Abstract
Mesenchymal stem cells (MSCs) have the potential to repair and regenerate ischemic heart tissue; however, the poor viability of transplanted MSCs in the ischemic region is a major obstacle to their therapeutic use. This cell death is caused by Fas and Fas ligand (FasL) interactions under harsh conditions. To investigate improving the survival and therapeutic effects of MSCs, we focused our research on Fas–FasL-mediated cell death. In this study, we found that the poor viability of transplanted MSCs was caused by Fas–FasL interactions between host ischemic myocardial cells and implanted MSCs. In addition, we found that increased Fas expression and the corresponding decrease of cell survival were in close relation to hypoxic MSCs treated with FasL and H2O2. When MSCs were treated with a recombinant Fas/Fc chimera (Fas/Fc) inhibiting Fas–FasL interactions, the expressions of proapoptotic proteins including caspase-8, caspase-3, Bax, and cytochrome-c were attenuated, and the survival of MSCs was recovered. In ischemia–reperfusion injury models, the interaction between FasL in ischemic heart and Fas in implanted MSCs caused a loss of transplanted MSCs, whereas the inhibition of this interaction by Fas/Fc treatment improved cell survival and restored heart function. Thus, our study suggests that Fas–FasL interactions are responsible for activating cell death signaling in implanted stem cells and could be a potential target for improving therapeutic efficacy of stem cells in treating ischemic heart diseases.
Introduction
Although various treatment modalities for treating cardiac diseases have been advanced, heart failure remains a serious disease (11) due to limited regenerative capacity after myocardial infarction (MI) (20, 23, 35). Recent therapeutic approaches have used genes, growth factors, or cells to simply improve myocardial replacement (22). Upon recent advances in repairing ischemic heart using mesenchymal stem cells (MSCs), understanding the tissue microenvironment will help optimize the therapeutic potential of these cells (5). MSCs transplanted into an infarcted region of ischemic heart, however, face harsh conditions, such as calcium overload, acidosis, and reactive oxygen species (ROS), which induce apoptotic signaling and inhibit survival (14, 21, 25, 30). Ultimately, cell death in both transplanted cells and cardiomyocytes is induced by ROS production in the damaged heart (3, 19, 28).
Apoptosis can be induced by an extrinsic death receptor pathway or an intrinsic mitochondrial pathway, which causes organ failure in response to various harmful stimuli (16). Fas is a classical death receptor that belongs to the tumor necrosis factor (TNF) family, whereas Fas ligand (FasL) is a type II membrane protein that binds Fas (34). The interaction between Fas and FasL can occur on many cell types and triggers apoptosis (10, 34). Specifically, the interaction between Fas and FasL leads to caspase-8 cleavage activation from procaspase-8, which in turn activates caspase-3. FasL-mediated apoptosis is closely connected to various pathophysiological conditions, such as ischemia–reperfusion (I/R) injury and congestive heart failure (7).
We examined the relationship between Fas and FasL in a MI model. The treatment of recombinant membrane FasL in cardiomyocytes can activate Fas–FasL signaling and cause cardiomyocyte death (7). In addition, Fas is an important apoptotic signaling mediator that induces myocardial cell death during I/R (18). We showed that the lower survival rate of transplanted MSCs was due to the interaction between Fas in hypoxic MSCs and FasL in ischemic heart tissue. The antiapoptotic activity of recombinant Fas/Fc chimera (Fas/Fc) correlated with increased survival of transplanted MSCs in ischemic hearts. While hypoxic conditions inhibited cell survival, the interruption of Fas–FasL by pretreatment with Fas/Fc restored not only MSC survival but also heart function. These results suggest that Fas–FasL-mediated apoptosis is a critical cell death inducer of transplanted MSCs in ischemic heart and a promising target for improving therapeutic efficacy of MSCs in clinical trials.
Materials and Methods
In Vitro Ischemic Condition
After overnight incubation in cell culture conditions, neonatal rat ventricular cardiomyocytes and MSCs were washed twice with fresh serum-free Minimum Essential Medium α (α-MEM; Life Technologies Corporation, Grand Island, N Y, USA) and Dulbecco's modified Eagle's medium (DMEM; Life Technologies Corporation), respectively, and then placed in a hypoxic chamber (Technomart, Inc., Seoul, Korea) for 12 h. Hypoxic conditions of all our experiments were created in an anaerobic system with 5% CO2, 10% H2, and remainder N2. The oxygen level was ~0.5%. Various concentrations of hydrogen peroxide (H2O2; Sigma-Aldrich, St. Louis, MO, USA) were added to MSCs in DMEM supplemented with 10% fetal bovine serum (FBS; Life Technologies Corporation) at 37°C for 6 h. Various concentrations of recombinant FasL (EMD Millipore Corporation, Billerica, MA, USA) were used to treat both types of cells in media supplemented with 5% FBS for 6 h.
