Abstract
To become potent T-cell stimulators, DCs need to mature. Treatment with soluble CD83 (sCD83) induces immune tolerance and protects against transplant rejection by maintaining dendritic cells in an immature, tolerogenic state. Until now, the mechanism through which sCD83 keeps DCs immature has not been investigated. The internalizing pathway of CD83 was screened by Western blot, and the direct interactions between internalized proteins were verified through coimmunoprecipitation (co-IP) and transmission electron microscopy (TEM). CD83 plasma membrane levels were detected by Western blot using a plasma membrane protein extraction protocol. The changes in CD83 surface levels in DCs were detected by flow cytometry. Caveolin-1 function was detected in a kidney transplant model. In this study, we demonstrated that caveolin-1 could affect CD83 level during endocytosis in mouse DCs. Caveolin-1 coprecipitates with CD83, as demonstrated by co-IP analysis. TEM morphometric analysis of the entire CD83 distribution associated with internalized caveolin-1 demonstrated a significant interaction in cellular vesicles. sCD83 reduces endogenous CD83 plasma membrane levels, and caveolin-1 knockdown reverts CD83 levels in plasma membrane. sCD83 treatment decreases CD83 surface levels in DCs. siRNA to caveolin-1 in DCs inhibits this effect of sCD83. The effects of sCD83-treated DCs were proved in CD1 mice. Knocking down caveolin-1 in DCs obstructs the effects of sCD83 on kidney transplant. In conclusion, our data indicated that a caveolin-dependent endocytic pathway is involved in CD83 internalization in DCs and that caveolin-1 is involved in the activity of DCs.
Introduction
Dendritic cells (DCs) are professional antigen-presenting cells with antigen processing and presentation capabilities that enable them to be the key actor in initiating and regulating immune responses. Life science techniques using a combination of granulocyte/macrophage-colony stimulatory factor and interleukin-4 (IL-4) allow for the propagation of CD11c-positive DC progenitors (11, 18, 19).
DCs play a dual role in both initiating and regulating immune responses. This dichotomous f unction is primarily attributed to distinct populations of DCs. These are mature DCs (mDCs) and immature DCs (iDCs). iDCs are tolerogenic because of deficient costimulation. They induce graft hyporeactivity and prevent autoimmune diseases. mDCs, on the other hand, are immunostimulatory and can induce autoimmune diseases and transplant rejection (12). DC immaturity can be maintained by exposure to some cytokines, such as transforming growth factor-β (TGF-β) (32). Along with others, we have shown that transplant survival increases through transfusion of iDCs along with an agent called sCD83, which prevents the DCs from maturing within the transplant recipient (11, 18, 19). In this fashion, the iDCs and deficient mDCs are able to prevent transplant rejection and prolong transplant survival. However, how sCD83 inhibits DC maturation has remained unclear.
For two decades, donor-specific transfusion of DCs has been shown to reliably improve allograft survival with iDC administration (11, 18, 19). Biological agents, such as CD154 monoclonal antibody, have been shown to act synergistically with iDCs and promote graft survival. However, despite various modifications of iDCs using biological agents and pharmacotherapy, iDCs administered in the absence of immunosuppressive therapy lead to only modest improvements in graft survival (3–4 weeks) (10). This has been associated with the eventual maturation of donor iDCs in the allograft recipient within several days to weeks. We hypothesize that early maturation of iDCs in the graft recipient limits their ability to maintain graft hyporesponsiveness, and this is why only modest improvements in graft survival have been achieved (3–4 weeks).
