Abstract
In this study, the effects of autologous islet transplantation (ITx) were compared to those of oral antidiabetic drugs (OAD) after distal pancreatectomy (NCT01922492). We enrolled nondiabetic patients who underwent distal pancreatectomy for benign tumors. In the ITx group, islets were isolated from the normal part of the resected pancreas and implanted via the portal vein. Patients who did not receive ITx were regularly monitored and were enrolled in the OAD group if diabetes mellitus developed. The OAD group was treated with metformin with or without vildagliptin. Metabolic parameters were monitored for 12 months postoperatively. Nine patients in the ITx group and 10 in the OAD group were included in the analysis. After 12 months, hemoglobin A1c significantly increased by 5% of the baseline in each group. Area under the curve for blood glucose (AUCglucose) of the 75-g oral glucose tolerance test increased similarly in the immediate postoperative period in both groups but significantly reduced only in the ITx group thereafter. Insulinogenic index (INSindex) significantly decreased from 25.6 ± 18.9 to 4.7 ± 3.7 in the OAD group, while no significant change was observed in the ITx group (from 15.0 ± 4.5 to 11.0 ± 8.2). In the multiple regression analysis, ITx was an independent factor for changes in AUCglucose and INSindex. In addition, changes in INSindex in the ITx group after postoperative 6 months were associated with the efficacy of islet isolation, amount of grafts, and peak serum HMGB1 and VEGF levels after ITx. ITx was superior to OAD in maintaining insulin secretory capacity and glucose tolerance after distal pancreatectomy.
Keywords
Introduction
The number of patients who are identified with benign pancreatic disease and subsequently undergo partial pancreatectomy (Px) has increased with the widespread use of imaging studies (3). During distal Px, approximately 40% to 80% of the pancreas is removed. It has been traditionally thought that postsurgical diabetes mellitus (DM) is uncommon after partial Px, unless more than 80% of the pancreatic tissue is resected (26,43). However, the incidence of postoperative DM after partial Px varies from 4.8% to 38%, varying not only according to the type of surgery but also the duration of postoperative follow-up, progression of the underlying pancreatic disease, and presence of certain preoperative factors, such as obesity and impaired glucose tolerance (5,16,32,38). More generalized use of imaging studies has enabled early diagnosis of disease in younger patients. In addition, society is rapidly aging, as the overall life expectancy is progressively increasing. These factors combine to prolong life expectancy after pancreatic resection and increase the incidence of eventual DM. In a recent study from Korea, the 7-year diabetes-free survival rate was only 45% after 50-60% distal Px (11). Therefore, DM after Px may become a new medical challenge.
There are no treatment guidelines for postsurgical DM after partial Px because of a paucity of clinical evidence. Usually, oral antidiabetic drugs (OAD) for type 2 DM are used for postsurgical diabetes, although impaired insulin secretory capacity is the primary cause of this glucose intolerance (23,32). Since 1978, when the group at University of Minnesota reported the first cases of autologous islet transplantation (ITx) (44), this procedure has been performed with favorable results also in patients who underwent partial Px for benign pancreatic disease (8,11,12). With distal Px for tumor located in the middle of the pancreas, the resected region distal to the lesion could be used for islet isolation and autologous transplantation. Originally, autologous ITx was performed after total or near total Px in patients with chronic pancreatitis; this effectively prevented or ameliorated postoperative DM (27). Compared to allogeneic ITx, autologous ITx produces better glycemic control, without the need for immune suppression (27).
This study was performed to compare the effects of OAD and autologous ITx after distal Px. No previous studies have examined this issue. Metformin is the most commonly prescribed OAD. It is recommended as an initial therapy for type 2 DM by the American Diabetes Association and the European Association for the Study of Diabetes (9). Metformin is an insulin-sensitizing drug, whose mechanism has not been fully established. There are some studies on the direct effects of metformin on the pancreatic β-cells, which may differ depending on the context (20,25,36). Dipeptidyl peptidase-4 (DPP-4) inhibitors enhance glucose-dependent insulin secretion by suppressing degradation of endogenous glucagon-like peptide-1. There are several studies that some DPP-4 inhibitors promoted β-cell proliferation and increased β-cell mass in rodent models of type 2 diabetes (2,34) and improved β-cell function in patients with type 2 DM (1,4). DPP-4 inhibitors improved the outcome of syngeneic ITx in a mouse model of partial Px (15), but the effects of DPP-4 inhibitors have not been evaluated in humans after partial Px.
