 60 h of culture. Wells without cells served as negative controls.
To examine the migration activity of MDPSCs, MBMSCs, and MADSCs by G-CSF, they were compared with colony-derived DPSCs, BMSCs, and ADSCs. Horizontal chemotaxis assay was performed using the TAXIScan-FL (Effector Cell Institute, Tokyo, Japan). The TAXIScan-FL consists of an etched silicon substrate and a flat glass plate, both of which form two compartments with a 6-mm-deep microchannel. Each cell fraction (1 μl of 105 cells/ml) was placed into the single hole with which the device is held together with a stainless steel holder, and 1 μl of 100 ng/ml of G-CSF was placed into the contra-hole. The video images of cell migration were taken every 3 h until 24 h.
To assess the antiapoptotic effect of G-CSF, MDPSCs, MBMSCs, and MADSCs were incubated with 500 nM staurosporine (Sigma-Aldrich) in DMEM supplemented with G-CSF (final concentration: 100 ng/ml). After 2 h, cells were harvested, and the cell suspensions were treated with annexin V-FITC and propidium iodide (PI) (Annexin-V-FLUOS Staining Kit; Roche Diagnostics) for 15 min and analyzed by FACS Canto II (BD Biosciences).
Real-Time RT-PCR Analysis
To further characterize the phenotype of MDPSCs, MBMSCs, and MADSCs, total RNA was extracted with TRIzol (Life Technologies) at the sixth passage of culture. First-strand cDNA syntheses were performed on the total RNA of these cells by reverse transcription using the ReverTra Ace-α (Toyobo, Tokyo, Japan) after DNase I treatment (Roche Diagnostics) at 37°C for 20 min.
Real-time RT-PCR was performed at 95°C for 10 s, 65°C for 15 s, and 72°C for 8 s using primers for the angiogenic and neurotrophic factors (Table 1), granulocytemonocyte colony-stimulating factor (GM-CSF), matrix metalloproteinase-3 (MMP3), vascular endothelial growth factor (VEGF), brain-derived neurotrophic factor (BDNF), glial cell-derived neurotrophic factor (GDNF), and nerve growth factor (NGF) were labeled with Light Cycler-Fast Start DNA master SYBR Green I (Roche Diagnostics) in a Light Cycler (Roche Diagnostics). The mRNA expression was normalized with β-actin. The RT-PCR products were subcloned into the pGEM-T easy vector (Promega, Madison, WI, USA) and confirmed by sequencing.
Canine Primers for Real-Time RT-PCR and In Situ Hybridization Probe Synthesis

GM-CSF, granulocyte-monocyte colony-stimulating factor; MMP3, matrix metalloproteinase-3; VEGF, vascular endothelial growth factor; BDNF, brain-derived neurotrophic factor; GDNF, glial cell-derived neurotrophic factor; NGF, nerve growth factor; MMP20, enamelysin; DSPP, dentin sialophosphoprotein.
Effect of MDPSC-, MBMSC-, and MADSC-Conditioned Media
At 60% confluence, the culture medium was switched to DMEM without serum, and the conditioned media (CM) from MDPSCs, MBMSCs, and MADSCs were collected 24 h later and concentrated approximately 80-fold by an Amicon Ultra-15 Centrifugal Filter Unit with an Ultracel-3 membrane (Millipore, Billerica, MA, USA). Protein concentration of the CM was determined by Coomassie (Bradford) Protein Assay Kit (Pierce Biotechnology, Rockford, IL, USA). Each CM was added to a final concentration of 5 μg/ml.
To assess the endothelial differentiation potential of the CM, HUVECs at the sixth passage were seeded at 1 × 104 cells/well in a 96-well plate on Matrigel (BD Biosciences) in EBM-2 (Lonza) containing 2% FBS, heparin, ascorbic acid, hydrocortisone (each supplement was attached to an endothelial growth medium EGM™-2 Bulletkit™, Lonza) and 5 μg/ml each CM from MDPSCs, MBMSCs, and MADSCs with or without G-CSF (100 ng/ ml). Network formation was observed after 4 h of cultivation. The total lengths of networks of cords and tube-like structures were measured under an inverted microscope (Leica DFC290HD, Leica Microsystems GmbH, Wetzlar, Germany) using Leica Application Suites V3 (Leica).