To become potent T-cell stimulators, DCs need to mature. CD83, the expression of which coincides with expression of costimulatory molecules CD40 and CD80, plays a key role in DC maturation and function (6, 14, 17, 20, 23, 26, 27, 30, 33). Upregulation of these costimulatory molecules has been associated with increased DC stimulatory capacity. The functional role of CD83 was demonstrated using the soluble extracellular domain of CD83 (sCD83), and it was determined that sCD83 inhibits both murine and human immune responses (11, 18, 19, 37). sCD83 blocks DC maturation in response to proinflammatory cytokines (IL-1, TNF-α, and PGE2) or Toll-like receptor ligands (lipopolysaccharide). Moreover, the exposure of mDCs to sCD83 downmodulates CD83 expression and causes considerable cytoskeletal rearrangement and morphological changes resembling iDCs. sCD83 also completely inhibits DC–T-cell clustering and T-cell proliferation by allogeneic DCs in vitro. However, sCD83 has no direct inhibitory effect on T-cells. In experimental autoimmune encephalomyelitis induced in the mouse (a model for multiple sclerosis), sCD83 prevented paralysis caused by the neuroantigen myelin oligodendrocyte glycoprotein (MOG). Tolerance was also induced, since mice were completely protected from disease induced by reimmunization with MOG 25 days after initial sCD83 treatment. IL-2 partially restored in vitro hyporesponsiveness of T-cells from these mice to MOG, suggesting that the induction of energy may be responsible for hyporesponsiveness rather than deletion. However, the molecular mechanisms of sCD83-mediated inhibition in DCs remain unclear (4, 36).
Mouse CD83 is a 45-kDa glycoprotein that plays a key role in DC maturation and function, the expression of which coincides with expression of costimulatory molecules CD40 and CD80. Mouse CD83 is a member of the immunoglobulin superfamily of receptors, expressed as a type I transmembrane glycoprotein containing a 114-amino acid (aa) extracellular region at N-terminal end, a 22-aa transmembrane segment, and a 39-aa cytoplasmic domain at C-terminal end (6). sCD83 contains only the extracellular region of CD83.
Endocytosis is a master organizer of signaling regulation and plays a major role in resolving signals in a spatial and timely manner, especially for cellular membrane protein (24, 33, 34). Coated pits from plasma membrane depend primarily on clathrin, but accumulating evidence indicates that non-clathrin-mediated endocytic pathways also regulate various membrane proteins. For example, receptor-mediated endocytosis can occur via a pathway involving caveolae. TGF-β receptors internalize into both caveolin-1- and clathrin-positive vesicles. It was also shown that the epidermal growth factor receptor is endocytosed via either clathrin-dependent or clathrin-independent routes, depending on the epidermal growth factor concentration. In CD8+ T-cells, caveolin-1 is an orchestrator of T-cell receptor signal specificity (35). However, as of now, we find no studies about CD83 endocytosis in DCs, though CD83 plays a key role in DC maturation and function.
Caveolae is the main vesicle being internalized from the cellular membrane (5, 22, 24, 25, 34). The caveolar membrane is a special lipid raft consisting of caveolin, cholesterol, glycosphingolipids, and signaling enzymes, such as Src kinase. Caveolin is the main effector of caveolae formation and maintenance. In mammals, there are three kinds of caveolin. Caveolin-1 is the representative one. It is the signature membrane protein in the internalized vesicle. It is involved in cell internalization and cell membrane recycling through its activity as a protein scaffold.
Materials and Methods
Animals and Reagents
CD1 mice were purchased from the Jackson Laboratory (Bar Harbor, ME, USA) and maintained in the animal facility of Zhengzhou University (Zhengzhou, China). The mice were maintained under specific pathogen-free conditions. Eighty-four mice were all male and 7 weeks old. Animal experiments were conducted in accordance with the Canadian Council on Animal Care guidelines under protocols approved by the Animal Use Subcommittee at our institution. Animals were housed in conventional conditions. They had free access to food and water.
