Abstract
Mononuclear stem cells have been studied for their potential in myocardial ischemia. In our previous published article, ReACT® phase I/II clinical trial, our results suggest that a certain cell population, promonocytes, directly correlated with the perceived angiogenesis in refractory angina patients. This study is ReACT's clinical update, assessing long-term sustained efficacy. The ReACT phase IIA/B noncontrolled, open-label, clinical trial enrolled 14 patients with refractory angina and viable ischemic myocardium, without ventricular dysfunction, who were not suitable for myocardial revascularization. The procedure consisted of direct myocardial injection of a specific mononuclear cell formulation, with a certain percentage of promonocytes, in a single series of multiple injections (24—90; 0.2 ml each) into specific areas of the left ventricle. Primary endpoints were Canadian Cardiovascular Society Angina Classification (CCSAC) improvement at the 12-month follow-up and ischemic area reduction (scintigraphic analysis) at the 12-month follow-up, in correlation with ReACT's formulation. A recovery index (for patients with more than 1 year follow-up) was created to evaluate CCSAC over time, until April 2011. Almost all patients presented progressive improvement in CCSAC beginning 3 months (p = 0.002) postprocedure, which was sustained at the 12-month follow-up (p = 0.002), as well as objective myocardium ischemic area reduction at 6 months (decrease of 15%, p < 0.024) and 12 months (decrease of 100%, p < 0.004) The recovery index (n = 10) showed that the patients were graded less than CCSAC 4 for 73.9 ± 24.2% over a median follow-up time of 46.8 months. After characterization, ReACT's promonocyte concentration suggested a positive correlation with CCSAC improvement (r = −0.575, p = 0.082). Quality of life (SF-36 questionnaire) improved significantly in almost all domains. Cost-effectiveness analysis showed decrease in angina-related direct costs. Refractory angina patients presented a sustained long-term improvement in CCSAC and myocardium ischemic areas after the procedure. The long-term follow-up and strong improvement in quality of life reinforce effectiveness. Promonocytes may play a key role in myocardial neoangiogenesis. ReACT dramatically decreased direct costs.
Keywords
Introduction
Refractory angina refers to patients who have disabling and continued angina, usually Canadian Cardiovascular Society Angina Classification (CCSAC) class III/IV, and objective evidence of reversible myocardial ischemia despite optimum medical therapy. Additionally, these patients are not candidates for revascularization, either by percutaneous transluminal coronary angioplasty (PTCA) or coronary artery bypass grafting (CABG).
In the US, as many as 1.7 million patients are believed to have refractory angina (13). Data from the Cleveland Clinic from 500 consecutive coronary angiograms showed that 12% of the cases were not suitable for conventional revascularization techniques in patients with evidence of myocardial ischemia (22). European center reports also suggest that approximately 15% of patients with chronic coronary artery disease have refractory angina (20).
Severe angina pectoris often results in a substantial decrease in quality of life. Surprisingly, the vast majority of patients with refractory angina (75%) have preserved left ventricular function and a mortality rate lower than the general coronary artery disease population (21). As a result of that, this group is rapidly growing.
Economical impact and costs are difficult to be measured, but if we consider that in 2006, $11.7 billion was paid to Medicare beneficiaries for in-hospital costs when coronary heart disease was the principal diagnosis (direct costs) (27), we could estimate an annual cost of approximately $1.75 billion (15% of total coronary heart disease costs) for refractory angina patients.
Symptom relief for these “no option” refractory angina patients is a complex and challenging process. Alternative therapies as described in the American Heart Association Guidelines, such as surgical laser transmyocardial revascularization, external counterpulsation, and spinal cord stimulation, have all provided modest results at best (11,16,27).
Previous preclinical and clinical studies have supported the feasibility, safety, and potential of stem cell therapy for myocardium tissue regeneration. These trials encompass patients presenting with a wide range of coronary artery disease diagnoses, from acute myocardial infarction to chronic ischemic heart disease (5,23,29,31,33).
Recent clinical studies involving refractory angina patients, including myocardial administration of bone marrow-derived mononuclear stem cells (BMMSCs), have been associated with improvement in angina symptoms and exercise capacity, as well as increase in myocardial perfusion (1,2,4,18,19,24,25,30,32). Most of these trials had their primary endpoints met at the 6-month follow-up. If we consider the possibility of placebo effect after any proposed treatment for a disease such as refractory angina, a longer follow-up time is certainly desired to assess therapeutic effectiveness. Additionally, direct intracoronary injection was the route of stem cell administration used by most of these studies, which may prove to be substantially less effective than the intramyocardial route, in terms of heart stem cell uptake. Standardized and good cell manipulation practices, in compliance with the Food and Drug Administration guidelines, is a fundamental guidance for clinical trials, as well as a cardinal requirement for future translation of clinical trial results to a routine medical practice.
