Abstract
Spinal cord injury (SCI) is a major cause of paralysis with no current therapies. Following SCI, large amounts of ATP and other nucleotides are released by the traumatized tissue leading to the activation of purinergic receptors that, in coordination with growth factors, induce lesion remodeling and repair. We found that adult mammalian ependymal spinal cord-derived stem/progenitor cells (epSPCs) are capable of responding to ATP and other nucleotidic compounds, mainly through the activation of the ionotropic P2X4, P2X7, and the metabotropic P2Y1 and P2Y4 purinergic receptors. A comparative study between epSPCs from healthy rats versus epSPCis, obtained after SCI, shows a downregulation of P2Y1 receptor together with an upregulation of P2Y4 receptor in epSPCis. Moreover, spinal cord after severe traumatic contusion shows early and persistent increases in the expression of P2X4 and P2X7 receptors around the injury, which are completely reversed when epSPCis were ectopically transplanted. Since epSPCi transplantation significantly rescues neurological function after SCI in parallel to inhibition of the induced P2 ionotropic receptors, a potential avenue is open for therapeutic alternatives in SCI treatments based on purinergic receptors and the endogenous reparative modulation.
Keywords
Introduction
Spinal cord injury (SCI) is one of the major causes of disability around the world, which causes irreversible loss of function distal to the lesion (paralysis) as a result of axonal damage, demyelination, and death of oligodendrocytes, astrocytes, and neurons, including both spinal cord interneurons and motor neurons (16,18). Although much work has been accomplished regarding understanding the pathophysiology of SCI, there is an important need to determine novel methods to replace the cell loss, integrating newly transplanted or generated cells into the spinal cord circuitry, and novel targets for therapeutic interventions.
Ependymal stem/progenitor cells (epSPCs) are multipotent stem cells found in the adult tissue surrounding the ependymal canal of the spinal cord (39). Frisen and colleagues showed that after SCI, epSPC progeny are recruited to the injury site, even when the injury does not affect the epSPCs or their processes, giving rise among others to new oligodendrocyte progenitors (25). The intrinsic potential of epSPCs to replace some of the cells in the spinal cord following injury opens up the opportunity for developing noninvasive therapies for patients with SCI, through activating the differentiation of epSPCs into various cell types. An in vitro study in mice has shown that the manipulation of epSPCs after SCI might be a viable strategy for restoring neuronal dysfunction in humans. Thus, using specific differentiation protocol, 90% of differentiated cultures of epSPCs obtained after SCI stain positive for the motor neuron-specific marker HB9, with 32% of these motor neurons displaying electrophysiological properties that resemble those of functional spinal motor neurons (28). Transplantation of adult spinal cord-derived neurospheres from healthy donors was first shown to slightly improve motor recovery with aberrant axonal sprouting associated with allodynia. In our previous work, acute transplantation of epSPCs cultured from spinal cord-injured donors (ependymal stem/progenitor cells after injury, epSPCis) was able to efficiently reverse the paralysis associated with SCI in rats (28).
The implication of purinergic signaling in stem cell biology, including regulation of proliferation, differentiation, and cell death, has become evident in the last decade (4). Nucleotides activate two different types of purinergic receptors called P2X and P2Y receptors. The P2X are ion-gated channels that lead to a fast calcium influx (ionotropic), whereas P2Y are G-protein-coupled receptors (metabotropic). To date, seven P2X ionotropic subunits (P2X1-7) and eight P2Y metabotropic receptors (P2Y1,2,4,6,11,12,13,14) have been cloned and characterized according to their agonist sensitivity, sequence identities, and signal transduction mechanism. All P2X receptors are formed by assembly of three P2X subunits and mediate calcium entry into the cell following activation by adenosine triphosphate (ATP), although affinity, desensitization, and recovery properties vary between different receptor subtypes (5). Concerning P2Y receptors, four subtypes are activated by adenine nucleotides (P2Y1,11,12,13), two exclusively respond to uracil derivatives (P2Y6,14), and two are activated by both adenine and uracil nucleotides (P2Y2,4) (51). P2Y12,13,14 are coupled to Gi proteins, whereas P2Y1,2,4,6,11 are coupled to Gq/11 proteins that mediate calcium release from intracellular stores and diacyl-glycerol production. Neurospheres obtained from fetal rat brain expressed P2X2-P2X7 receptor subunits, as well as P2Y1, P2Y2, P2Y4, and P2Y6 receptors (4,42). Functional purinergic receptors were also detected in adult progenitor cells from both the subventricular zone (SVZ) and the dentate gyrus of the hippocampus (43). Neurospheres derived from the adult mouse SVZ express P2Y1 and P2Y2 nucleotide receptors, whose activation increases cell proliferation in the presence of growth factors, suggesting a synergistic activation of several intracellular pathways (15,27). Following SCI, large amounts of ATP and other nucleotides are released by the traumatized tissue leading to the activation of purinergic receptors that, in coordination with growth factors, induce lesion remodeling and repair (4,13). Moreover, blockade of purinergic receptors after SCI reduced the gliosis response and diminishes also the formation of a glial scar (38).
The first aim of this study was to analyze the expression profile of purinergic receptors in ependymal-derived neurospheres and to determine which purinergic receptors are functional via the analysis of intracellular Ca2+ concentration. Our study demonstrated for the first time that epSPCs express functional ionotropic P2X4 and P2X7 and metabotropic P2Y1 and P2Y4 receptors, able to respond to ATP, adenosine diphosphate (ADP), and other nucleotidic compounds. Interestingly, afterward, a comparative study between epSPCs cultured from healthy rats versus epSPCis, obtained from rats that 1 week earlier suffered an SCI, revealed that a downregulation of P2Y1 receptor together with an upregulation of P2Y4 receptor occur in epSPCis. Following the time course expression of purinergic receptors in spinal cord after injury, we showed that severe traumatic contusion induced early and persistent increase in the expression of P2X4 and P2X7 receptors around the injury, which was completely reversed by acute transplantation of epSPCis when a significant locomotion was restored.
