Abstract
Stem cell therapy is currently being studied with a view to rescuing various neurological diseases. Such studies require not only the discovery of potent candidate cells but also the development of methods that allow optimal delivery of those candidates to the brain tissues. Given that the blood-brain barrier (BBB) precludes cells from entering the brain, the present study was designed to test whether hyperosmolar mannitol securely opens the BBB and enhances intra-arterial cell delivery. A noninjured normal canine model in which the BBB was presumed to be closed was used to evaluate the feasibility and safety of the tested protocol. Autologous adipose tissue-derived pericytes with platelet-derived growth factor receptor β positivity were utilized. Cells were administered 5 min after mannitol pretreatment using one of following techniques: (1) bolus injection of a concentrated suspension, (2) continuous infusion of a diluted suspension, or (3) bolus injection of a concentrated suspension that had been shaken by repeated syringe pumping. Animals administered a concentrated cell suspension without mannitol pretreatment served as a control group. Vital signs, blood parameters, neurologic status, and major artery patency were kept stable throughout the experiment and the 1-month posttreatment period. Although ischemic lesions were noted on magnetic resonance imaging in several mongrel dogs with concentrated cell suspension, the injection technique using repeated syringe shaking could avert this complication. The cells were detected in both ipsilateral and contralateral cortices and were more frequent at the ipsilateral and frontal locations, whereas very few cells were observed anywhere in the brain when mannitol was not preinjected. These data suggest that intra-arterial cell infusion with mannitol pretreatment is a feasible and safe therapeutic approach in stable brain diseases such as chronic stroke.
Keywords
Introduction
Stroke remains the leading cause of death and disability in developed countries (28,32). Numerous attempts have been made to develop neuroprotective therapies that interrupt the ischemic cascades (9). However, no clinical trials have demonstrated the efficacy of neuroprotective therapies, despite promising results from both in vitro and animal experiments (1). Stem or progenitor cells have enormous potential for neuroregeneration (30,34), and cell-based therapies for treating cerebral ischemia have been researched using various sources, including bone marrow (8), peripheral blood (17,18), cord blood (21), and adipose tissue (14,20). In particular, adipose tissue is known to harbor many stem cell populations, such as pericytes, vascular progenitor cells, and mesenchymal stem cells (7,24,25).
Pericytes, which are characterized by their morphology and close contact with endothelial cells (17,19,23), can differentiate into multiple mesodermal lineage cells (10,40). Moreover, they are a subset of tissue-resident stem cells with the potential to direct vascular neurogenesis (7,10,17) and have been regarded as one of the most promising cell sources for neurovascular diseases. Although specific markers are not available to define the pericyte, α-smooth muscle actin, platelet-derived growth factor receptor β (PDGFRβ), and chondroitin sulfate proteoglycan NG2 have been used to identify pericytes (10,29,35,41,42). The results of our recent work suggest that pericytes that are positive for PDGFRβ (PDGFRβ+) can function as multipotent stem cells for tissue repair (17).
Despite the discovery of potent cell sources, the clinical application of cell-based therapy is limited by two major obstacles. First, several weeks are required to expand autologous stem cells isolated from a patient's tissue in order to obtain a sufficient number for delivery. On that timescale, acute stroke evolves to the subacute stage, with the loss of signals for stem cell recruitment and closing of the blood-brain barrier (BBB) (15,33,44). Therefore, the ability to open the BBB would be a prerequisite for placing the cells into the brain tissue. Second, intra-arterial administration has the advantage of improved physiological engraftment relative to other routes of administration, but restricted blood flow through the occluded artery may greatly diminish or even prevent the delivery of stem cells to the brain parenchyma. Although arterial infusion through an intact artery—even on the contralateral side—may be more effective for delivery in this situation, it is questionable whether the unilateral injection of stem cells would achieve effective bilateral engraftment.