To assess the stimulatory effect of CM on neurite outgrowth, human neuroblastoma cell line, TGW cells were used. For quantification of neurite outgrowth, TGW cells at the eighth passage were serum-starved and stimulated with 5 μg/ml of each CM from MDPSCs, MBMSCs, and MADSCs with or without G-CSF (100 ng/ml) for 48 h. The mean neurite length was measured under an inverted microscope (Leica DFC290HD) using Suite V3 (Leica). At least 100 cells were counted per sample (n = 3).
To assess the proliferative effect of the CM, bone marrow CD31- SP cells were seeded at 1 × 103 cells per well in a 96-well plate in DMEM (Sigma-Aldrich) supplemented with 0.2% bovine serum albumin (BSA) with or without 5 μg/ml of CM from MDPSCs, MBMSCs, and MADSCs. Ten microliters of Cell Counting Kit-8 was added to the 96-well plate, and the cell numbers were measured using a spectrophotometer at 450 nm absorbance at 2, 12, 24, 36, 48, and 60 h of culture. Wells without cells served as negative controls.
To examine migration activity of bone marrow CD31-SP cells in 5 μg of CM from MDPSCs, MBMSCs, and MADSCs with or without 100 ng of G-CSF, horizontal chemotaxis assay was performed using the TAXIScan-FL.
To assess the antiapoptotic effect of the CM, bone marrow CD31- SP cells were incubated with 500 nM staurosporine (Sigma-Aldrich) in DMEM supplemented with 5 μg/ml of CM from MDPSCs, MBMSCs, and MADSCs. After 2 h, bone marrow CD31- SP cells were harvested, and the cell suspensions were treated with annexin V-FITC and PI for 15 min and then analyzed by flow cytometry.
Differentiation of Odontoblast Lineage
Canine DPSCs were cultured in DMEM supplemented with 10% FBS, 50 μg/ml l-ascorbic acid 2-phosphate (Wako Pure Chemical Industries, Ltd., Osaka, Japan), 1 mM inorganic phosphate (Pi) (Sigma-Aldrich), and bone morphogenetic protein (BMP2; kindly provided by Astellas Pharma Co., Ltd., Tokyo, Japan) at a final concentration of 100 ng/ml (8). To compare the mineralization followed by odontoblast differentiation of CM from MDPSCs, MBMSCs, and MADSCs, 5 μg/ml of CM was added instead of BMP2. The medium was changed twice a week. After 21 days, the cell layer was fixed with 4% para-formaldehyde (Sigma-Aldrich), and Alizarin red (Wako) staining was performed. To characterize mRNA expression of odontoblastic differentiation marker, enamelysin (MMP20) (Table 1), real-time RT-PCR was performed described above.
Experimental Model of Pulp Regeneration
An experimental model of pulp regeneration in the permanent teeth with complete apical closure in female dogs (Kitayama Labes) at 9–11 months of age (6) was used. The whole pulp tissue was removed, and the root canals were enlarged to open the apical foramen to 0.6 mm in width in incisors. Autologous transplantation of MDPSCs, MBMSCs, and MADSCs, 5 × 105 cells in each, at the sixth passage was performed with G-CSF (NEUTROGIN®) at the final concentration of 7.5 ng/ μl and atelocollagen scaffold (Koken, Tokyo, Japan) at 20 μl/site (n = 5, day 14, extracted = 20). The cavity was sealed with zinc phosphate cement (elite Cement, GC, Tokyo, Japan) and composite resin (Clearfil FII, Kuraray, Tokyo, Japan) following treatment with a bonding agent (Clearfil Mega Bond, Kuraray).
Developed Monoclonal Antibody Against Canine Thyrotropin-Releasing Hormone Degrading Enzyme (TRH-DE)
A monoclonal antibody against canine TRH-DE was newly developed by Medical & Biological Laboratories (MBL) Co., Ltd. (Nagano, Japan) by immunization of purified protein. A mouse was injected percutaneously with protein emulsified in TiterMAX® adjuvant (Sigma-Aldrich), followed by four booster injections of 100 μg/ head of antigen at 2-day intervals. Two weeks later, lymphocytes from the immunized mice were fused with myeloma cells, P3-X63Ag8-U1 (P3U1) cells (JCRB). After centrifugation, the cells were pelleted, washed, and resuspended in DMEM supplemented with 15% FBS, hypoxantine–aminopterin–thymidine (Sigma-Aldrich), and 50 ng/l of mouse interleukin (IL)-6 (PeproTech, London, UK), and plated in flat-bottomed 96-well culture palates (Asahi Technoglass). Hybridoma clones were cultured for an additional 10 days, and then the culture media were assayed for specific antibody production using the ELISA method with the antigen protein. A hybridoma culture supernatant was added to antigen-coated 96-well microplate (Thermo Fisher Scientific, Waltham, MA, USA) and incubated for 60 min. After washing, horseradish peroxidase-conjugated goat Fab′ (1:10,000; 330; MBL, Nagoya, Japan) to mouse IgG reagent was added and incubated for 60 min. After washing, enzyme chromogenic substrate was added. The absorbance was measured automatically using a plate reader at 492 nm absorbance (Bio-Rad Laboratories, Inc., Berkeley, CA, USA). The ELISA-positive hybridoma cells were cloned by limiting dilution in 96-well culture plates and established as stable hybridoma cells. Culture supernatants of these cells were used for immunostaining.