Materials and Methods
Antibodies and Chemicals
P2X and P2Y receptor antibodies were purchased from Alomone Labs (Jerusalem, Israel), with the exception of the P2X6 receptor that was obtained from Chemicon (Temecula, CA, USA). Horseradish peroxidase-conjugated secondary antibodies were from Dako (Barcelona, Spain). Cy3™-conjugated donkey anti-rabbit IgG was from Jackson ImmunoResearch (West Grove, PA, USA), and Oregon Green-Alexa 488-conjugated goat anti-mouse IgG was from Molecular Probes (Leiden, The Netherlands). ATP, ADP, uridine triphosphate (UTP), UDP, α,β-methylene-ATP (α,β-meATP), 2′,3′-O-(4-benzoyl)-benzoyl ATP (BzATP), diadenosine tetraphosphate (Ap4A), diadenosine pentaphosphate (Ap5A), suramin, Brilliant Blue G (BBG), 2-aminoethoxydiphenyl borate (2-APB), and ethylene gly-col tetraacetic acid (EGTA) were all from Sigma-Aldrich (St. Louis, MO, USA). Pyridoxalphosphate-6-azophenyl-2′,4′-disulfonic acid (PPADS) was from Tocris (Bristol, UK) and Fura 2-AM was from Molecular Probes. Primers and other analytical-grade reagents were purchased from Sigma-Aldrich.
Ependymal/Progenitor Cell Isolation and Culture
epSPCs or epSPCis were harvested from adult female Sprague-Dawley rats, SD-Tg(GFP)2BalRrrc (enhanced green fluorescent protein gene under the control of the human ubiquitin-C promoter with the woodchuck hepatitis virus posttranscriptional regulatory element; RRRC, Columbia, MO, USA) (23), isolated and cultured as previously described (28,37). eGFP+/+ rats were used for cell transplantation experiments and eGFP-/- rats for in vitro assays. epSPCs were obtained from a noninjured rat, and epSPCis were isolated 1 week after severe contusion of spinal cord (250 kdyn at T8-T9).
Spinal Cord Contusion, epSPCis Transplantation, and Functional Evaluation
Adult female Sprague-Dawley rats were obtained from the animal house of “Príncipe Felipe” Research Center (Valencia, Spain). SCI by traumatic contusion was performed as previously described (28). Briefly, anesthetized rats (of an average of 200 g) with isofluorane (B. Braun, Barcelona, Spain) were subjected to laminectomy at the T8-T9 level to induce a severe traumatic lesion by applying 250 kdyn using the “Infinitive Horizon Impactor” (Precision Systems, Kentucky, IL, USA). Then 106 epSPCis were transplanted by stereotaxis at a distance of 2 mm rostral and caudal to the lesion. All animals were subjected to postsurgery care, passive, and active rehabilitation protocols as was previously described (28). Open-field locomotion was evaluated by using the 21-point Basso, Beattie, and Bresnahan (BBB) locomotion scale (1) after blind visualization of digitally individually videotaped animals once a week. The experimental protocol was approved by the Animal Care Committee of the Research Institute Principe Felipe (Valencia, Spain) in accordance with the National Guide to the Care and Use of Experimental Animals (Real Decreto 1201/2005).
Calcium Microfluorimetric Analysis
Neurospheres were placed on coverslips pretreated with poly-L-lysine and laminin (Life Technologies, Madrid, Spain). After 24 h, some cells of the outer layers of the neurosphere migrated over the coverslip, and isolated cells can be observed. Microfluorimetric studies were performed as previously described (10,14). Briefly, cells were loaded with the calcium dye Fura 2-AM (5 μM) for 45 min at 37°C. Afterward, coverslips were placed in a superfusion chamber (Warner Instruments, Hamden, CT, USA) on a microscope (Nikon), and the cells were stimulated for 30 s with different agonists: ATP (1 μM to 1 mM), α,β-meATP (100 μM), BzATP (10 μM-100 μM), ADP (100 μM), UTP (100 μM), UDP (100 μM), Ap4A (100 μM), and Ap5A (100 μM). In other studies, the compounds EGTA (to reduce extracellular calcium to 200 nM; 6 mM), PPADS (30 μM), suramin (100 μM), 2-APB (100 μM), and BBG (1 μM) were preincubated for 5 min before agonist superfusion. In some experiments, a Mg2+ (Merck, Madrid, Spain)-free solution was used, replacing MgSO4 by glucose (Merck) at a concentration that conserved the solution osmolarity. The data are represented as the normalized fluorescence ratio F340/F380 that increases when [Ca2+]i increases.
RT-PCR and Quantitative Real-Time PCR (Q-PCR)
Total RNA was extracted from either epSPCs or rat spinal cord. In the latter case, four adult rats per group (sham with no injury; 1 week, 2 weeks, 1 month, and 2 months after SCI) were sacrificed under CO2 exposition and immediately after the spinal cords were dissected. Approximately 1.5 cm of the thoracic segments including the injured or equivalent (in control group) area was removed and immediately frozen in liquid nitrogen for total RNA isolation. Total RNA was extracted and reverse transcribed as previously described (12). PCR reactions were carried out using AmpliTaq Gold® PCR Master Mix (Applied Biosystems, Foster City, CA, USA) and specific primers. Reactions involved an initial denaturation step at 94°C for 5 min, followed by 40 cycles of amplification (94°C for 30 s; 60°C for 30 s; and 72°C for 30 s) conducted with a thermocy-cler GeneAmp PCR System 2400 (Applied Biosystems). Fast thermal cycling was performed using a StepOnePlus® Real-Time System (Applied Biosystems). The results were normalized as indicated by parallel amplification of glyceraldehyde 3-phosphate dehydrogenase (GAPDH). Table 1 summarizes primers and Taqman MGB probes (Roche, Madrid, Spain) employed in these studies.