Mouse sCD83 protein powder was obtained from Dr. Yongchu Huang at Huazhong University of Science and Technology, China. sCD83, the extracellular domain of mouse CD83 (aa 20–114), was purified from bacteria, and the glutathione S-transferase purification tag was removed before stimulating DCs (11). Anti-caveolin-1 and anti-actin and anti-Na+/K+-ATPase (α1 subunit) antibodies, protein A/G, control siRNA, and caveolin-1 siRNA were purchased from Sigma-Aldrich (St. Louis, MO, USA). Tris lysis buffer, protease inhibitor cocktail, FBS, MEM, sample buffer, paraformaldehyde, glutaraldehyde, and sodium dodecyl sulfate (SDS) were also purchased from Sigma-Aldrich. Anti-CD83 (C14), anti-CD83 (N15) antibodies, GAPDH antibody, horseradish peroxidase secondary antibody, and bovine serum albumin were purchased from Santa Cruz Biotechnology Inc. (Dallas, TX, USA). Mouse CD11c-FITC, PE-conjugated anti-mouse CD83, CD40, CD80, or appropriate isotype antibodies were purchased from eBioscience (San Diego, CA, USA). CD11c microbeads were purchased from Miltenyi Biotec Inc. (Auburn, CA, USA). Fish gelatin, saponin, uranyl acetate, lead citrate, and 200-mesh formvar-coated nickel grids were purchased from Electron Microscopy Sciences (Hatfield, PA, USA). Rhombine, ketamine, and isoflurane were purchased from Wuhan Boster Biological Technology Co. Ltd. (Wuhan, China).
Western Blot Analysis and Coimmunoprecipitation (co-IP)
We have previously demonstrated techniques to derive mature and immature CD11c+ DCs with 90% purity. To find the relative proteins in CD83 endocytosis and to detect the CD83 expression level, splenic CD11c+-enriched DC subpopulations from CD1 mice were purified using CD11c microbeads. Purity of DCs was confirmed by flow cytometry, and >94% cells were CD11c+ (11, 18, 19).
After 24 h of caveolin-1 transfection, cells were cultured in PBS (Wuhan Boster Biological Technology, Co. Ltd.) with 10 μg/ml sCD83 protein for another 24 h (18, 23). Then, cells were harvested for either Western blot or co-IP analysis. Total cell protein was obtained by treating cells with a Tris lysis buffer in the presence of protease inhibitor cocktail and PMSF (Sigma-Aldrich). Cell lysate at 15 mg/lane was separated on SDS-PAGE gels, transferred onto a PVDF membrane (VWR International, Edmonton, Canada), and then blocked for 1 h with 5% nonfat milk (Wuhan Boster Biological Technology, Co. Ltd.). For experiments where two gels ran simultaneously, one membrane was probed with anti-CD83 and the other membrane was probed with anti-caveolin-1. Anti-actin antibody served as a loading control. The blots were incubated with the primary antibody for 1 h at room temperature and then with horseradish peroxidase secondary antibodies. The blots were visualized with Kodak film using ECL plus (General Electric, Baie-D'Urfe, QC, Canada).
In order to detect CD83 expression, plasma membrane proteins in DCs were enriched using a plasma membrane protein extraction kit (Biovision, Rockland, MA, USA). Protein lysate at 15 μg/lane was separated on SDS-PAGE gels to detect CD83 expression.
For co-IP, the total cell lysate was extracted using protease inhibitor cocktail and PMSF. Samples of 0.5 mg of protein were incubated with the first antibody overnight at 4°C, and they were subsequently precipitated with protein A/G plus agarose beads for 4 h at 4°C. The samples were washed three times with lysis buffer. The beads were then resuspended, and all the lysates in 2× sample buffer in each lane were separated on SDS-PAGE gel as described above.
siRNA and Transfection
Knockdown of basal expression levels of caveolin-1 in DCs was performed using respective siRNAs as well as scrambled control siRNA (Santa Cruz, Santa Cruz, CA, USA). Cells were grown in 60-mm dishes (BD Biosciences, Franklin Lakes, NJ, USA) at 60–70% confluence, and 80 pmol of duplex siRNA was transfected into cells using Lipofectamine 2000 (Invitrogen, Burlington, ON, Canada). After 24 h of transfection and subsequent treatment, cells were harvested for either Western blot or co-IP analysis.