Immunohistological and In Situ Hybridization Analysis
For morphological analysis, a total of 20 teeth from five dogs were used. After extraction, they were fixed in 4% paraformaldehyde (Sigma-Aldrich) at 4°C overnight and were embedded in paraffin wax (Fisher Scientific, Pittsburgh, PA, USA) after demineralization with Kalkitox™ (Wako). The paraffin sections (5 μm in thickness) were morphologically examined after staining with hematoxylin and eosin (H&E; Muto Pure Chemicals Co., Ltd. Tokyo, Japan). For examining relative amounts of regenerated tissue, three sections at 150-μm intervals from a total of five teeth on day 14, each transplanted with MDPSCs, MBMSCs, and MADSCs with G-CSF, were examined. On-screen image outlines of newly regenerated tissue were traced by capturing images of the histological preparations on a binocular microscope (Leica M 205 FA; Leica Microsystems GmbH), and the surface area of these outlines was determined by using Leica Application Suite software (version 3.4.1; Leica). The ratio of the regenerated area to the root canal area on day 14 was calculated in three sections of each tooth.
For quantitative analysis of matrix formation, each three paraffin sections from five teeth 14 days after cell transplantation of MDPSCs, MBMSCs, and MADSCs with G-CSF were stained with Masson trichrome staining (Muto Pure Chemicals Co., Ltd.). On-screen image outlines were traced by capturing images of the histological preparations on a binocular microscope (Leica M 205 FA; Leica). Positive area was quantitatively analyzed using Leica Application Suite software (version 3.4.1; Leica).
For neovascularization and innervation analyses, 5-μm-thick paraffin sections were deparaffinized and stained with fluorescein Bandeiraea simplicifolia lectin 1/ fluorescein Galanthus nivalis (snowdrop) lectin (BS-1 lectin; 20 μg/ml; Vector Laboratories, Inc., Youngstown, OH, USA) and anti-protein gene product 9.5 (PGP9.5) antibody (1:10,000; Ultra Clone, Ltd., Wellow, UK), respectively (6). The ratio of the BS-1 lectin+ newly formed capillaries and PGP9.5+ nerve fibers to the root canal area was calculated using Dynamic cell count BZ-HIC (KEYENCE, Osaka, Japan).
To confirm that the regenerated tissue is functional pulp tissue, 5-μm-thick paraffin sections were deparaffinized and immunostained with anti-canine TRH-DE antibody (undiluted solution of culture supernatants of hybridoma cells described above), a pulp tissue biomarker (37).
For examining odontoblastic differentiation in the regenerated tissue, in situ hybridization was performed using antisense probe of enamelysin and dentin sialophosphoprotein (DSPP) in the 5-μm-thick paraffin sections. Canine enamelysin (151 bp) and DSPP (190 bp) probes were constructed out of the pGEM-T easy (Promega) after subcloning the PCR products using the same primers as those designed for real-time RT-PCR (Table 1). Canine cDNA of enamelysin and DSPP linearized with NcoI were transcribed in vitro in the presence of digoxigenin (DIG) for production of antisense probes with Sp6 RNA polymerase (Roche Diagnostics). The DIG signals were detected by a Tyramide Signal Amplification system (PerkinElmer, Waltham, MA, USA). To further examine matrix formation followed by odontoblastic differentiation process, double staining of DSPP mRNA and aggrecan was performed. In situ hybridization and immunohistochemical double staining were performed as described previously (37). Polyclonal rabbit anti-aggrecan antibody (1:1,000; Novus Biologicals, Littleton, CO, USA) and anti-rabbit IgG-Alexa Fluor 488 secondary antibody (1:200; Life Technologies) were used. They were examined by confocal laser microscopy (TCS SP5 conventional inverted microscope; Leica).