The present study was conducted to test the feasibility and safety of a cell delivery technique for clinical translation, specifically, intracarotid arterial delivery following mannitol-induced BBB opening in a moderate size animal model (3,12,45). Autologous, adipose tissue-derived PDGFR+ (AT-PDGFR+) cells were chosen based on our previous observations suggesting that PDGFRβ+ cells can be harvested more easily and abundantly from adipose tissue than from other sources (17).
Materials and Methods
Animals
This study was designed to evaluate the feasibility and safety of a cell delivery procedure in 12 mature (age: 24-36 months), morphologically normal, male mongrel dogs of similar sizes and weights (range: 22-28 kg). Each of the dogs was designated with its own admission number by the Animal Care and Use Committee of Seoul National University Hospital. All animals were cared for according to routine procedures approved by the Institutional Animal Care and Use Committee of Seoul National University Hospital (SNUH-IACUC number: 12-0046). Three dogs underwent cell injection without mannitol pretreatment (control condition), and nine dogs were subjected to various cell delivery techniques following pretreatment with a hyperosmolar mannitol solution. The latter nine dogs were subjected to safety monitoring with blood tests, imaging, and neurologic tests.
Preparation of Autologous AT-PDGFRβ+ Cells
Canine adipose tissue was obtained directly from individual dogs. As described previously in humans (17,24,25,31), stromal vascular fractions were first acquired from canine subcutaneous adipose tissue by digestion with 0.075% collagenase type I (Invitrogen, Carlsbad, CA, USA) for 1 h at 37°C. After removing the mature adipocyte fraction from the stromal vascular fractions by centrifuging at 1,200 χ g for 10 min, the remaining fractions were treated with red blood cell lysis buffer (Sigma-Aldrich, St. Louis, MO, USA) and then filtered through a 100-μm nylon mesh (Anping County Bolin Metal Wire Mesh Co., Ltd., Hengshui, China) to remove the erythrocyte debris. The samples were recentri-fuged at 1,200 χ g for 10 min. The obtained cells were incubated with endothelial growth medium (EGM-2; Clonetics, Walkersville, MD, USA) for 1 h and then maintained in Dulbecco's modified Eagle'smedium and nutrient mixture F-12 (Sigma-Aldrich) plus 5% fetal bovine serum (Sigma-Aldrich) plus N2 supplement without growth factors (Gibco, Invitrogen, Carlsbad, CA, USA), which allowed the cells to maintain their PDGFRβ positivity, as shown in our previous study (17). The PDGFRβ positivity was identified by flow cytometry with a FACScan device (Becton Dickinson, San Diego, CA, USA) using mouse monoclonal anti-human PDGFRβ-phycoerthyrin (BD Biosciences, San Jose, CA, USA). The percentage of PDGFRβ+ cells, which was estimated to be about 40% in the original cell suspensions, increased to 83% in the enriched cell suspensions (Fig. 1A-C). Beginning 3 days before transplantation, AT-PDGFRβ+ cells were harvested using 0.25% trypsin (Sigma-Aldrich) and cocultured with green fluorescent protein-labeled lentivirus (107 plaque-forming units/ml medium; Seoulin Bioscience, Gyeonggi-do, Korea) for 3 days. Reporter gene expression was examined using fluorescence microscopy and flow cytometry prior to transplantation (Fig. 1D, E). The percentage of PDGFRβ+ cells infected with GFP-lentivirus was found to be 98% (Fig. 1E).

Isolation and culture of PDGFRβ+ cells from canine adipose tissue and flow cytometry analysis. (A) The original cell suspensions were stained with anti-PDGFRβ antibodies (y-axis) and run on a flow cytometry system. The percentage of PDGFRβ+ cells was estimated to be 43%. (B) The PDGFRβ+ cells were sorted from canine adipose tissue and seeded in culture. The cells formed a monolayer of homogeneous bipolar, spindle-like cells at 2 weeks. (C) The strong expression of PDGFRβ+ without differentiation into mature pericytes was achieved using Dulbecco's modified Eagle's medium/F-12 plus 5% fetal bovine serum plus N2 supplement without growth factors. (D) Fluorescence showing the GFP-labeled lentivirus (lenti-GFP)-transfected AT-PDGFRβ+ cells. (E) The percentage of sorted PDGFRβ+ cells infected with lenti-GFP was determined by flow cytometry. Scale bars: 25 μm.