Transmission Electron Microscopy (TEM)
Live DCs were treated with a goat anti-CD83 antibody [anti-CD83 (N-15); Santa Cruz] for 1 h in 10% FBS MEM at room temperature. This antibody targeted the N-terminal region of the CD83 protein and thus served to label the cell surface CD83 protein. The cells were fixed with 2% paraformaldehyde and 0.1% glutaraldehyde in 0.1 M phosphate buffer (pH 7.2) for 2 h at room temperature and then left overnight in 0.1 M phosphate buffer containing 2% paraformaldehyde.
After dehydration in ethanol, the cells were embedded in LR White resin (Canemco Inc., St. Laurens, Quebec, Canada), and the blocks were polymerized with UV light (20 h, 4°C). Ultra film sections were cut and mounted on 200-mesh formvar-coated nickel grids. For immunocytochemistry, the grids were blocked with 1% bovine serum albumin, 0.2% fish gelatin, and 0.05% saponin in 0.1 M phosphate buffer for 1 h, and then incubated with rabbit anti-caveolin-1 antibody for 1 h at room temperature. After immunolabeling, the grids were blocked for 1 h and incubated for 45 min at room temperature with an anti-goat antibody conjugated with colloidal gold partials (25 nm; 1:20; Sigma-Aldrich, Mississauga, Canada) mixed with anti-rabbit antibody conjugated with colloidal gold (5 nm; 1:20; BBInternational, Llanishen, UK). Finally, the sections were counterstained with uranyl acetate and lead citrate, washed in double distilled water, dried, and examined under a TEM (Hitachi 7000; Hitachi, Twinsburg, OH, USA). Control samples were incubated with control IgG antibody; no immunolabeling was observed.
Flow Cytometry
DCs were harvested and divided into three groups: the control group without treatment, the sCD83-treated group, and caveolin-1-negative and sCD83-treated group. DCs in the last group were transfected by caveolin-1 siRNA for 24 h prior to stimulation by PBS with 10 μg/ml sCD83 protein. For surface marker staining of DCs, cells were incubated for 15 min at 4°C with PE-conjugated anti-mouse CD83, CD40, CD80, or appropriate isotypes (eBioscience).
FCM analysis was performed using FACSCalibur and CellQuest software (BD Biosciences) and analyzed using FlowJo software (Treestar, Ashland, OR, USA). Mean fluorescent intensity was expressed as the geometric mean channel fluorescence minus the appropriate isotype control.
Renal Transplant Model
Anesthesia was induced by using intramuscular rhombine (0.04 mg/kg intramuscular) and ketamine (0.1 mg/kg intramuscular) and was kept with 2% isoflurane. Left renal transplantations were executed as described previously (31).
Purified CD1 mouse spleen-derived DCs were stimulated by PBS with 10 μg/ml sCD83 protein for 24 h. The DC solution was adjusted to 1 × 106/ml. CD1 mice were pretreated with sCD83-treated or nontreated DCs [0.05 ml/kg, intraperitoneal (IP)] 18 h before kidney procurement. In the experiments involving pretreatment of recipients with sCD83-treated DCs, murine recipients were randomly allocated to one of the four following groups: (1) normal saline treated (0.05 ml/kg, IP, given 18 h before organ procurement), (2) control DCs treated (0.05 ml/kg, IP, given 18 h before organ procurement), (3) sCD83-stimulated DCs treated (0.05 ml/kg, IP, given 18 h before organ procurement), and (4) caveolin-1-knocked-down, sCD83-stimulated DCs treated (0.05 ml/kg, IP, given 18 h before organ procurement). Animals survived until study termination.
Serum creatinine levels were used to evaluate renal function after organ procurement. Serum creatinine was measured by colorimetric enzymatic means using the Roche Modular Analyzer (Hoffmann-La Roche, Mississauga, ON, Canada).
