Abstract
Standardized islet characterization assays that can provide results in a timely manner are essential for successful islet cell transplantation. A critical component of islet cell quality is β-cell function, and perifusion-based assessments of dynamic glucose-stimulated insulin secretion (GSIS) are the most informative method to assess this, as they provide the most complex in vitro evaluation of GSIS. However, protocols used vary considerably among centers and investigators as they often use different low- and high-glucose concentrations, exposure-times, flow-rates, oxygen concentrations, islet numbers, analytical methods, measurement units, and instruments, which result in different readouts and make comparisons across platforms difficult. Additionally, the conditions of islet storage and shipment prior to assessment may also affect islet function. Establishing improved standardized protocols for perifusion GSIS assays should be an integral part of the ongoing effort to increase the rigor of human islet studies. Here, we performed detailed evaluation of GSIS of human islets using a fully automated multichannel perifusion instrument following various warm-up recovery times after cold storage that corresponds to current shipping conditions (8°C). We found that recovery times shorter than 18 h (overnight) resulted in impaired insulin secretion. While the effects were relatively moderate on second-phase insulin secretion, first-phase peaks were restored only following 18-h incubation. Hence, the biphasic profile of dynamic GSIS was considerably affected when islets were not allowed to recover for a sufficient time after being maintained in cold. Accordingly, while cold storage might improve islet cell survival during shipment and prolong the length of culture, functional assessments should be performed only after allowing for at least overnight recovery at physiological temperatures.
Keywords
Introduction
Patients with type 1 diabetes (T1D) 1 require lifelong administration of exogenous insulin, and even intensive insulin replacement therapy can control blood glucose levels only to certain extent 2,3 . Despite considerable technological improvements, including continuous glucose monitoring and insulin infusion systems 4 , diabetes-related complications are still common especially in long-time patients 5 . This is due to the fact that T1D is a chronic disease resulting from a progressive destruction of pancreatic β-cells, and their lack eventually leads to unavoidable metabolic abnormalities 6,7 . Because in T1D, β-cells are selectively destroyed by an autoimmune attack, cell replacement strategies offer attractive treatment options. Clinical trials clearly demonstrated that islet transplantation offers a viable option for patients with severe forms of T1D and can successfully treat the disease 8 –11 . However, due to the limited number of available donor organs and the requirement for life-long immunosuppression, transplantation of pancreatic islet cells is currently available only to patients with brittle diabetes, i.e. the most severe forms of T1D refractive to any other treatment options 9,12 –15 .
The success of the Edmonton protocol, improvements made in the islet isolation procedure, and the application of low-dose immunosuppressive regiments led to improved long-term islet graft survival and insulin independence 8,16 –20 . Islet cell transplantation is now approved as “standard-of-care” in Canada, Australia, and countries of the European Union, and will most likely achieve the same status in the United States for a selected cohort of T1D patients 9,15,21 . Nevertheless, challenges persist and are well documented. Loss of islet mass and/or function prior to transplantation constitutes a major obstacle 22,23 . To minimize this loss, which is mainly due to the hypoxia caused by oxygen diffusion limitations in the avascular isolated islets 24 , islets are shipped under cold conditions (8°C) from the isolation centers to the destination clinical or research facilities 25 . Several studies have shown that cold culture can preserve islet function 26 –29 ; however, cooling and hypothermia are known to inhibit not only glucose utilization and oxidation, but also glucose-induced Ca2+ rise, Ca2+-activated exocytosis, and glucose-stimulated insulin secretion (GSIS) 30,31 , and it is unclear what recovery warm-up times are needed to fully restore islet function before assessment and transplant.
To ensure that the islet isolation process is consistent and reproducible and the final product is safe, pure, and potent, strict control is paramount 32 . Several qualitative and quantitative tests are used to verify the final human islet cell product 32 –34 . Pretransplant criteria that must be met include the determination of the total islet cell mass (>5,000 islet equivalents (IEQ)/kg recipient body weight), total pellet volume of the final preparation (<7 ml of tissue), islet cell purity (>30%), viability (>70%), and function (GSIS or static incubation index >1) 35 . Additional assays that evaluate β-cell fractional viability and content as well as function are available. They can offer meaningful information predictive of in vivo function 36 –38 and are essential to ensure progress in our understanding of islet function and pathogenesis of T1D 39,40 . However, due to the lack of standardized protocols, the need for highly specialized equipment, and the time required to obtain results, they are not currently utilized as lot release criteria.