Endovascular Procedure
After acquiring adipose tissue and an acclimatization period of 3 weeks, 12 animals were subjected to an endovascular procedure. All of the dogs were fasted from the night before the procedure. Anesthesia was initially induced with intramuscular zoletil (15 mg/kg zolazepam and tiletamine; Virbac AH, Fort Worth, TX, USA), and then inhaled anesthesia was applied with enflurane (Arylane; Ilsung Pharmaceuticals, Seoul, Korea). The animals were ventilated mechanically using an anesthesia device (GE Healthcare, Chalfont St. Giles, UK) during the procedure. Heparin (100 units/kg; JW Pharmaceutical, Seoul, Korea) was injected intravenously before the endovascular procedure to prevent clot formation within the catheter. A 5F sheath (Arrow International, Cleveland, OH, USA) was inserted into the right femoral artery after it was cut, and a 5F Davis catheter (Cook, Bloomington, IN, USA) was advanced to the aortic arch. The internal carotid artery was superselected by using a microcath-eter (Prowler-10; Cordis, Miami, FL, USA) guided by a microwire (Agility-10; Cordis). A carotid angiogram was performed to verify the position of the catheter. Cells were injected into three dogs [5 χ 106 cells in 2 ml of phosphate-buffered saline (PBS); Sigma-Aldrich] without mannitol pretreatment. Cells were injected into another nine dogs at 5 min after mannitol pretreatment, whereby a hyperosmolar mannitol solution was infused (20% mannitol, total 50 cm3, 1 g/kg, for 1 min; JW Pharmaceutical) via a micro-catheter positioned at the internal carotid artery.
Cell Delivery Technique
The cells were prepared and delivered using one of three techniques. The cell solution was dissociated from the culture plate with hand shaking after the addition of a solution of ethylene diaminetetraacetic acid (EDTA)/ trypsin (Sigma-Aldrich). In the first protocol (protocol #1, n=2), 5 χ 106 autologous AT-PDGFRβ+ cells in 2 ml of PBS were slowly hand injected over a 1-min period. After experiencing a thromboembolic complication, the infusion protocol was modified (protocols #2 and #3). In protocol #2 (n=4), 5 χ 106 autologous AT-PDGFRβ+ cells were mixed with 50 ml of normal saline plus 1,000 units of heparin solution, and the mixture was dripped slowly using a pressure bag over a 10-min period. In protocol #3 (n=3), the 5 χ 106 autologous AT-PDGFRβ+ cells were mixed with 5 ml of normal saline plus 1,000 units of heparin solution, and the mixture was injected with continuous pumping out using two syringes connected by a three-way valve over a 2-min period (Fig. 2A). Since shaking of the syringe could affect the cell behavior, this procedure was performed very gently and without a significant pressure change.
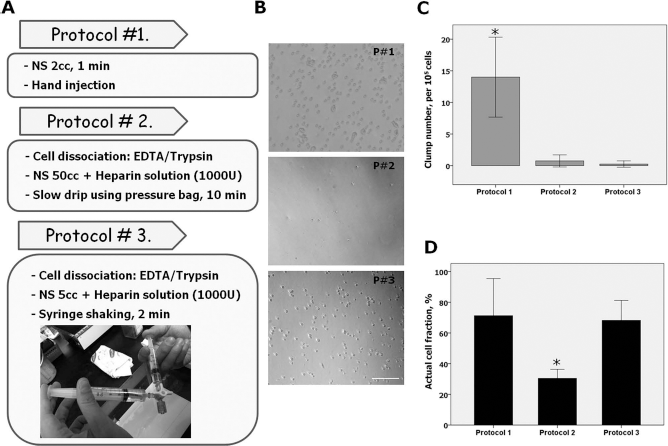
Ex vivo testing of the cell injection protocol. (A) Three protocols with different concentrations, injection methods, and durations were tested (P#1, P#2, and P#3 in B). (B) Cell dripping was performed onto a culture dish ex vivo. Clumped cells were occasionally detected with protocol #1 on microscopic evaluation. After completion of ex vivo dripping, the number of clumped cells per 105 cells (C) and the actual cell fraction (D; number of cells on the dish after dripping/initial number of cells) was measured. Scale bar: 100 μm (B). *p <0.05 versus the other two groups (n = 4, ANOVA followed by post hoc testing).
