Abstract
An increasing number of patients have refractory angina despite optimal medical therapy and are without further revascularization options. Preclinical studies indicate that human CD34+ stem cells can stimulate new blood vessel formation in ischemic myocardium, improving perfusion and function. In ACT34-CMI (N = 167), patients treated with autologous CD34+ stem cells had improvements in angina and exercise time at 6 and 12 months compared to placebo; however, the longer-term effects of this treatment are unknown. ACT34 was a phase II randomized, double-blind, placebo-controlled clinical trial comparing placebo, low dose (1 × 105 CD34/kg body weight), and high dose (5 × 105 CD34/kg) using intramyocardial delivery into the ischemic zone following NOGA® mapping. To obtain longer-term safety and efficacy in these patients, we compiled data of major adverse cardiac events (MACE; death, myocardial infarction, acute coronary syndrome, or heart failure hospitalization) up to 24 months as well as angina and quality of life assessments in patients who consented for 24-month follow-up. A total of 167 patients with class III–IV refractory angina were randomized and completed the injection procedure. The low-dose-treated patients had a significant reduction in angina frequency (p = 0.02, 0.035) and improvements in exercise tolerance testing (ETT) time (p = 0.014, 0.017) compared to the placebo group at 6 and 12 months. At 24 months, patients treated with both low-and high-dose CD34+ cells had significant reduction in angina frequency (p = 0.03). At 24 months, there were a total of seven deaths (12.5%) in the control group versus one (1.8%) in the low-dose and two (3.6%) in the high-dose (p = 0.08) groups. At 2 years, MACE occurred at a rate of 33.9%, 21.8%, and 16.2% in control, low-, and high-dose patients, respectively (p = 0.08). Autologous CD34+ cell therapy was associated with persistent improvement in angina at 2 years and a trend for reduction in mortality in no-option patients with refractory angina.
Introduction
There are increasing numbers of patients with advanced coronary artery disease who are not amenable to surgical or percutaneous revascularization1–4. These patients frequently have symptoms refractory to standard medical therapy and are left with limited treatment options. Positive preclinical data and encouraging early clinical trials suggest that cell therapy is an attractive treatment option for these patients5–8. In particular, preclinical studies in which subjects were transplanted with autologous CD34+ cells have demonstrated that these cells have the ability to restore the microcirculation and improve myocardial tissue perfusion in the presence of occluded epicardial vessels9–11. An initial phase I/IIa, double-blind, placebo-controlled trial in 24 patients with refractory angina demonstrated excellent safety with improvement in angina frequency, nitroglycerin use, and exercise time in patients receiving intramyocardially delivered CD34+ stem cells 12 . This was followed by ACT34-CMI, a larger phase II, double-blind, placebo-controlled trial in which 167 patients with refractory angina were demonstrated to have a significant improvement in angina frequency and exercise time at 12 months, which was accompanied by a trend for a decrease in major adverse cardiac events (MACE) 13 . To determine the long-term safety and effects of autologous CD34+ stem cell therapy on angina and quality of life (QoL), we conducted a 24-month follow-up study of ACT34-CMI patients. We report the 2-year safety and efficacy results of the ACT34-CMI trial, which remains the largest, randomized, placebo-controlled stem cell trial in patients with refractory angina.
Materials and Methods
Study Population and Design
The ACT34-CMI study was a phase II, prospective, double-blind, randomized, placebo-controlled clinical trial in patients aged 21–80 years with Canadian Cardiovascular Society (CCS) class III–IV chronic refractory angina despite optimal medical management 13 with a planned follow-up of 12 months. Eligible patients were not candidates for revascularization and experienced a minimum of seven angina episodes per week as well as exercise limitation between 3 and 10 min on a modified Bruce protocol due to angina.