In this study, we aimed to compare the effects of OAD and autologous ITx after distal Px in terms of glucose tolerance, insulin secretory function, and insulin resistance and tried to find out a role of autologous ITx in partially pancreatectomized patients for benign diseases. Also, we tested the potential markers as a predictor for the efficacy of ITx (NCT01922492).
Materials and Methods
Patients and Study Design
The inclusion criteria were nondiabetic patients who underwent distal Px at Seoul National University Hospital between 2008 and 2011 for the pathologic diagnosis of pancreatic mass. DM was defined as fasting plasma glucose levels >126 mg/dl, 75-g oral glucose tolerance test (OGTT) 2-h plasma glucose levels >200 mg/dl, or a hemoglobin A1c (HbA1c) >6.5%. Patients who accepted ITx were allocated to the ITx group. If a subject refused ITx or if the pancreatic tissue distal to the mass was less than 25 g, the individual did not undergo ITx. Along with education on healthy diet and exercise, the participants were followed every 3 months until 12 months postoperatively to assess metabolic changes. Metformin (Diabex®; Daewoong Pharmaceutical Co. Ltd., Seoul, Korea) 500 mg/day was started if DM developed, and vildagliptin (Galvus®; Novartis, Seoul, Korea) was added 50 mg/day up to 100 mg/day if the patients were still in the diabetic range. If vildagliptin was refused, metformin was increased instead. Doses were adjusted to maintain glycemia under the diabetic range. Patient interviews were performed by a trained nurse to obtain a medical history and measure anthropometric parameters. The study was approved by the Institutional Review Board of Seoul National University Hospital (IRB No. H-0710-007-221), and written informed consent was obtained from all patients. The study was performed in accordance with the principles of the Declaration of Helsinki, as revised in 2000. The patients did not pay for their inclusion or transplantation treatment in this study.
Autologous Islet Transplantation
During distal Px, the region of the pancreas distal to the mass and appearing normal on gross inspection was transferred to the laboratory for islet isolation maintaining a cold ischemic time of less than 1 h. Islets were isolated as previously described with modifications (22). Briefly, after organ distension with intraductal collagenase (SERVA Electrophoresis GmbH, Heidelberg, Germany), digestion was performed using a modified Ricordi method (37). Islet purification was performed using a COBE 2991 processor (COBE, Lakewood, CA, USA). Except for two cases with purity <10%, the purity of most cases was >80%. Viable islets were counted as islet equivalent (IEQ) volume. Islet preparations were cultured in the Miami Medium-1 (Mediatech Inc., Herndon, VA, USA) containing 0.5% human serum albumin (Sigma-Aldrich, St. Louis, MO, USA) in a 5% CO2 humidified incubator at 37°C overnight, thereafter at 22°C for the remaining time before transplantation. With the final culture media, endotoxin activity was assessed by a commercial quantitative chromogenic Limulus amebocytes lysate assay (Lonza, enders, Belgium). For microbiologic evaluation with the final culture media, Gram stains and cultures using blood agar plate, MacConkey agar, Brucella agar, and phenyl-ethyl alcohol agar were performed. Endotoxin activity was not detected, and the culture results were negative. To evaluate islet function, 20 islets with a diameter of 100 μm were picked in an Eppendorf tube with 1 ml of Krebs-Ringer bicarbonate 2-[4-(2-hydroxyethyl)piperazin-1-yl] ethanesulfonic acid buffer (Gibco, Seoul, Korea) containing 0.2% BSA (Sigma-Aldrich) with low (5 mM) and high (20 mM) glucose (Sigma-Aldrich) serially for 60 min at each concentration. The soup from each incubation period was collected, and the insulin levels were determined with an ELISA kit (Sigma-Aldrich). Stimulation index was calculated as the ratio of insulin released from the islets in high to that in low glucose. ITx was performed on the day after surgery, only after a benign tumor was histologically confirmed. Histologic confirmation was done for surgical margins as well as the primary mass to be sure tumor cells were not processed and potentially transplanted. The islets were infused into the liver through percutaneous transhe-patic portal vein catheterization, with a concomitant intravenous heparin infusion.