The efficacy and possible complications of each protocol were investigated through ex vivo simulation of the in vivo injection technique. After dripping cells onto the culture dish as per each protocol (n=4 per protocol), the numbers of clumped and lost cells were measured during the procedure.
Canine Monitoring and Brain Magnetic Resonance Imaging
Changes in the blood pressure, heart rate, oxygen saturation, and electrocardiogram were monitored throughout the procedure. Angiography was performed to assess arterial patency after injecting the mannitol and cells. Since these injections could induce early complications, such as shock, electrolyte imbalance, inflammation, and thromboembolism, blood sampling and brain imaging were conducted. Blood samples were taken prior to and at 1 day and 1 week after intracarotid injection. Samples of blood (5-10 ml) were collected into EDTA and serum separation tubes (Jaewoo Corporation, Seoul, Korea) and analyzed for complete cell count using automated counters, liver, and rental functions by spectrophotog-raphy and coagulation status by measuring prothrombin time and activated partial thromboplastin time.
The presence of a potential embolic infarct was examined at 1 day and 1 week after cell injection. Anesthesia with an intramuscular injection of zoletil (15 mg/kg zolazepam and tiletamine; Virbac AH) was adjusted to maintain immobility during the MRI scan. T1- and T2-weighted images, fluid-attenuated inversion recovery (FLAIR) images, and diffusion-weighted images (DWIs) were obtained in sagittal, transverse, and dorsal planes using a 3.0-tesla scanner (Magnetom Vision; Siemens, Erlangen, Germany). An infarct area was defined as an area with an intensity of more than 25% higher than a normal area.
Neurobehavioral Scoring for Dogs
A neurobehavioral test was done serially using a standardized rating scale, which is compatible with mongrel experiments (6); scoring was performed for motor function, consciousness level, head turning, circling behavior, and hemianopsia. The total neurobehavioral score ranged from 2 (completely normal) to 11 (most severe deficits: comatose or dead). Each dog was evaluated daily for the first week and then weekly until sacrifice at 4 weeks.
Immunohistochemistry of Brain Tissues
The dogs were euthanized at 28 days after transplantation. The brain was harvested, fixed for 24 h in 4% paraformaldehyde (Sigma-Aldrich) and then divided into ipsilateral and contralateral hemispheres and the brainstem; the hemispheres were further dissected into frontal, parietal, temporal, occipital, and deep brain regions. The specimens were cryoprotected by soaking them in 30% sucrose (Sigma-Aldrich) for 24 h, cut into 1-cm3 blocks, and then frozen. Each block was sectioned at 40 μm using a cryostat (CM 1900; Leica, Deerfield, IL, USA). For each animal, corresponding coronal sections were chosen for comparison at thickness intervals of 1 mm, and cells were counted in at least 10 serial sections per region. Sections were incubated with primary antibody against GFP (1:500; AB290; Abcam, Cambridge, MA, USA) at 4°C overnight, and then with TRITC-conjugated secondary antibody (Abcam) for 1 h at room temperature. They were counterstained with 1 μg/ml DAPI (Sigma-Aldrich). The number of labeled cells in each section was traced and analyzed using an image-analysis system (Image-Pro Plus; MediaCybernetics, Silver Spring, MD, USA). Cells were localized according to their relationship to vessels as follows: outside vessel (i.e., parenchymal), perivascular, and within vessel (intravascular).