All patients (N = 174) underwent synthetic granulocyte colony-stimulating factor (G-CSF; 5 μg/kg per day for 4–5 days, subcutaneously; Neupogen, Amgen, Thousand Oaks, CA, USA)-mediated stem cell mobilization followed by apheresis on day 5. Mononuclear cells (MNCs) were enriched for CD34+ cells using a commercially available device (Isolex 300i Magnetic Cell Selection System; Baxter Healthcare, Deerfield, IL, USA). Following completion of release testing on the final cell preparation, 167 patients were randomly assigned to undergo intramyocardial injection of 1 × 105 adult autologous CD34+ cells/kg, 5 × 105 autologous CD34+ cells/kg, or placebo. Electromechanical endocardial mapping was performed with the NOGA Map system (Biologics Delivery Systems, Diamond Bar, CA, USA) as previously described to identify viable, ischemic areas of the myocardium (12,13), and study product was delivered via intramyocardial injection with a NOGA Myostar® catheter.
All patients completing 12 months of follow-up (n = 156) were offered the opportunity to participate in this continued 12- to 24-month follow-up study (NCT00545610), and, if interested, these patients gave consent again prior to continued follow-up. The institutional review board (IRB) at each center approved the protocol. All patients signed written informed consent and were aware that the study was experimental. The ability to pay was not a requirement for inclusion, and patients were not compensated for participation. All patients were informed of their rights to withdraw consent at any time. Baxter Healthcare sponsored the study and was responsible for the conduct of the investigation, with oversight provided by the principal investigator and the Scientific Advisory Board (SAB).
Safety Endpoints
Patients were contacted via telephone at months 15, 18, 21, and 24 postinjection to collect information on adverse events (AEs), hospitalizations, and concomitant medication use. All suspected MACE [death, myocardial infarction (MI), acute coronary syndrome (ACS), hospitalization, worsening congestive heart failure, or stroke] were adjudicated by an independent clinical events committee blinded to treatment assignment.
Efficacy Endpoints
Data on angina frequency, CCS angina classification, and Seattle Angina Questionnaire (SAQ), Short Form-36 (SF-36), and QoL were assessed at 18 and 24 months. Angina frequency was documented on a daily basis for 28 days in a handwritten diary during the 5 weeks preceding the 18- and 24-month follow-up visits, but was documented using an interactive voice response system (IVRS) at the 3-, 6-, and 12-month follow-up visits. As in the original trial, patients were instructed to report ischemic-related chest pain. Patients with nonischemic chest pain including noncardiac or pain related to aortic stenosis were excluded.
Statistical Analysis
Sample Size Determination
The sample size for this study was to be determined by the number of subjects who completed the 12-month visit (final study visit) of the core therapeutic study. The statistical analysis was performed by statisticians employed by Baxter Healthcare. The raw data and analysis were available and reviewed by the SAB and principle investigator.
Efficacy
The efficacy (angina and QoL) endpoints in this study were compared to the values at baseline of the core therapeutic study. Because the core therapeutic study includes 3-, 6-, and 12-month follow-up visits, the 6- and 12-month visits for this study were to be designated as 18 and 24 months, respectively, for the analysis. A log linear model (Poisson regression) was performed on the frequency of angina at baseline and at months 6, 12, 18, and 24. The independent parameters in the model were the treatment group (as randomized) and visit (baseline, 6, 12, 18, and 24 months) and the interaction between treatment group and visit. The baseline value was used as a covariate. Contrasts were to be constructed on the difference between 6, 12, 18, 24 months and baseline and differences between treatment groups at each follow-up period. Since this analysis was to be done on the log scale, these contrasts took the form of relative risks.
Descriptive statistics are displayed as means and standard deviations (SDs) for continuous variables; number and percentage with characteristics are given for categorical variables. Repeated-measures analysis of variance (ANOVA) was performed on continuous data.
Generalized linear models were used to analyze ordinal and categorical data. The independent parameters in the model were the treatment group and visit (baseline, 6, 12, 18, and 24 months) and the interaction between treatment group and visit. Contrasts were to be constructed on the differences between 6, 12, 18, 24 months and baseline and differences between treatment groups at each follow-up period.