Our group recently published the initial clinical results of a proprietarily designed protocol, the CellPraxis Refractory Angina Cell Therapy (ReACT®) Protocol, in which a single series of multiple intramyocardial injections of a specific BMMSC formulation was performed as the sole surgical therapy in eight patients, treated from September 2005 to July 2007 (10). Inclusion criteria for this study required patients to be properly diagnosed with refractory angina pectoris, with viable myocardium by stress technetium scintigraphy, preserved left ventricular function (ejection fraction > 45%), and who were not suitable for myocardial revascularization (either PTCA or CABG). Almost all patients presented progressive improvement in angina class by 3 months (p = 0.008) postprocedure, which was sustained at the 18-month follow-up (p = 0.004), as well as objective myocardium ischemic area reduction at 12 months (decrease of 84.4%, p < 0.004). Moreover, specific stem cell type analysis revealed a positive correlation between bone marrow promonocyte concentration and CCSAC improvement at the 18-month follow-up (r = −0.759, p < 0.05), supporting a dose-related efficacy and suggesting neoangiogenesis occurred as a direct result of stem cell administration. This first study, phase I/IIA, also assured the procedure safety.
The myocardial angiogenic effect of monocyte stem cell lineages has also been documented by other reports. Bouchentouf et al. (3) demonstrated that transplanted monocyte derivatives and their secretome participate in preserving functional myocardium after ischemic insult and attenuate pathological remodeling in a rat model of myocardial infarction.
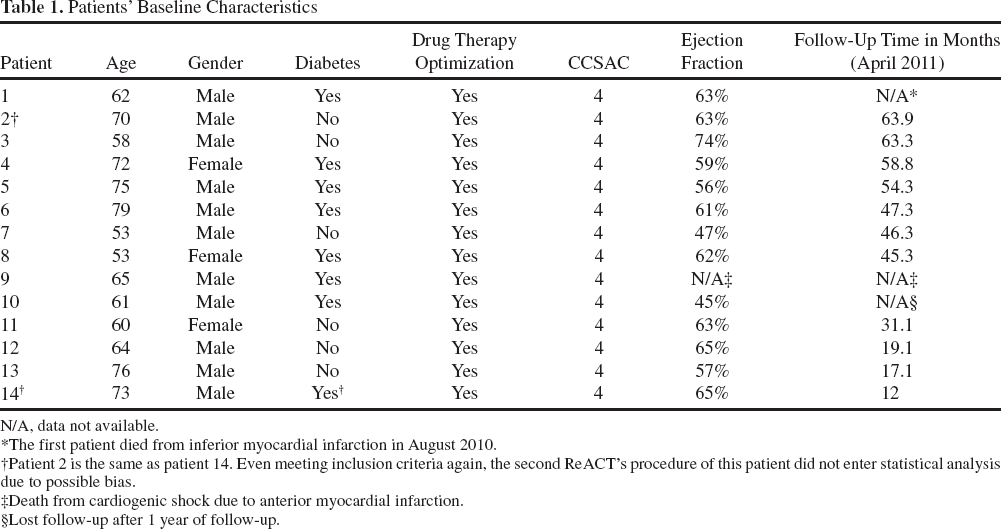
We now present the results of a larger number of patients, with follow-up time greater than 1 year, for all patients enrolled between September 2005 and April 2011. Patients' baseline characteristics are described in Table 1.
Patients' Baseline Characteristics
N/A, data not available.
The first patient died from inferior myocardial infarction in August 2010.
Patient 2 is the same as patient 14. Even meeting inclusion criteria again, the second ReACT's procedure of this patient did not enter statistical analysis due to possible bias.
Death from cardiogenic shock due to anterior myocardial infarction.
Lost follow-up after 1 year of follow-up.
Materials and Methods
We conducted a Phase IIA/B noncontrolled open-label clinical trial and included a further five patients and a longer follow-up time (48 months) (10). The Federal University of São Paulo ethical committee approved the study, and all patients gave informed consent prior to enrollment.
Study Population
Refractory angina patients, routinely undergoing treatment at the São Paulo Hospital, in São Paulo, Brazil, a tertiary referral Federal University Hospital for coronary heart disease, were included in the study. Refractory angina was defined as patients presenting with angina class IV (symptoms at rest) according to the CCSAC despite optimum medical therapy, not amenable for conventional myocardial revascularization, and viable myocardium confirmed by baseline nuclear imaging. Ineligibility for revascularization—-either percutaneous or surgical—-had been determined by at least two cardiologists and two cardiovascular surgeons based on the most recent (within 6 months) patient's coronary angiogram. Exclusion criteria were (1) LVEF < 45% on transthoracic echocardiogram; (2) absence of viable myocardium on cardiac nuclear imaging; (3) positive serologic tests for HIV, types A, B, and C hepatitis, human T-cell lymphotropic virus, or Chagas disease; (4) significant heart valve disease; (5) chronic renal disease on dialysis; (6) abusive use of alcohol or drugs; (7) any other medical condition with estimated survival <5 years; (8) participation in prior cell therapy studies in the past 2 years; and (9) pregnancy.