Specific Primers and TaqMan MGB Probes Used

Note: fw, forward; rv, reverse; nt, nucleotides; P2X, purinergic ionotropic receptor X; P2Y, purinergic metabotropic receptor Y; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Western Blot
Western blot analyses were performed as previously described (12). Briefly, total protein extracts from either epSPCs or epSPCis were electrophoresed on sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gels (Bio-Rad, Madrid, Spain), transferred to nitrocellulose membranes (Whatman, Maidstone, UK), and incubated overnight at 4°C with the appropriate primary antibody diluted in blocking solution, including 5% skinned dry milk, at the dilution specified in parentheses: P2X1 (1:200), P2X2 (1:200), P2X3 (1:500), P2X4 (1:500), P2X6 (1:200), P2X7 (1:250), P2Y1 (1:200), P2Y2 (1:200), P2Y4 (1:500), P2Y6 (1:200), α-tubulin (1:10,000). Protein bands were visualized by using horseradish peroxidase secondary antibodies (diluted in blocking solution at 1:5,000) followed by enhanced chemoluminescence detection (PerkinElmer, Waltham, MA, USA).
Immunocytochemical and Immunohistochemical Studies
Neurospheres placed on coverslips for 24 h were fixed with 4% paraformaldehyde (PFA; Sigma-Aldrich) for 15 min. Cells were permeabilized with 0.1% Triton X-100 (Sigma-Aldrich), blocked with 5% goat serum (Sigma-Aldrich), and incubated overnight with primary antibodies (diluted to 1:50 in blocking solution). Afterward, cells were incubated for 1 h with either Cy3™-conjugated or Oregon Green-Alexa 488-conjugated secondary antibodies (1:400). Nuclei were counterstained with 4′,6-diamidino-2-phenylindole (DAPI; Invitrogen). For immunohistochemical studies, control (noninjured), SCI (subjected to severe SCI and sacrificed 2 months later), and SCI+epSPCi (subjected to severe SCI and transplanted with epSPCis and sacrificed 2 months later) were transcardially perfused first with 0.9% NaCl and then with 4% PFA in phosphate buffer. The spinal cord was dissected and cryopreserved by overnight incubation in 30% sucrose (Merck). The medullar tissue cryosectioned at 10 μm, in the longitudinal axis, was processed as previously described for neurosphere staining. Confocal images were acquired with a TCS SPE microscope from Leica Microsystems (Wetzlar, Germany), and fluorescence signal was quantified by using the software Leica LAS AF.
Statistical Analysis
Data were analyzed using one way-ANOVA with the Dunnett's post hoc test or unpaired t-test for two-group comparisons. For multiple comparisons, one-way ANOVA analyses were corrected with Sidak's post hoc test (Graph Pad Prism 5; Graph Pad Software Inc., San Diego, CA, USA). Statistical analyses used are indicated in figure legends. Data are expressed as mean ± SEM of a minimum of three independent experiments for in vitro studies and four different animals per group within in vivo assays. A value of p <0.05 was considered statistically significant.
Results
Expression of Functional Purinergic Receptors in epSPCs
Neurospheres were cultured onto coverslips treated with poly-L-lysine and laminin to allow for their adhesion. After 24 h, some cells of the outer layers of the neurospheres migrated over the coverslip, and isolated cells can be observed (Fig. 1Aa). Cells were loaded with the calcium dye Fura 2-AM (Fig. 1Ab), and the coverslips were placed in a superfusion chamber on an inverted microscope. Then, cells were stimulated with ATP, and the change in intracellular-free calcium concentration ([Ca2+y was monitored in single cells (Fig. 1B). Application of growing doses of ATP resulted in a concentration-dependent increase in [Ca2+]i (Fig. 1C). Since the application of 100 μM ATP was enough to produce a strong calcium response in all cells analyzed (n = 150), this ATP concentration was fixed for subsequent experiments. The reduction of extracellular Ca2+ concentration to 200 nM using an EGTA buffer decreased in part the ATP-elicited calcium response (Fig. 1D). A similar effect was observed when epSPCs were treated with 2-aminoethoxydiphenyl borate (2-APB), an inhibitor of inositol trisphosphate receptor (Fig. 1D). These results showed that the increase in [Ca2+]i elicited by ATP was dependent in some extent on the entry of Ca2+ from the extracellular medium, and partially dependent on the mobilization of Ca2+ from intracellular stores, pointing to the participation of both ionotropic P2X and metabotropic P2Y receptors in the calcium response exerted by ATP in epSPCs.

ATP induces intracellular calcium transients in epSPCs that are mediated by both ionotropic P2X and metabotropic P2Y receptors. (A) Phase-contrast image of neurospheres cultured onto coverslips for 24 h (a, scale bar: 300 μm) and a detail of the same field viewed with fluorescein optics that shows isolated epSPCs loaded with the calcium dye Fura-2 (b, scale bar: 75 μm). (B) Fluorescence recorded from a single cell (white circle at the images) was used to calculate changes in the F340 nm/F380 nm ratio before and after stimulation with 100 μM ATP. Scale bar: 75 μm. (C) Cultures of epSPCs were superfused for 30 s with graded doses of ATP ranging from 1 μM to 1 mM. Representative traces (left panel) and mean ± SEM of the increment at the fluorescence ratios observed in ATP responding cells (right panel) are shown (n=166 cells from four independent experiments). (D) Cultures of epSPCs were challenged once with 100 μM ATP and subsequently exposed 100 μM ATP in the absence or presence of either 6 mM EGTA (reduces extracellular [Ca2+] to 200 nM) or 100 μM 2-aminoethoxydiphenyl borate (2-APB; (IP3 receptor inhibitor). Left panel: representative traces of the fluorescence ratios are shown. Right panel: mean ± SEM of the increment at the fluorescence ratios observed in ATP-responding cells are shown. Amplitudes of the second ATP response in the absence (black bar) or presence (gray bars) of either EGTA or 2-APB are represented as a percentage of the first ATP stimulation, which was considered as 100% (n = 218 cells from four independent experiments); one-way ANOVA with Dunnett's post hoc test, **p <0.01, ***p <0.001; or Sidak's post hoc test, ns: p>0.05.