Statistics
For Western blot, co-IP, TEM imaging, and flow cytometry analyses, at least three independent experiments were performed to ensure the consistency of the findings. All data are expressed as the mean ± the standard deviation of the mean. Statistical analysis of the in vitro data was performed by one-way ANOVA and Tukey's post hoc tests to detect statistical significance between the control and test groups. Animal survival analysis was performed with log-rank test using GraphPad Prism software (GraphPad Software Inc., La Jolla, CA, USA). A value of p ≤ 0.05 was considered significant.
Results
The Involvement of Caveolin-1 in CD83 Internalization and Degradation
sCD83 is at the position of N-terminal end of CD83. With CD83 (C14) antibody, only the levels of endogenous CD83 were detected. sDC83 was not detected. DCs to be treated by PBS with 10 μg/ml sCD83 for 24 h expressed significantly less caveolin-1 levels. DCs treated by control siRNA were detected with no changing of the caveolin-1 level. CD83 level was not significantly changed when just prohibiting the caveolin-1 expression. On the other hand, when overexpressing caveolin-1 by plasmid transfection, significantly less CD83 was detected in DCs in comparison with the control. When caveolin-1 expression was inhibited by the siRNA and sCD83 stimulation was used, the CD83 protein levels were reverted significantly (Fig. 1). These results suggest that caveolin-1 mediates CD83 endocytosis in DCs only under sCD83 stimulation.

CD83 endocytosis through a caveolin-1-dependent pathway in DCs. There is caveolin-1 expression (+) in normal DCs. Caveolin-1 overexpression by caveolin-1 plasmid is indicated by “††™. The condition of caveolin-1 inhibition by caveolin-1 siRNA is demonstrated by ”–™. The condition of control siRNA is indicated by “±”. Purified DCs to be treated by PBS with 10 μg/ml sCD83 for 24 h express less CD83 and less caveolin-1 levels compared to those in the nontreated DC group. Caveolin-1 overexpression enhances CD83 levels, and knocking down caveolin-1 significantly lessens sCD83 function in reducing CD83 levels.
Physical Association and Colocalization Between CD83 and Caveolin-1
Since caveolin-1 may be involved in CD83 endocytosis, we studied the direct association between endogenous CD83 and caveolin-1 proteins using co-IP analysis.
Purified mouse spleen-derived DCs were directly harvested, and cell lysates were extracted. An anti-caveolin-1 antibody was used to precipitate caveolin-1 and its associated proteins, and the immunoprecipitated proteins were detected using a CD83 (C14) antibody. As shown in Figure 2A left, the endogenous CD83 band was detected in the caveolin-1-precipitated proteins. In the second set of experiments, total cell lysates were immunoprecipitated with a CD83 (C14) antibody, and the immunoprecipitate was detected using an anti-caveolin-1 antibody by Western blot analysis. As shown in Figure 2A right, caveolin-1 was detected in endogenous CD83 precipitated proteins. These results indicate a physical interaction between CD83 and caveolin-1.

Colocalization between CD83 and caveolin-1 in DCs. (A) Coimmunoprecipitation (co-IP) analysis. DCs were lysed, and samples of 0.5 mg protein were used for immunoprecipitation using caveolin-1 antibody (left) or anti-CD83 (C14) antibody (right). GAPDH antibody was used as the control antibody. Actin served as a loading control for co-IP. The immunoprecipitated protein was then immunoblotted using anti-CD83 (C14) (left) or anti-caveolin-1 antibody (right). (B) Representative TEM images of caveolin-1-dependent CD83 internalization. Live DCs at 24 h after transfection with plasmid caveolin-1 were treated with an anti-CD83 (N15) antibody. The cells were fixed for TEM. Representative caveolin-1 colloidal gold particles are highlighted with solid line arrows. Representative CD83 colloidal gold particles are highlighted with dashed line arrows. Scale bar: 100 nm.