Study Procedures
For each patient, a total of 100 cc of bone marrow was aspirated from the iliac crest under general anesthesia and stored in a saline solution (Baxter, São Paulo, Brazil) with 80 IU heparin/ml (Heptar; Eurofarma, São Paulo, Brazil). Mononuclear cells were isolated by Ficolllike density gradient (Cellpraxis, São Paulo, Brazil) and diluted to a final concentration of 107 cells/ml, in accordance with the protocol's operational guidance, using autologous plasma as vehicle for cell injection. Laminar flow: FLV—CL II—A1—KE (TROX Technik, Neukirchen-Vluyn, Germany); centrifuge: Jouan CR 3.12 (Thermo Scientic, Waltham, MA, USA). Cell viability was analyzed using trypan blue dye exclusion method (microscope: Nikon, YS100, Tokyo, Japan). Total mononuclear and leukocyte differential cell counting were performed with Hematologic Analyzer CELM (Mythic 18; Barueri, São Paulo, Brazil). Safety tests, including aerobe and anaerobe microbiology tests, were conducted through BacT/ALERT system (Biomérieux, Durham, NC, USA). Samples for future evaluation were stored and frozen. After manipulation and appropriate tests, cells were delivered in a sterile bag to the operating room and then drawn up into a series of syringes. The heart was exposed via a left lateral thoracotomy. Stem cell administration consisted of a series of epicardial injections into the left ventricular myocardium under direct observation as follows: 0.2 ml (2 × 106 cells)/injection, 1 cm distance between injections, and 1 cm epimyocardial depth per injection. Since the ReACT protocol addresses the number of injections and dose as a direct relation to the extent of viable ischemic myocardium area determined by nuclear imaging tests, left ventricle chamber size and the number of injections differed for each patient.
Monocyte and Promonocyte Characterization
Fluorescence-Activated Cell Sorter Analysis
Flow cytometry (FACSCanto; BD Biosciences, San Jose, CA, USA) with software analysis (FACSDiva software version 6.1.1; BD Biosciences) was performed in all patients in order to characterize monocyte and promonocyte cell populations (including frozen and fresh samples). Fresh samples were obtained immediately after bone marrow cell manipulation. Frozen samples were the remaining stem cells not used for injection or analysis. They were stored for all patients. These samples were thawed according to previously described manipulation practice protocols for frozen umbilical cord blood stem cells.
Monocyte and promonocyte fractions isolated from human mononuclear bone marrow cells were sorted by flow cytometry using specific surface antigens. Cells were incubated for 20 min at 4°C with monoclonal antibodies (concentration 5 μl/106 cells for all antibodies) conjugated with fluorescein isothiocyanate (FITC): phycoerythrin (PE), allophycocyanin (APC), peridinin chlorophyll protein, and phycoerythrin cyano 7. The markers tested included pan-leukocyte CD45 APC (cat 340942, clone 2D1, Immunostep; BD Biosciences), promonocyte CD64 FITC (cat 555527, clone 10.1, Immunostep; BD Biosciences), monocyte CD14 PE (cat 555398, clone M5E2, Immunostep; BD Pharmingen, Franklin Lakes, NJ, USA), and CD34 PE (cat 348057, clone 8G12, Immunostep; BD Biosciences). After staining, erythrocytes were lysed with Lysis Buffer Solution (BD). Data acquisition was performed on BD FACSARIA II Cytometer (BD) and analyzed with the BD FACSDiva version 6.1.3 software.
Hematoxylin and Eosin (H&E) Stain
The sorted cell suspension was mounted on slides after a citospin centrifugation (400 × g for 10 min; Jouan CR 3.12; Thermo Scientific). H&E staining (Sigma-Aldrich, St. Louis, MO, USA) was performed according to standard protocol. Briefly, cells were fixed for 15 min in ethanol 70% and stained with hematoxylin (5 min) and eosin (2 min).
Follow-Up
Patient follow-up examination included CCSAC evaluation, objective myocardium perfusion analysis (stress nuclear imaging testing), functional changes, safety, quality of life questionnaire (SF-36), and cost-effectiveness analysis, as follows.
CCSAC Evaluation
Clinical evaluation of CCSAC was carried out at baseline and at 3, 6, and 12 months postprocedure for all patients. For patients with a follow-up greater than 1 year, the average CCSAC was evaluated over time, until April 2011. The recovery index was then defined as the percentage of time with a CCSAC grade of less than 4. A score of zero corresponds to CCSAC 4 for all time periods.
Stress Nuclear Imaging Testing
Technetium stress-induced myocardium perfusion scintigraphy (GE Infinia; GE Healthcare, Wauwatosa, WI, USA) (12) was performed at baseline and 6 and 12 months after the procedure to evaluate the percentage of myocardium ischemic area. It is important to point out, regarding the objective analysis of myocardium ischemic area change over time that all patients were considered to have 100% of ischemic area in those particular left ventricular walls previously identified as reversible ischemia areas by stress technetium scintigraphy. These reversible ischemic areas constituted the baseline comparison to 6- and 12-month follow-up scintigraphic analysis.
Functional Changes and Safety
Echocardiogram (GE Vivid; GE Healthcare) was performed at baseline and 12 months after surgery to assess ventricular function through the Simpson's method (15).
Safety was evaluated by EKG-documented arrhythmia and myocardial enzyme [creatine kinase-MB (CK-MB) fraction and troponin] tests. CK-MB was measured through enzymatic reaction (Cobas 6000 analyzer, module c501; Roche, Indianapolis, IN, USA) and troponin levels with immunoassay test (Cobas 6000 analyzer module 601; Roche). Other adverse events were also monitored.
Quality of Life Questionnaire (SF-36)
The SF-36 quality of life questionnaire was given at baseline and 12-month follow-up.