Characterization of P2X Receptors in epSPCs
In order to investigate the presence of native ionotropic purinergic receptors in epSPCs, the expression of rat P2X subunits was analyzed at both the transcriptional and the protein level. RT-PCR experiments showed a single PCR product of the predicted size for P2X2, P2X4, P2X5, P2X6, and P2X7, whereas P2X1 and P2X3 transcripts were undetectable (Fig. 2A). To characterize the relative amount of each P2X transcript, Q-PCR assays were performed with P2X4 and P2X7 transcripts being the most abundant ones (Fig. 2B). Western blot analyses confirmed that P2X4 and P2X7 proteins were strongly expressed in epSPC lysates (Fig. 2C, blots). The sizes obtained for P2X4 (~60 kDa) and P2X7 subunits (~75 kDa) are consistent with the molecular weights reported for the glycosylated monomers (30). Immunocytochemical assays confirmed the presence of P2X4 and P2X7 receptors in epSPC-forming neurospheres (Fig. 2C).

Functional P2X4 and P2X7 receptors are simultaneously expressed in epSPCs. (A) RT-PCR was used to screen the expression of inotropic P2X receptors in epSPCs. Bands corresponding to P2X2, P2X4, P2X5, P2X6, and P2X7 subunits were amplified from cDNA coming from neurosphere cultures. No amplification products were observed in parallel assays carried out without template (not shown). (B) Quantitative real-time PCR showing the expression levels of P2X subunits in epSPCs. Values were normalized by the content of the housekeeping transcript GAPDH (n = 3 in triplicate). (C) The expression of P2X4 and P2X7 proteins in neurospheres was confirmed by Western blot and immunostaining with specific antibodies. Scale bars: 50 μm. (D) Cultures of epSPCs were challenged with ATP, the P2X1 and P2X3 agonist α,β-meATP, and the P2X7 agonist BzATP. All compounds were tested at 100 μM. The percentages of cells responding to each agonist are shown (n = 243 cells from four independent experiments). (E) Cultures of epSPCs were challenged once with 100 μM ATP and subsequently exposed 100 μM ATP in the absence or presence of the antagonists PPADS (30 μM), suramin (100 μM), and BBG (1 μM). Left panel: representative traces of the fluorescence ratios and percentages of cells responding to each agonist are shown. Right panel: means ± SEM of the increment at the fluorescence ratios observed in ATP-responding cells are shown. Amplitudes of the second ATP response in the absence (black bar) or presence (gray bars) of antagonists are represented as a percentage of the first ATP stimulation, which was considered as 100% (n = 197 cells from three independent experiments); one-way ANOVA with Dunnett's post hoc test, *p <0.05.
The functional characterization of P2X4 and P2X7 receptors was carried out analyzing changes in [Ca2+]i elicited by several P2X purinergic agonists and antagonists. First, epSPCs were stimulated with 100 μM ATP, which is able to activate all P2X receptors, with the exception of P2X7 that requires millimolar ATP concentrations (5). As expected, α,β-meATP, which activates P2X1 and P2X3 receptors, did not modify [Ca2+]i, in epSPCs (Fig. 2D). On the contrary, BzATP, which is able to activate the P2X7 receptor at micromolar concentrations elicited a strong calcium response in more than 90% of tested epSPCs (Fig. 2D). The calcium influx induced by 100 μM ATP was affected by neither the general P2X antagonist PPADS nor the P2X7 antagonist BBG (Fig. 2E). Suramin slightly reduced ATP calcium response (Fig. 2E), this effect being possibly explained by inhibition of some P2Y receptors, since suramin is a general P2 antagonist able to inhibit both P2Y1 and P2Y2 receptors.
To go in depth into the characterization of the calcium response elicited by BzATP, some pharmacological approaches were performed. As shown in Figure 3A, the increase in [Ca2+]i induced by BzATP was significantly reduced when the Ca2+ concentration in the extracellular medium was reduced to 200 nM. Moreover, the calcium response elicited by BzATP was not affected by 2-APB, but was completely blocked by BBG (Fig. 3A). It is well known that a reduction in the extracellular concentration of cations is able to potentiate P2X7 receptor-mediated calcium responses (50). Thus, epSPCs were challenged with submaximal BzATP concentrations (10 μM) prepared in either standard solution (containing 1.2 mM MgCl2) or in a free Mg2+ medium. As shown in Figure 3B, the calcium influx induced by BzATP was enhanced fivefold in the absence of Mg2+. Immunocytochemical assays using two different antibodies that recognize either intracellular or extracellular epitopes of P2X7 subunit confirmed the presence of P2X7 receptors in isolated epSPCs (Fig. 3C). Summarizing: ATP activates a P2X receptor insensitive to α,β-meATP, PPADS, suramin, and BBG; and BzATP activates a P2X receptor that is completely inhibited by BBG and potentiated in the absence of Mg2+ in the extracellular medium. All together, these results supported the presence of functional P2X4 and P2X7 receptors in epSPCs.

Pharmacological characterization of functional P2X7 receptor in epSPCs. (A) Cultures of epSPCs were challenged once with 100 μM BzATP and subsequently exposed to 100 μM BzATP in the absence or presence of 6 mM EGTA or 100 μM 2-APB or 1 μM BBG (P2X7 receptor antagonist). Left panel: representative traces of the fluorescence ratios and percentages of cells responding to each agonist are shown. Right panel: means ± SEM of the increment at the fluorescence ratios observed in BzATP-responding cells are shown. Amplitudes of the second BzATP response in the absence (black bar) or presence (gray bars) of EGTA, 2-APB, or BBG are represented as a percentage of the first ATP stimulation, which was considered as 100% (n = 209 cells from four independent experiments); one-way ANOVA with Dunnett's post hoc test, **p <0.01, ***p <0.001. (B) Potentiation of calcium responses induced by 10 μM BzATP in the absence of extracellular Mg2+ ions. Mg2+-free medium was superfused 5 min before and during BzATP application. The percentages of cells responding to each agonist are shown (n = 106 cells from three independent experiments). (C) Immunostaining of P2X7 receptors in isolated epSPCs using two different antibodies that recognize either intracellular (P2X7i) or an extracellular (P2X7e) epitope. Scale bar: 15 μm.