Ultrastructure of Internalized Vesicles in DCs
To further our understanding of the interactions between whole CD83 and caveolin-1, the ultrastructure of the endocytosis process was examined using TEM. Anti-CD83 (N-15) antibody is used to tag the extracellular domain of CD83 in live cells before internalization. We used 25-nm colloidal gold particles to tag CD83 protein and 5-nm colloidal gold particles to tag caveolin-1 protein. As shown in Figure 2B, caveolin-1 molecules (indicated by solid line arrows) form some vesicle shapes in the cell plasma at high magnification. CD83 molecules (indicated by dashed line arrows) are mostly on the vesicle membrane.
Whole CD83 Molecule Expression Level on DC Plasma Membrane
Since sCD83 inhibits DC maturation, we assumed that CD83 expression levels on the DC plasma membrane had been changed by sCD83 overexpression. To confirm this assumption, we extracted plasma membrane proteins from DCs and detected the entire CD83 molecule expression level using Western blot. As shown in Figure 3, treating DCs with sCD83 protein significantly decreased CD83 expression levels compared to the control in DC plasma membrane components. Interestingly, CD83 expression levels were reverted significantly on the cellular membrane after treatment with caveolin-1 siRNA compared to the control. At the same time, clathrin levels on DC plasma membrane were not significantly changed by sCD83 or cavelin-1 siRNA.
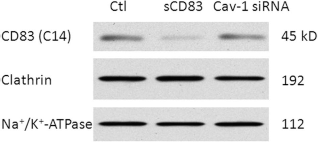
CD83 expression levels on DC plasma membrane. DCs were divided into three groups: (1) nontreated DC group as the control, (2) sCD83-treated DC group, and (3) sCD83-treated DCs after transfection with caveolin-1 siRNA (Cav-1 siRNA). Cellular membrane protein was then extracted and detected by Western blot. CD83 (C-14) antibody was used to detect whole CD83 molecules by their C-terminal domain. Na+/K+-ATPase served as a loading control for plasma membrane proteins.
Effect of sCD83 on Plasma Membrane Proteins in DCs
In order to confirm sCD83 function on DCs, the plasma membrane markers CD83, CD40, and CD80 were detected using flow cytometry. Isotype controls were used to identify staining control and help to gate before FlowJo analysis. As shown in Figure 4, with sCD83 protein treatment, the DC surface levels of CD83, CD40, and CD80 declined significantly compared to the nontreated DC control group. However, when DCs were treated by sCD83 protein and caveolin-1 siRNA, the DC surface levels of CD83, CD40, and CD80 reverted to the levels similar to those in nontreated DC groups. The levels between sCD83-treated DC group and caveolin-1-negative sCD83-treated (caveolin-1--sCD83-treated) DC group were significantly different (p < 0.05), while the levels between the nontreated DC group and caveolin-1--sCD83-treated DC group were not significantly different (p > 0.05).

The effect of sCD83 on plasma membrane proteins on DCs. (A) DCs were harvested and stimulated by PBS with 10 μg/ml sCD83 protein for 24 h. For surface marker staining of DCs, cells were incubated for 15 min at 4°C with PE-conjugated anti-mouse CD83, CD40, CD80, or appropriate isotypes (eBioscience). DCs were divided into three groups: (1) nontreated DC group as the control, (2) sCD83-treated DC group, and (3) sCD83-treated DCs after transfection with caveolin-1 siRNA. The DC surface levels of CD83, CD40, and CD80 were significantly reduced in the sCD83-treated group compared to those in the nontreated DC control group. When DCs were treated with sCD83 protein and caveolin-1 siRNA, the DC surface levels of CD83, CD40, and CD80 reverted in comparison with the levels in sCD83-treated DC groups. (B) Values are mean ± SEM. *p < 0.05 (for control DC vs. sCD83-treated DC, sCD83-treated DC vs. caveolin-1--sCD83-treated DC). sCD83 proteins significantly reduced CD83, CD40, and CD80 surface levels in DCs. Knockdown of caveolin-1 significantly inhibited this reducing process.