Safety
Rates for MACE and key MACE, individually and in composite, were calculated for each treatment group based on all 167 patients. The probability of being MACE free for each treatment group was also calculated as well as the time to first hospitalization for all causes.
All statistical calculations and plots were done with SAS (Cary, NC, USA).
Results
Patient Disposition
Of the 156 patients who completed the 12-month follow-up in the ACT34-CMI trial, 148 patients enrolled in the 2-year follow-up study, and 130 patients completed the 24-month follow-up efficacy study (Fig. 1).

Subject flow during ACT34 and the follow-up period.
Baseline Characteristics
Baseline characteristics were similar for the 167 patients who initially enrolled and the 148 patients who were enrolled in the 2-year study (Table 1). Baseline characteristics were also similar for all three groups in both the 1- and 2-year study. The 2-year patient population (n = 148) included 20 (13.5%) females and 128 (86.5%) males with a mean age of 60.9 (range, 41 to 91) years. Previous coronary artery bypass grafting (CABG) and previous percutaneous coronary intervention (PCI) had been performed in 93.9% and 83.8% of patients with a mean number of prior PCI procedures of 3.7 (range, 1 to 23) per subject. Medications, left ventricular ejection fraction (LVEF), and baseline angina episodes per week were also similar between groups (Table 1).
Baseline Characteristics of Study Population Enrolled in 1-Year and 2-Year Follow-up Studies

MACE During the 2-Year Follow-Up
All 167 patients were included in the safety (MACE) study. There were seven deaths during the 12- to 24-month period: four in the control group, one in the low-dose group (1 × 105 cells/kg), and two in the high-dose group (5 × 105 cells/kg).
At 2 years, 7 of 56 (12.5%) patients in the control group had died compared to 1 of 55 (1.8%) in the high-dose group and 2 of 56 (3.6%) in the low-dose group, for an overall 2-year mortality of 5.4% (Table 2). There was no difference in the rate of MI, ACS hospitalization, worsening heart failure, or stroke, although cell-treated patients had consistently lower rates of MI and composites of these endpoints as well as cardiovascular (ACS and heart failure) hospitalizations (Table 2). Freedom from MACE was higher in cell therapy-treated patients (Fig. 2). There was a significant reduction in the analysis of time to first hospitalization for all causes for the low-dose group when compared to the placebo group (p = 0.012, estimate 0.53; 95% CI: 0.32–0.87) and a trend toward the relative risk of time to first hospitalization in the high-dose group compared to placebo (p = 0.055, estimate 0.63; 95% CI: 0.39–1.01). The relative risks for first hospitalization for cardiac causes were 0.75 and 0.79 for the low- and high-dose groups, respectively, but did not reach statistical significance.
Percentage of Subjects With MACE by Treatment Group

Subjects with MACE from the start of mobilization through 24 months.
Represents significance as determined by Fisher's exact test.

Freedom from MACE during 2-year follow-up period.
Angina Frequency
In addition to the reduction in angina observed at 12 months in patients treated with CD34+ cell therapy, cell-treated patients continued to experience significant reductions in the mean number of angina episodes per week at both 18 and 24 months (Figs. 3–5). Importantly, the reduction in angina episodes per week at 12 months persisted to 24 months in both the low- and high-dose cell therapy groups (Figs. 3 and 4). This was true comparing all patients that entered the postinjection follow-up period as well as those patients specifically enrolled in the 2-year follow-up study (Figs. 3 and 5).