Cost-Effectiveness Analysis
Cost-effectiveness analysis compared the number of interventional coronary procedures (coronary artery bypass procedures, coronary angiographic examinations, and coronary artery stenting) and hospital length (in days) by unit admittance (emergency room, ward, and intensive care) 3 years prior to ReACT enrollment and post-ReACT (until April 2011) in 10 patients who had a follow-up time greater than 12 months.
Statistical Analysis
The Friedman nonparametric test was used to assess changes from baseline in angina CCSAC class at 3, 6, and 12 months and in the myocardium ischemic area at 6 and 12 months of follow-up. The Wilcoxon test with Bonferroni's correction was used for post hoc comparisons. Correlations between outcomes (angina class and myocardium ischemic area after the procedure) and stem cell formulation, as well as the number of myocardium punctures, were assessed by the Spearman test. The Wilcoxon nonparametric test was used to compare the left ventricle function on echocardiogram at baseline and 12 months follow-up. SF-36 questionnaires and cost-effectiveness data were also analyzed with the Wilcoxon nonparametric test.
Results
Patients' Baseline Characteristics
Baseline data is presented in Table 1. Drug therapy optimization was required for all patients for at least 3 months prior to meeting inclusion criteria, being defined as repeated attempts of optimization and combination, including maximum dosage of β-blockers, angiotensinconverting enzyme or angiotensin inhibitors, calcium channel blockers, short- and long-lasting action nitrates, statins, and antiplatelets (Table 2).
Drug Therapy Optimization and Clinical Data for All Patients
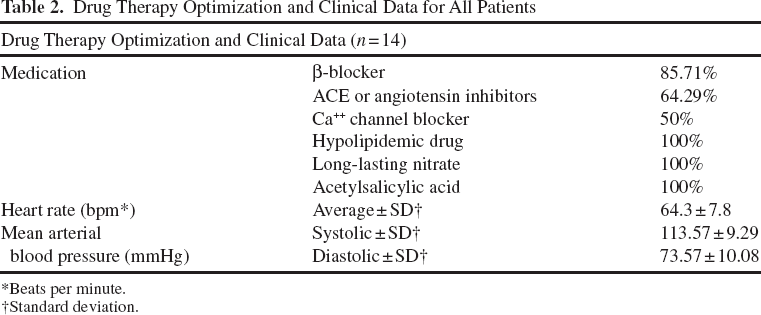
Beats per minute.
Standard deviation.
There were no ReACT procedure-related complications, including arrhythmias, need for reoperations due to bleeding, or other cardiac adverse events.
The first patient died from inferior myocardial infarction in August 2010, approximately 4 years postprocedure. Another patient (number 9) died from cardiogenic shock due to anterior myocardial infarction after 5 months of follow-up.
Patient number 2 returned to refractory angina class IV after 47.3 months of follow-up and met inclusion criteria again. This patient was referred and reenrolled to a new ReACT procedure, being listed as patient number 14 (even meeting inclusion criteria again, the new enrollment was not included in the statistical analysis due to possible bias).
Subjective Improvement in Myocardial Ischemia
After the ReACT myocardial injection procedure, there was a progressive improvement in the median angina classification, varying from class 4 at baseline to 2.0 ± 1.1 (p = 0.002), 1.0 ± 1.3 (p = 0.003), and 0.5 ± 0.9 (p = 0.002) at 3, 6, and 12 months of follow-up, respectively (Fig. 1).
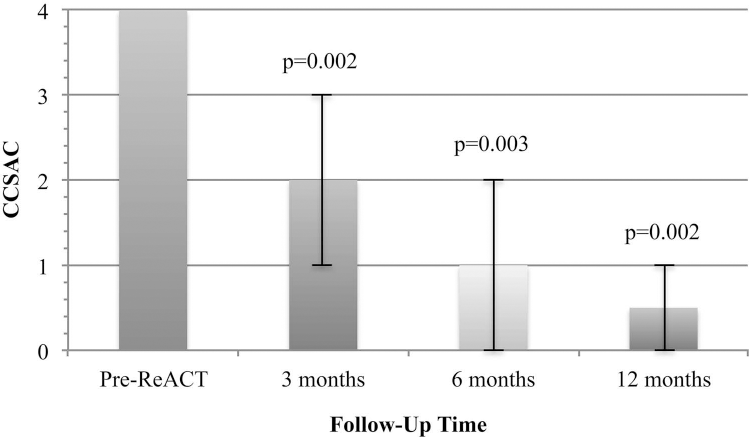
Variations in CCSAC in 12-month follow-up. Related to baseline, variation in angina class was statistically significant if p < 0.017 (0.05/3), according to Bonferroni's correction. Error bars represent the range between quartiles 25 and 75.
The Recovery Index (used for patients with more than 1-year follow-up, n = 10) showed a median percentage of time with CCSAC grade lesser than 4 of 73.9 ± 24.2%, for a median follow-up time of 46.8 ± 17.1 months (Fig. 2).

CCSAC evolution and recovery index (for patients with more than 1-year follow-up) for each patient (n = 10). **This patient (number 2) presented refractory angina (CCSAC 4) after 47.3 months in CCSAC 0, after the first ReACT's procedure. As this patient met ReACT's inclusion criteria again, he was enrolled in our protocol for the second time (number 14), but the second ReACT's procedure did not enter statistical analysis due to possible bias.