Assessment of Metabolic Parameters and Markers of Islet Damage
A 75-g OGTT was performed preoperatively and at 1 week, 6 months, and 12 months postoperatively. After at least an 8-h overnight fast, a 75-g oral glucose load was taken. Blood samples were obtained by venipuncture before and at 30, 60, 90, and 120 min after loading. Fasting glucose (hexokinase method; Cobas Integra800; Roche, Basel, Switzerland), insulin/c-peptide (radioimmunoassay; Izotope, Budapest, Hungary), 1,5-anhydroglucitol (1,5-AG; enzymatic method; BM6010; JEOL Ltd., Tokyo, Japan), and HbA1c (high-performance liquid chromatography; Variant II Turbo; Bio-Rad, San Francisco, CA, USA) levels were evaluated preoperatively and every 3 months postoperatively. Homeostasis model assessments for insulin resistance (HOMA-IR) and β-cell function (HOMA-B) were calculated as previously described (30). INSindex was calculated as Δ insulin30min (μIU/ml)/Δ glucose30min (mmol/L) during the 75-g OGTT (39). Serum levels of high mobility group box 1 protein (HMGB1), soluble receptor for advanced glycation end product (sRAGE), and vascular endothelial growth factor (VEGF) were measured using ELISA Kits (MyBioSource Inc., San Diego, CA, USA, for HMGB1; R&D Systems Inc., Minneapolis, MN, USA, for sRAGE and VEGF) before and after islet infusion.
Assessment of Pancreatic Volume
Pre- and postoperative computed tomography (CT; Philips Medical Systems, Cleveland, OH, USA) scans were performed as a routine practice at the surgery department for a preoperative diagnosis and a postoperative drain management and complication evaluation. After administering intravenous radiocontrast media using the pancreatic protocol, 3-mm sliced CT images were obtained. Image data were transferred to a computer workstation (Rapidia, Infinitt, Seoul, Korea) for measurement of pancreas volume. On every CT slice, the borders of the pancreatic parenchyma and resection lines were outlined excluding tumors, cystic lesions, and dilation of the pancreatic duct and vessels. The volume of pancreatic parenchyma per slice was calculated as the pancreas area (in mm2) times the slice thickness (in mm). The total pancreas volume was the sum of the volume of each slice. The percent resected volume (PRV) was determined using the following formula: PRV(%) = [(preoperative pancreas volume - postoperative pancreas volume)/preoperative pancreas volume] χ 100% (42). Pancreatic volumes were assessed by both an endocrinologist and radiologist.
Statistical Analysis
Data are expressed as mean ± standard deviation or mean ± standard error of mean (in the figures) and were analyzed using SPSS Windows version 14.0 (IBM, Armonk, NY, USA). For comparisons between independent samples, Student's t-test was applied for continuous variables and Fisher's exact test/chi-square test were used for categorical variables. One-way analysis of variance (ANOVA) with Tukey's honestly significant difference (HSD) post hoc analysis was used to compare means of continuous variables among three groups. Changes in paired parameters were evaluated using paired t-test and repeated-measures ANOVA. Spearman correlation analysis was used to assess the association between two continuous variables. Multiple linear regression analysis was performed to measure the linear relationship between clinical outcomes and possible predictors. A value of p <0.05 was considered statistically significant.
Results
Subjects
Of the 31 patients screened, one refused to participate, and two had DM before surgery. Among the other 28, one experienced a postoperative complication, and two were lost to follow-up. Nine patients were enrolled into the ITx group after Px, and 10 patients were diagnosed as DM and started OAD (OAD group) during follow-up. None of the ITx group developed DM during the 1 year. These 19 subjects were included in the final analysis, but we also presented the data from the six patients who did not develop DM and therefore were not on OAD (non-OAD group) for a reference. Statistical analysis using the non-OAD group was performed only at baseline because inclusion of them in the analysis was not compliant with the primary endpoint, which is a comparison between ITx and OAD.