Statistical Analyses
Data are reported as means ± SD. The p values were calculated using Tukey's multiple comparison test method using SPSS 21.0 (IBM, Armonk, NY, USA).
Results
Isolation and Characterization of MDPSCs, MBMSCs, and MADSCs
The subsets of BMSCs and ADSCs (MBMSCs and MADSCs) were isolated with the same optimal conditions of G-CSF-induced mobilization as previously described in human MDPSCs. The isolation efficiency of MBMSCs and MADSCs from each primary colony-derived MSCs (2.5 ± 0.5% and 2.9 ± 0.3%, respectively) was similar to that of MDPSCs (3.3 ± 0.6%). The isolated MBMSCs and MADSCs formed colonies in several days, containing spindle-shaped cells (Fig. 1A–C). The proliferation rate, however, was slower in MBMSCs compared with that in MDPSCs (Fig. 1G). The frequency of colony-forming units (CFUs) in MBMSCs and MADSCs at the fourth passage (73.6 ± 5.0% and 79.1 ± 7.7%, respectively) was significantly higher compared to colony-derived BMSCs and ADSCs (59.2 ± 3.1% and 59.6 ± 3.8%, respectively) (p < 0.05). The CFUs in MDPSCs (79.8 ± 5.6%) was similar to MBMSCs and MADSCs. The G-CSF-induced migratory activity was much higher in MBMSCs and MADSCs compared to colony-derived BMSCs and ADSCs. The migration activity of MBMSCs and MADSCs was lower than MDPSCs (Fig. 1H). Antiapoptotic effect of G-CSF in MBMSCs and MADSCs was similar to MDPSCs (Fig. 1Ia–c).
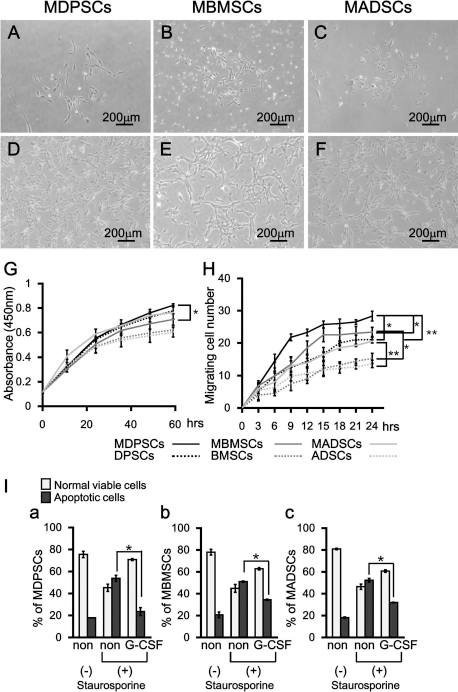
Characterization of mobilized MSCs by G-CSF from DPSCs, BMSCs, and ADSCs. (A) Mobilized dental pulp stem cells (MDPSCs), (B) mobilized bone marrow-derived stem cells (MBMSCs), and (C) mobilized adipose-derived stem cells (MADSCs) after isolation on day 5. (D) MDPSCs, (E) MBMSCs, (F) MADSCs at the sixth passage. (G) The proliferation in the presence of 10% canine serum (*p < 0.05). (H) Migration in the presence of granulocyte-colony stimulating factor (G-CSF) (*p < 0.05, **p < 0.01). (I) Antiapoptotic effect of G-CSF on MDPSCs (a), MBMSCs (b), and MADSCs (c). The relative percentage of viable and apoptotic cells analyzed by flow cytometry after annexin V staining (*p < 0.05). (G-I) The experiments were repeated three times, and data are expressed as mean ± standard deviation at three determinations. Tukey's multiple comparison test method was performed.
To examine “stemness” of MBMSCs and MADSCs, their positive rates of cell surface antigen markers were compared to MDPSCs. The percentages of CD105-positive cells in MBMSCs and MADSCs were similar to MDPSCs. The percentage of CXCR4+ cells and G-CSFR+ cells was significantly lower in MBMSCs compared to MDPSCs, and that in MADSCs was similar to MDPSCs (Table 2). The rates of CXCR4+ and G-CSFR+ cells in MDPSCs, MBMSCs, and MADSCs were higher than those in DPSCs, BMSCs and ADSCs, respectively (Table 2).
