Abstract
Islet transplantation is a promising cellular therapy for the treatment of type 1 diabetes (T1D). The immunogenicity of isolated islets has been of interest to the transplant community for many years, as upon transplantation, islets are damaged or destroyed through specific and nonspecific inflammatory and immune events. Antigen presenting cells (APC) are crucial intermediates in the generation of both innate and specific immune responses, and it has long been understood that some APC are resident in islets in situ, as well as after isolation. Our aim was to identify and characterize intraislet resident populations of APC and other immune cells in islets from nonhuman primates (Macaca fascicularis) in situ (pancreas biopsies obtained prerecovery) and after isolation using immunohistochemistry, confocal microscopy, and flow cytometry. The numbers of cells obtained in situ are similar to those in islets postisolation. Each isolated islet equivalent contains an average of 21.8 immune cells, 14.7 (67%) of which are APC. Many of these APC are dentritic cells and, surprisingly, 50% are B lymphocytes. The number of islet-resident immune cells increases with islet size, with greater numbers in large versus small islets (p < 0.001). The APC were localized around the exterior or spread evenly throughout the islets, with no definitive orientation identified. This knowledge will be useful to develop tailored modulation strategies to decrease immunogenicity, enhance engraftment, and ultimately prevent islet rejection.
Keywords
Introduction
Islet transplantation is a promising cellular therapy for the treatment of type 1 diabetes (T1D) (20,24,59). Its widespread application is limited by several factors, including a limited number of organs and loss of islets throughout the processes of organ recovery, islet isolation, and culture (36,40,65). Upon transplantation in the liver, islets are destroyed through nonspecific inflammatory events and specific immune reactions and are further affected by the toxicity of obligate immunosuppression (14).
Antigen presenting cells (APC) are crucial intermediates in the generation of both innate and specific immune responses, and APC and other passenger leukocytes reside within islets in situ and after isolation (17, 41). To the best of our knowledge, no comprehensive analysis of resident immune cells in islets from larger animals or humans has been reported, as the majority of studies have been conducted in rodent islets and tended to focus on a particular immune cell type (27,32).
There are three broad classes of APC: dendritic cells (DC), macrophages, and B lymphocytes. Though their roles in antigen capture and presentation frequently overlap, they differ in their mobility, tissue distribution, and ability to elicit an immune response. DC have the broadest range of antigen-presenting skills and trafficking (37), and macrophages are generally more stationary and act as key scavengers in the culmination of a specific immune response and tissue destruction (50). B lymphocytes function as APC by directly stimulating the T-cell response in the presence or absence of other APC (12). Their diverse roles in both auto- and alloimmunity have made B lymphocytes the focus of hundreds of varied studies. It has been reported that in NOD mice, they are the earliest infiltrating lymphocytes (61). In T1D, B lymphocytes produce autoantibodies and it has been suggested that they act as potent APC in the development of the T-cell inflammatory response (16,48,56).
Islet resident DC were originally reported by Lacy and colleagues (18) with an estimation of 10–15 DC per islet. Recently, 2–5 DC per islet were observed in nondiabetic mouse strains (11); the DC were classified as immature, based on chemokine receptor expression, and were shown to present high levels of β-cell-derived peptides. Other cells have been visually identified and quantified as putative APC in islets through use of Ia and MHC II labeling (21,58). The presence of macrophages in islets from humans and rats with type 2 diabetes has also been demonstrated (15). An increased number of islet-associated macrophages was detectable in rats prior to disease onset, and these cells were shown to induce an increase in endocrine cell release of the proinflammatory cytokines IL-6, IL-8, GCSF, and MIP-1α (15). In rodent models, B lymphocytes have been shown to be critical APC for the initiation of autoimmunity (56). In this regard, treatment with an anti-CD20 antibody prevented disease progression (26), and mice missing B cells did not develop T1D (2,55,70).
Several studies in rodents have demonstrated the importance of APC in the acute rejection of a transplant. In many transplant models, including islet allografts, rejection has been associated with the activation status and number of DC, with a higher DC content leading to an increased likelihood of graft rejection (51,57). It has been demonstrated that removal or attenuation of the function of intraislet immune cells leads to decreased immunogenicity, both in vitro and in vivo (6,13,17,18,39,47,52,68). Conversely, donor APC may be necessary for allograft tolerance through the priming of the direct processing and presentation pathway. In this context, the migrating APC provide a critical source of donor antigen to T cells in the lymph node (LN), which, when confronted with the appropriate stimuli and cytokines, can create a tolerogenic, nonrejecting environment (3).
