Abstract
Muscular dystrophy is a genetic disorder with no definite cure. A study was carried out on 150 patients diagnosed with muscular dystrophy. These included Duchenne muscular dystrophy, limb-girdle muscular dystrophy, and Becker muscular dystrophy variants. They were administered autologous bone marrow-derived mononuclear cells intrathecally and intramuscularly at the motor points of the antigravity weak muscles followed by vigorous rehabilitation therapy. No significant adverse events were noted. Assessment after transplantation showed neurological improvements in trunk muscle strength, limb strength on manual muscle testing, gait improvements, and a favorable shift on assessment scales such as the Functional Independence Measure and the Brooke and Vignos Scales. Furthermore, imaging and electrophysiological studies also showed significant changes in selective cases. On a mean follow-up of 12 ± 1 months, overall 86.67% cases showed symptomatic and functional improvements, with six patients showing changes with respect to muscle regeneration and a decrease in fatty infiltration on musculoskeletal magnetic resonance imaging and nine showing improved muscle electrical activity on electromyography. Fifty-three percent of the cases showed an increase in trunk muscle strength, 48% showed an increase in upper limb strength, 59% showed an increase in lower limb strength, and approximately 10% showed improved gait. These data were statistically analyzed using Student's paired t test and found to be significant. The results show that this treatment is safe and efficacious and also improves the quality of life of patients having muscular dystrophy. This manuscript is published as part of the International Association of Neurorestoratology (IANR) supplement issue of Cell Transplantation.
Introduction
Muscular dystrophy is a group of heterogeneous disorders that are characterized by progressive weakness, a wasting of skeletal muscle, and possible cardiac complications (9). It is caused by mutations of different components of the dystrophin–glycoprotein complex, which links the extracellular matrix in muscle to the intracellular cytoskeleton and varies in terms of severity, age of onset, and selective involvement of muscle groups (7,14). It has been postulated that one of the pathogenetic mechanisms of progression in muscular dystrophy is an imbalance between muscle damage or degeneration and muscle repair through stem cell-mediated regeneration (22). The molecular mechanisms of the disease have been extensively investigated, but there is currently no effective treatment (5). Stem cell transplantation therapy may offer an approach to enhance the regenerative ability of damaged and degenerating muscle cells in patients with muscular dystrophy (10,23). Stem cells can be obtained from a variety of different sources that can harbor myogenic potential (8).
We present the data of a clinical trial conducted on 150 muscular dystrophy patients to study the efficacy of autologous bone marrow-derived mononuclear cells (MNCs) as a therapy. These MNCs were administered intrathecally and intramuscularly in specific motor points of the muscles. The treatment was found to be safe with significant improvements in the quality of life of the muscular dystrophy patients.
Materials and Methods
Patient Selection
One hundred fifty muscular dystrophy patients were included in the study. One hundred twenty-five patients had Duchenne muscular dystrophy (DMD), 20 had limb-girdle muscular dystrophy (LGMD), and 5 had Becker muscular dystrophy (BMD). The age of the patients ranged from 2.11 to 48 years with an average of 11.53 years in the DMD group, 34.40 years in the LGMD group, and 27.25 years in the BMD group. In both the DMD and the BMD groups, 100% of the total cases were males, whereas in the LGMD group, only 72% of the cases were males. Patients were selected on the basis of the inclusion criterion in paragraph 35 of the World Medical Association Declaration of Helsinki—Ethical Principles for Medical Research Involving Human Subjects (3). The protocol had been reviewed and approved by the Institutional Committee for Stem Cell Research and Therapy (IC-SCRT) in accordance with the Indian Council of Medical Research (ICMR) guidelines. The exclusion criteria were presence of respiratory distress, presence of acute infections such as HIV/HBV/HCV, malignancies, and other acute medical conditions such as respiratory infection, fever, and others and pregnancy or breastfeeding. An informed consent was obtained from the patients and their families. Before therapy, all subjects underwent extensive evaluation by medical and rehabilitation experts. The evaluation included a complete neurological examination, psychological examination, Manual Muscle Testing (MMT), and evaluation on scales such as the Functional Independence Measure (FIM) and the Brooke and Vignos Scales for DMD. Patients were assessed for pretherapy fitness through routine blood tests, X-rays, and others. Specific tests such as serum creatine phosphokinase (CPK), electromyography (EMG), and magnetic resonance imaging (MRI) of the musculoskeletal system were also performed. The mean follow-up period was 12 ± 1 months.
Neurological Evaluation
All patients underwent a thorough neurological evaluation as well as evaluation on various scales such as the Brooke and Vignos Scales and FIM along with MMT.