Flow Cytometric Analysis of Cell Surface Markers on MDPSCs, MBMSCs, MADSCs, DPSCs, BMSCs, and ADSCs at the Sixth Passage of Culture

Data are expressed as mean ± SD of four determinations (four lots). MDPSCs, mobilized dental pulp stem cells; MBMSCs, mobilized bone marrow-derived stem cells; MADSCs, mobilized adipose-derived stem cells; DPSCs, dental pulp stem cells; BMSCs, bone marrow-derived stem cells; ADSCs, adipose-derived stem cells; CXCR4, chemokine (C-X-C motif) receptor 4; G-CSFR, granulocyte-colony-stimulating factor receptor.
p < 0.01,
p < 0.05 compared with colony-derived stem cells.
p < 0.05 compared with MBMSCs. Tukey's multiple comparison test method.
The expression of angiogenic/neurotrophic factors, GM-CSF, MMP3, VEGF, and NGF, was slightly less in MBMSCs and MADSCs compared to MDPSCs. BDNF, however, was expressed slightly higher in MBMSCs compared to MDPSCs. GDNF was expressed slightly higher in MADSCs compared to MDPSCs (Table 3).
Relative mRNA Expression of Angiogenic and Neurotrophic Factors in MBMSCs and MADSCs Compared With MDPSCs
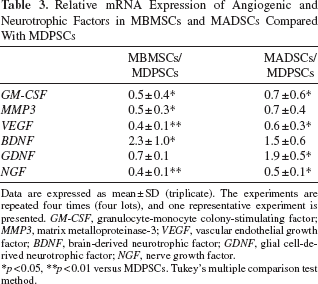
Data are expressed as mean ± SD (triplicate). The experiments are repeated four times (four lots), and one representative experiment is presented. GM-CSF, granulocyte-monocyte colony-stimulating factor; MMP3, matrix metalloproteinase-3; VEGF, vascular endothelial growth factor; BDNF, brain-derived neurotrophic factor; GDNF, glial cell-derived neurotrophic factor; NGF, nerve growth factor.
p < 0.05,
p < 0.01 versus MDPSCs. Tukey's multiple comparison test method.
In Vitro Effects of Conditioned Medium
We next examined the in vitro effects of CM of MBMSCs and MADSCs compared to MDPSCs with or without G-CSF. In the presence of each CM, HUVECs formed extensive networks of cords and tube-like structures as early as 4 h, a phenotype typically associated with endothelial cells (Fig. 2B–D). The CM of MDPSCs had higher angiogenic potential than the CM of MBMSCs and MADSCs, and their effects were enhanced in the presence of G-CSF (Fig. 2E). Neurite outgrowth in human neuroblastoma TGW cells was also enhanced by exposure to each CM (Fig. 2G–I). The CM of MDPSCs had higher stimulatory effects on neurite outgrowth than the CM of MBMSCs, and its effects were enhanced with G-CSF. There was no significant difference in the stimulatory effect of CM between MADSCs and MDPSCs (Fig. 2J).

Combinatorial effect of G-CSF and CM of MDPSCs, MBMSCs, and MADSCs. (A–D) The endothelial differentiation potential using the Matrigel assay. (E) Quantification and statistical analyses of total tube formation of human umbilical vein endothelial cells (HUVECs) on Matrigel (*p < 0.05, **p < 0.01). (F–I) The stimulatory effect of conditioned media (CM) on neurite outgrowth of TGW cells. (J) Quantification and statistical analyses of neurite outgrowth of TGW cells (*p < 0.05, **p < 0.01). (A and F) non, (B and G) MDPSCs CM with G-CSF, (C and H) MBMSCs CM with G-CSF, (D and I) MADSCs CM with G-CSF. (K) The proliferation activity of bone marrow CD31- SP cells using CM with or without G-CSF [*p < 0.05 vs. 0.2% bovine serum albumin (BSA)]. (L) The migration activity of bone marrow CD31- SP cells using CM with or without G-CSF (*p < 0.05, **p < 0.01). (M) Antiapoptotic effect of CM with or without G-CSF on bone marrow CD31- SP cells. The relative percentage of viable and apoptotic cells analyzed by flow cytometry after annexin V staining (*p < 0.05 vs. the percentage of only staurosporine added apoptotic cells, #p < 0.05). The experiments were repeated three times, and data are expressed as mean ± standard deviation at three determinations (three lots). Tukey's multiple comparison test method was performed.