Recent imaging of the architecture of islets from various species demonstrated that rodent islets are distinctly different from human and nonhuman primate (NHP) islets (8,9). These interspecies structural differences have significant implications for islet physiology, as they translate into marked disparities in islet responses to physiological stimuli (10). The similarity in islet architecture and metabolism between NHP and humans make NHP islets a good model to study and understand human islets, in particular considering the paucity of transplant-quality human islets available for research. In this study we characterized APC, including DC, macrophages, and B lymphocytes, as well as T lymphocytes, in NHP islets in the native pancreas and after isolation.
Materials and Methods
Nonhuman Primates
Pancreata were recovered from eight male cynomolgus monkeys (Macaca fascicularis) of 4.4–7.8 kg (mean 6.6 ± 1.0) and 5–9.75 years of age (mean 6.8 ± 1.9). All animals were specific pathogen free, screened negative for TB, herpes B, SRV, SIV, and STLV-1, and were obtained from Charles River BRF, Inc. (Houston, TX) or the Mannheimer Primatological Research Foundation, Inc. (Homestead, FL). The University of Miami complies with the Animal Welfare Act of 1966 (PL89-544) as amended by the Welfare Act of 1970 (PL91-279), adheres to the principals stated in the NIH guidelines for the care and use of laboratory animals, and is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC). All donors demonstrated normal c-peptide responses to intravenous glucose tolerance testing prior to organ recovery.
Perifusion
In vitro functional capacity of all isolated islets was determined via assessment of dynamic glucose-stimulated insulin secretion using a column perifusion assay (29). The stimulation index (SI) was calculated as the ratio of insulin released under high glucose (11 mM) over insulin released under low glucose (3 mM) concentrations. The average SI for isolated islet preparations used was 6.8 ± 5.2, with a range of 1.9–15.7. We have previously shown a positive correlation between SI and early in vivo c-peptide levels, with an SI ≥ 2 resulting in functional insulin release (4).
Immunohistochemistry and Immunofluorescence
Pancreatic biopsies were collected prior to organ recovery from the tail of the pancreas, fixed in 4% paraformaldehyde (PFA; Electron Microscopy Sciences) in Hank's buffered salt solution (HBSS) (Invitrogen) for 6 h at 37°C, cryoprotected with increasing sucrose gradients, and frozen in Optimum Cutting Temperature compound (OCT; Tissue-Tek). The donor pancreas was recovered and flushed with UW Solution (Viaspan™, Belzer UW), and NHP islet isolation was performed as previously described (4,38). Immediately after isolation, islets were collected, washed in HBSS, and fixed in Bouins (Sigma) for 1 h, dehydrated in 70% ethanol, and embedded in paraffin. All sections (5 μm) were placed on glass slides, deparaffinized, and blocked with Universal Blocker Reagent (Biogenex) and Protein Block (Biogenex). Thereafter, sections were incubated with combinations of antibodies against: insulin, CD1a, CD8α, CD31 (AbCam), Ck19, HLA DR (Biogenex) (to measure MHC II), CD4, CD45, CD83 (Becton Dickinson), CD11b, CD20 (Beckman Coulter), CD68 (DakoCytomation), and IgG + IgM (Nordic Immunology). Immunostaining was visualized using Alexa Fluor conjugated secondary antibodies (Molecular Probes), and cell nuclei were stained with DAPI (Molecular Probes). Slides were mounted and coverslipped with ProLong Gold antifade reagent (Molecular Probes). As a negative control, the primary antibodies were omitted. Sections were viewed at 40x magnification using a Zeiss LSM 510 confocal microscope.
Cell Enumeration and Localization
Islets were selected randomly (3–5 per section) using nuclear staining (DAPI) as a guide. Only islets with an area of ≥ 1000 μm2 were included in the analysis (1000 μm2 is equal to 17.6 islet equivalents, defining one IEQ as an islet with a diameter of 150 μm, and therefore an area of 17662.5 μm2, assuming the islet is a circle and using the equation a = πr2). The average islet area was 11,709 μm2. All images from which cells were counted were digitally acquired and islet areas calculated using LSM Image Browser (9) from manually drawn borders. Positive cells were identified based on staining levels of ≥50% above background surrounding an identifiable nucleus.