The patients' baseline characteristics are presented in Table 1. Age, sex ratio, body mass index (BMI), and waist circumference were not different between the ITx and the OAD groups. Plasma glucose levels while fasting and after oral glucose loading, as well as HbA1c levels, were slightly higher in the OAD group (p = 0.002, 0.019, and 0.012, respectively). The HOMA-IR was also higher in the OAD group than in the ITx group (p = 0.012), suggesting greater insulin resistance would be the cause of hyperglycemia in the OAD subjects. The most common tumors were mucinous cystic tumor and solid pseudopapillary tumor in the OAD and ITx group, respectively. PRV was significantly greater in the ITx group, which was a consequence of our decision to exclude patients from the ITx group if the volume of normal pancreatic tissue distal to the mass was small. As for the non-OAD group, preoperative OGTT 2-h glucose was lower than that of the OAD group (p <0.05). It could have resulted from a difference in insulin resistance according to the BMI, waist circumferences, and HOMA-IR (Table 1) and resulted in no development of DM.
Baseline Patient Characteristics

Continuous variables are expressed as mean ± standard deviation.
BMI, body mass index; ET, endocrine tumor; HDL, high density lipoprotein; HMGB1, high-mobility group box-1; HOMA-IR, homeostasis model assessment for insulin resistance; HOMA-B, homeostasis model assessment for β-cell function; IEQ, islet equivalent; INSindex, insulinogenic index; IPMT, intraductal papillary mucinous tumor; ITx, islet transplantation; MCT, mucinous cystic tumor; NA, not applicable; OAD, oral antidiabetic drugs; OGTT, oral glucose tolerance test; PRV, percent resected volume; SCA, serous cystadenoma; SPT, solid pseudopapillary tumor.
Values were determined by Student's t-test for continuous variables and Fisher's exact test for categorical variables between the OAD and the ITx groups.
Values were determined by ANOVA for continuous variables and chi-square test for categorical variables among all three groups. Superscript lower case letters indicate homogeneous subsets determined by post hoc analysis using Tukey's HSD test.
In the OAD group, the mean dose and duration of metformin treatment were 670 ± 395 mg/day and 303 ± 66 days. Vildagliptin was added in six OAD subjects, at a mean dose of 67 ± 26 mg/day for 246 ± 40 days. In the ITx group, islet isolation yield was 2,507 ± 970 IEQ/g of pancreas on the day of isolation. After overnight culture, the islet amount became 2,314 ± 1,707 IEQ/g of pancreas, and the stimulation index was 4.46 ± 5.83, which were comparable to those of previous reports (6). The mean quantity of islet grafts were 1,173 ± 839 IEQ/kg body weight of the subjects, which was obtained from a small volume of pancreatic tissue (mean 35.7 g). Total number of transplanted islets was 74,487 ± 52,924 IEQ. None of the participants in the ITx group had severe adverse events during or after the ITx.
Control of Blood Glucose Levels
Even with ITx and OAD treatment, HbA1c levels increased significantly at 1 year postoperatively by approximately 5% of the preoperative values (Fig. 1A, B). The final HbA1c was 6.2 ± 0.6% in the OAD group and 5.9 ± 0.3% in the ITx group (p <0.05 between the groups). At 3 months postoperatively, the percent change of HbA1c was greater in the OAD group than in the ITx group with marginal significance (p = 0.083). However, the percent change of HbA1c after this time was similar for the groups, likely because of adjustments in OAD after 3 months (Fig. 1B). Changes in fasting plasma glucose levels were similar for the two groups; they increased by 14% of preoperative values at 12 months.

Comparisons of glucose homeostasis throughout the 12-month study period after distal pancreatectomy. HbA1c levels (A), percent changes of HbA1c from the baseline (B), 1,5-AG (C), and AUC lucose during 75-g OGTT (D). White circles represent the OAD group, closed circles the ITx group, and inverted triangles in (A) and (D) the non-OAD group. The data are means with standard error. *p <0.05 using repeated-measures ANOVA; #p <0.05 using Student's f-test between the ITx and the OAD groups.