The CM of the three cell populations stimulated proliferation activity of bone marrow CD31- SP cells similarly with or without G-CSF (Fig. 2K). The CM of three cell populations also stimulated migration activity of bone marrow CD31- SP cells. The CM of MDPSCs had higher migration activity compared to CM of MBMSCs and MADSCs, and its effect was enhanced by G-CSF, especially CM of MDPSCs (Fig. 2L). The antiapoptotic effect of CM of three cell populations without G-CSF was similar, and that of MDPSCs was superior to MBMSCs and MADSCs with G-CSF (Fig. 2M).
We further analyzed in vitro effects of CM of three cell populations on odontoblastic differentiation of DPSCs. Odontoblastic differentiation was induced by BMP2 with ascorbic acid and Pi on day 21 as demonstrated by Alizarin red staining (Fig. 3A), and was not induced by ascorbic acid and Pi without BMP2 (Fig. 3B) or by FBS only (Fig. 3C). On the other hand, odontoblastic differentiation was also induced by CM of three cell populations without BMP2 in the presence of ascorbic acid and Pi on day 21. The Alizarin red+ area was larger and induced by CM of MBMSCs compared to CM of MDPSCs and MADSCs (Fig. 3D–F). Odontoblastic differentiation marker, enamelysin, was more highly induced by the CM of MBMSCs compared to the CM of MDPSCs and CM of MADSCs (Fig. 3G).
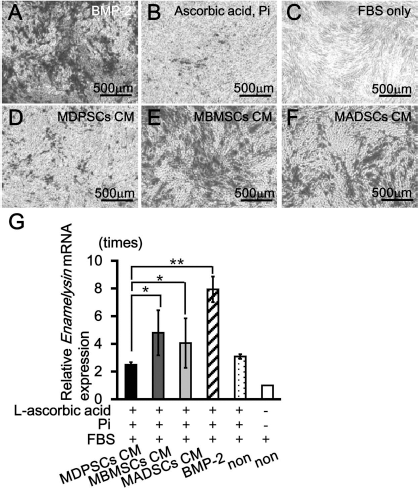
Odontoblastic differentiation of DPSCs induced by CM of MDPSCs, MBMSCs, and MADSCs. (A–F) Alizarin red staining 21 days after induction. (A) l-ascorbic acid, inorganic phosphate (Pi), and fetal bovine serum (FBS) with bone morphogenetic protein 2 (BMP2); (B) l-ascorbic acid, Pi, and FBS; (C) FBS only; (D) l-ascorbic acid, Pi, and FBS with MDPSCs CM; (E) l-ascorbic acid, Pi, and FBS with MBMSCs CM; (F) l-ascorbic acid, Pi, and FBS with MADSCs CM. (G) The relative mRNA expression of enamelysin. Data are expressed as mean ± standard deviation at four determinations (*p < 0.05, **p < 0.01). The experiments were repeated three times (three lots), and one representative experiment is presented. Tukey's multiple comparison test method was performed.
Pulp Regeneration After Cell Transplantation with G-CSF
Next, the efficacy for pulp regeneration was examined after autologous transplantation of MDPSCs, MBMSCs, and MADSCs with G-CSF into the root canals of pulpectomized teeth in dogs (Fig. 4). Pulp-like loose connective tissue with vasculature was regenerated 14 days after autologous transplantation of MDPSCs, MBMSCs, and MADSCs with G-CSF (Fig. 4Aa-c, Ba-c). Statistical analysis showed that the regenerated area was significantly larger in MDPSC transplantation compared to MBMSC and MADSC transplantation (1.9-fold, 1.4-fold, respectively) (Fig. 4Ad). Masson trichrome staining, however, revealed that fibrous matrix formation was observed in part of the MBMSC and MADSC transplantation (Fig. 4Cb, c). Statistical analysis showed that the Masson trichrome+ area was increased 2.3-fold and 1.6-fold in MBMSCs and MADSCs compared to that in MDPSC transplantation, respectively (Fig. 4Cd). Double staining of DSPP mRNA and aggrecan demonstrated that DSPP mRNA was expressed in some part of the regenerated pulp tissue where aggrecan was strongly expressed in MBMSC and MADSC transplantations (Fig. 4Da–c), indicating odontoblastic differentiation and its matrix formation leading to mineralization and future obliteration. Immunostaining analysis with BS-1 lectin and PGP9.5 demonstrated neovascularization and nerve fibers in the regenerated pulp tissue after all the cell transplantations, respectively (Fig. 4Ea–c, Fa–c). Statistical analysis showed that both vascularization area and innervation area were higher in MDPSC transplantation compared to that in MBMSC and MADSC transplantations, respectively (Fig. 4Ed, Fd). Immunostaining analysis of TRH-DE, a marker for pulp tissue, demonstrated that TRH-DE+ area was observed in the regenerated pulp tissue of MBMSC and MADSC transplantations, similar to MDPSC transplantation (Fig. 4Ga–c), indicating that the regenerated pulp-like tissue had a pulp tissue phenotype. The odontoblast-like cells lining the dental wall in the root canal were positive for DSPP and enamelysin, markers for odontoblasts (Fig. 4Ha–c, Ia–c).