Perifusion studies, which have been developed in the late 1960s 41 –43 , challenge islets with glucose and allow the dynamic measurement of GSIS. Although not part of currently accepted final islet product release criteria, such studies represent the most complex in vitro assay to assess the quality and function of isolated pancreatic islets and provide considerably more physiological data than those obtainable from static GSIS and corresponding stimulation indices (SIs). Improved equipment and analytical techniques now allow the quantitative assessment of insulin release kinetics with customizable temporal resolution and under fully controllable incoming concentrations of glucose and/or other secretagogues of interest 36,37,44 –49 . Dynamic perifusion studies are now routinely used to assess the quality and function of islets 46,50,51 , and microfluidic chip technologies make possible even the quantitative monitoring of single islet insulin secretion with high time resolution 52,53 . However, various nonstandardized systems and protocols are being used including variations in (i) low- (basal) and high- (stimulating) glucose concentrations (e.g., 3 mM→11 mM, 5.6 mM→16.7 mM, 2.8 mM→28 mM, and others), (ii) exposure times to stimulating glucose (typically, 10 to 30 min), (iii) flow rates (e.g., 30 to 1,000 μl/min), (iv) oxygen concentrations (from 21% atmospheric up to 95%), (v) quantity of islets per channel, (vi) analytical methods used to quantify insulin concentrations (enzyme-linked immunosorbent assay [ELISA], radioimmunoassay, immunochemiluminometric assay, and others), (vii) measurement units used to express results (pg/IEQ/min, ng/100islets/min, mU/l, μU/ml/ngDNA, μU/ml/ngDNA, relative values compared to baseline, percent insulin content, and others), and (viii) perifusion systems utilized. As a first step toward more informative and standardized perifusion assays, we have previously investigated the suitability of various glucose steps utilized to assess islets 37 .
Variability in assay methodology is further compounded by the fact that islets are now regularly shipped under cold storage conditions 25 , which is followed by various warm recovery times before assessment. With a limited number of studies addressing this subject, the effects of cold storage on islet preservation and function are poorly understood 29 , and the influence of the length of subsequent recovery at physiological culture conditions on the assessment of islet function remains unclear. The aim of the present study was to investigate the effects of cold storage and subsequent culture (recovery) at physiological conditions (37°C) on the dynamic GSIS assessment of human islet function. To evaluate these parameters, freshly isolated human islets from several preparations were exposed to cold storage conditions and allowed to recover for 2, 6, and 18 h at physiological culture conditions 25 . For increased comparability, perifusion studies were performed in parallel for all samples from each preparation using a fully automated perifusion machine with software-controlled customizable input for multiple parallel channels at high-resolution sample collection (every minute).
Materials and Methods
Human Islets
Human pancreatic islet samples were procured from the cGMP (current Good Manufacturing Practice) Human Islet Cell Processing Facility at the Diabetes Research Institute (DRI), University of Miami (UM), Miami, FL, USA. Human islets were isolated using a modification of the automated method according to the protocol standardized as part of the Clinical Islet Transplant (CIT) consortium and under the exemption issued by the UM Institutional Review Board 15,54 . Human pancreata were obtained from deceased multiorgan donors with the consent for transplant and/or research obtained from the family or next of kin of the donor by a responsible Organ Procurement Organization at the time of organ donation. All samples utilized in this study were from normal, nondiabetic donors; corresponding characteristics are summarized in Table S1. Data were collected using standard forms utilized by the cGMP Cell Processing Facility per previously established standard operating procedure (SOP) for human islet cell isolation.