Isolation and Culture of MSCs
MSCs were purified as previously described (32). Briefly, bone marrow from the femurs and tibias of 4-week-old male Sprague–Dawley rats (100 g, Samtako, OSan, Korea) was aspirated in 10 ml of DMEM supplemented with 10% FBS and 1% antibiotic-penicillin/streptomycin solution (Life Technologies Corporation). Mononuclear cells were recovered by centrifuging in Ficoll (GE Healthcare, Munich, Germany), washing twice, resuspending in 10% FBS supplemented DMEM, and plating on 100-mm plates (Corning Incorporated, Corning, NY, USA). Cultures were maintained at 37°C in a humidified atmosphere containing 5% CO2. After 72 h, nonadherent cells were removed, and adherent cells were thoroughly washed twice with DMEM. The 10% FBS medium was changed every other day for 7 to 10 days. Cells were harvested after incubation with 0.25% trypsin-EDTA (Life Technologies Corporation) for 3 min at 37°C, replated onto 100-cm2 plates by 1 × 105 cells per plate, and then grown for approximately 5 days.
Isolation of Neonatal Rat Ventricular Cardiomyocytes
Cardiomyocytes were prepared from Sprague–Dawley neonatal rat hearts as previously described with minor modifications (8, 39). Isolated hearts were washed with Dulbecco's PBS solution (pH 7.4; Life Technologies Corporation) to deplete red blood cells. Using microdissecting scissors (Kent Scientific Corporation, Torrington, CT, USA), the hearts were minced to approximately 0.5 mm3 and treated with 4 ml of collagenase II (1.4 mg/ml, 270 unit/mg; Life Technologies Corporation) for 5 min at 37°C. The supernatant was removed, and the remaining tissue was treated with fresh collagenase II for an additional 5 min and repeated until the tissue was completely digested. The resulting supernatants were collected and centrifuged at 448 × g for 3 min at room temperature. The cell pellet was resuspended in 5 ml of cell culture medium and plated onto culture dishes (Corning Incorporated), then incubated for at least 2 h in a 37°C incubator containing 5% CO2. The adherent cells were fibroblasts, and the nonadherent cells were cardiomyocytes. Cardiomyocytes were plated on 100-mm culture plates (3 × 106 cells/ml) and incubated in α-MEM supplemented with 10% FBS. Cells were cultured in a 37°C incubator containing 5% CO2. To reduce fibroblast contamination, we used α-MEM with 0.1 mM 5-bromo-2′-deoxyuridine (Sigma-Aldrich) (17, 28–30).
Cell Viability
Cell viability was determined using a CCK-8 kit and a WST-8 [2-(2-methoxyl-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium, monosodium salt] assay (Dojindo, Kumomoto, Japan). MSCs were plated by 5 × 103 per well in triplicate wells in 96-well plates (Corning Incorporated) and incubated for 24 h. Media were changed using DMEM supplemented with 10% FBS. Absorbance was measured at 450 nm with a spectrophotometer (Bio-Rad, Hercules, CA, USA).
Coculture of MSCs with Cardiomyocytes
Hypoxic or normoxic MSCs and hypoxic cardiomyocytes were cocultured indirectly in two chambers separated by Falcon Cell Culture Inserts (BD Falcon, Franklin Lakes, NJ, USA) to allow secreted FasL from hypoxic cardiomyocytes to interact with Fas in MSCs. The semipermeable insert (0.4-μm pore size) allowed FasL to diffuse but inhibited cell migration between the two populations. Cardiomyocytes were plated in an upper chamber insert, and MSCs were cultured in a lower chamber of a six-well culture plate. MSCs were pretreated for 1 h with or without 1 μg/ml of recombinant rat Fas/TNFRSF6/Fc chimera (Fas/Fc; EMD Millipore Corporation) in 10% FBS to inhibit Fas–FasL interactions (38). After pretreatment with Fas/Fc, the upper chamber containing cardiomyocytes was inserted into the MSC-cultured plate. MSCs were cocultured with cardiomyocytes in a hypoxic chamber or normoxic cell culture incubator for 12 h. Then the survival of hypoxic and normoxic MSCs was measured.