Changes in plasma glucose levels after oral glucose loading at 12 months were more prominent in the OAD group; they increased 38% from the preoperative value in OAD subjects (which was significant), but only 19% in the ITx group (which was not significant). In contrast to the HbA1c changes, the reduction in 1,5-AG (reflecting an aggravation of postprandial hyperglycemia) during the 12 months after surgery was greater in the OAD group (29% decrease, from 21.3 ± 5.1 to 15.0 ± 7.5 μg/ml) than the ITx group (8% decrease, from 28.7 ± 6.7 to 26.2 ± 6.3 μg/ml) (Fig. 1C). Area under the curve for the blood glucose levels (AUCglucose), another measure of glucose intolerance, was comparable for the two groups preoperatively and at 1 week postoperatively, but showed significant recovery only in the ITx group (Fig. 1D). As a result, AUCglucose at 12 months postoperatively in the ITx group was significantly lower than that in the OAD group (18,653 ± 3,558 vs. 21,402 ± 8,055 mg/dl120 min, respectively; p <0.05). This suggests that glucose intolerance and postprandial hyperglycemia after distal Px were prevented by ITx but not by OAD. AUCglucose in the non-OAD group increased at 1 week postoperatively and then recovered at 6 months.
Changes in Basal Insulin Secretion
Basal insulin secretion was evaluated by HOMA-B and fasting insulin/glucose ratios. From preoperatively to 12 months postoperatively, HOMA-B decreased from 118.6 ± 50.2 to 77.0 ± 42.9 (36% reduction) in the ITx group and from 124.5 ± 37.1 to 52.5 ± 27.5 (57% reduction) in the OAD group (Fig. 2A). Fasting insulin/glucose ratios showed a similar pattern to HOMA-B, with a 32% reduction at 12 months from the preoperative value in the ITx group (which was not significant) and a 55% reduction at 12 months from the preoperative value in the OAD group (which was significant) (Fig. 2B).

Comparisons of insulin secretion throughout the 12-month study period after distal pancreatectomy. HOMA-β-cell function (A), fasting serum insulin to glucose ratio (B), insulinogenic index (C), and individual changes of insulinogenic index between 6 and 12 months postoperatively (D). White circles represent the OAD group, closed circles the ITx group, and inverted triangles in (A) and (C) the non-OAD group (only shown for preOp and 12 months and are obscured by the closed circles). The data are means with standard error. *p <0.05 using Student's t-test between the ITx and the OAD groups; #p <0.05 using paired t-test.
Changes in Stimulated Insulin Secretion
Stimulated insulin secretion was evaluated with the INSindex. In the OAD group, the INSindex decreased significantly, from 25.6 ± 18.9 U/mol preoperatively to 10.4 ± 6.9 U/mol at 6 months postoperatively (a 51.8% reduction from preoperatively) and to 4.7 ± 3.7 U/mol at 12 months after surgery (a 54.8% reduction from 6 months) (Fig. 2C). By contrast, no significant change was observed in the ITx group during the 12 months after surgery (15.0 ± 4.5 U/mol preoperatively and 11.0 ± 8.2 U/mol at 12 months) (Fig. 2C). Differences in INSindex between the groups occurred after 6 months postoperatively. When tracings of the INSindex for each individual were examined, the tracings of most OAD subjects exhibited a substantial decrease from 6 to 12 months postoperatively, whereas the tracings of ITx subjects showed variable responses, including some patients with an increase in INSindex over this time period (Fig. 2D). Consequently, the INSindex at 12 months was significantly higher in the ITx group than the OAD group, but similar for the groups at baseline and 6 months postoperatively (Fig. 2C). Even when compared to the non-OAD group where a smaller part of pancreas had been resected, INSindex in the ITx group was well preserved. INSindex at postoperative 1 year in the ITx group was 77% of the baseline and that in the non-OAD group was 59% of the baseline.
Changes in Insulin Resistance
Unlike insulin secretion, preoperative insulin resistance was significantly higher in the OAD group than in the ITx group (Fig. 3). It is likely that the difference in preoperative glucose homeostasis between groups was at least partly due to this difference in insulin resistance. With OAD administration, HOMA-IR decreased by 35% from 3.70 ± 1.07 preoperatively to 2.40 ± 1.20 at 12 months (p = 0.028); however, HOMA-IR in the ITx group did not change. HOMA-IR was similar for the two groups at 6 and 12 months. This suggests that the difference in insulin secretion was the main contributor to the difference in glucose tolerance between groups at 12 months.

Comparison of insulin resistance using HOMA-IR. White circles represent the OAD group, closed circles the ITx group, and inverted triangles the non-OAD group (only shown for preOp and 12 months). The data are means with standard error. *p <0.05 using Student's t-test between the ITx and the OAD groups.