The Brooke and Vignos Scale were used to assess the functional limitation in DMD cases. The Brooke Scale was used for upper extremities ranging from grade 1 to 6, showing increment in the weakness in upper extremities from ability to raise upper extremity completely to not being able to raise hands to the mouth or have any purposeful hand functions. The Vignos Scale was used for lower extremities ranging from grade 1 to 10, showing increment in the weakness of lower extremities from ability to walk and climb stairs without assistance to being totally confined to bed or immobile. Patients were evaluated on the basis of these two scales because they are widely accepted assessment tools and clearly show both functional and neurological improvements posttherapy (2,20).
The FIM scale was used to assess the changes in functional abilities thereby improving the quality of life of patients after stem cell transplantation. The FIM assesses the levels of independence from level 1 to 7, ranging from complete independence (timely, safely) in all Activities of Daily Living (ADL) to total assistance required. The areas of ADL that are assessed in detail are self-care, bladder and bowel management, transfers and locomotion, comprehension and expression, social interaction, problem solving, and memory (6).
Radiological Evaluation
Before the treatment, all the patients underwent MRI for musculoskeletal system. For all MRI studies, a General Electric 1.5 Tesla closed system (GE 1.5 Tesla Excite 8 channel MRI system; General Electric, Milwaukee, WI, USA) was used. The MRI was also carried out on follow-up to compare the changes.
Electrophysiological Evaluation
EMG was done to assess the extent of damage to the muscles and also as a preoperative outcome tool for specific muscles, in which MNCs were to be injected intramuscularly. EMG was also carried out on follow-up to compare the changes.
Motor Point Plotting
Physiotherapists selected individual muscles for intramuscular injection of stem cells with manual muscle testing. Proximal girdle muscles or weak antigravity muscles such as deltoid, biceps, triceps, abdominals, glutei, quadriceps, hamstrings, tibialis anterior, peroni longus, and brevis were selected.
For plotting the motor points, a low-frequency electronic stimulator with automatic surgery was used. A faradic current with pulse duration of 0.1–1 ms and a frequency of 50–100 Hz was used.
Isolation and Administration of Autologous Bone Marrow Cells
Granulocyte colony-stimulating factor (G-CSF) injections were administered 48 h and 24 h before the treatment to help in stimulation, survival, and multiplication of CD34+ cells (13). Bone marrow aspiration was carried out under local or general anesthesia depending on the individual case. Approximately 100 ml (varying between 80 and 120 ml based on the age and body weight) of bone marrow was aspirated from the anterior superior iliac crest bone under aseptic precautions using a bone marrow aspiration needle. MNCs were obtained after Ficoll density gradient separation (Sigma-Aldrich, St. Louis, MO, USA). The viable count of the isolated MNCs was taken using the trypan blue exclusion test (S D Fine Chem Ltd., Mumbai, India) and confirmed by a TALI image cytometer using propidium iodide (both Life Technologies, Grand Island, Ny, USA). The MNCs were checked for CD34+ [CD34-phycoerythrin (PE) antibody; BD Biosciences, San Jose, CA, USA] by fluorescence-activated cell sorting (FACS; BD FACSCalibur Flow cytometer; BD Biosciences). Body weight x 106 MNCs were injected immediately postseparation, intrathecally in L4–L5 using an 18-gauge Tuohy needle and catheter (Portex, system 1, Epidural minipack, Smiths Medical, Ashford, Kent, UK). Along with an intrathecal route, MNCs were also injected intramuscularly at the specific motor points on the muscles. For this, MNCs were diluted in the patient's own cerebrospinal fluid as it is known to harbor growth factors that help the growth of the cortical epithelium and promote vascularization in the nervous system (11). Simultaneously, the patient was administered methylprednisolone (30 mg/kg over 1 h; Solumedrol; Pfizer, Berlin, Germany) intravenously during the MNC transplantation.
Neurorehabilitation
All patients underwent extensive rehabilitation, which included physiotherapy, occupational therapy, and psychological therapy under the guidance of an expert team of neurorehabilitation specialists over a period of 6 months to 1 year. The exercise protocol was planned out specifically for individual patients as per the detailed assessment done before the therapy.
Statistical Analysis
Statistical analysis was carried out for DMD and LGMD groups using Student's paired t test. Data are expressed as mean ± SD (standard deviation). A significance level of p < 0.005 was used. In the case of BMD, the sample size was too small for the statistical analysis.
Results
One hundred fifty patients diagnosed with muscular dystrophy underwent autologous bone marrow-derived MNC transplantation. These included 125 Duchenne muscular dystrophy cases, 20 LGMD cases, and 5 BMD cases. All patients were administered body weight x 106 MNCs. After the stem cell transplantation, the patients had no major adverse effects. However, some procedure-related adverse effects such as headache, nausea, vomiting, and backache were noticed in a few cases. All these minor adverse effects were self-limiting and were relieved within 1 week.