Objective Improvement in Myocardial Ischemia
Ischemic myocardium reversible area presented a progressive reduction through stress technetium scintigraphy assessment after 6 months [median decrease of 15% (0—100%), average decrease of 42.3 ± 48%; p = 0.024] and 12 months postprocedure [median decrease of 100% (0—100%), average decrease of 72.7 ± 27.3; p = 0.004] (Figs. 3 and 4).

Variations in myocardial ischemic area evaluated by stress technetium scintigraphy in 12-month follow-up. Related to baseline, variation in ischemic myocardium area was statistically significant if p < 0.025 (0.05/2), according to Bonferroni's correction. Error bars represent the range between quartiles 25 and 75.

Comparison between myocardial ischemic area evaluated by stress technetium scintigraphy pre-ReACT and at 12-month follow-up in different patients.
Monocyte and Promonocyte Characterization
Flow cytometry showed that, in previously frozen samples, a promonocyte population (CD45+, CD64+, CD14-, CD34-) was detected (3.11 ± 1.94%), while a monocyte population (CD14+/CD34-) was absent. In fresh samples, both promonocytes (5.34%) and monocytes (12.67%) were detected. However, cellular details obtained by H&E (Fig. 5) confirmed that monocyte cell integrity was highly compromised. Monocytes were actually disrupted (Fig. 5A), while promonocyte cell integrity was not affected (Fig. 5B).

Hematoxylin and eosin stain. (A) Microscopy of sorted monocyte cells illustrated that the surface membrane is not preserved. (B) Promonocyte cell membrane is preserved. Scale bar: 10 μm.
Correlation Between Angina Improvement and Cell Type
ReACT's promonocyte concentration (12.9 ± 3.3%; range 8.0—18.4%) (Table 4) suggests a positive correlation (n = 10, r = −0.575, p = 0.082) with clinical response, particularly improvement in CCSAC at 12-month follow-up (Tables 3 and 4).
Percentage of Promonocytes and Lymphocyte Concentration in ReACT Formulation for Each Patient
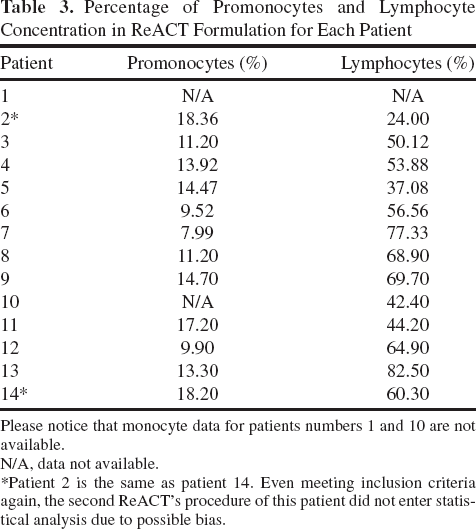
Please notice that monocyte data for patients numbers 1 and 10 are not available.
N/A, data not available.
Patient 2 is the same as patient 14. Even meeting inclusion criteria again, the second ReACT's procedure of this patient did not enter statistical analysis due to possible bias.
Spearman's Correlation Coefficients (rs) Between ReACT Promonocytes Content, Lymphocytes, and CD34+ Cells and Improvement of Angina Classification and Percentage of Myo cardium Ischemic Area in the Follow-Up

According to Canadian Cardiovascular Society Angina Classification.
Improvement assessed by stress
ReACT's lymphocyte concentration (56.0 ± 17.4%; range 24—82.5%) (Table 3) also appears to be correlated (n = 11, r = −0.561, p = 0.073) with CCSAC improvement at 12-month follow-up.
Other stem cell types, such as CD34+ cells, did not show any correlation with either subjective (n = 11, r = −0.111, p = 0.746) or objective improvement (n = 10, r −0.314, p = 0.377) at 12-month follow-up.
Correlation Between Number of Myocardium Punctures and Improvement
The protocol indicates that the number of myocardium punctures varies according to the extent of ventricular ischemic area, as determined by stress technetium scintigraphy. In this study, myocardial punctures varied from 24 to 90 (54.8 ± 14.9). No significant correlation was found between the number of punctures (the “act of introducing the needle” into the heart muscle) and angina classification improvement at 3 (n = 13, r = −0.315, p = 0.295), 6 (n = 12, r = −0.285, p = 0.37), and 12 months (n = 12, r = −0.362, p = 0.247) of follow-up. Also, there was no correlation between the number of punctures and objective changes in ischemic myocardium area at 6 (n = 11, r = −0.160, p = 0.638) and 12 months of follow-up (n = 11, r = −0.090, p = 0.792).
Functional Changes and Safety
The difference in LVEF (Fig. 6) assessed by echocardiogram was not statistically significant, according to Wilcoxon test, varying from 59.2 ± 8.9% at baseline to 61.6% ± 6.3% at 12 months follow-up (p = 0.533).