Islet Culture and Perifusion
Following isolation and culture for 48 h, islet samples containing 350 to 400 IEQ were collected and prepared as described below. Some samples were maintained under standard culture conditions of 37°C in PIM(R)® media (Prodo Laboratories, Aliso Viejo, CA, USA) in accordance with the Integrated Islet Distribution Program (IIDP) SOPs for islet culture and preparation for shipment 25 ; others were exposed to the conditions of cold storage following the IIDP SOP for packaging and cold shipping of human islets 55 . Briefly, following purification, islet cells were washed and resuspended in PIM(R)® media supplemented with 1% PIM-G (Prodo Laboratories), 5% AB serum (Gemini Bioproducts, Sacramento, CA, USA), and 1% ciprofloxacin (Cellgro Inc., Manassas, VA, USA). Islets of >70% purity were cultured at a density of 20,000 IEQ per non-culture-treated T-175 cm2 tissue culture flask (Corning, Tewksbury, MA, USA) in a standard 37°C incubator with 5% CO2 and atmospheric O2 for 48 h. Up to six different samples containing 350 to 400 IEQ each were prepared. Two were placed back in the 37°C incubator (with 5% CO2 and atmospheric O2) for overnight culture. The others were packaged in 30 ml shipping bottles (Fisher, Miami, FL, USA), placed in an inner corrugated cardboard shipping box (7″×5″×5″) (Uline, Pleasant Prairies, WI, USA) surrounded with six 8°C cold packs (Uline), placed in an insulated shipping container (Uline) with two CryoPak pouches (Edison, NJ, USA), four 8°C cold packs (Uline), and two frozen ice packs (Uline) 25 . Temperature-monitoring devices were utilized to assure that the temperature in each box was within the acceptable limits. Each sample was packaged in a different insulated shipping container and stored as such. Boxes were then opened, and their sample content transferred to a standard 37°C incubator with 5% CO2 and atmospheric O2 after 6, 18, and 22 h of cold storage for 18, 6, and 2 h recovery at physiological culture conditions, respectively. Perifusion experiments (dynamic GSIS) were performed using a PERI4-02 machine (Biorep Technologies, Miami, FL, USA) that allows parallel perifusion for up to 12 channels (up to 4 independent conditions) as described before 36,37 . For each experiment, 100 human IEQ from the same islet isolation batch were handpicked and loaded in Perspex microcolumns between 2 layers of acrylamide-based microbead slurry (Bio-Gel P-4, Bio-Rad Laboratories, Hercules, CA, USA) by the same experienced operator. Perifusion buffer containing 125 mM NaCl, 5.9 mM KCl, 1.28 mM CaCl2, 1.2 mM MgCl2, 25 mM HEPES, and 0.1% bovine serum albumin (Sigma-Aldrich, St. Louis, MO, USA) at 37°C with selected glucose or KCl (25 mM) concentrations was circulated through the columns at a rate of 100 μl/min. After 60 min of washing with low-concentration glucose solution to stabilize the samples, islets were stimulated as follows: 10 min low-concentration glucose (3 or 5.6 mM), 20 min high-concentration glucose (11 or 16.7 mM), 15 min low glucose, 10 min KCl (25 mM), and 10 min of low glucose. Samples of 100 μl were collected every minute from the outflow column tubing in an automated fraction collector designed for a multi-well plate format. Islet samples as well as glucose and KCl solutions were maintained at 37°C in a built-in temperature-controlled chamber; the perifusate in the collection plate was maintained at <4°C to preserve the integrity of the analytes during perifusion. Samples were stored at –20°C until analysis. Insulin concentrations were determined using commercially available human insulin ELISA kits (Mercodia Inc., Winston Salem, NC, USA) and converted to μg/l using the formula provided by the manufacturer (1 µg/l = 23 mU/l). Because accurate assessment of islet mass (IEQ) is challenging, to account for possible differences among parallel channels, values were adjusted by up to 30% based on the response to KCl as described before using the area under the curve (AUC) in each column for normalization 36,37,48,49,56,57 . All responses are scaled to 100 IEQ.
Statistical Analyses
All statistical comparisons were done using Prism 8.3 (GraphPad, La Jolla, CA, USA). Data used here are averages of at least three samples for each condition. Dynamic stimulation indices (dSI) were calculated as the ratio between average insulin secretions during high- and low-glucose stimulation. Average stimulated insulin secretion and AUCs were calculated for the entire 20 min of high glucose (minutes 12 to 32 to account for the tubing delay) as well as for phase 1 (first 7 min) and 2 (last 13 min) separately.