Flow Cytometry
Isolated NHP islets were dissociated for flow cytometry by enzymatic digestion with 400–600 units Accutase (Innovative Cell Technologies) per 1000 IEQ (28). Single cell suspensions were incubated with antibodies for: CD3, CD4, CD11b, CD14, CD45, CD83 (Becton Dickinson), CD8, CD20, HLA DR (Beckman Coulter), CD1a (AbCam), and with 7AAD (Beckman Coulter) for viability. Control samples included: unstained cells, isotype antibody-stained cells, and single fluorochromestained cells. These controls were critical for defining the correct voltages and color compensation settings and accounting for the high amount of autofluorescence in islets. Cells were analyzed using a Beckman Coulter FC500 Flow Cytometer.
Statistical Analysis
Unless otherwise indicated, data represent mean ± SD. Statistical significance was calculated using the two-tailed Student's t-test or ANOVA, with a Tukey-Kramer Multiple Comparisons posttest; p < 0.05 was considered significant. Statistics were calculated using InStat statistical analysis software (GraphPad Software, Inc., La Jolla, CA).
Results
Identification of Immune Cells in Islets In Situ and in Isolated Islets
Molecules Used for Immune Cell Identification

Antibodies against cell surface molecules were used for both confocal imaging and flow cytometry.
CD11b and CD3 staining was utilized by flow cytometry, though not in fixed sections, as high nonspecific background limited exclusive identification. CD68 (unavailable for flow cytometry) was used to identify macrophages by confocal microscopy.
Number of APC and T Lymphocytes in Isolated NHP Islets

Values are mean ± SD from n = 5 isolated islet preparations, n = 2 pancreatic biopsies. Phenotype and quantity per IEQ based on enumeration by confocal microscopy (No. cells/area, including islets with zero per area) and flow cytometry (% of dissociated islet preparation). Values in parenthesis indicate relative proportion of each population within the total number of immune cells.
Counts in acinar tissue scaled to area of average islet (150 μ m diameter). NK cells were not included as they represent <0.4 cells/IEQ.
p < 0.05 compared to acinar tissue, by ANOVA.
Islets in situ in the native pancreas contained numerous MHC II+ (HLA DR) cells (Fig. 1). MHC II was a useful visualization tool in tissue sections, though it was not used to conclusively identify APC because its expression levels vary in DC populations and it is also present on endothelial cells. Panels I and III show staining in islets; panels II and IV the corresponding acinar tissue. The majority of the immune cells identified were mature DC or B lymphocytes (CD83+, CD20+, IgG + IgM+). Immature DC, macrophages, and T lymphocytes were also identified, though in limited numbers in comparison to mature DC and B lymphocytes. CD4 and CD8 were used to identify T lymphocytes, and CD16 to identify NK cells. CD31 and Ck19 were used to visualize endothelial cells and pancreatic ductal cells, respectively. The distribution of immune cells throughout the surrounding acinar tissue was consistent with that seen in islets, with mature DC and B lymphocytes being the primary tissue-resident APC populations. Macrophages (CD68+) were also present in both the in situ islets and acinar tissue. There were few T lymphocytes (CD4+ or CD8+) in the acinar tissue or in the endocrine islets in the native pancreas. A parallel immune cell composition and population distribution was seen in isolated NHP islets.