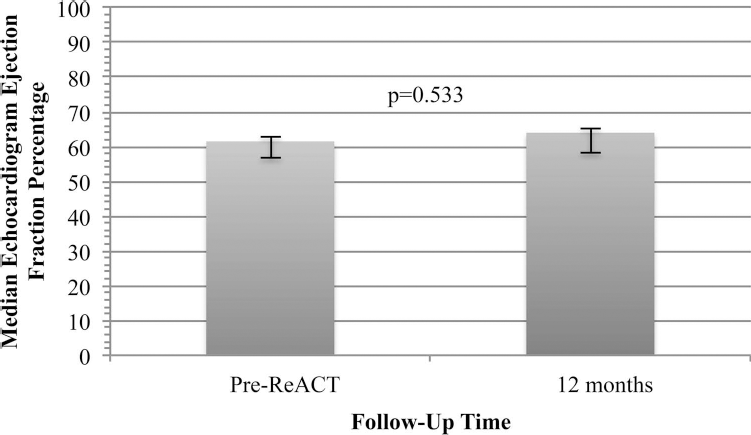
Comparison between LVEF, as assessed by transthoracic echocardiogram, at baseline and 12-month follow-up. The left bar represents baseline LVEF, while the right one represents the ejection fraction at 12 months. The x-axis corresponds to echocardiographic LVEF (in %), and the y-axis corresponds to time point, in months (where 0 = baseline). Error bars represent the range between quartiles 25 and 75.
Postoperative myocardial infarction diagnosis did not occur after ReACT's procedure, considering clinical symptoms, electrocardiogram analysis, and CK-MB fraction and troponin levels. Also, no major adverse events were noted after cell therapy.
Quality of Life Questionnaire (SF-36)
The quality of life questionnaire (SF-36) demonstrated a statistically significant improvement in seven out of eight domains (Figs. 7 and 8). Only the role-emotional domain (mental health domains) did not differ from baseline to 1-year follow-up questionnaire (n = 10, p = 0.317).

Comparison between SF-36 quality of life questionnaire physical health domains at baseline and 12-month follow-up. Error bars represent the range between quartiles 25 and 75.

Comparison between SF-36 quality of life questionnaire mental health domains at baseline and 12-month follow-up. Error bars represent the range between quartiles 25 and 75.
Cost-Effectiveness Analysis
Cost-effectiveness analysis showed a significant decrease in interventional coronary procedures such as CABG, coronary angiographic examinations (CAT), and coronary artery stenting (Fig. 9 and Table 5), as well as important reduction in median number of hospital stay length (in days), regardless of the admittance unit (emergency room, ward, or intensive care) with a median follow-up time of 46.8 ± 17.1 months (Fig. 10 and Table 5) in 10 patients (follow-up greater than 12 months).

Comparison between interventional coronary procedures pre-ReACT and post-ReACT. Comparison between interventional coronary procedures—-CABG, CAT, and coronary artery stenting—-pre-ReACT and post-ReACT, with a median follow-up time of 46.8 months, in 10 patients, who had longer than 12-month follow-up. Error bars represent the range between quartiles 25 and 75.

Comparison between hospital stay length (in days) by admittance unit pre-ReACT and post-ReACT. Comparison between hospital stay length (in days) by admittance unit—-emergency room, ward, and intensive care—-pre-ReACT and post-ReACT, with a median follow-up time of 46.8 months, in 10 patients, who had longer than 12 months follow-up. Error bars represent the range between quartiles 25 and 75.
Cost-Effectiveness Analysis, Considering Number of Coronary Interventional Procedures—- CABG, CAT, and Coronary Artery Stenting—-and Hospital Stay Length (in Days) by Admittance Unit—-Emergency Room, Ward, and Intensive Care—-Pre-ReACT and Post-ReACT, With a Median Follow-Up Time of 46.8 Months, in 10 Patients

n, number.
Discussion
These results in 13 patients enrolled in the ReACT Phase III protocol reinforce the beneficial effects reported in our preliminary refractory angina Phase II trial (10).
This update expands on the previously published article (10) as it called for a much longer follow-up time (average of 48 months in 10 patients) with reproducible improvement results, either in CCSAC or objective myocardial perfusion. Since refractory angina patients can experience relief of severe symptoms over a long period of time, as part of the disease's natural history (26), clinical evidence of CCSAC and myocardial perfusion recovery over a longer follow-up time was of paramount importance to support our findings. Furthermore, the isolation and characterization of a bone marrow monocyte precursor population (promonocytes), instead of a mature monocyte population, really suggests a stem cell-based myocardial recovery. Additionally, quality of life and cost-effectiveness analysis was never addressed as endpoints in other myocardial stem cell clinical trials.
As previously reported (10), angina symptom relief initiated as early as 3 months postprocedure with continuing improvement through the 12th month, suggesting that angiogenesis began early and that it kept evolving 12 months after the procedure (Fig. 1). Furthermore, symptom relief progressively improved in all patients, continuing to suggest that the effect is sustained and not transitory, different from other reports (4). However, some patients experienced return of angina symptoms after the initial 12-month follow-up time, probably due to the natural evolutionary history of the atherosclerotic disease.
It is important to point out that all patients had their medical drug treatment optimized for at least 3 months prior to meeting inclusion criteria. Nevertheless, since CCSAC analysis is a subjective endpoint that is prone to bias, an objective endpoint analysis (scintigraphic stress myocardial perfusion) was crucial to support our results.
Different from most trials involving the use of stem cell formulations in refractory angina patients in which endpoints were evaluated for at most 1 year, our trial has a median follow-up time of 46.8 ± 17.1 months.