Statistical Analysis
Data of continuous variables are expressed as mean ± SD values. Repeated-measures ANOVA followed by the post hoc test (Tukey B) were used to analyze multiple parametric data using SPSS 17.0 (Chicago, IL, USA). The cutoff for statistical significance was set at p <0.05.
Results
Changes in the Cell Delivery Protocol
The technique of hand injection of a concentrated cell pellet was initially used for cell delivery (n=2). After experiencing a thromboembolic complication, the infusion technique was modified to protocol #2, with a slow injection of a diluted cell pellet (n=4). Protocol #2 was associated with a low risk of thromboembolism, but histological evaluation revealed a low rate of cell engraftment. Taking into account the advantages and disadvantages of protocols #1 and #2, the technique was further refined to protocol #3 (n=3) to reduce the cell clumping and cell loss observed in the in vitro simulation study (see below).
Development of the Cell Preparation and Injection Techniques
In vivo cell injection may be complicated by cell clumping within the syringe during the period between dissociation and injection, as well as cell loss, presumably due to cells adhering to the syringe wall and piston. Therefore, three protocols were adopted with different dilutions, injection durations, and techniques, and cell clumping and cell loss were tested by simulating the in vivo experiment in an ex vivo design (Fig. 2A). When cells were dripped on the culture dish according to each protocol, cell clumping was occasionally noted on microscopy in protocol #1 (Fig. 2B, C). After the completion of cell dripping, the actual number of cells on the dish had decreased significantly to 30% compared with the initial number of cells in protocol #2. Protocol #3, with a repeated two-syringe shaking technique, appeared to circumvent the problems of cell loss and cell clumping (Fig. 2D).
Feasibility and Safety of Intracarotid Injection of Autologous AT-PDGFRβ+ Cells in Dogs
Serial common carotid angiograms of nine dogs obtained after autologous AT-PDGFRβ+ cell injection revealed good arterial patency without evidence of thromboembolic occlusion. The vital signs of mean arterial pressure, heart rate, and oxygen saturation remained stable during each step of the procedure (Table 1. The blood analysis indicated that hemoglobin, serum calcium, serum potassium, total protein, and albumin levels were reduced after infusion compared to baseline (Table 2, but the results of other blood count, electrolyte, liver, renal, and coagulation batteries were within the normal ranges. C-reactive protein (CRP) was also within the normal range. The vital signs and laboratory findings did not differ significantly between the three protocols. All nine dogs exhibited intact motor skills and consciousness on the neurologic scales (Table 3. Abnormal movements of head turning, circling, and hemianopsia were not observed. Brain MRI performed at days 1 and 7 revealed acute small infarcts in a few dogs; the incidence of acute infarct was 50% with protocol #1, 25% with protocol #2, and 0% with protocol #3 (Table 4. In dog #1, small areas of diffusion restriction were found on the frontal lobe contralateral to the injected side in the 1-day DWI and on the ipsilateral temporoparietal lobe at 1 week in the FLAIR images (Fig. 3A, B). A tiny lesion was observed in dog #5 on day 1, which disappeared in the follow-up FLAIR images performed on day 7 (Fig. 3C, D).
Mean Arterial Pressure (MAP), Heart Rate (HR), and Oxygen Saturation (SpO2) at Baseline During the Endovascular Procedure

The monitored MAP, HR, and SpO2 values of nine dogs were compared at each step of the procedure using ANOVA. Data are mean ± SD values. AT-PDGFRβ+, adipose tissue-derived, platelet-derived growth factor receptor β-positive cells.