Factors Predicting Glucose Tolerance and Insulin Secretion After Distal Px
Although the effects of ITx and OAD after distal Px on HbA1c were similar, insulin release and glucose levels after an oral glucose load differed between groups. Multiple regression analysis was conducted to examine the relationships between these responses and potential predictors (Table 2). Preoperative glucose homeostasis, BMI, and PRV are known risk factors for postoperative DM in pancreatectomized patients (32,42). In our study, the change of AUCglucse at 12 months postoperatively (increase in AUCglucose) was positively correlated with age, preoperative HbA1c, and PRV (p = 0.047, 0.067, and 0.062, respectively) and was negatively correlated with the change in 1,5-AG at 1 month postoperatively and with ITx (p = 0.018 and 0.006, respectively). The change in INSindex over 12 months (increase in INSindex) was positively correlated with ITx (p = 0.052). The PRV might also contribute to the increased INSindex (p = 0.081).
We subsequently performed subgroup analysis of the ITx group to examine factors correlated with INSindex changes from 6 to 12 months postoperatively. During this period, some subjects in the ITx group showed no change or even an increase in the INSindex, contrary to the decrease noted in the OAD group (Fig. 2D). As shown in Figure 4, the recovery of INSindex from 6 to 12 months postoperatively was significantly correlated with islet yield and graft quantity (Fig. 4A). Peak serum HMGB1 and VEGF levels were related to the recovery of INSindex, but sRAGE levels were not (Fig. 4B). Serial changes in serum HMGB1, VEGF, and sRAGE levels are depicted in Figure 4C. Other factors, such as preoperative HbA1c, HOMA-B, INSindex, PRV, stimulation index of isolated islets, and HOMA-B at 1 month, were not correlated with the recovery of INSindex in the ITx group (data not shown).
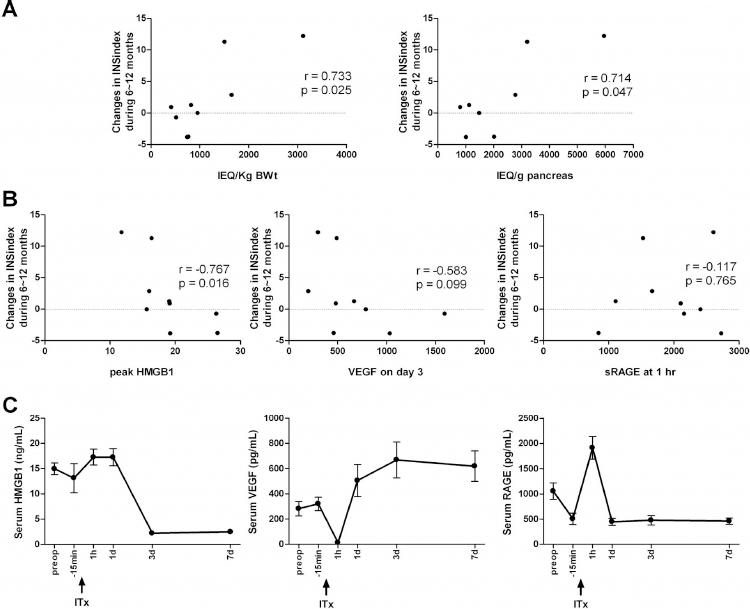
Factors associated with the recovery of insulin secretion in the ITx group. Islet yield per pancreas weight and graft amount per body weight (A) and serum markers involved in early graft loss (B). Serial changes of serum HMGB1, VEGF, and sRAGE in the ITx group during peri-islet infusion are presented in (C). The data in (C) are means with standard error. Spearman correlation analysis was applied in (A) and (B).
Predicting Factors for the Change of AUCglucose and INSindex at 1 Year Postoperatively: Results From Multiple Linear Regression Analysis

Abbreviations: AG, anhydroglucitol; AUCglucose, area under the curve for the blood glucose; BMI, body mass index; HbA1c, hemoglobin A1c; INSindex, insulinogenic index; ITx, islet transplantation; PRV, percent resected volume.
Increase at 1 year postoperatively, compared to the baseline.