Confocal fluorescent analysis of NHP islets in the native pancreas stained for the different immune cells. For a visualization of the immune cell distribution in the whole pancreas, two representative images are shown for each cell type (a close-up view of an islet, columns I and III, and a view of the same tissue containing both acinar and islet tissue, columns II and IV; all at 40x magnification). Islets are identified with insulin (red), the nuclei with DAPI (blue), and the immune molecules with varying antibodies (MHC II, CD83, CD68, CD1a, CD20, IgG + IgM, CD4, CD8, CD16) or other cell populations (Ck19, ductal; CD31, endothelial) (green). Scale bars: 100 μm. Primary antibodies omitted in unstained control (scale equal to columns II and IV). H&E of corresponding pancreatic tissue, acquired at 50x (scale equal to columns I and III).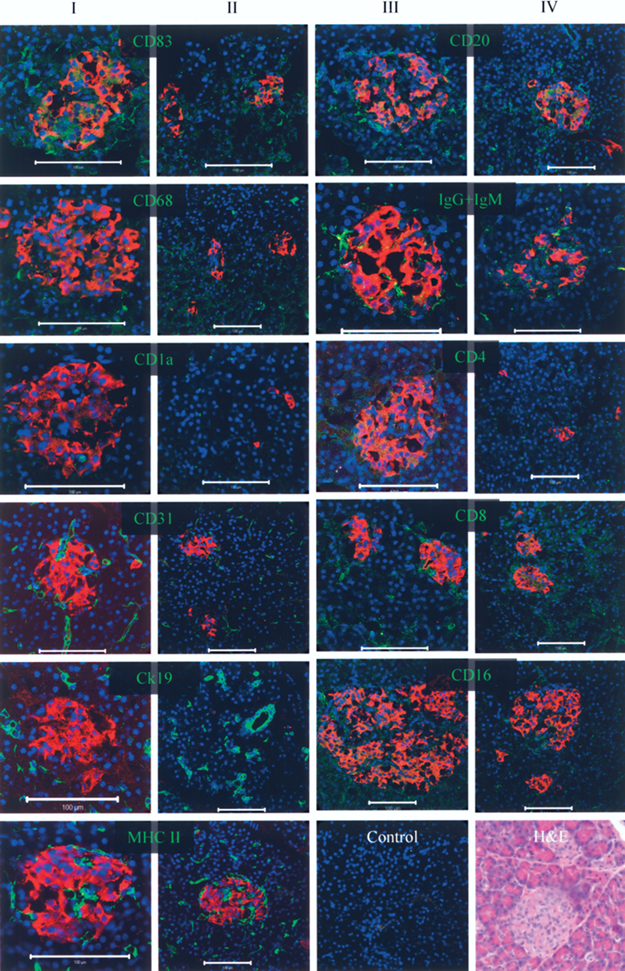
Enumeration of Immune Cells in Isolated Islets
Following identification of both APC and T lymphocytes in situ we next analyzed immune cells in isolated NHP islets. To thoroughly evaluate these populations, we carried out both confocal enumeration of whole isolated islets and four-color flow cytometric analysis of dispersed islets. Data from both microscopy and flow cytometry were essential to cell identification because we recognized that extra processing required to generate a single cell suspension (enzymatic dissociation) might alter the activation status, and thus modulate receptor expression on immune populations. Furthermore, reagent availability limited some dual staining for immune molecules by microscopy. The resulting data from the two methods were very similar. We determined (by averaging the values from confocal and flow cytometric evaluations) that one IEQ contained an average of 21.8 immune cells, 14.7 of which were APC (Table 2). One IEQ was defined as an islet of 150 μm in diameter containing 500 cells. Each IEQ contained an average of 7.6 DC (immature and mature), 2.9 macrophages, 3.8 B lymphocytes, and 7 T lymphocytes. In comparison, an equal area of pancreatic acinar tissue lacking islets contained an average of 2.8 DC, 2.4 macrophages, 1.5 B lymphocytes, and 3.5 T lymphocytes (Table 2). CD3−CD8+CD16+ NK cells comprised <0.4 cells per IEQ. CD45 was not used because despite testing numerous clones, we were not confident that CD45 monoclonal antibodies cross-reacted well in isolated or in situ islets. Similar to previously published results (11,18,66), we observed an average of 11.7 MHC II+ cells per IEQ, of which 60.4± 1.6% were CD31+, as determined by flow cytometry (data not shown).
An equal area of nonislet acinar tissue contained fewer numbers of all immune cells; each acinar IEQ area contained an average of 2.8 DC (immature and mature), 2.4 macrophages, 1.5 B lymphocytes, and 3.5 T lymphocytes. There were significantly fewer B lymphocytes in the acinar tissue, as compared to islets (p < 0.05).
Number of Immune Cells with Relation to Islet Size
As has been employed for various islet assessment strategies, isolated islets were grouped by diameter as small (50–150 μm) or large (150–300 μm) (43). We found that small islets had fewer immune cells compared to large isolated islets (Fig. 2); all individual immune cell populations increased with islet size (data not shown).
Immune cells in small and large isolated NHP islets. Counts in small islets (50–150 μm diameter, n = 353) and large (150–300 μm diameter, n = 56). ***p < 0.001 by unpaired t-test compared to small islets. Mean ± SD from n = 5 isolated islet preparations.