Characterization of P2Y Receptors in epSPCs
To characterize the metabotropic purinergic receptors that are expressed in epSPCs, we followed an analogous experimental approach, first analyzing the expression of P2Y transcripts. We focused the study on Gq/11-coupled P2Y receptors that mediate the release of Ca2+ from intracellular stores. RT-PCR experiments showed a single PCR product of the predicted size for P2Y1, P2Y2, and P2Y4, whereas the P2Y6 transcript was undetectable (Fig. 4A). Q-PCR studies showed that P2Y2 was the most abundant transcript, followed by P2Y1 and P2Y4 (Fig. 4B). However, Western blot analyses showed that only P2Y1 and P2Y4 proteins were expressed in epSPCs, since anti-P2Y2 receptor antibody recognized a weak band at ~52 kDa that was significantly higher than the predicted molecular weight of 42 kDa (Fig. 4C). Immunoreactive bands of ~60 kDa and ~41 kDa were consistent with the molecular weights reported for the glycosylated P2Y1 and unglycosylated P2Y4 receptors, respectively (44).

Functional P2Y1 and P2Y4 receptors are expressed in epSPCs. (A) RT-PCR was used to screen the expression of Gq/11-coupled (metabotropic) P2Y receptors in epSPCs. Bands corresponding to P2Y1,2, and 4 receptors were amplified from cDNA coming from neurosphere cultures. No amplification products were observed in parallel assays carried out without template (not shown). (B) Quantitative real-time PCR showing the expression levels of P2Y receptors in epSPCs. Values were normalized by the content of the housekeeping transcript GAPDH (n=3 in triplicate). (C) Total protein isolated from epSPCs were subjected to SDS-PAGE, transferred onto nitrocellulose membrane and incubated with anti-P2Y1, P2Y2, or P2Y4 antibodies. (D) Cultures of epSPCs were challenged with ATP, the P2Y1 agonist ADP, the P2Y2,4 agonist UTP, the P2Y6-preferred agonist UDP, and Ap4A, a full agonist of P2Y2. All compounds were tested at 100 μM. The percentages of cells responding to each agonist are shown (n = 102 cells from three independent experiments). (E) Cultures of epSPCs were challenged with ATP and Ap5A, both at 100 μM. The percentages of cells responding to each agonist are shown. Then, cells were stimulated once with Ap5A and subsequently exposed to Ap5A in the absence or presence of either 6 mM EGTA or 100 mM 2-APB. Left panel: representative traces of the fluorescence ratios are shown. Right panel: means ± SEM of the increment at the fluorescence ratios observed in Ap5A-responding cells are shown. Amplitudes of the second Ap5A response in the absence (black bar) or presence (gray bars) of either EGTA or 2-APB are represented as a percentage of the first ATP stimulation, which was considered as 100% (n = 182 cells from three independent experiments); one-way ANOVA with Dunnett's post hoc test, ***p <0.001. (F) Immunocytochemical detection of P2Y1 and P2Y4 receptors in neurospheres. Scale bar: 50 μm. (G) The expression of P2Y1 and P2Y4 receptors in isolated epSPCs was confirmed by immunostaining with specific antibodies. Scale bar: 15 μm.
In order to characterize the functionality of P2Y receptors expressed in epSPCs, we analyzed the ability of several P2Y agonists to increase [Ca2+]i in these cells. ADP, a P2Y1 receptor agonist, and UTP, a P2Y2 and P2Y4 receptors agonist, induced an increase in [Ca2+]i comparable to that elicited by ATP. However, UDP, a P2Y6-preferred agonist, only behaved as a partial agonist (Fig. 4D). Diadenosine polyphosphates (ApnA) comprise a group of compounds formed by two adenosine nucleosides joined by their ribose 5′ ends to a variable number of phosphates (n), which can range from 2 to 6. These compounds can activate with variable potency both P2Y (P2Y1, P2Y2, and P2Y4) and P2X (P2X1, P2X2, P2X3, P2X4, and P2X5) receptors (5,6,21,36). As shown in Figure 4D, Ap4A elicited a poor calcium response in epSPCs, probably because these cells do not express functional P2X2, P2X3, and P2Y2 receptors, which are fully activated by Ap4A. Ap5A is a full agonist at P2X3 receptor and a partial agonist at P2X1, P2X5, and P2Y4 receptors. In addition, Ap5A could be activating specific ionotropic receptors termed dinucleotide receptors, which are insensitive to other nucleotides or nucleosides (35). When Ap5A was assayed at epSPCs, this compound evoked a small increase in [Ca2+]i compared to ATP that was not modified when the Ca2+ concentration in the extracellular medium was reduced to 200 nM, but was completely dependent on the mobilization of Ca2+ from intracellular stores, pointing to the participation of P2Y4 receptors (Fig. 4E). Immunocytochemical studies showed the presence of P2Y1 and P2Y4 receptors in both neurospheres (Fig. 4F) and isolated epSPCs (Fig. 4G), corroborating the existence of functional P2Y1 and P2Y4 receptors in epSPCs.
Differential Expression of P2Y1 and P2Y4 Receptors in epSPCs Versus epSPCis
Previous studies revealed that epSPCs cultured from rats with severe spinal cord contusion proliferate faster, display enhanced self-renewal, and differentiate better into oligodendrocytes and motor neurons than epSPCs derived from healthy animals (28). Since several purinergic receptors can influence the proliferation, differentiation, and migration of a variety of stem cells (49), we analyzed whether functional P2 receptors expressed in control epSPCs are equally found in epSPCs derived from rats with SCI (epSPCis). First, we analyzed the ability of ATP, BzATP, ADP, UTP, UDP, Ap4A, and Ap5A to increase [Ca2+]i in epSPCis. Most of them exerted the same pharmacological properties than those observed in epSPCs (data not shown), with two exceptions: ADP and Ap5A. As shown in Figure 5A, the elevation of cytoso-lic [Ca2+] induced by ADP in epSPCs was significantly higher than that observed in epSPCis, whereas the calcium response elicited by Ap5A in epSPCs was smaller than that obtained in epSPCis. As expected, a reduction in the effect of ADP in epSPCis was associated with a decrease in the expression of P2Y1 mRNA (Fig. 5B) and protein (Fig. 5C) in these cells, whereas the increase in the calcium response induced by Ap5A in epSPCis correlated with a higher expression of P2Y4 (Fig. 5B, C).