Discussion
In this study, we directly compared 1-year changes in insulin secretory function of subjects who underwent preemptive ITx and those who were treated with OAD for postoperative DM after partial Px. We demonstrated that autologous ITx maintained insulin secretory capacity after 55% distal Px and thereby prevented aggravation of glucose tolerance and the development of diabetes within 1 year after surgery. By contrast, metformin with or without a DPP-4 inhibitor did not prevent the progression of insulin deficiency in postsurgical diabetes after distal Px.
During this period, control of blood glucose levels estimated by HbA1c was comparable between groups, with the HbA1c increasing in each group by 5% from baseline (Fig. 1B). Therefore, the administration of OAD successfully mimicked ITx with respect to mean serum glucose levels. Metformin seemed to improve insulin resistance in the OAD group (Fig. 3). Vildagliptin was expected to enhance insulin secretory function (1,2,4,34) because β-cell regeneration is well-known after partial Px in rodent models, although this has not been shown in humans (33). However, although preservation of glucose-stimulated insulin secretion occurred in the ITx group, it was not observed in the OAD group (Fig. 2). Consequently, glucose tolerance and variability were significantly impaired in the OAD group at 1 year, compared to the ITx group (Fig. 1C, D).
The difference in glucose tolerance between groups originated from a difference in glucose-stimulated insulin secretion, rather than basal insulin secretion (Fig. 2), although a longer follow-up may have also revealed improvement in basal insulin secretion (12). INSindex did not change significantly during the first year after surgery in the ITx group. In the OAD group, INSindex decreased by 51.8% during the first 6 months (Fig. 2C). High preoperative insulin resistance and its improvement by OAD would have contributed to this substantial reduction. After 6 months postoperatively, INSindex further decreased in the OAD group, although OAD were administrated from a very early stage of DM (Fig. 2C, D). During this period, insulin resistance was comparable between the two groups (Fig. 3). Therefore, the difference in INSindex between groups at 1-year follow-up was determined by whether or not the group was treated with ITx.
Nevertheless, long-term recovery of β-cell function after partial Px without ITx has been reported (31). Indeed, some subjects in our OAD group exhibited improved insulin secretion at 1 year postoperatively, compared to the secretion at the time they were diagnosed with diabetes (usually within 3 months after Px). Therefore, more evidence was sought regarding an association between ITx and insulin secretory function. Multiple linear regression analysis revealed that ITx was an independent factor for the changes in AUCglucose and the changes in INSindex during the first year postoperatively (Table 2). Additionally, we found that islet yield and graft quantity were significantly correlated with changes in INSindex after 6 months in the ITx group (Fig. 4A), when the difference between groups was most prominent (Fig. 2C, D). This finding is consistent with a recent report of the importance of islet yield in distal Px with ITx (11). In addition to these graft factors, two markers of islet damage, HMGB1 and VEGF, were also negatively associated with changes in INSindex after 6 months (Fig. 4B). By contrast, preoperative glucose homeostasis and PRV were not associated with changes in INSindex at 6 months after ITx. These findings indicate that ITx affected insulin secretory capacity and glucose tolerance after distal Px.
Although peak serum HMGB1 levels after ITx were closely associated with changes in INSindex after 6 months, peak levels of sRAGE, a putative decoy receptor of HMGB1 (18), were not related (Fig. 4B). HMGB1 has been suggested as a prototype of the emerging damage-associated molecular pattern molecule released from innate immune cells (40). It is released from damaged islets into the circulation soon after ITx in the liver (10,29), and it mediates initial events of early graft loss after ITx and β-cell apoptosis (19,21,29). Similar to the findings of Itoh et al. in patients receiving autologous ITx after total Px for chronic pancreatitis (10), serum HMGB1 reached peak levels at 1 h or at 1 day after ITx in our study (Fig. 4C) and were negatively correlated with ITx outcome (Fig. 4B). However, peak serum sRAGE levels, which showed one peak point at 1 h after ITx, were not associated with ITx outcome (Fig. 4C). It is likely that sRAGE was not sufficiently protective against the influence of HMGB1, especially around 1 day after ITx, which could have contributed to the lack of association between peak sRAGE and ITx outcome (Fig. 4B).