Laboratory Findings at Baseline, Immediately Postinfusion (Post-I.), and on Days 1 and 7 After Autologous AT-PDGFRβ+ Cell Infusion

The values of the blood profiles of nine dogs were compared among baseline, and post-I. and at day 1 and day 7 after infusion of AT-PDGFRβ+ cells (ANOVA followed by Tukey B). WBC, white blood cell count; Hb, hemoglobin; PLT, platelet; Ca, calcium; P, phosphate; GLU, glucose; BUN, blood urea nitrogen; AST, aspartate transaminase; ALT, alanine transaminase; CK, creatine kinase; LD, lactate dehydrogenase; CRP, C-reactive protein; Na, sodium; K, potassium; Cl, chloride; PT, prothrombin time; aPTT, activated partial thromboplastin time.
Scores on the Neurologic Scales for Nine Dogs at Days 1, 7, and 28 After Infusion of AT-PDGFRβ+ Cells

The scores were to evaluate motor function (no deficit=1, hemipa-retic but able to walk=2, stands only with assistance = 3, hemiplegic and unable to stand=4), consciousness (normal = 1, mildly reduced=2, severely reduced=3, comatose=4), head turning (absent=0, posturing and turns toward the side of the infarct = 1, does not lift head, comatose, or dead = 1), circling (absent = 0, present = 1, does not ambulate or dead = 1), and hemianopsia (absent=0, present=1, unable to assess due to decreased consciousness or death). All nine dogs exhibited intact motor skills and consciousness on the neurologic scales. Abnormal movements of head turning, circling, and hemianopsia were not observed.
MRI Findings for Nine Dogs on Days 1 and 7 After Infusion of AT-PDGFRβ+ Cells

In protocol #1 (n = 2), approximately 5 χ 106 autologous AT-PDGFRβ+ cells in 2 ml of PBS was slowly injected by hand. In protocol #2 (n=4), the cell solution was dissociated by hand shaking after the addition of EDTA/trypsin. The cells were mixed with 50 ml of normal saline plus 1,000 units of heparin solution, and then dripped slowly using a pressure bag over a 10-min period. In protocol #3 (n = 3), the cells were mixed with 5 ml of normal saline plus 1,000 units of heparin solution, and the mixture was injected with continuous pumping out of two syringes connected by a three-way valve, over a 2-min period.
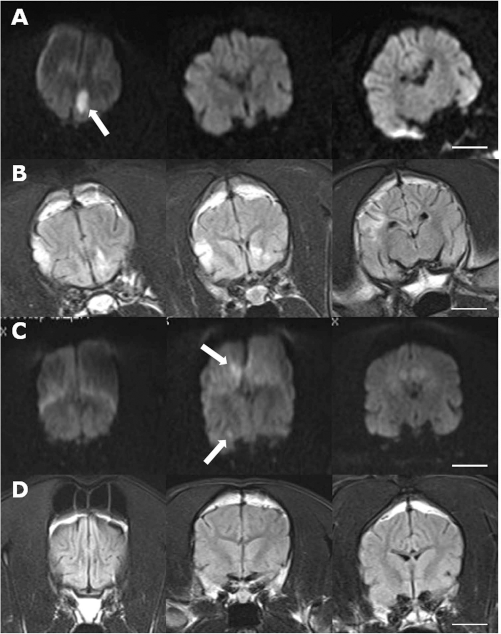
Brain MRI findings in dogs #1 and #5 at days 1 and 7 after autologous AT-PDGFRβ+ cell injection. (A) Coronal views of DWIs at 1 day after cell injection according to protocol #1, via the right internal carotid artery, revealed small areas of diffusion restriction at the left anterior frontal lobe (arrow). (B) Coronal views of a FLAIR sequence after 1 week revealed new hyperintense lesions at the right temporoparietal lobe. (C) Coronal views of DWIs at 1 day after cell injection according to protocol #1, via the right internal carotid artery, revealed two small diffusion restriction lesions (arrows) in the right frontal lobe ipsilateral to injected side. (D) The previous diffusion restriction disappeared, and no further hyperintense lesions were found on coronal views of a FLAIR sequence after 1 week. Scale bars: 10 mm.