Trichloroacetic Acid Protein Precipitation
Trichloroacetic acid (TCA; Sigma-Aldrich) precipitates were generated by adding two volumes of ice-cold 20% TCA while vortexing and then incubating for 30 min. Precipitates formed after 15 min at 4°C. The supernatant was removed and neutralized by adding 200 μl acetone (Sigma-Aldrich). The supernatant was centrifuged for 5 min at 4°C. Pellets were dried at room temperature for 10 min before adding SDS buffer. After heating at 65°C for 3 min, samples were loaded onto 10% SDS-PAGE gels.
RT-PCR Analysis
RNA was extracted using 1 ml TRIzol reagent (Sigma-Aldrich). Total cellular RNA was extracted by chloroform (Sigma-Aldrich), isopropanol (Sigma-Aldrich), and DEPC (Sigma-Aldrich)-treated 75% ethanol (Sigma-Aldrich). RNA was resuspended in 30 μl nuclease-free water. The quantity and quality of isolated RNA were determined by OD260/OD280 with a DU 640 spectrophotometer (Bio-Rad). Complementary DNA (cDNA) was synthesized with a Reverse Transcription System (Promega Corporation, Fitchburg, WI, USA) according to the manufacturer's instructions. One microgram of RNA was reverse transcribed in a 20-μl reaction containing 5 mM MgCl2, 10 mM Tris-HCl (pH 9.0 at 25°C), 50 mM KCl, 0.1% Triton X-100, 1 mM dNTP, 20 U RNase inhibitor, 0.5 μg oligo-(dT) primer, and 10 U reverse transcriptase for 15 min at 42°C, and then ended at 99°C for 5 min. PCR oligonucleotides were synthesized by Bioneer (Bioneer Corporation, Daejeon, Korea). To synthesize cDNA, an aliquot of the RT reaction mixture was subjected to PCR using GAPDH primers: 5′-ACCACAGTCCATGCCATCAC-3′ and 5′-TCCACC ACCCTGTTGCTG-3′ (450 bp). The Fas primers were 5′-TGCCTCCACTAAGCCCTCTA-3′ and 5′-CAAGACT GACCCCGGAAGTA-3′ (420 bp). PCR conditions were set to 95°C for 3 min; 95°C for 30 s with 35 cycles of dena-turation; 62°C for 30 s, 72°C for 90 s, and 72°C for 10 min as the final extension. During the first round of PCR, 1 μl of cDNA from the reverse transcription was added. PCR mix was made by adding 10 nM of each primer (Bioneer Corporation), 10× buffer [200 mM Tris-HCl (pH 8.8), 100 mM KCl, 1.5 mM MgSO4, 1% Triton X-100, 0.1 mM dNTP (Takara Bio, Otsu, Japan)], and 1.25 U Taq polymerase (Takara Bio) in a total mixture of 25 μl. All RT-PCR products were detected by electrophoresis in a 1.5% agarose gel (Bio-Rad) containing ethidium bromide (Sigma-Aldrich) and visualized using a Gel-Doc (Bio-Rad) (4).
Immunoblot Analysis
Cells were washed once in PBS and extracted using a lysis buffer (Cell Signaling Technology, Beverly, MA, USA) containing 20 mM Tris (pH 7.5), 150 mM NaCl, 1 mM Na2-EDTA, 1 mM EGTA, 1% Triton X-100, 2.5 mM sodium pyrophosphate, 1 mM β-glycerophosphate, 1 mM Na3VO4, 1 mg/ml leupeptin, and 1 mM phenylmethylsulfonyl fluoride (PMSF), mixed with a protease inhibitor (Roche, Basel, Switzerland), and a phosphatase inhibitor cocktail (Roche) at 4°C for 25 min. To separate cytoplasmic proteins, cells were first harvested by scraping in cold PBS. The cells were lysed in 10 mM hydroxyethyl piperazineethanesulfonic acid, 1.5 mM MgCl2, 10 mM KCl, 0.5 mM dithiothreitol (DTT), 0.5 mM PMSF, and 0.3% NP-40 (Sigma-Aldrich). The supernatant was collected by centrifugation (10,000 × g, 5 min, 4°C). Protein concentrations were determined using a BCA assay (Pierce Biotechnology, Rockford, IL, USA). Proteins were separated on 10% or 12% SDS-PAGE gels (Bio-Rad) and transferred to polyvinylidene difluoride membranes (Millipore, Billerica, MA, USA). After blocking with Tris-buffered saline (Sigma-Aldrich) with 0.1% Tween-20 (TBS-T; Sigma-Aldrich) and 5% nonfat dried milk (BD Biosciences, San Jose, CA, USA) for 1 h at room temperature, the membranes were incubated with primary antibody for 1 h at room temperature or overnight at 4°C. Antibodies for immun blots: monoclonal β-actin antibody obtained from Sigma-Aldrich; polyclonal caspase-8 antibody from Abcam (Cambridge, UK); polyclonal Fas antibody from Millipore; monoclonal FasL; monoclonal cytochrome-c, monoclonal Bcl-2, and polyclonal Bax antibody from Santa Cruz Biotechnology (Santa Cruz, CA, USA); and mouse or rabbit horseradish peroxidase-conjugated secondary antibodies from Santa Cruz Biotechnology were purchased. All antibodies were diluted by 1:1,000 with TBS-T and 5% nonfat dried milk. Membranes were washed three times with TBS-T for 5 min and incubated for 1 h at room temperature with horseradish peroxidase-conjugated secondary antibodies (Santa Cruz Biotechnology). After extensive washing, bands were detected by enhanced chemiluminescence reagent (Amersham Biosciences, Amersham Pharmacia Biotech, Tokyo, Japan). Band intensities were detected with a photo image system (Molecular Dynamics, Sunnyvale, CA, USA).