The recovery index (developed for patients with more than 1-year follow-up) with a median follow-up time of 46.8 ± 17.1 months in our data demonstrates a long-term clinical evolution after stem cell therapy. All of the patients were graded less than CCSAC 4 for 73.9 ± 24.2% over a median follow-up time of 46.8 months (Fig. 2).
Even though a quantitative score of exercise capacity to evaluate angina symptoms was not included in our study, stress scintigraphy objective analysis can provide a direct myocardial perfusion assessment.
Myocardial ischemic stress area analysis (assessed through stress technetium scintigraphy) showed a progressive decrease over time, reaching statistical significance at both 6 and 12 months (Figs. 3 and 4). The median stress ischemic area showed a decrease of 100% at 12 months of follow-up, meaning that more than half of the patients showed 100% recovery of the myocardial stress ischemic area. The average stress ischemic area showed a decrease of 72.7 ± 27.3, which also supports a high percentage of myocardial ischemic area recovery. These significant myocardial reperfusions rule out any possibility of myocardium necrosis or fibrosis promoted by the intramyocardial ReACT injections and, consequently, CCSAC improvement due to placebo effect.
Since our protocol indicates that the number of myocardial punctures varies according to the extent of ventricular ischemic area, as determined by stress technetium scintigraphy, possible correlations between number of myocardial punctures and endpoints had to be addressed. Myocardial punctures varied from 24 to 90 (54.8 ± 14.9) per patient. Nevertheless, the number of myocardial punctures or injections did not correlate with CCSAC improvement in all three follow-up time points: 3 (n = 13, r = −0.315, p = 0.295), 6 (n = 12, r = −0.285, p = 0.37), or 12 months (n = 12, r = −0.362, p = 0.247). In addition, there was no correlation with myocardial ischemic area reduction at 6 (n = 11, r = −0.160, p = 0.638) or 12 months of follow-up (n = 11, r = −0.090, p = 0.792).
Improvement in angina class and symptoms with corresponding reduction in myocardial ischemic area suggests angiogenesis was directly related to stem cell infusion and not myocardial punctures promoting secondary angiogenesis. Consequently, these results reassure our primary therapeutic goal: myocardial perfusion improvements using ReACT formulation from a subjective (CCSAC) and objective (stress-induced myocardium imaging testing) perspective.
Functional changes and safety evaluation, as assessed through echocardiogram, showed no significant change in LVEF at 12 months (p = 0.533) (Fig. 6). Since our inclusion criteria enrolled patients without left ventricular dysfunction, increase in LVEF was not expected; nevertheless, safety concerns regarding LVEF worsening after an intramyocardial stem cell administration had to be noted. The sustained LVEF (median LVEF varied from 61.5 ± 8.9% at baseline to 64 ± 6.3% at 12 months follow-up) reinforces ReACT's safety.
Only one patient died from myocardial infarction 5 months postprocedure. We could not expect reduction in deaths from major cardiovascular events, since stem cell neoangiogeneses is based upon increases in capillary density and improved microcirculation. Unfortunately, an autopsy was not performed.
The quality of life questionnaire (SF-36) showed a marked improvement in all SF-36 physical-health domains (physical function, role-physical, bodily pain, and general health) and in three out of four SF-36 mental health domains (vitality, social function, and mental health) (Figs. 7 and 8). The nonsignificant difference in the role-emotional domain (one of SF-36 mental health domains) suggests that emotional distress background (i.e., depression or other similar disorders) did not contribute as a major element of confusion in the patients' interpretation of angina limitation. Therefore, problems with work or other daily activities was not a result of emotional problems, but secondary to refractory angina. This finding completely rules out any possibility of placebo effect and reinforces somewhat the protocol's angiogenic property.
Monocyte and promonocyte characterization by flow cytometry showed that, in previously frozen human mononuclear cells, only a promonocyte population (CD45+, CD64+, CD14-, CD34-) was present (3.11 ± 1.94%). In fresh samples, both promonocytes (5.34%) and monocytes (12.67%) were detected. However, cellular detail obtained by H&E (Fig. 5) showed that monocyte integrity was hugely compromised (Fig. 5A) in comparison to promonocytes (Fig. 5B). These data suggest that promonocytes are more resistant to standard harvesting, preparation, and storage than monocytes. The suggested correlation between number of promonocytes and improvement of clinical response after 12 months of follow-up (n = 10, r = −0.575, p = 0.082) supports a stem cell-related effect. Since cell therapy relies on cell quality, these data support that promonocytes, and not mature monocytes, might be responsible for ReACT's angiogenic properties.
Furthermore, lymphocyte concentration also appears to be correlated to CCSAC improvement at 12 months follow-up (n = 11, r = 0.561, p = 0.073) (Tables 3 and 4). Bone marrow CD34+ stem cell concentration was not correlated to either CCSAC improvement (n = 11, r = −0.111, p = 0.746) or myocardial ischemic area reduction (n = 10, r = −0.314, p = 0.377) at 12 months follow-up, which was different from other studies (17), since CD34+ cells are well recognized as vascular lineage-compromised stem cells.