P2Y1 and P2Y4 receptor expression is modified in epSPCs from rats subjected to severe spinal cord contusion (epSPCis). (A) Calcium responses elicited by ADP and Ap5A, both at 100 μM, in epSPCs (black) and epSPCis (gray). Left panel: representative traces of the fluorescence ratios are shown. Right panel: mean ± SEM amplitudes of the calcium responses elicited by ADP and Ap5A in epSPCs (n = 137 cells from three independent experiments) versus epSPCis (n = 171 cells from three independent experiments) are represented; unpaired t-test, *p <0.05. (B) Quantitative real-time PCR showing the expression levels of P2Y1 and P2Y4 receptors in both epSPCs and epSPCis. Values were normalized by the content of GAPDH transcript (n = 3 in triplicate); unpaired t-test, *p <0.05, **p <0.01. (C) Total protein isolated from epSPCs and epSPCis were subjected to SDS-PAGE, transferred onto nitrocellulose membrane, and incubated with anti-P2Y1 or anti-P2Y4 antibodies. Specific bands were normalized by the content of α-tubulin (n = 3); unpaired t-test, **p <0.01.
Acute Transplantation of Undifferentiated epSPCis After SCI Prevented Injured-Dependent Induction of P2X4 and P2X7 Receptors
Preceding studies reported that the expression of P2X4 and P2Y2 receptors can be significantly increased during the acute and chronic stages of SCI (38,41). Here we analyzed the expression of P2X4, P2X7, P2Y1, and P2Y4 transcripts at different times after induction of a severe traumatic lesion in the spinal cord of rats at T8 level. P2X4 and P2X7 transcripts were significantly expressed in the spinal cord of sham animals (laminectomy with no lesion), while P2Y1 and P2Y4 transcripts were almost undetectable (Fig. 6A, B, C, and D; black bars). Spinal cord contusion induced an early (2 weeks postinjury) and persistent increase in the expression of P2X4 and P2X7 mRNAs, with them remaining overexpressed at least 2 months after injury (Fig. 6A, B, gray bars). In the case of P2Y1 transcript, a weak and transitory increase was observed 2 weeks after lesion, returning to basal levels at longer time periods (Fig. 6C, gray bars). P2Y4 expression was not modified in rats with SCI at any time studied compared to sham animals (Fig. 6D, gray bars).

Transplantation of epSPCis after SCI reversed spinal cord upregulation of P2X4 and P2X7 expression induced by injury. Adult rats were subjected to laminectomy at the T8-T9 level followed by severe spinal cord contusion and acute transplantation of epSPCis. The expression of P2X4 (A), P2X7 (B), P2Y1 (C), and P2Y4 (D) transcripts in the spinal cord was analyzed by quantitative real-time PCR at different times after SCI: 1 week, 2 weeks, 1 month, and 2 months after injury (gray bars). Control animals (sham group): rats with laminectomy but without lesion (black bars). The effect of epSPCis transplantation was analyzed 2 months after injury (white bars). Values were normalized by the content of GAPDH transcript (n=4 mice per group); one-way ANOVA with Dunnett's post hoc test for sham versus SCI groups comparison, *p <0.05, **p <0.01, ***p <0.001, ns: p>0.05; or Sidak's post hoc test for 2 m SCI versus 2 m SCI + epSPCi comparison, ###p <0.001, ns: p>0.05.
Although preceding approaches based on spinal cord-derived SPCs from either fetal or adult healthy donors reported only modest or no significant functional improvements during the acute phase of transplantation (31,34), we previously demonstrated that acute transplantation of epSPCis obtained from injured tissue reverses the paralysis associated with spinal cord contusion in rats (28). Moreover, a blockade of P2 nucleotide receptors after SCI reduces the gliosis response and spared tissue (38). Based on these findings, we studied whether epSPCi transplantation immediately after SCI is able to decrease P2X4 and/or P2X7 expression in the spinal cord of injured rats. The acute transplantation of epSPCis at a distance of 2 mm rostral and caudal to the lesion was able to completely reverse the upregulation of P2X4 and P2X7 transcripts observed 2 months after SCI (Fig. 6A, B, white bars), while it did not modify the expression of both P2Y1 and P2Y4 mRNAs (Fig. 6C, D, white bars). Immunohistochemical studies corroborated that SCI induced a strong and persistent increment in P2X4 and P2X7 receptors in spinal cord slices, which could be significantly reversed by acute transplantation of epSPCis (Fig. 7A, B). P2Y1 and P2Y4 immunostaining was not modified by injury or acute epSPCi transplantation (Fig. 7A, B). All together, these results suggested that the control of P2X4 and P2X7 receptor expression exerted by epSPCi transplantation could be related with the functional capacity of epSPCis to recover neurological function following injury in epSPCi-transplanted animals (Fig. 7C), which, in fact, occurs in parallel with inhibition of the ionotropic receptor induction. An important avenue is then open for potential therapeutic alternatives in SCI treatments based on purinergic receptor modulation.