Fluorescence-Activated Cell Sorting
MSCs were prepared according to the fluorescence-activated cell sorting (FACS) staining procedure described below. Briefly, cells were detached from the plate with 0.25% trypsin-EDTA, washed in PBS, and fixed in 70% ethanol at 4°C for 30 min with agitation. Cells were washed twice in PBS and resuspended in blocking buffer (1% BSA, 0.1% FBS) containing antibodies against Fas and FasL. The cells were then washed twice and labeled with FITC-conjugated goat anti-rabbit IgG (Jackson ImmunoResearch, West Grove, PA, USA) and PerCP-conjugated goat anti-mouse IgG (Jackson ImmunoResearch) for 20 min in a dark area. After two more washes, flow cytometry was performed on a FACS Calibur system using CellQuest™ software (Becton Dickinson, San Jose, CA, USA) with 10,000 events recorded for each sample.
Caspase-3 Assay
Relative caspase-3 activity was determined by using caspase-3 activity assay kit (Roche). In brief, after different treatments, the cultured MSCs (2 × 106) were extracted in 1 × DTT for 1 min. Lysates were collected and loaded onto a plate coated with 20× monoclonal caspase-3 antibody. Upon substrate cleavage, free fluorescent AFC (7-amino-4-trifluoromethylcoumarine) was quantified using a microplate reader (Bio-Rad).
Induction of Myocardial Infarction and Transplantation
MI was produced in male Sprague–Dawley rats (250 ± 30 g) by surgical occlusion of the left anterior descending coronary artery (LAD) as previously described (31). Cells to be transplanted (1 × 106 cells) were suspended in 30 μl PBS and injected into the border zone of the injured region using a Hamilton syringe with a 30-gauge needle (Hamilton Company, Hamilton, OH, USA). Throughout the operation, animals were ventilated with 95% O2 and 5% CO2 using a Harvard ventilator (Harvard Apparatus, Holliston, MA, USA). Five animals per group (ligation, MSCs, and Fas/Fc-MSCs) were used for morphologic and functional analyses after occluding the left coronary artery. DAPI staining was performed for 30 min. The cells were rinsed six times in PBS to remove all excess DAPI (Life Technologies Corporation). Labeled cells were then detached with 0.25% (w/v) trypsin and suspended in PBS for grafting. All experimental procedures of our animal studies were approved by the Institutional Animal Care and Use Committee (approval No. 2012-0197) at Yonsei University College of Medicine and were performed in accordance with the Committee's Guidelines and Regulations for Animal Care.
Left Ventricular Catheterization
To determine invasive hemodynamics, left ventricular catheterization was performed 3 weeks after the infarction. A Millar Mikro-tip 2 F pressure–volume transducer (model SPR-838; Millar Instruments, Houston, TX, USA) was introduced into the left ventricle via the right carotid artery under anesthesia. All data were analyzed off-line with PVAN 3.5 software (Millar Instruments).