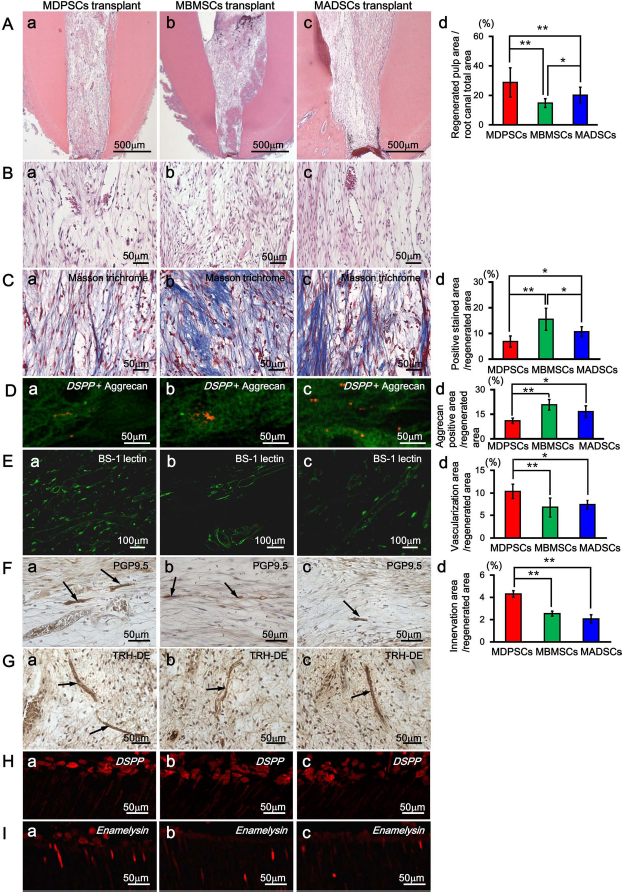
Regeneration of pulp tissue after autologous transplantation of MDPSCs, MBMSCs, and MADSCs with G-CSF in pulpectomized teeth of dogs (n = 5). (A–I) 14 days after transplantation. (A, B) Hematoxylin and eosin (H&E) staining. (C) Masson trichrome staining. (D) In situ hybridization of dentin sialophosphoprotein (DSPP) and immunostaining with aggrecan double staining. (E–G) Immunostaining with BS-1 lectin (E), PGP9.5 (F), and TRH-DE (G). (H, I) In situ hybridization analyses of DSPP (H) and enamelysin (I). (a) MDPSC transplantation. (b) MBMSC transplantation. (c) MADSC transplantation. (d) Morphometric statistical analyses. Data are mean ± standard deviation of five determinations (five lots) (*p < 0.05, **p < 0.01, Tukey's multiple comparison test method).
Discussion
Accumulating data have demonstrated the therapeutic effects of MSCs in animal models and clinical application of various diseases (35). Among these, MSCs derived from bone marrow or adipose tissue comprise a population of cells that exhibit extensive proliferative and multidifferentiation potential in vitro and that have trophic and paracrine functions with the greatest therapeutic impact in vivo (2,4,31). We have previously demonstrated CD31- SP cells derived from bone marrow or adipose tissues isolated by flow cytometer as possible alternative cell sources for pulp regeneration (12). The safety of these CD31- SP cells, however, has not been established. Some reports have characterized “stemness” as populations containing a higher rate of CXCR4+ cells (16,18,28) and with higher migratory activity (17,19,32). In this report, the safety isolation method using G-CSF-induced mobilization optimized for human DPSC subsets (20) was applied to isolate MSC subsets from bone marrow and adipose tissue. The isolation efficiency of MBMSCs and MADSCs was similar to MDPSCs. The enhanced frequency of CFUs, enhanced migratory activities, and increased rate of CXCR4+ cells and G-CSFR+ cells were demonstrated in MBMSCs and MADSCs compared to nonisolated BMSCs and ADSCs, as demonstrated in MDPSCs to DPSCs. All three populations showed similar antiapoptotic effects by G-CSF and only a slight difference in expression of angiogenic/neurotrophic factors. Furthermore, combinatorial trophic effects of their CM with G-CSF on accelerating angiogenesis, neurite extension, migration, and antiapoptosis were similar in all three populations. Thus, the potential utility of this device under the same optimized condition as human MDPSCs was demonstrated for isolation of canine MSC subsets from bone marrow and adipose tissue. The optimal isolation conditions including the plating cell number in the upper chamber, the final concentration of G-CSF in the lower chamber, and the incubation time with G-CSF might be dependent on tissue origin of MSCs.