Results
Islets were isolated following SOPs used for clinical islet transplantation 35 . For the present experiments, samples of 350 to 400 IEQ were collected after 48 h of culture in a standard 37°C tissue culture incubator 25 . Samples that served as control were maintained at all times in the standard 37°C tissue culture incubator; all others were packaged and stored following the IIDP SOP for cold shipment (storage) of human islets 25 . They were then opened at different times and moved to standard 37°C culture conditions so as to allow for recovery period of 2, 6, and 18 h prior to dynamic GSIS perifusion assessment. All samples were perifused at the same time using a fully automated programmable multichannel perifusion apparatus that allowed for direct parallel comparison of the responses for up to 12 parallel channels with customizable inflow and collection conditions. As before, the standard perifusion protocol of our center was used: preincubation in 3 mM basal glucose solution (G3), followed by 11 mM high glucose (G11) for 20 min, G3 low glucose for 15 min, and KCl for 10 min. To fully capture both first- and second-phase insulin secretions, outflow samples were collected every minute 36,37,48,49 . This stepwise glucose stimulation challenge results in a classic biphasic insulin response 36,37 ; a corresponding average profile obtained by us from a large number of human islet samples (n = 55) is included as a gray line in Fig. 1 and is shown in detail in Figure S1.
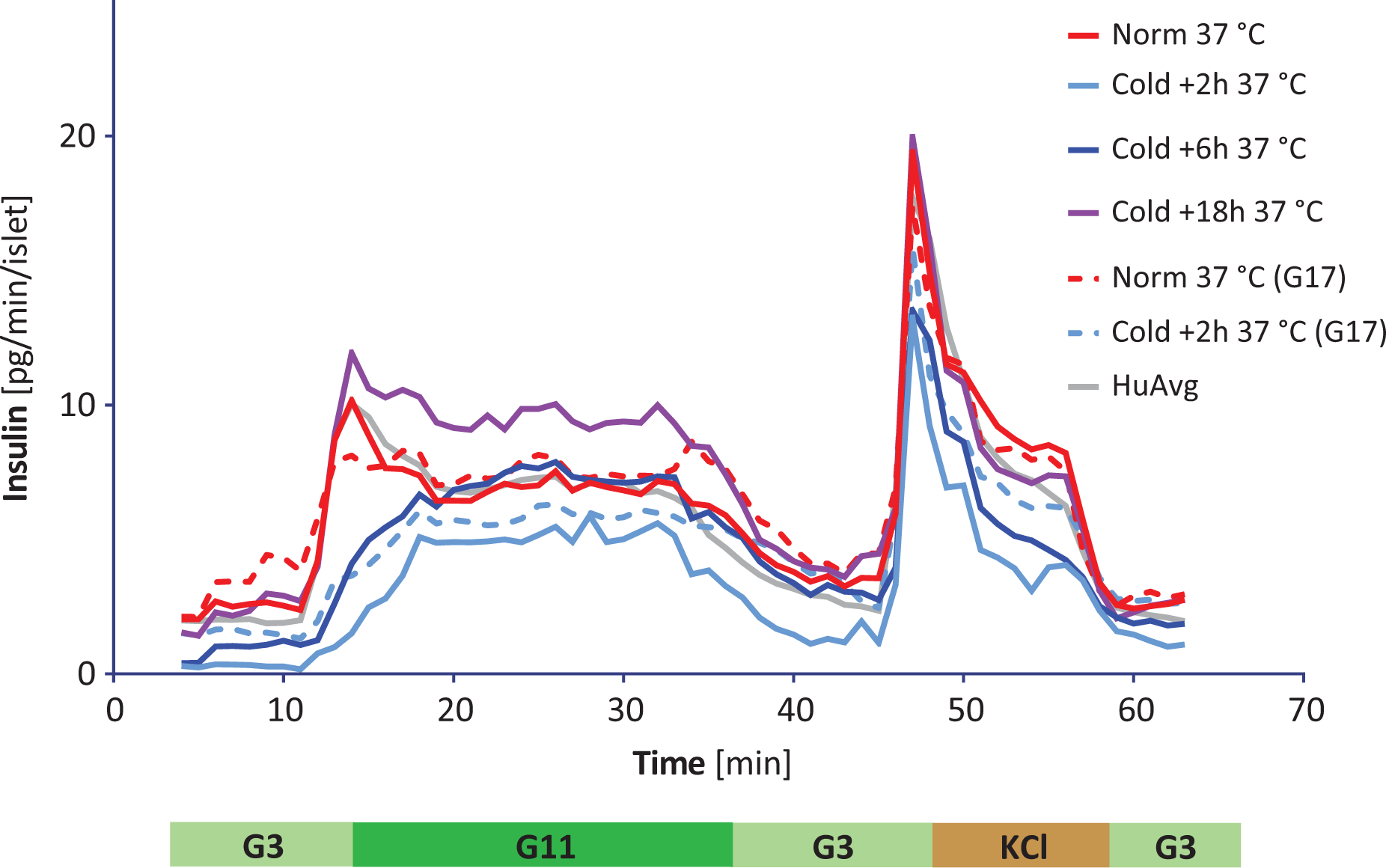
Glucose-stimulated insulin secretion (GSIS) profile of human islets following various warm-up recovery times. Dynamic GSIS obtained in perifusion studies with human islets after various warm-up recovery times (2, 6, and 18 h) following standard cold shipping and culture condition (8°C). Continuous lines indicate perifusion with our standard protocol (G3→G11→G3), while dashed lines indicate perifusion with glucose levels used by the HIPP IIDP Islet Assessment Center (G5.6→G16.7→G5.6). Red lines denote data obtained with control islets stored at 37°C for the entire time; blue to purple lines denote data after various recovery times as indicated; the gray line denotes the average of all data obtained by us so far with human islets (G3→G11, n=55; Figure S1), and it is included for comparison purposes. For clarity, error bars have been omitted; see Figure 2 for a scatter plot indicating range and number of data points for each condition.