The correlation between ReACT's promonocyte and lymphocyte concentration and the sustained improvement in angina class could indicate improvement not directly related to a single supporting cell type, either alone or in combination (9,14,28). Monocytes have been shown to contribute to neovascularization in heart ischemic diseases, regardless of the mechanism involved (either arteriogenesis and angiogenesis) (3,28). Losordo et al. (17) conducted a randomized controlled clinical trial, involving dose-escalating intramyocardial injections and catheter delivery via NOGA® eletromechanical mapping system with CD34+ stem cells in patients with refractory angina. The low CD34+ dose group experienced significantly less weekly angina episodes and improvement in exercise tolerance, compared to the placebo group or the CD34+ high-dose group. The causal mechanism of this difference is not yet known, but we could speculate that a single stem cell lineage is not sufficient for sustained neoangiogenesis. Stem cell microenvironment probably plays a critical role in angiogenesis. To date, however, the optimal number or formulation of stem cells to promote myocardial regeneration remains unidentified (6—8,17).
Several patients exhibited a recurrence or worsening of their CCSAC angina class over time, with 6 out of 10 patients (who improved to CCSAC classes 1 or 0 after ReACT's procedure) developing higher CCSAC classes after an average of 36 months. This worsening tendency could be explained by either loss of cellular effect (angiogenesis) over time or by natural historical evolution of the coronary artery disease in an area not treated by the stem cell therapy. One of our patients (numbers 2 and 14) presented progressive refractory angina (CCSAC 4) after being asymptomatic (CCSAC 0) for 47.3 months after the first ReACT procedure. As this patient met ReACT's inclusion criteria again, he was reenrolled in our protocol for the second time, although this procedure did not enter statistical analysis due to possible bias. Six months after the second cell therapy, this patient progressively improved from CCSAC 4 to 0, remaining angina free after 1 year of follow-up, as demonstrated by the recovery index graph (Fig. 2). Regardless of the underlying cause, we could speculate that refractory angina patients will most probably need more than one cell transplantation procedure over time.
Cost-effectiveness analysis of coronary interventional procedures showed a significant decrease in CABG, CAT, and coronary artery stenting in 10 patients who had been followed for at least 12 months. Nevertheless, these pre- and post-ReACT differences have to be analyzed with extreme caution. Since all patients met ReACT's inclusion criteria, well-established refractory angina was present. As a result of that, patients included in ReACT's protocol were theoretically unable to be referred to any type of coronary interventional procedures at the time of enrollment. Consequently, this significant difference in coronary procedures does not represent reduction in costs, but only reinforces the magnitude of refractory angina itself. Nevertheless, one patient (patient number 7) underwent CABG after the ReACT procedure. This particular CABG operation, not performed by our group, can be considered a desperate attempt to relieve refractory angina symptoms in a patient who did not improve favorably after the stem cell procedure (CCSAC class 3 or 4 at all time points). Despite the operation, this patient continued to present angina (CCSAC 3). In addition, patient number 3 underwent an unsuccessful PTCA and stenting attempt after an isolated chest pain episode.
The second part of the cost-effectiveness analysis, hospital stay length (in days) by admittance unit (emergency room, ward, and intensive care) pre-ReACT (past 3 years prior to enrollment) and post-ReACT, with a median follow-up time of 46.8 ± 17.1 months, really translates itself into an important reduction in refractory angina direct costs. Considering the median hospital stay length, regardless of the admittance unit, virtually no patient underwent hospital admittance after the ReACT procedure, with a median follow-up time of 46.8 months (n = 10). The ReACT procedure costs approximately $20,000. If we consider only the median ICU length of 10.5 days before enrollment, the ReACT procedure reduced refractory angina direct costs. Other hospital admittance variable analysis (ward and emergency room) just reinforces cost reduction.
Study Limitations
Our study has several limitations. Owing to ethical aspects and the inability to justify the use of an isolated surgical intramyocardial placebo in this population, this was a nonrandomized, open study. Several reports have already studied the natural history and symptom evolution of medically treated refractory angina (26). However, the clinically important improvements in CCSAC both short and long term (recovery index with sustained CCSAC improvement for a median follow-up time of approximately 4 years) as well as quality of life domains correlated with objective reduction in myocardial ischemic area. This seems to rule out any possibility of placebo effect.
Additionally, our protocol was limited by the small sample size and potential investigator bias in assessing CCSAC angina grade or nuclear scan interpretation, as these tests were performed by the principal investigator and were not blinded. Finally, there were no follow-up nuclear scans in those patients that redeveloped angina to confirm whether this represented gradual failure of the initial angiogenesis or, more likely, new angina in an area not initially treated. Also, no coronary angiograms were obtained to demonstrate whether cell therapy-induced angiogenesis was present at the macro- or only micro-vascular level.
In conclusion, this clinical update strongly reinforces ReACT's safety and effectiveness in terms of promoting progressive and sustained improvement in patients with refractory angina. The study included a long-term follow-up analysis. Promonocytes, lymphocytes, and other supporting stem cells may play a key role in cardiological stem cell therapy.
Footnotes
Acknowledgments
This study was supported by Cellpraxis Bioengeneering, in a scientific cooperation with Paulista School of Medicine, Federal University of São Paulo. Nelson Americo Hossne, Jr., Adriana Luckow Invitti, and Paul R. Sanberg are inventors on patents submitted in regard to the CellPraxis Refractory Angina Cell Therapy (ReACT®) Protocol. Nelson Americo Hossne, Jr. and Paul R. Sanberg are also consultants for Cryopraxis. Paul R. Sanberg was not involved in the review process for this manuscript.