Localization of Immune Cells in Isolated Islets
Distribution of Immune Cells Within Small or Large Islets

Percent of islets per localization pattern, grouped by islet diameter: small (Sm): 50–150 μm, large (Lg): 150–300 μm. n = 5 isolated islet preparations.
Discussion
With a goal of identifying islet resident cells that can play a role in immunity, we characterized NHP islets for identity, quantity, and location of immune cells, including APC and lymphocytes. Because APC can potentiate both innate and specific immune responses, multiple approaches to altering the immunogenicity of isolated islets have been used to modulate or remove these cells. Such strategies include the use of UVB irradiation (39), anti-class II (anti-Ia) (17), or antidendritic cell-specific antibodies (18) and complement, CTLA4-Ig expression in isolated islets via gene transduction (18), culture of islets in high oxygen (6,13), at low temperature (47,52), or for an extended period of time (68). These diverse APC modification processes have resulted in the generation of tolerance in the rodent islet transplant model; while prolonged graft survival has been observed in canine recipients of UV modulated islets, tolerance was not achieved.
Rodent transplant studies suggest that allorejection is greatly dependent on the interaction of recipient CD8 cells with donor cells through direct antigen presentation, with the source of donor antigen on the surface of islet resident APC (23,60). A recent study indicated that these donor APC were present in the spleen of recipient mice long after the rejection of islet allografts, demonstrating their extended survival and potential role in the stimulation of tissue rejection (19). This trafficking of donor APC to lymphoid organs can trigger the stimulation of a specific response, which is further amplified if these migratory cells die and are phagocytosed and presented on host LN-resident APC (31).
In this study we identified all three classes of APC in isolated NHP islets, with roughly double the number of DC as compared to macrophages. This result was expected, as DC have the broadest range of antigen-presenting skills and trafficking, and macrophages are generally more stationary and are crucial scavengers and active in the culmination of a specific immune response (1). Our results of 11.7 MHC II+ cells per IEQ correspond to the number of MHC II+ or APC previously reported in islet studies (11,18,66).
When immature, DC continuously traffic through tissues, capturing soluble and particulate antigens (25). These particles are presented, primarily through receptor-mediated pathways, until the cells receive a combination of maturation stimuli including inflammatory cytokines, LPS, CpG, or CD40L. DC maturation and migration may also be induced by ischemia and/or transplantation, as indicated in the skin graft model (42). Once maturation occurs, DC reshape their chemokine receptors, decrease endocytosis, and produce the T-cell-stimulating factor IL-12 (53,64). Mature DC stimulate more T-cell proliferation than immature DC, as demonstrated by the mixed lymphocyte response (35). T cells that have been stimulated by immature DC are functionally impaired, in part because they lose their capacity to be further stimulated by mature DC and subsequently produce the anti-inflammatory cytokine IL-10 (35). Additionally, mature DC are more immunogenic when used to immunize mice (30,54,63). It is therefore possible that a transplant's potential for rejection can be associated with the activation status of the donor DC. The quantity of such DC may also play a role in tissue rejection. We identified both immature and mature DC in NHP islets. The average number of DC per IEQ was 7.8, half of which were immature, although dissociation of islets for flow cytometry resulted in a higher number of mature as opposed to immature DC, presumably due to activation during the dissociation process. Therefore, measures taken to prevent maturation of these cells through the islet isolation and transplant processes have the potential to decrease the immunogenicity of islets. An important observation in these studies was an increased number of all immune cells, most notably DC and B lymphocytes, in islets, as compared to the surrounding acinar tissue. This finding might be explained by the disproportionately large blood flow to islets (10–20%) within the pancreas relative to their low pancreatic mass (1–2%) (33,44,49). Studies of the islet vascular system have demonstrated this large blood flow, resulting from the glomerular-like structure, glucose-regulated blood flow control, and the vasoactive roles of some pancreatic hormones (5,7,34).
A recent report indicated an increase in the number of islet-associated macrophages in humans and rats with type 2 diabetes (15), which induced endocrine cell release of multiple proinflammatory cytokines. In islets from healthy NHP, we did not find much greater a number of islet-associated macrophages compared to acinar resident macrophages, with an average of 3.1 macrophages per NHP IEQ and 2.4 in the equivalent acinar area. The presence of macrophages in islets, as in all tissues, and their ability to induce a powerful cytokine release, indicates that in the right environment, they might potentiate the immune response by activating islet-resident immature DC. Additionally, macrophages are avid phagocytosers and central players in the induction and maintenance of chronic inflammation.