Results following various recovery periods after cold (8°C) storage back to physiological temperature (37°C) are shown in Fig. 1. They indicate that recovery time periods of less than 18 h (overnight) resulted in somewhat impaired insulin-secreting ability when compared to those of control islets maintained at 37°C at all times. Pictures illustrating representative islet morphologies are included in Figure S2. While effects were relatively moderate on second-phase insulin secretion, which exhibited an essentially normal profile after a 6-h recovery period, the first-phase peak was restored only after the 18-h recovery period. Average 11 mM high-glucose (G11) stimulated insulin secretions were 4.42, 6.54, and 9.76 pg/min/IEQ for the 2-, 6-, and 18-h recovery time periods, respectively, versus 7.33 pg/min/IEQ for the control islet samples. While differences were relatively small during the second-phase plateau secretion (5.15, 7.30, and 9.48 vs 6.91 pg/min/IEQ), they were quite substantial for the first-phase secretion (calculated as the average secretion during the first 7 min of G11 high-glucose challenge). These were 3.06, 5.13, and 10.27 pg/min/IEQ for the 2-, 6-, and 18-h recovery period compared to 8.12 pg/min/IEQ for the control. Corresponding AUCs also show the same trend with the first-phase AUCs of the 2 and 6 h and the overall AUC of the 2-h recovery conditions being significantly different from those of the control—they are summarized in Fig. 2. Note that the control profile obtained here (n = 5, red line) agrees well with our average profile obtained on a much larger sample (n = 55, gray line), confirming the representative nature of the experiments discussed here. Because basal secretions were also relatively low following 2- and 6-h recovery periods, corresponding dSIs were higher, i.e. 14.45, 7.36, and 4.36 for the 2-, 6-, and 18-h recovery periods, respectively, compared to 3.00 for the control sample, highlighting the difficulty of using such SI-based comparisons to characterize the function of islet cells 37 .

Areas under the curve (AUCs) for glucose-stimulated-insulin-secretion profiles obtained under the different conditions as indicated and separated into phase 1 and 2, respectively. AUCs for the dynamic perifusion profiles shown in Fig. 1 were calculated in GraphPad Prism separately for first- (12 to 19 min) and second-phase (20 to 32 min) secretions and are shown in red and orange, respectively, as scatter plots as well as mean ± SE in pg/islet equivalent (IEQ) units. Asterisks denote significant differences versus the normal (37°C) control (one-way analysis of variance followed by Dunnett’s multiple comparison test; * P < 0.05, *** P < 0.001).