Differential expression profiles of trophic factors and growth factors among MSC populations have been demonstrated (26,27). Some differences between the BMSCs and ADSCs are also demonstrated in their immunophenotype, differentiation potential, transcriptome, proteome, and immunomodulatory activity, although they share many biological characteristics. These differences may be derived from specific properties and inherent heterogeneity of the two populations or related to different isolation and culture methods (33). Thus, in this investigation, the stem cell properties of the MDPSCs, MBMSCs, and MADSCs that were isolated from the same individuals and by the same isolation and expansion method were compared. The higher expression rate of CXCR4 and G-CSFR, higher migratory activity, higher trophic effect of CM on angiogenesis, neurite extension, and migration were demonstrated in MDPSCs compared with MBMSCs. On the other hand, the similar expression rate of CXCR4 and G-CSFR and similar trophic effect on neurite extension were demonstrated in MDPSCs compared with MADSCs. These results suggested that these differences might be derived from specific properties depending on their origin (36) and heterogeneity of the three populations.
The present investigation further demonstrated that transplantation of MBMSCs or MADSCs together with G-CSF was also capable of regenerating pulp/dentin, although less volume of regenerated pulp tissue, less angiogenesis, and less reinnervation compared with MDPSCs with G-CSF were observed on day 14. Tissue regeneration effects of BMSCs and ADSCs have been compared in various tissues, including tendon (1), bone (3,5,15,25,38), and muscle (14). Although MSCs derived from different sources are able to support regeneration of variety of damaged tissues, extensive investigations are still needed to determine which cell sources are the best for specific diseases (29). Furthermore, the mechanism of distinct regenerative potential has not been elucidated yet. MSCs have been demonstrated to enhance angiogenic and neurogenic recovery, although the therapeutic effects may be derived from an indirect paracrine effect (29), rather than direct cell replacement. MSCs are regulated with the local stimuli and secrete bioactive factors and signals at variable concentrations in response to local microenvironmental cues (21). Our recent report showed that G-CSF and secretory factors from transplanted MDPSCs have a stimulatory effect on antiapoptosis and anchor the transplanted cells in the root canal. They further induce endogenous stem cell homing and survival and accelerate angiogenesis, reinnervation and pulp/ dentin regeneration (6). In this study, we showed that the antiapoptotic effect of G-CSF was the highest in MDPSCs. In addition, CM from MDPSCs had the highest induction of migration, antiapoptosis, endothelial differentiation, and neurite extension in the presence of G-CSF among the three populations. These results suggest the mechanism of the MDPSCs to regenerate pulp tissue superior to MBMSCs and MADSCs.
Transplantation of MBMSCs and MADSCs induced superior matrix formation in the regenerated pulp tissue compared to transplantation of MDPSCs. After induction of odontoblastic differentiation in vitro, higher staining with Alizarin red, and higher mRNA expression of enamelysin were demonstrated in the CM of MBMSCs and MADSCs compared to the CM of MDPSCs, suggesting that MBMSCs and MADSCs may secrete some odontoblastic differentiation factor more than MDPSCs.
In conclusion, transplantation G-CSF-mobilized MBMSCs and MADSCs are potential alternatives to MDPSCs, for therapeutic pulp/dentin regeneration.
Footnotes
Acknowledgments
We thank Mr. Masaaki Shimagaki from Toray Industry Inc. for supplying the chemically treated transmembrane. This work was supported by the Budget for promoting science and technology in Japan, which directly follows the policy of the Council for Science and Technology Policy (CSTP), chaired by the Prime Minister (M.N.), and the Research Grant for Longevity Sciences (23-10) from the Ministry of Health, Labour and Welfare (M.N.). The authors declare no conflicts of interest.