Though primarily responsible for the antibody-mediated clearance of extracellular pathogens, B lymphocytes are also APC. They can function by directly stimulating the T-cell response in the presence or absence of other APC. CD20 is expressed on the surface of B lymphocytes at all developmental stages, minus that of pro-B or plasma cell. In this study, CD20 was used to identify non-plasma cell B lymphocytes. In the setting of allogeneic transplantation, plasma cells could serve as an unwanted potent responding population, given the correct stimulus. The presence of numerous B lymphocytes in NHP islets, both in situ and after isolation, is intriguing, especially as the number of islet-associated B lymphocytes was significantly greater than the number found in an equivalent area of acinar tissue. B lymphocytes phocytes may act as a central player in the cell-mediated rejection of islets by directly stimulating recipient T cells. In rodent models, B lymphocytes have been shown to be critical APC for the initiation of autoimmunity (56). The ongoing use of anti-CD20 in the treatment of various autoimmune diseases, including T1D (48,62), may be effective in directly targeting these B lymphocytes and prevent progression to T1D, as has been shown to prevent progression to T1D in mice (26). Additionally, anti-CD20 therapy promoted long-term islet allograft survival in NHP (45). Even though “immunocytes” of isolated rat islets have previously been reported to contain B cells (66), our study shows the first detailed characterization of B lymphocytes in NHP islets in both the native pancreas and after isolation, with an average of 3.8 cells per IEQ.
There are a small number of T lymphocytes in NHP islets. While the concern for graft versus host disease is minimal due to the small population size, stimulation by host and islet-resident APC could result in immediate intraislet inflammation. Additionally, these T cells have the potential to act as immunogens for host APC. It is likely that T lymphocytes, NK cells, and B lymphocytes play a role in the inflammatory processes that lead to initiation of an islet directed immune response and progression to T1D.
Distribution of islet resident immune cells was explored. A polarity might indicate differential adhesion molecule expression or cytokine secretion. We have shown that the endocrine populations of NHP islets are distinctly different than those in rodent islets, with a lack of centralization of insulin + cells in NHP islets (8,9). Similarly, here we have shown the lack of an APC localization in islets. All islets were prepared for analysis immediately following isolation, therefore, the impact of culture on immune populations was avoided and it is unlikely that the lack of interior-only located APC or T cells was due to culture-related hypoxia or lack of nutrients, as has been previously documented (22).
In summary, we determined that there are an average of 14.7 APC per NHP IEQ. This value is similar to the number of APC reported in mouse islet studies. Moreover, to the best of our knowledge, this is the first study to delineate the contributions of all three classes of APC as well as T lymphocytes in a relevant preclinical model such as NHP islets. We have also shown that mature DC and B lymphocytes are the largest APC populations in isolated NHP islets, and that their numbers are greater than in equal sized areas of pancreatic acinar tissue. Finally, the number of islet-resident APC and lymphocytes increases with islet size. This knowledge will be useful to develop tailored modulation strategies to decrease immunogenicity, enhance engraftment, and ultimately prevent islet rejection. Experimental approaches to altering the immunogenicity of islets have been designed to block one or more of the myriad interactions that occur between T-cells and APC or to interfere with additional molecules on the immune cells. Based on these studies, demonstrating the cell types present in islets, we propose the use of agents to prevent dendritic cell maturation combined with the depletion of B lymphocyte populations to enhance islet engraftment.
Footnotes
Acknowledgments
This work was funded by the Diabetes Research Institute Foundation and was presented in part at the Immunology of Diabetes Society Conference of the American Diabetes Association in Miami, Florida, in 2007. The authors gratefully acknowledge the expert technical assistance of the following individuals: Dr. Norman Kenyon (surgeon); Mr. Alex Rabassa (islet isolations); Mr. Waldo Diaz and Mr. James Geary (veterinary team); the staff of the Imaging Core Facility (UM); Mr. Kevin Johnson (histology); Drs. Alejandro Caicedo and Over Cabrera (confocal staining and analysis guidance). The authors also gratefully acknowledge the input and manuscript review of Dr. Luca Inverardi.