Histology and Determination of Fibrosis Area
Rats were sacrificed at various time points after implantation, and their hearts were excised. Tissues were fixed in 3.7% buffered formaldehyde (Sigma-Aldrich) and embedded in paraffin. Tissue sections, 5 μm thick, were deparaffinized, dehydrated, and rinsed with PBS. Antigen retrieval was performed with 10 mM sodium citrate (pH 6.0; Sigma-Aldrich) in a microwave for 10 min. Sections were incubated in 3% H2O2 to quench endogenous peroxidase activity. Samples were blocked in 2.5% normal horse serum (Vector Laboratories, Burlingame, CA, USA) and incubated with monoclonal CD31 antibody (Santa Cruz Biotechnology). Finally, slides were biotinylated with a pan-specific universal secondary antibody (Vector Laboratories). The sections were stained with methyl green (Sigma-Aldrich) to assess cytologic details, such as nuclei, cytoplasm, and connective tissue. Capillaries were counted in 10 randomly chosen fields from two slides. The mean number of capillaries per field in the infarcted myocardium was used for statistical analysis. Additionally, fibrosis was analyzed by Masson's trichrome staining kit (Sigma-Aldrich). The images for the trichrome-stained whole heart sections were obtained by a stereomicroscope (SZ6045TR/DP12; Olympus, Tokyo, Japan), and the magnified images for the trichrome-stained heart sections, and the images for CD31 were obtained by virtual microscopy (BX51/dot Slide; Olympus). Serial sections were analyzed with anti-mouse CD90 and anti-rabbit FasL from Santa Cruz Biotechnology. All antibodies were diluted in 1:500 by 1.5% normal horse serum. FITC-conjugated goat anti-mouse IgG (Jackson ImmunoResearch Laboratories) and rhodamine-conjugated goat anti-rabbit IgG (Jackson ImmunoResearch Laboratories) were used as secondary antibodies. All images were made using an excitation filter under laser scanning confocal microscopy (LSM 700; Carl Zeiss, Thornwood, NY, USA) and transferred to a computer with MetaMorph software version 4.6 (Universal Imaging Corporation, Downtown, PA, USA). Results are expressed as a percentage of the total left ventricle.
Terminal Deoxynucleotidyl Transferase-Mediated dUTP Nick-End Labeling (TUNEL) Assay
The TUNEL assay was performed according to the manufacturer's instructions (Chemicon International, Temecula, CA, USA). In brief, excised heart tissues were fixed in 3.7% buffered formaldehyde and embedded in paraffin. Tissue sections were deparaffinized, rehydrated, and rinsed with PBS. A positive control sample was prepared from a normal heart section by treating the section with DNase I (10 U/ml; Roche) for 10 min at room temperature. The sections were pretreated with 3% H2O2 (Sigma-Aldrich), subjected to a reaction with the TdT enzyme (Vector Laboratories) at 37°C for 1 h, and incubated with digoxigenin-conjugated nucleotide substrate (Vector Laboratories) at 37°C for 30 min. Nuclei exhibiting DNA fragmentation was visualized after staining with 3,3-diaminobenzidine (Vector Laboratories) for 5 min. The nuclei of apoptotic cardiomyocytes were stained dark brown. The sections were counterstained with methyl green, coverslipped, and observed by light microscopy. All images were obtained using a virtual microscopy.
Statistical Analysis
All quantified data are an average of at least triplicate samples and analyzed by Prism (GraphPad Software, Inc., La Jolla, CA, USA). Error bars represent the standard errors of the mean. Statistical significance was confirmed by Student's t-test, with p < 0.05 considered significant. Comparisons between more than two groups were performed by one-way ANOVA using Bonferroni's correction. A value of p < 0.05 was considered significant.
Results
FasL Is Secreted Under Hypoxic Condition
The soluble FasL is a cytokine that mediates apoptosis by interacting with Fas (12). To determine whether expression and secretion of FasL increased in hypoxic cardiomyocytes, FasL expression was examined in hypoxic and normoxic cardiomyocytes by TCA protein precipitation and immunoblot. Increased FasL expression and secretion were detected in I/R heart tissue and hypoxic cardiomyocytes, respectively (Fig. 1A, B). Next pro- or antiapoptotic signals were investigated in hypoxic and normoxic cardiomyocytes. The proapoptotic protein cytochrome-c was present in more abundance in FasL-treated hypoxic cardiomyocytes than in normoxic or hypoxic controls. The ratio of Bcl-2/Bax expression also decreased in FasL-treated cells (Fig. 1C, D).