We also measured pre- and post-ITx serum VEGF levels. VEGF is a cytokine that plays a central role as a regulator of vascular formation in physiological and pathological conditions (41). Serum VEGF levels decreased prominently during islet infusion, and increased thereafter, reaching peak concentrations on day 3 after ITx (Fig. 4C). The initial suppression of serum VEGF was likely due to the large quantity of heparin used during angiography and islet infusion (13). The subsequent increase in VEGF was not associated with serum HMGB1 levels, although a previous report found that VEGF secretion from isolated human islets was correlated with HMGB1 release in vitro (35). The main cause of the induction of circulating VEGF after ITx is unclear, but HMGB1 or the islet graft itself is not likely a significant contributor, according to a previous study (10). Induction of circulating VEGF has also been found in other ischemic conditions, including acute myocardial infarction (13,17) and ischemic stroke (28); in these conditions, elevated VEGF levels were related with a poor prognosis, which is similar to our finding. However, the influence of circulating VEGF is not conclusive, as local overexpression of the VEGF gene enhanced islet vascular engraftment and preserved islet graft function in a mouse model (24).
Interestingly, the AUCglucose at 1 year seemed to be determined by the early postoperative 1,5-AG changes, which suggests the possibility of being able to predict the 1-year prognosis for glucose homeostasis by early postoperative glucose variability (Table 2). 1,5-AG is a naturally occurring 1-deoxy form of glucose, which is mostly reabsorbed by the kidneys after excretion and whose reabsorption is competitively inhibited by glucose (7). 1,5-AG is a marker for postprandial hyperglycemia in well-controlled diabetic patients (7,14).
Taking both the changes in insulin secretory function and insulin resistance into account, the primary contributor of the difference in glycemic control between the OAD and the ITx groups was insulin resistance preoperatively and insulin secretory function at 1-year follow-up. In spite of improved insulin resistance, progressive decline of insulin secretory function was not prevented in the OAD group. Conversely, ITx preserved insulin secretory function and glucose tolerance at 1 year postoperatively, independent of preoperative glucose homeostasis and PRV. Significant improvement in basal insulin secretion by ITx was not observed in this study. However, long-term follow-up may reveal a benefit on basal insulin secretion, as reported previously (12).
This study has several limitations. Initially, subjects in the ITx group were less insulin resistant and had a better glucose profile than those in the OAD group. Therefore, direct comparisons of insulin secretion between groups may be confounded, although the baseline values between the groups were not significantly different. Additional enrollment of subjects with prediabetes in the ITx group would be required to confirm the effects of ITx observed in this study. Furthermore, PRV was significantly higher in the ITx group because subjects whose lesion was located at the end of the pancreas tail were excluded from the ITx group. Usually, the quantity of pancreas resection is estimated in relation to a resection border, such as the portal vein. However, even with the same resection margin, there is significant individual variation of resected pancreas volume depending on the size and shape of the pancreatic tissues and tumors (42). Therefore, we quantitatively measured pancreas volume by CT scan and calculated the PRV to improve the accuracy and reliability of the resected volume assessment. Additionally, an insufficient dose of vildagliptin possibly contributed to the failure to maintain insulin secretory function in the OAD group. Preventive administration of vildagliptin before the development of DM, like preventive ITx, might be beneficial.
Nonetheless, we followed real-world practice after distal Px in this study design. In nondiabetic patients who underwent distal Px, transplantation of properly isolated islets from the normal part of the resected pancreas maintained glucose-stimulated insulin secretion and glycemic variability within the first postoperative year. Conversely, in nondiabetic patients who became diabetic after distal Px, metformin with or without vildagliptin improved insulin resistance, but neither restored insulin secretory function nor maintained glucose tolerance. This observation may be useful for the management of pancreatogenic DM after partial Px.
Footnotes
Acknowledgment
J.W.Y. analyzed and interpreted the data and wrote the manuscript. H.S.J. designed the study, analyzed and interpreted the data, and wrote the manuscript. J.Y.J., M.J.K., J.H.K., J.H.O., H.M.L., K.B.L., and S.K. contributed to the management of patients and acquisition of data. S.A.C. researched data, and J.H.K. contributed to the discussion. K.S.P designed the study, contributed to the discussion, and reviewed the manuscript. All authors approved the final version of the manuscript. H.S.J. is the guarantor of this work. This study was supported by a grant from the Innovative Research Institute for Cell Therapy (A062260) by the Ministry of Health and Welfare, Republic of Korea, and Novartis Inc. (CLAF237AKR02T). The authors declare no conflicts of interest.