Because a somewhat different glucose step (G5.6→G16.7 vs G3→G11) is used for the perifusion-based assessment of human islets by the Human Islet Phenotyping Program (HIPP) of the IIDP 51,58 , we also evaluated insulin secretion profiles obtained with this protocol (G5.6 → G16.7) for control and 2-h recovery period conditions. Overall, profiles were quite similar with baselines slightly more elevated, normal first-phase responses slightly more diminished, and second-phase plateaus quite similar (Fig. 1, dashed lines)—confirming again that GSIS responses after a 2-h recovery time period are diminished. Average G16.7 high-glucose insulin secretions were 5.46 vs 7.61 pg/min/IEQ for the 2-h sample vs control, whereas first-phase rates were 4.69 vs 7.85 pg/min/IEQ for the 2-h sample vs control. Due to the higher baselines, dSIs were lower in this set of experiments, i.e. 2.29 and 3.55 for the 2-h and control samples, respectively.
Discussion
Methods for large-scale isolation of adult human pancreatic islet cells have significantly improved as a result of advances made in the process 10,16,18,35 . As part of the CIT collaborative work, efforts were recently undertaken to standardize the assessment methods utilized to release final islet products for transplant 35 . This resulted in the definition of common criteria for the manufacture and testing of islet cell products. These include assays for the assessment of islet cell viability by fluorescein diacetate/propidium iodide (FDA/PI), identity by dithizone (DTZ) staining, purity by DTZ and endotoxin, safety by 14-day sterility testing, and biological potency as determined by static GSIS with results expressed as an SI 35 . Several assays, such as islet cellular composition and β-cell fractional viability 38 and high-throughput dynamic perifusion studies 36,46 , were not included in the standardized lot release criteria. Although these assessment methods have been around for quite some time and offer critical information predictive of islet function in vivo, they were omitted due to the need for specialized equipment, lack of standardized methods, and the time required to obtain the results. Nevertheless, these assays remain critical for assessing islet function/quality and for extending our knowledge and understanding of β-cell function and pathogenesis of T1D.
Because dynamic GSIS perifusion assays are the most complex and informative in vitro assay that can be used to characterize the functionality of isolated human islets, it is critical to establish reproducible and standardized experimental conditions that allow accurate assessment. Nevertheless, few efforts have been made to do so or to investigate what conditions provide the most functionally relevant information 37 . Accurate quantitative assessments of the mass and quality of isolated pancreatic islets that are predictive of biological activity in vivo are of obvious interest for anyone working with islets and are particularly important for clinical applications, as they determine the success of transplantation. We, as well as others, have already shown that the insulin secretion dynamics of human and rodent islets are considerably different 37 , and it is now well recognized that studies with human islets are needed because of their clinical relevance 39 . This, however, is challenging since there is always considerable variability among human samples, and three- to fivefold differences in responses are not uncommon 37,50 .
Here, we found that the biphasic profile of dynamic GSIS was considerably affected when islets were not allowed to recover at physiological culture conditions for a sufficiently long period of time following cold storage and/or shipment (Figs. 1 and 2). Insulin secretion in response to a sharp increase in glucose has been long known to be biphasic: a transient first-phase peak followed by a sustained second phase 36,59 –61 , and this has been confirmed in perifusion studies starting in the late 1960s 41,43 . While in vivo blood glucose levels might never increase fast enough to induce such a biphasic secretion following oral food intake, the pattern is a sensitive indication of an adequate β-cell function 62 . A sharp first phase may not be fully evident after an oral challenge (as the corresponding increase in plasma glucose is not steep enough), but it is clearly present in intravenous glucose tolerance tests 63,64 or hyperglycemic clamp studies 65 , and it is well reproduced in perifusion studies. The ability of pancreatic β-cells to generate a rapidly increasing insulin profile is important to restrain hepatic glucose production 62 , and its physiological relevance is well illustrated by the accelerated loss of the first-phase insulin response found as a first sign in those progressing toward T1D 66 –69 as well as T2D 70,71 . Hence, presence of an adequate first-phase response is important, and its lack has long-term physiological consequences 36,72 .