FasL is secreted under hypoxic conditions. (A) FasL was identified in an ischemia–reperfusion injury model. (B) Hypoxic cardiomyocytes secreted FasL into hypoxic cardiomyocyte-conditioned media. Cardiomyocytes were incubated in hypoxic conditions for 12 h. (C, D) FasL-mediated apoptosis signals were detected in normoxic and hypoxic cardiomyocytes. Expression levels of (C) cytochrome-c, (D) Bcl-2, and Bax were detected by Western blot; 100 nM FasL was used for treatment. Values are the average of three measurements with the SD indicated by error bars. *p < 0.05 versus control.
Fas Is Activated Under Hypoxic Conditions
Hypoxic conditions produced cellular ROS that is known to induce cell death. However, it is well known that low levels of ROS modulate cell survival (37). Thus, to confirm whether ROS changed Fas expression and cell survival, cells were incubated with H2O2 in various concentrations. Fas expression increased in a dose-dependent manner up to 250 μM H2O2 (Fig. 2B). To detect activated Fas expression in MSCs under hypoxic conditions, cells were incubated in hypoxia. Increased Fas expression was observed until 6 h in hypoxic conditions (Fig. 2C). The results indicate that Fas is activated in 250 μM H2O2-pretreated MSCs; however, cell survival still increased in Fas-activated cells.

Fas is expressed under hypoxic conditions. (A) The effect of FasL on the survival of MSCs pretreated with 250 μM H2O2 was measured by CCK-8. (B, C) Fas expression in (B) H2O2-treated MSCs or (C) hypoxic MSCs was detected by RT-PCR. Values are the average of three measurements with the SD indicated by error bars. *p < 0.05 versus control.
Inhibiting Fas–FasL Interaction Regulates Survival of Hypoxic MSCs
To confirm FasL-induced apoptosis, MSCs were treated with FasL (FasL-MSCs). FasL induced apoptosis in H2O2-pretreated MSCs; however, MSCs without H2O2 treatment showed cell viability similar to that of the control even with FasL treatment (Fig. 3A). These results indicate that the interaction between Fas and FasL induces MSC apoptosis. In MSCs treated with Fas/Fc (Fas/Fc-MSCs), cell survival improved compared to FasL-treated MSCs (Fig. 3B). In addition, the ratio of Fas-positive to FasL-positive cells was lower in Fas/Fc-MSCs than in FasL-MSCs (Fig. 3C). To confirm the effects of Fas/Fc at the cellular level, the survival of hypoxic MSCs was measured using an in vitro coculture system. The results showed an insignificant effect on cell survival under normoxic conditions, whereas higher survival rates were portrayed in hypoxic Fas/Fc-MSCs than in hypoxic MSCs with hypoxic cardiomyocytes (Fig. 3D). Next, we confirmed that blocking the Fas system in hypoxic MSCs changed the expression of apoptosis-related proteins. Caspase-8 expression and caspase-3 activity increased in FasL-MSCs and were attenuated in Fas/Fc-MSCs (Fig. 4A, B). Further, expression of proapoptotic proteins, such as cytochrome-c and Bax, increased in FasL-MSCs and decreased in Fas/Fc-MSCs. Also, expression of an antiapoptotic protein, Bcl-2, was higher in Fas/Fc-MSCs than in FasL-MSCs (Fig. 4C-E).

The inhibition of Fas–FasL interaction by rat Fas/TNFRSF6/Fc chimera (Fas/Fc) modulated the expression and activity of apoptosis-related proteins and increased MSC survival. (A) Survival of FasL-MSCs was measured by CCK. MSCs were pretreated with H2O2. (B) Fas and FasL expression was detected by FACS. (C) The survival was calculated by CCK assay. (D) FasL-induced apoptosis was detected in hypoxic or normoxic MSCs cocultured with hypoxic cardiomyocytes. Cells were pretreated with 1 μg/ml Fas/Fc and then with 100 ng FasL. Then, all samples were treated with 250 μM H2O2. Values are the average of three measurements with the SD indicated by error bars. *p < 0.05 versus control.

Apoptotic signaling in Fas/Fc-treated MSCs. Expressions of apoptotic signals including (A) caspase-8, (B) caspase-3, (C) cytochrome-c, (D) Bcl-2, and Bax were detected by Western blot. Cells were pretreated with 1 μg/ml Fas/Fc in 10% DMEM and then with 100 ng FasL. Then, all samples were treated with 250 μM H2O2. Values are the average of three measurements with the SD indicated by error bars. *p < 0.05 versus control.