Localization of Transplanted Autologous AT-PDGFRβ+ Cells Within the Canine Brain
The transplanted autologous AT-PDGFRβ+ cells were detected throughout the entire brain (Fig. 4A), but more were present in the ipsilateral hemisphere (13 and 8 cells/mm2 in the ipsilateral and contralateral frontal lobes, respectively). The AT-PDGFRβ+ cells were distributed as follows: 70% around vessels, 20% outside vessels, and 10% within vessels, regardless of the injection protocol or brain region (Fig. 4B, C). The mean numbers of AT-PDGFRβ+ cells were 1.5- to 1.7-fold higher in protocols #1 and #3 than in protocol #2 (p<0.001) (Fig. 4D). Approximately 7% of the injected cells survived in the brain at 4 weeks. Very few cells were observed in the brains of the control group without mannitol pretreatment. The AT-PDGFR β+ cells were found most frequently in the frontal lobe, followed by the parietal lobe, temporal lobe, brainstem/thalamus, and occipital lobe in the ipsilateral and contralateral hemispheres (Fig. 4E). The distribution patterns were similar for the three different protocols.

Engraftment of transplanted autologous AT-PDGFRβ+ cells within the canine brain. (A, B) The frontal cortex was examined in a section of the canine brain using GFP (green) and DAPI stain (blue). (C) The AT-PDGFRβ+ cells were located in the perivascular area (70%), outside vessels (parenchymal; 20%), and within vessels (intravascular; 10%). (D) The mean number of AT-PDGFRβ+ cells was greater with protocols #1 and #3 than with protocol #2 and the control condition. Regarding the cell distribution (E), the AT-PDGFRβ+ cells were found most frequently in the ipsilateral frontal lobe, followed by the ipsilateral parietal lobe, contralateral frontal lobe, ipsilateral temporal lobe, occipital lobes, and brainstem/thalamus (BG/Th). Rt., right; Lt., left. Scale bars: 100 μm (B) and 20 μm (D). *p <0.01.
Discussion
While pericytes have been recognized as potential candidates for cell therapy due to their multipotent characteristics (7,10,17), the clinical feasibility of pericyte transplantation in chronic disease models has not received sufficient attention. This study achieved a stable preparation of highly purified autologous canine AT-PDGFRβ+ cells within 3 weeks from adipose tissue acquisition, and their optimal culture conditions were clarified, which indicates the clear feasibility of autologous AT-PDGFRβ+ cell preparation for clinical applications.
The transplantation into the brain via an intra-arterial route is critically limited by the BBB, especially when the disease enters the chronic phase and homing signals are extinguished. Very few AT-PDGFRβ+ cells were found throughout the brain in the control condition (without mannitol preloading) in the present study. It is postulated that acute stroke evolves to the subacute stage, with loss of homing signals for stem cell recruitment or BBB closure, within the 2 or 3 weeks that may be required for autologous AT-PDGFRβ+ cell preparation. Remarkably, hyperosmolar mannitol allowed cells to enter even into the normal canine brain. Various BBB disruption methods are available, including mannitol, hyperventilation, hypothermia, hypotension, and hypoperfusion (16,27,38,43). We adopted mannitol treatment for transient opening of the BBB for several reasons. Mannitol is a sugar alcohol that has been used safely in clinical practice to decrease cerebral edema and intracranial pressure (26,37). This osmotic agent is known to open the BBB by shrinking the tightly linked endothelial cells temporarily (22) and to dilate small vessels or capillaries, thus improving their flow (4). The dosage of mannitol and the timing of the subsequent AT-PDGFRβ+ cell injection used in the current study were taken from the literature of cerebral enzyme and cell delivery (27,38).