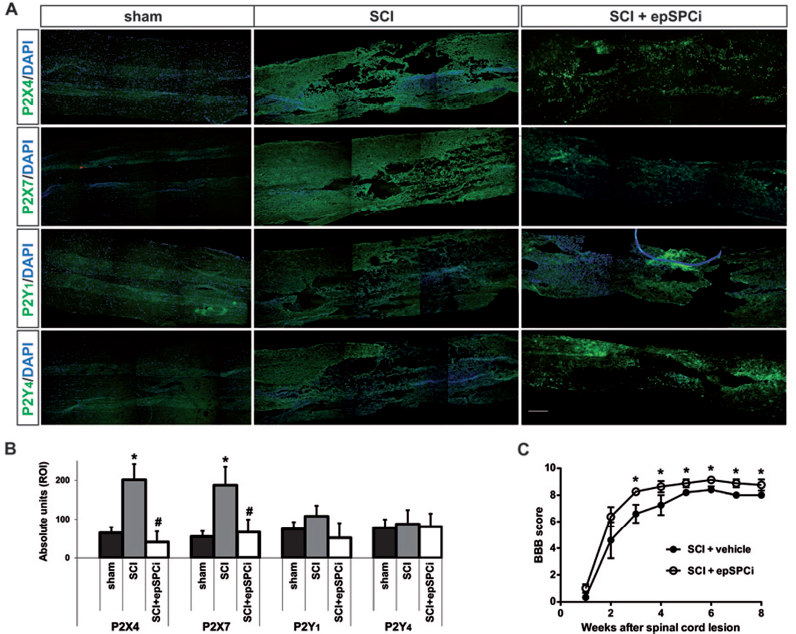
Transplantation of epSPCis after SCI prevented injury-dependent induction of P2X4 and P2X7 proteins in the spinal cord. (A) Immunohistochemical analysis was performed in spinal cord cryopreserved tissue of control animals (sham group), SCI (subjected to severe SCI by traumatic contusion and sacrificed 2 months later), and SCI + epSPCi (subjected to severe SCI by traumatic contusion and intramedullar transplanted with epSPCis, and sacrificed 2 months later) for P2X4, P2X7, P2Y1, and P2Y4 receptors using specific antibodies. Representative confocal images are shown for each experimental group at equivalent (control, left panels) or at the epicenter of the lesion (SCI or SCI + epSPCi; central and right panels); Scale bar: 200 μm. Nuclei are counterstained with DAPI. (B) Quantitative analysis of the protein expression by using ImageJ software for each experimental group from three different animals showed significant differences on P2X4 and P2X7 expression; one-way ANOVA with Dunnett's post hoc test for sham versus SCI group comparison, *p <0.05; or Sidak's post hoc test for SCI versus SCI + epSPCi comparison, #p <0.05. Green fluorescent protein-positive fluorescence from transplanted cells was subtracted. (C) Locomotor performance was evaluated weekly by BBB scoring, and a significant improvement was observed in rats transplanted epSPCis when compared to nontransplanted ones (vehicle) (n = 4 mice per group); one-way ANOVA with Dunnett's post hoc test for SCI + vehicle versus SCI + epSPCi groups comparison, *p <0.05. BBB scores are only represented up to 10 in y-axes (for better visualization) being the regular maximum score is 21.
Discussion
The importance of purinergic signaling in developmental and adult neurogenesis, including regulation of proliferation, differentiation, and cell death, has become evident in the last years (4,13,49). Purinergic receptors are expressed in both neural stem cells and neural precursor cells from very early stages of frog, chick, and mammalian embryonic development, suggesting their participation in the regulation of proliferation and lineage specification (46). Purinergic activation has been associated with proliferation and neurogenesis in neonatal and adult mouse olfactory epithelium (19,20). Functional purinergic receptors were also identified in the adult mammalian brain in specific neurogenic niches, the SVZ of the lateral ventricle and the subgranular layer of the hippocampal dentate gyrus (43).
Our study demonstrates that epSPCs respond to nucleotidic compounds through the activation of ionotropic P2X4 and P2X7, and metabotropic P2Y1 and P2Y4 receptors. As a first approach to investigate the presence of functional P2X and Gq-coupled P2Y receptors, we performed microfluorimetric techniques using the calcium dye Fura-2. To our knowledge, this is the first report of ATP-elicited Ca2+ entry signals in epSPCs. The partial dependence on extracellular Ca2+ together with the incomplete effect of IP3 receptor inhibition indicated that both P2X and P2Y receptors were mediating ATP-elicited Ca2+ entry in epSPCs. Previous studies reported that primary neurospheres prepared from the SVZ zone expressed P2X4 and P2X7 transcripts (45). In our cellular model, the quantification of mRNA and protein levels of P2X1-7 subunits also indicated that epSPCs mainly express P2X4 and P2X7 subunits. Pharmacological properties of endogenous P2X receptors found in epSPCs resembled that showed by homomeric P2X4 and P2X7 receptors. P2X7 receptor arouses special interest due to its dual function as an inhibitor of neurogenesis and axon outgrowth and inductor of cell death in other cases. In cultured hippocampal neurons axonal growth and branching was induced following P2X7 receptor inhibition (9), whereas a strong activation of P2X7 receptor causes necrosis/apoptosis in both embryonic and adult neural precursor cells (7,26). Interestingly, it is suggested that the cell death elicited by P2X7 receptor activation may counterregulate progenitor cell survival after CNS injury, where excessive neuro- and gliogenesis is induced (26).
Regarding metabotropic receptors, previous investigations support a role of P2Y1 and P2Y2 receptors in stimulating progenitor cell migration, which may be of relevance in situ for the local movement of cells in the neurogenic niches (15,40). Moreover, P2Y1 receptor antagonists reduced the size and frequency of primary neurospheres and permitted differentiation into neurons and glia (45). The quantification of mRNA and protein levels of Gq-coupled P2Y receptors, in combination with the pharmacological studies, showed that epSPCs mainly express functional P2Y1 and P2Y4 receptors. The involvement of P2Y2 receptor was excluded since Ap4A, a potent agonist for this receptor (32), induced a weak calcium response. Furthermore, intracellular calcium increase elicited by Ap5A was mediated by P2Y4 activation, since this dinucleotide is inactive at the rat P2Y1, P2X4, and P2X7 receptors (5,36,53).
Nanomolar concentrations of Ap5A and other diadenosine polyphosphates have been recently quantified in cerebellar dialysates as a consequence of the normal brain activity of the rat, reinforcing the role of dinucleotides as signaling molecules in the central nervous system (17).