Effect of sCD83-Treated DC Perfusion on Isogeneic Transplantation
CD1 mice were pretreated with sCD83-treated, caveolin-1--sCD83-treated, or nontreated DCs (0.05 ml/kg, IP) 18 h prior to kidney procurement. In the transplant model (31), a low dose of tacrolimus (0.2 mg/kg intravenously) was given to allograft recipients postoperatively. As shown in Figure 5, without immunotherapy, mice died within 12 days (serum creatinine 700 ± 140 μmol/L, detected on day 3). In the nontreated DC group, improved graft function was noted on day 50 (serum creatinine 170 ± 38 μmol/L). In the sCD83-treated DC group, graft function was almost normal on day 50 (serum creatinine 50 ± 7 μmol/L). At the same time, animals treated with nontreated DCs had lived about 55 days. In the sCD83-treated DC group, all animals survived beyond 80 days. Interestingly, in the caveolin-1 siRNA-treated and sCD83-treated DC groups, graft function was noted on day 50 (serum creatinine 210 ± 46 μmol/L). The animals in the caveolin-1 siRNA-treated and sCD83-treated DC group lived about 53 days. Caveolin-1 siRNA-treated and sCD83-treated DCs significantly reduced the improvement of lifetime and graft function compared to the improvement in sCD83-treated group. Thus, knocking down caveolin-1 blocks the function of sCD83. The function of sCD83 on CD83 internalization needs caveolae.

The effect of sCD83-treated DCs perfusion on isogeneic transplantation. DCs were stimulated by 10 μg/ml sCD83 for 24 h. Kidney transplant recipient preconditioning with 1 × 106/ml DCs (0.05 ml/kg, IP) 18 h before kidney procurement provided significant recipient protection against transplantation injury based on (A) survival (sCD83-treated DCs) and (B) graft function. (A) After University of Wisconsin solution flush, grafts failed, and mice sacrificed within 12 days. Animals treated with nontreated DCs had lived about 55 days. In contrast, if recipients were pretreated with sCD83-treated DCs, animals had superior renal function and survival for more than 80 days. When DCs were treated by sCD83 protein and caveolin-1 siRNA, animal lifetime reduced back to about 53 days. (B) Serum creatinine improved significantly in the sCD83-treated DC group compared to that in the nontreated DC group. Caveolin-1 knockdown significantly weakened the improvement of serum creatinine by sCD83. Values are mean ± SEM. *p < 0.05 (for lifetime and creatinine levels in control DCs vs. sham, sCD83-treated DCs vs. control DCs and sCD83-treated DCs vs. caveolin-1–-sCD83-treated DCs).
Discussion
Our results indicated that active DCs release sCD83 in vivo and in vitro (14). sCD83 is also the maturation marker of DCs. To further our understanding of iDCs, we selected CD83 internalization as our point of interest. For now, only a very limited number of studies involved DC endocytic processes, though iDC is very important in immune-related diseases (13, 16). In this study, the treatment with sCD83 reduced caveolin-1 protein levels (Fig. 1), and overexpression of caveolin-1 reduced CD83 levels. Conversely, knockdown of caveolin-1 reverted CD83 levels. An antibody of CD83 (C14) was used to detect CD83 rather than sCD83. There is a link between caveolin and CD83, which is proven by co-IP. To investigate CD83 endocytosis in DCs, we must distinguish the internalizing process from the recycling process because it is uncertain whether the recruitment and endocytosis of different types of cargo by caveolin-1 can be differentially regulated (9). In the TEM experiment, we used anti-CD83 (N-15) antibody to tag the extracellular domain of CD83 in live cells. After the internalization of CD83, but not recycling, CD83 and caveolin-1 appeared in a vesicular structure together.