Various routes for cell therapy in neurologic disease have been studied. An intravenous injection is a simple approach, but a higher dose of stem cells would be required for clinical application (2). Concerns about ineffective transplantation and poor safety are also raised because most intravenously injected cells are trapped in the capillary beds of the lungs and liver (36). Local intracerebral injection is invasive and may cause adjacent tissue damage, and multiple craniotomy sessions might not be clinically tolerated (11). In this regard, the intracarotid route for stem cell delivery is an attractive approach because cells can be delivered directly to the injured tissue, offering greater homing and seeding potential, particularly in the case of autologous transplantation. Endovascular techniques that are essential for intracarotid cell delivery are applicable to larger animal models than rodents. The sizes of the carotid arteries in dogs are similar to those in humans, and endovascular techniques in the canine model are well established (5,13,39). A stroke model might have been more appropriate for confirming our hypothesis, but no standardized chronic stroke models are yet available in dogs for direct evaluation of the feasibility and safety of intracarotid injection of autologous AT-PDGFRβ+ cells.
The safety of intracarotid injection of AT-PDGFRβ+ cells was evaluated by catheter angiography, vital signs, blood tests, neurobehavioral score, and brain MRI in the canine subjects. Vital signs were stable after injecting mannitol and AT-PDGFRβ+ cells, and the laboratory tests did not reveal any clinically significant adverse reactions. In particular, CRP, a surrogate marker of inflammation, was within the normal range, indicating that autologous AT-PDGFRβ+ cell infusion was not associated with a significant inflammatory or immunologic reaction. Although catheter angiography confirmed arterial patency in all dogs, acute small infarcts were observed on brain MRI in one of the two dogs in protocol #1, which used a concentrated cell pellet (multiple infarcts with an incidence of 50%), and in one of the four dogs in protocol #2, which used a diluted cell pellet (single tiny infarct with an incidence of 25%). Protocol #3, a technique that is currently used for the particle embolization of hypervas-cular intracranial tumors, produced no thromboembolic events. In terms of cell engraftment, the number of cells was greater with protocols #1 and #3 than with protocol #2. Although protocol #1 would be preferred due to its greater therapeutic effect, the thromboembolic risk of the higher concentration of cell pellets should not be overlooked. Optimal balancing of the thromboembolic risk and cell engraftment suggests that protocol #3 represents the best option, but additional well-controlled experiments are required before human application.
Arrested flow through an occluded carotid artery may result in AT-PDGFRβ+ cells not being delivered to the brain parenchyma, despite hyperosmolar mannitol preloading. An arterial infusion into the patent contralateral carotid or basilar artery would permit cells to enter the brain tissue. The current study demonstrated that AT-PDGFRβ+ cells administered into the unilateral carotid artery could effectively engraft into the bilateral hemispheres. These findings are in accordance with those from a previous study of basilar artery injection of umbilical cord-derived stromal cells after 1 day of acute middle cerebral artery occlusion (6). The detailed mechanism underlying cell engraftment in the bilateral hemispheres should be studied in future investigations.
The current study was subject to several limitations. First, the AT-PDGFRβ+ cells were engrafted into normal canine brains following pretreatment with hyperosmolar mannitol, but their clinical efficacy was not assessed in the disease model. We are now developing a reliable and reproducible model that simulates human stroke. Second, the optimal dosage of mannitol pretreatment was not determined because of the difficulty of quantitatively measuring the magnitude of BBB breakdown. Moreover, the optimal dose of AT-PDGFRβ+ cells was not determined, since only a single cell dosage was tested.
Conclusions
In summary, pretreatment with hyperosmolar mannitol enhanced the delivery of autologous AT-PDGFRβ+ cells into the normal canine brain by transiently opening the BBB. The only significant adverse reaction associated with the intracarotid injection of autologous AT-PDGFRβ+ cells was silent thromboembolism. Improvement in cell pellet preparation is required to overcome this complication. Together the results of this study indicate that the intracarotid infusion of AT-PDGFRβ+ cells following pretreatment with hyperosmolar mannitol can be a feasible and safe cell-based neurorestorative therapy for stable brain diseases such as chronic stroke.
Footnotes
Acknowledgment
This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education, Science and Technology (grant number: 2012R1A1A2002081). The authors declare no conflict of interest.