In the present study, a dynamic perifusion system was used to evaluate the influence of various warm-up recovery times on islet function following cold shipment as standardized by IIDP 58 . In contrast to static GSIS, which is not a good predictor of in vivo islet function 18,50,73 –75 , the dynamic assay allows detailed evaluation of the biphasic insulin secretory pattern. Despite SIs being widely used for islet assessment in both research and clinical applications, there is no uniform standard, and various low- to high-glucose steps are used without clear evidence as to which one provides better characterization of islet function. For example, static GSIS involving a large G2.8→G28 step is used for clinical (transplantation) assessment 15,35 as well as for islet assessment by IIDP. Other versions (e.g., G5.6→G16.7 50,58 or G3.3→G16.7 75 ) are also used. For dynamic perifusion, our group (DRI, Miami) has been using G3→G11 36,37,49 , whereas G5.6→G16.7 is used by Powers and coworkers 50 , including the centralized HIPP/IIDP 58 , and different, more or less arbitrary steps are also used. In addition to differences in the glucose step, there are also differences in exposure times to high glucose, flow rates, oxygen concentrations, the islet numbers per channel, analytical methods used to quantitate insulin concentrations, measurement units used to express readout, and perifusion systems utilized, which all make comparisons across platforms difficult. Our results here show that the first-phase response is significantly suppressed following insufficient recovery times and the biphasic pattern is fully recovered only after >18 h (Figs 1 and 2). Second-phase GSIS and even KCl-induced membrane depolarization are impaired at the shortest recovery times, likely due to the time needed to recover a normal physiological metabolic rate following storage at cold (8°C). The islet group evaluated after a warm-up period of 18 h consistently displayed a slightly increased GSIS, possibly indicating a rebounce as well as improved preservation of islet function by reversibly slowing islet metabolism compared to continuous culture at physiological temperature (37°C) 76 . Hence, we have shown that with the cold shipment conditions currently utilized, even the recovery culture times used can have a significant impact on the assessment of islet function.
In conclusion, while cold storage might prolong the culture time of isolated islets, there is clear evidence that functional assessments should be performed only after allowing for at least overnight recovery at physiological temperatures. As demonstrated by the CIT consortium, standardization of the islet isolation process and assessment assays led to the manufacture of 75 clinical islet cell products that met the prespecified characteristics of safety, purity, potency, and identity and were successfully transplanted into 48 subjects 35 . It can be argued that the same harmonization strategies should be applied to the assessment of islet function by dynamic perifusion among different centers. As part of the effort to increase the rigor of human islet studies 39 , establishment of improved, standardized, and highly reproducible protocols for the dynamic perifusion GSIS assay is essential to allow for meaningful cross-platform analyses of results and has the potential to impact the use of human isolated islets for both clinical applications and research.
Supplementary Material
Supplementary_Tables - The Effect of Recovery Warm-up Time Following Cold Storage on the Dynamic Glucose-stimulated Insulin Secretion of Isolated Human Islets
Supplementary_Tables for The Effect of Recovery Warm-up Time Following Cold Storage on the Dynamic Glucose-stimulated Insulin Secretion of Isolated Human Islets by Oscar Alcazar, Alejandro Alvarez, Camillo Ricordi, Elina Linetsky and Peter Buchwald in Cell Transplantation
Footnotes
Author Contributions
PB, EL, CR, and OA conceived the study; OA and AA performed the islet culture and perifusion experiments; OA and PB analyzed the data; PB and EL wrote the main part of manuscript; OA, AA, and CR contributed to the manuscript. All authors read and approved the final manuscript.
Ethical Approval
Human pancreatic islet samples were procured from isolations performed at the cGMP Cell Processing Facility at the Diabetes Research Institute (DRI), University of Miami (UM, Miami, FL, USA). The islet isolation was done according to the common batch record established for the CIT consortium and under the exemption issued by the UM IRB.
Statement of Human and Animal Rights
Human Rights: Human pancreatic islet samples were procured from isolations performed at the cGMP Cell Processing Facility at the DRI, UM (Miami, FL, USA).
Statement of Informed Consent
Human pancreata were collected from deceased multiorgan donors for whom consent for transplantation and/or research was obtained by accredited OPOs from the donors’ families or next of kin, at the time of donation.
Declaration of Conflicting Interests
The author(s) declare that they have no competing interests with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported with funding from National Institute of Diabetes and Digestive and Kidney Diseases (1UC4DK104208).
Supplementary Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