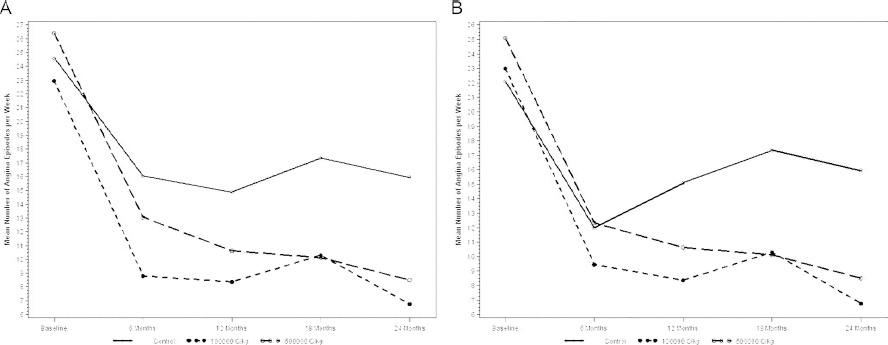
Mean number of angina episodes over time for ACT34 subjects who entered the postinjection follow-up period (A) and those enrolled in the 2-year follow-up study (B).

Angina frequency at 6, 12, 18, and 24 months for subjects entering postinjection follow-up period.

Angina frequency at 6, 12, 18, and 24 months for subjects enrolled in the 2-year follow-up study.
With regard to QoL overall, patients in all treatment groups improved at both 12 and 24 months (data not shown). There was a trend toward improvement in the angina stability score at 24 months in the low-dose group and at 18 months in the high-dose group, compared to control patients, but this did not reach statistical significance. There were trends toward improvement in the SF-36 score at 18 months in the low- and high-dose-treated patients compared to controls as well as trends toward improvement in physical functioning at 24 months in the low- and high-dose-treated patients, although this did not meet statistical significance.
Discussion
Autologous CD34+ stem cells obtained by apheresis after 5 days of G-CSF (5 μg/kg per day) and delivered into the ischemic zone using NOGA electrical mechanical mapping followed by intramyocardial injection was associated with persistent trends toward lower rates of mortality, time to first hospitalization, and MACE in cell therapy-treated patients when tested in a randomized, double-blind clinical trial. In addition, the significant improvements in angina seen at 6 and 12 months persisted at 18 and 24 months. The trial was not powered to evaluate MACE, but certainly the trend is encouraging and allays any safety concerns. Notably, the 2-year mortality in the cell-treated patients (1.8% and 3.6%) is lower than expected for this high-risk patient population1,14–16. These results enhance the 1-year results of ACT34-CMI that showed significant reductions in angina and significant improvement in exercise time in CD34+ cell-treated patients 13 . Patients with refractory angina remain a challenging group of patients to identify and treat. Over a decade ago, the European Society of Cardiology Task Force on Refractory Angina recognized this problem and indicated there was an urgent need to understand the natural history of this patient population 3 . Recent studies indicate that up to 16% of consecutive patients undergoing cardiac catheterization have evidence of myocardial ischemia, yet are suboptimal candidates for revascularization16,17. Previously, it was reported that these patients were at high risk for death 14 , but with advances in medical therapy and secondary prevention, it appears the mortality is lower than previously reported. Recent data from a dedicated refractory angina clinic reported 3.9% 1-year and 28.4% 9-year mortality 1 . For patients with class III or IV angina, 5-year mortality was 16.3% and 18.2% 1 . Similarly, a cohort of patients modeled to mimic entry criteria for cell and gene therapy studies, including ACT34-CMI, demonstrated 3-year mortality of 13.0%, while 3-year MACE rates exceeded 50% 15 . While the control group in ACT34-CMI had mortality similar to the expected, the mortality in CD34+ stem cell-treated patients was lower than expected. This finding was also suggested in three recent meta-analyses examining cell therapy in “no-option” patients but needs to be confirmed in larger and adequately powered trials5–7.