Regarding the source of extracellular nucleotides, real-time bioluminescence imaging of ATP release revealed that neural progenitor cells themselves were the source of local ATP, which they spontaneously released in brief burst events (22). Moreover, after CNS hypoxia and trauma, or under other pathological conditions, large amounts of ATP and other nucleotides accumulate at the sites of injury and cooperate with growth factors to induce lesion remodeling and repair (3,13). We have previously reported that epSPCis cultured from rats with a severe spinal cord contusion display a higher rate of proliferation, more robust self-renewal, and better yields of oligodendrocytes and motoneurons when subjected to direct differentiation in vitro (28). Now we demonstrated that the expression and functionality of P2Y1 and P2Y4 receptors were modified in epSPCis compared to neurospheres derived from control animals. Thus, in epSPCis, a downregulation of P2Y1 receptor expression was accompanied by a decrease in the calcium response induced by ADP. On the contrary, epSPCis showed an upregulation of P2Y4 mRNA and protein that correlated with a higher Ap5A calcium response. These findings suggest that the expression levels of P2Y receptors may play a critical role in the modulation of neural progenitor cell expansion. In support of this idea, some reports demonstrated that purinergic signaling might be required not only for developmental progenitor cell expansion and neurogenesis, but also to maintain neural stem/progenitor cell niches in the adult brain. External addition of ATP or its analogs increased the mitotic index and rate of neural progenitor cells, whereas P2Y antagonists suppressed both neurosphere expansion and the mitotic index of cells within those neurospheres (22). Furthermore, purinergic receptors are localized in regions of active mitotic progenitor cell expansion and neurogenesis in the adult brain (2,43), suggesting that neural progenitor cells release ATP and respond to it with an increase in proliferation.
In response to spinal cord lesion, the proliferation of ependymal cells increases significantly, and these cells migrate to the injured zone (25). Previously, we demonstrated that the number of epSPCs that form neurospheres increases dramatically after SCI, and acute transplantation of these neurospheres into rats with SCI promotes early functional locomotor recovery (28). Here we showed that severe contusion induces early and persistent increase in the expression of P2X4 and P2X7 receptors in the injured spinal cord, which can be completely reversed by acute transplantation of undifferentiated epSPCis. The overexpression of P2X4 receptors following spinal cord lesion was previously reported, identifying the majority of P2X4-positive cells as activated microglia/macrophages and surviving neurons/neurites (41). P2X4 receptors are also elevated in spinal cord microglia after peripheral nerve injury (47,48), and P2X4 knockout mice have lower levels of neuroinflammation after SCI, resulting in significant improvement in tissue sparing and functional recovery, especially during the first week after injury (8). Concerning P2X7, postischemic, time-dependent upregulation of this receptor on neurons and glial cells has already been reported (11), but the relationship between its inhibition and the pathogenesis of contusive spinal cord injury remains controversial. Some evidences indicate that antagonism of P2X7 receptor improves both the functional and pathological consequences of SCI (33,52), whereas others conclude that P2X7 receptor blockade failed to improve motor recovery or histopathological outcome (24). We observed that a reduction in P2X4 and P2X7 receptors expression correlates with a functional locomotor recovery in a rat model of SCI acutely transplanted with epSPCis. However, further work will be necessary to know whether both phenomena are directly connected.
Conclusion
In summary, we have demonstrated that epSPCs of the adult mammalian spinal cord respond to ATP and other nucleotidic compounds, mainly through the activation of P2X4, P2X7, P2Y1, and P2Y4 receptors. The expression and functionality of P2Y1 and P2Y4 receptors were altered in epSPCis compared to neurospheres derived from control animals, suggesting that changes in purinergic signaling could be playing a relevant role in the regenerative capacities of epSPCis. Furthermore, acute transplantation of undifferentiated epSPCis following severe contusion completely reversed the overexpression of P2X4 and P2X7 receptors in spinal cord observed after injury. An interesting open question for further investigation is how P2Y1 and P2Y4 expression levels can be modulated within a balance between the P2X expression in epSPCis upon transplantation in the injured spinal cord. Previous studies showed that P2Y1 receptor antagonists reduced the size and frequency of primary neurospheres and permitted differentiation into neurons and glia (45). Other investigations support a role of P2Y1 and P2Y2/4 receptors in stimulating progenitor cell migration, which may be of relevance in situ for the local movement of cells in the neurogenic niches (15,40). Thus, P2Y1 receptor downregulation could be facilitating the differentiation of epSPCis into neuronal/glial cells that participate in the improvement of functional locomotor recovery observed after epSPCi transplantation, while an increment in P2Y4 receptor expression in epSPCis could increase the expansion and mitotic index of neural progenitor cells within those neurospheres. Moreover, it is well known that the neural precursor cells possess an immune modulatory effect (29), suggesting here an important influence of the epSPCi transplantation into the P2X-positive cells including, among others, the macrophages and microglia. Since ATP appears to increase the proliferation of neural stem/progenitor cells in the postnatal neurogenic niches of the brain (2,43), it is plausible that the ATP released in conditions of neuronal injury could mobilize endogenous epSPCs and induce them to undergo proliferation and neuronal differentiation. If this is the case, being able to manipulate this purinergic modulation would enable a greater control over the neurogenic capacity of epSPCs.
Footnotes
Acknowledgment
This work was supported by research grants from the Marcelino Botín Foundation, the Ministry of Science and Innovation (BFU2011-24743), the Spanish Ion Channel Initiative (CSD2008-00005), and the Instituto de Salud Carlos III (PI10/01683 and PI13/00319). Author contributions: Rosa Gómez-Villafuertes: conception and design, collection and/or assembly of data, data analysis and interpretation, and manuscript writing; Francisco Javier Rodríguez-Jiménez: collection and/or assembly of data, data analysis and interpretation; Ana Alastrue-Agudo: collection and/or assembly of data; Miodrag Stojkovic: financial support, data analysis and interpretation, final approval of manuscript; María Teresa Miras-Portugal: conception and design, data interpretation, manuscript writing, financial support, and final approval of manuscript; Victoria Moreno-Manzano: conception and design, data interpretation, manuscript writing, financial support, and final approval of manuscript. The authors declare no conflicts of interest.