Intramyocardial Injection of Fas/Fc-MSCs Restores the Cardiac Function of Infarcted Myocardium
To determine the therapeutic effects of Fas/Fc-MSCs in ischemic heart tissue, we implanted DAPI-stained Fas/Fc-MSCs into the border region after LAD ligation (Fig. 5A). Survival of DAPI-stained Fas/Fc-MSCs was approximately twofold higher than that of DAPI-stained MSCs (Fig. 5B). Additionally, FasL expression and survival of transplanted MSCs were detected by fluorescence immunostaining. The expression of FasL decreased, and the survival of transplanted MSCs, which was detected by the MSC marker CD90, increased in Fas/Fc-MSC-injected heart compared to those of MSC-injected heart (Fig. 5C). Indeed, fibrosis decreased in the ischemic heart injected with Fas/Fc-MSCs (Fig. 6A). TUNEL-positive cells were also reduced in Fas/Fc-MSCs than in MSCs of the sham operation (Fig. 6B). The number of CD31-positive cells per field in the infarcted myocardium was higher in the heart transplanted with Fas/Fc-MSCs than in other groups (Fig. 6C). Moreover, heart function was better in rats injected with Fas/Fc-MSCs than those injected with untreated MSCs. At baseline, left ventricular pressure and volume did not differ significantly between the control groups. Fas/Fc-MSC-implanted heart had better stroke volumes, end diastolic volumes, end-systolic volumes, rise in maximum rates of pressure (dV/dt max), and rise in minimum rates of pressure (dV/dt min). Transplanting Fas/Fc-MSCs improved the ejection fraction as an indicator of heart function (Table 1). Finally, we demonstrated that Fas–FasL interaction is a major cause of apoptosis among transplanted MSCs in the ischemic heart. When MSCs are injected into the ischemic heart, ROS is increased, which in turn stimulates Fas expression in implanted cells. Then, secreted FasL from ischemic heart tissue binds with Fas in transplanted cells and results in cell death. Thus, inhibiting the Fas–FasL interaction by pretreating MSCs with Fas/Fc increased survival of implanted cells and improved heart function after myocardial infarction. These results indicate that blocking Fas–FasL interaction plays an important role in restoring donor cell and host cell survival (Fig. 7).
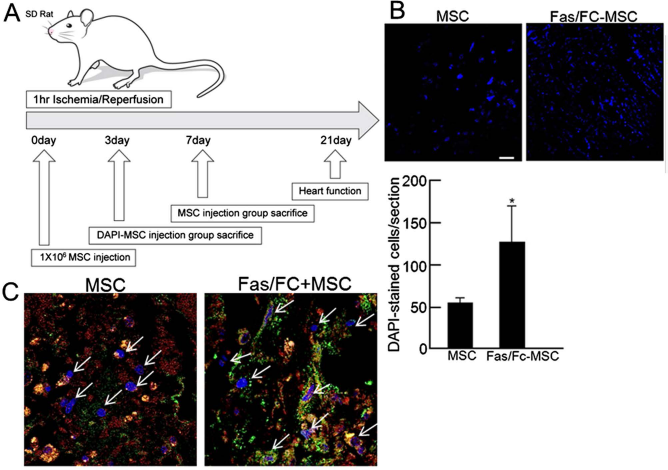
Transplantation of Fas/Fc-MSCs into the ischemic heart improved heart function. (A) Schematic diagram of MSC transplantation in Sprague–Dawley rat. (B) After 3 days, DAPI-stained MSCs were detected in the ischemic myocardium. Scale bars: 50 μm. Values are the average of three measurements with the SD indicated by error bars. *p < 0.05 versus control. (C) FasL in ischemic myocardium interacted with implanted MSCs. DAPI: Blue, CD90: Green, FasL: Red (image magnification: 400×).

Histological analysis of Fas/Fc-transplanted ischemic heart. (A–C) After 7 days, histology was studied in the I/R models. (A) Masson's trichrome staining showed fibrotic tissue (purple) in the ischemic myocardium. Sclae bars: 1 mm. (B) Normal nuclei (green) were stained using methyl green, and TUNEL-positive cells were stained brown (image magnification: 200x), indicating apoptotic cells. (C) CD31+ cells were stained brown (image magnification: 200×). Scale bars: 50 μm. Values are the average of three measurements with the SD indicated by error bars. *p < 0.05 versus control.

The Fas–FasL interaction between transplanted MSCs and ischemic myocardium. Transplanted MSCs expressed Fas that binds FasL secreted from the ischemic myocardium. This mechanism induces cell death in implanted cells. However, blocking the Fas–FasL interaction by Fas/Fc increased the survival of transplanted MSCs and restored heart function.