Given the improved and relatively low overall mortality for patients with refractory angina, the primary focus of therapy has centered on improvement in angina and QoL. Angina is more common than expected and has been both underrecognized and understudied. For example, in the COURAGE trial, at 12 months, 42% of patients in the medical treated arm and 31% of patients in the PCI-treated arm had persistent angina 18 . On the basis of the recent National Health and Nutrition Examination Survey (NHANES) data, it is estimated that 10–12 million Americans have angina 19 . For those patients who are suboptimal candidates for percutaneous or surgical revascularization, the options are currently limited with only two approved treatments including enhanced external counter pulsation (EECP) and surgical transmyocardial laser revascularization2,3,19. As expected, there was a reduction in angina in the placebo group, which was most prominent at 6 months (Fig. 3). The “placebo effect” is well documented in refractory angina trials 1 and was less prominent at 18 and 24 months, providing greater separation between the cell-treated and placebo groups. Cell therapy appears to be a promising treatment strategy for this challenging group of patients. In addition to the improvement in mortality, the three recent meta-analyses of patients with refractory angina/refractory ischemia with and without left ventricular dysfunction demonstrated improvement in angina, exercise time, and QoL, as well as improvements in MACE5–7. Our findings in ACT34-CMI extend our previous observations to 24 months and are consistent with the results of the meta-analyses.
There are theoretical reasons why CD34+ cells are an attractive cell type for patients with refractory angina. While there appears to be modest efficacy with bone marrow-derived MNCs (BM-MNCs), the quality of autologous stem cells declines with age. In the recent National Institutes of Health (NIH)-sponsored FOCUS trial, the improvement in LVEF was inversely related to age and directly related to the percentage of CD34+ cells 20 . Recent studies have pointed to the importance of the CD34+ cell content in the bone marrow of patients with risk factors for coronary disease in predicting not only baseline but also future exercise capacity21,22. In addition, in a recent study of patients with dilated cardiomyopathy (N = 110), treatment with intracoronary delivery of CD34+ stem cells resulted in an improvement in ejection fraction and 6-min walk, and a reduction in N-terminal brain natriuretic peptide (BNP) compared to placebo. Patients with higher myocardial homing had more substantial improvement in LVEF, and total mortality was also lower in the cell-treated group 23 . Most recently, the results of the PreSERVE-AMI trial reported a CD34+ cell dose-dependent improvement in LVEF as well as trends toward a dose-dependent reduction in MACE in post-STEMI (ST-segment elevation myocardial infarction) patients (N = 161) treated with intracoronary delivery of CD34+ cells versus placebo 24 .
Our study is even more provocative given that this represents the effect of a single treatment. Recent reports suggest that in patients with recurrence of symptoms, repeated cell administration may replicate initial results 25 . With refinement of cell preparation/isolation procedures, such repetitive therapy may be possible without additional mobilization/apheresis procedures, potentially offering an opportunity for additional augmentation of benefit.
In conclusion, in patients with CCS functional class III/IV angina refractory to conventional medical therapy who are not candidates for revascularization, intramyocardial injection of CD34+ cells resulted in a persistent improvement in angina at 2 years posttreatment. In addition, there was a significant reduction in the time to first hospitalization in cell-treated patients, with a trend for reduction in mortality. These results extend the primary results of ACT34-CMI to 2 years and are consistent with several recent meta-analyses, which reported improvement in symptoms, exercise time, mortality, and MACE in “no-option” refractory angina patients compared to placebo.
Footnotes
Acknowledgment
Baxter Healthcare sponsored the study and was responsible for the conduct of the investigation, with oversight provided by the principal investigator and the SAB. T. D. Henry was coprincipal investigator and steering committee member for the ACT34 trial and RENEW trial. G. L. Schaer is a steering committee member for the ixCELL-DCM trial, which he is paid an honoraria from Vericel Corporation, and is a steering committee member for the RENEW trial, which he is paid an honoraria from Baxter Healthcare. T. J. Povsic has received research funding at his institution from Baxter Healthcare and Baxalta US Inc., and he has received personnel fees from Pluristem and Capricor and Celyad. C. Junge was a former employee of Baxter Healthcare. C. White was a steering committee member for the ACT34 trial and RENEW trial. R. A. Harrington was a steering committee member for the ACT34 trial and RENEW trial. D. W. Losordo is employed by Caladrius Biosciences. A. Hunt was a former employee of Baxter Healthcare.