To confirm that CD83 is exclusively undergoing caveolar internalization, we clarified that sCD83 reduces endogenous CD83 plasma membrane levels using a plasma membrane protein extraction protocol. Using the same method, caveolin-1 knockdown enhances CD83 plasma membrane levels (Fig. 3). Additionally, sCD83 proteins significantly reduced CD83, CD40, and CD80 surface levels in DCs using flow cytometry. Conversely, knockdown of caveolin-1 inhibited this reducing process (Fig. 4). In the results of flow cytometry, there is still some extent of CD83, CD40, and CD83 expression on DCs in the condition of appropriate sCD83 stimulation. It implies DCs did not undergo cell death, such as apoptosis (18). It suggests to us that the function of iDC or adaptive immunity was not spoiled.
Finally, to confirm the in vivo involvement of caveolae in CD83 endocytosis, we clarified that mice given sCD83-treated DCs prior to kidney transplantation show improved kidney function and survival. Interestingly, knocking down caveolin-1 in DCs inhibited the effects of sCD83 on kidney transplant. The mice under the treatment of sCD83-treated DCs can survive more than 80 days from kidney transplantation. There is no evidence that the transferred DCs could survive for 80 days and have the ability to inhibit rejection. We tend to believe that this treatment contributes to the survival of acute kidney graft rejection in mice. This result is consistent with those in other studies (18, 31).
However, caveolin is on the cytoplasmic face of membranes and does not span the membrane. sCD83 is outside of the plasma membrane and should not be in direct contact with caveolin. Hence, in the next step, we examined whether there is an intermediate transmembrane protein involved in this endocytosis. On the other hand, though clathrin plays a key role in endocytosis, we could not clarify whether clathrin is involved in the CD83 internalization or not, though clathrin levels were not varied by sCD83 and caveolin-1-siRNA in the plasma membrane experiment. In the next step, we plan to select different conditions to verify the involvement of clathrin in CD83 endocytosis. Hence, we need to examine whether there is an intermediate transmembrane protein involved in this endocytosis. We have a hypothesis that there is a protein or some proteins to recognize the sCD83 structure, and then caveolin-involved endocytosis is activated aiming at this structure.
Tolerogenic DCs are prospective cellular mediators, and they can complement or even substitute immunosuppressive agents, which have obvious side effects (1). However, the greatest challenge is to resist DC maturation (2, 8, 15, 29). Our previous study demonstrated that sCD83 injection significantly improved DC function in vitro and slightly improved murine lifetime (11, 18, 19). In this study, sCD83 stimulated DCs in vitro. Treated DCs were injected as a vaccine rather than just sCD83 stimulation. This treatment significantly prevented DC maturation, enhanced sCD83 function, and extended murine lifetime in a renal transplant model. Importantly, without caveolin-1, the function of the vaccine is inhibited. It was further confirmed that caveolin is involved in CD83 endocytosis in vivo.
We first investigated CD83 endocytosis in DCs and showed that CD83 is internalized through a caveolin-1-dependent pathway. If we used sCD83 as a reagent to treat DCs, we should also consider the caveolin-1-dependent endocytosis. It provides us evidence that adjustment in the caveolin-1 pathway might alter the DC maturation process and vary the immune levels.
Conclusion
This article provides strong evidence that CD83 is internalized at least through a caveolin-1-dependent pathway. sCD83 can affect CD83 expression on the DC plasma membrane at least through caveolin-1-dependent endocytosis. Caveolin-1 is involved in DC activity (21). For the first time, we demonstrated that CD83 and caveolin-1 in this pathway hold significant promise for therapeutic use in future clinical immune treatments. Targeting endocytosis, a new therapeutic benefit is proposed for autoimmune diseases and transplantation (3, 7, 22, 28).
Footnotes
Acknowledgments
The authors are thankful to Ying Liu and Xiujuan Li for the administrative and mouse work, respectively. We also thank Dr. Jun Liu for critical reading of the manuscript and for helping with figure preparation. This work was supported by Dr. Tianfang Xie and was supported by a grant from the Chinese 5451 project of Henan Province Health Department. The authors declare no conflicts of interest.