Effect of Fas/Fc-MSCs on Cardiac Function in Ischemia-Reperfusion (I/R) model

EF, ejection fraction; ESV, end-systolic volume; EDV, end-diastolic volume; SV, stroke volume; maximum dV/dt and minimum dV/dt. All values are expressed as mean ± SEM (*p < 0.05 vs. I/R group, †p < 0.001 vs. I/R. group).
Discussion
Recent investigations have suggested adult stem/progenitor cells as a promising source for stem cell therapy. Stem cell therapy regenerates ischemic myocardium and has been clinically effective in human trials (41). However, a high percentage of donor cells died within few hours or days after transplantation, which must be resolved to improve therapeutic potential and clinical outcomes for successful stem cell therapies (24). Engrafted stem cells encounter harsh conditions that induce apoptosis due to loss of interaction between cells and the extracellular matrix (21). Thus, poor cell survival in the harsh conditions of the transplantation site remains a major problem. Recently, various researches attempted to improve stem cell survival by various techniques, such as overexpressing chitosan (19) or periostin (6) or modifying stem cells with atorvastatin (2). Despite the improved myocardial regeneration within these systems, the major apoptotic scheme of transplanted stem cells in the ischemic heart is not clear and there has not been an effective method to avoid programmed cell death due to the loss of matrix attachment (13). Therefore, our study focused on the enhanced survival of transplanted MSCs by avoiding cell death in the ischemic heart.
Hypoxia induces apoptosis by increasing Fas expression in various cell types (7, 31, 40). In addition, hypoxia induces ROS generation, which stimulates the Fas-mediated apoptotic signaling pathway (27). During oxidative stress, apoptosis is induced by Fas, and caspase subtypes are activated by proteolytical cleavage of critical cellular proteins, ultimately leading to cell death (26, 36). The present study demonstrates that the inhibition of Fas in stem cells significantly downregulates expression of caspases and apoptosis-related proteins (Bax, cytochrome-c) relative to nontreatment. Accordingly, we found that Fas–FasL interactions in hypoxic myocardium are responsible for the death of transplanted MSCs. The Fas–FasL complex was found also to be a critical mediator in myocardial injury (1, 15, 19). Fas-mediated apoptosis is sparked by FasL binding, which initiates the caspase cascade (9). In our study, we proved that although exogenous FasL was treated into the stem cell-cultured medium, stem cell survival was inevitably regulated by Fas expression. When Fas–FasL interaction was blocked using Fas/Fc, despite FasL pretreatment, caspase-3 activity decreased, and the expressions of proapoptotic proteins, including caspase-8, Bax, and cytochrome-c were attenuated. Additionally, antiapoptotic protein Bcl-2 expression increased, and cell survival of Fas/Fc-MSCs improved compared to that of FasL-MSCs. Indeed, when cocultured with cardiomyocytes and MSCs, survival of MSCs treated with Fas/Fc increased in hypoxic conditions. Thus, the blocking of Fas–FasL interaction on MSCs was important for survival. The results of in vivo experiments using I/R models showed that Fas expression in MSCs, which were injected into ischemic rat hearts, was increased. Importantly, blocking Fas–FasL interactions by pretreatment of Fas/Fc increased survival of implanted MSCs and improved heart function after myocardial infarction. The enhanced cardiac function of ischemic heart by transplanted MSCs is supplemented not only by direct differentiation into cardiomyocytes but also by providing various secreted factors (33). These results indicated that Fas–FasL interaction plays an important role for transplanted stem cell death in ischemic heart.
The new finding of this study is that the main cause of apoptosis of transplanted MSCs into the ischemic heart is from Fas–FasL interactions. Treating stem cells with Fas/Fc might make them a better therapeutic source for regenerating ischemic myocardium. While genetic modifications and exogenous growth factors are possible methods to enhance cell survival, poor survival is still a major problem. Moreover, the rate of Fas and FasL generation in other cellular responses and pertaining side effects in the ischemic zone are not well known. Further study is required to improve solutions to these problems. Therefore, a new strategy for inhibiting the Fas–FasL interaction is a good therapeutic target for improving stem cell survival and desirable clinical outcomes in ischemic human heart.
Footnotes
Acknowledgments
This research was supported by a Korea Science and Engineering Foundation grant funded by the Korean government (MEST) (2011-0019243, 2011-0019254) and a grant from the Korea Health 21 R&D Project, Ministry of Health & Welfare, Republic of Korea (A120478). The authors thank Dong-Su Jang for his support with medical illustrations. The authors declare no conflicts of interest.