Along with the stem cell therapy, patients underwent intensive neurorehabilitation consisting of physiotherapy, occupational therapy, and psychological therapy. Neurorehabilitation has been shown to promote recovery and independence through neurofacilitation (4). Changes were documented in the Brooke and Vignos Scales. Quality of life was improved, which was noted by a shift on the FIM scale.
To quantify the improvements observed, a grading system was devised, which graded the improvements as follows:
Deterioration: On reassessment, the patient showed clinical/neurological worsening or death.
No improvement: On reassessment, the patient was the same as before the therapy.
Mild improvement: Patient reported subjective improvements/caregivers reported ease in handling but no demonstrable neurological improvement.
Moderate improvement: Patient showed shift in the Brooke and Vignos Scales.
Significant improvement: Improvement in FIM scale and/or improvement in the radiological (MRI) or electrophysiological (EMG) findings.
Symptoms commonly observed among the cases were abnormal gait, scoliosis, hip knee flexion deformity, Achilles tendon tightness, and talipes equinovarus. On mean follow-up at 12 ± 1 months stem cell administration, 86.67% of cases showed improvement (Fig. 1), with 80 (53.33%) of 150 cases showing improvement in trunk strength and 89 (59.33%) of 150 cases showing improvement in lower limb strength (Fig. 2).
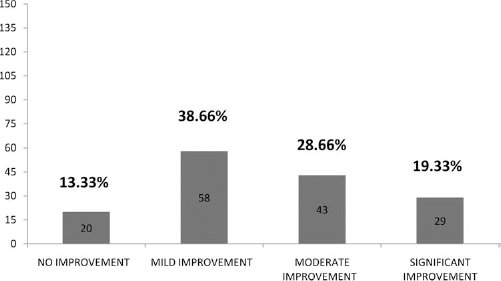
Graph showing improvements in muscular dystrophy patients after stem cell therapy. y-axis = number of patients (n = 150).

Graph showing symptomwise improvements in muscular dystrophy patients after stem cell therapy. Number of patients showing improvements with respect to trunk strength, upper limb (UL) strength, lower limb (LL) strength, gait, and standing are shown. y-axis = number of patients (n = 150).
Among 125 DMD cases, 111 (88.8%) showed improvements (Fig. 3), with 70 cases (56%) showing improvement in trunk strength and lower limb strength and 57 cases (45.6%) showing improvement in upper limb strength (Fig. 4).
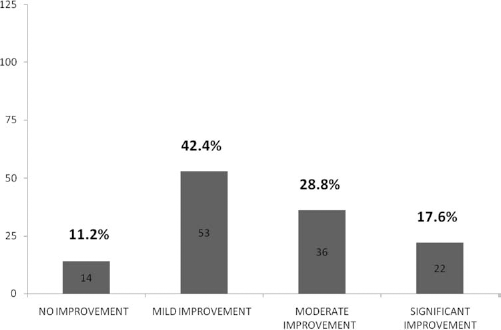
Graph showing improvements in Duchenne muscular dystrophy (DMD) patients after stem cell therapy. y-axis = number of patients (n = 125).

Graph showing symptomwise improvements in DMD patients after stem cell therapy. Number of patients showing improvements with respect to trunk strength, upper limb (UL) strength, lower limb (LL) strength, gait, and standing are shown. y-axis = number of patients (n = 125).
On reassessment, serum CPK was reduced in 85 cases (68%), with 50 cases (40%) shifting on the FIM scale, showing improved independence in activities of daily living. Sixty cases (48%) showed change on the Brooke and Vignos Scales. On comparing the MRI (musculoskeletal system) before the transplantation and on follow-up, 3 cases showed minimal muscle regeneration, generally in the proximal muscles of the limbs, in which MNCs were injected. On comparing the EMG carried out before the transplantation and on follow-up, improvements were recorded in 5 patients in antigravity muscles and also in the muscles that were injected intramuscularly, for example, vastus medialis, tibialis anterior, glutei, deltoid, and others. Interference patterns in the muscles that were injected with MNCs had improved, thereby confirming increase in muscle strength and activity. In addition, the clinical findings were consistent with the electrodiagnostic results, thereby confirming halt in the progression of the disorder and also bringing about improvement in clinical condition of the patients. The statistical data showed significant improvement in upper limb, lower limb, and trunk strength (Table 1). The table reveals that in DMD, the mean pretreatment upper limb strength was 2.11, whereas lower limb and trunk strength was 1.66. After the treatment, the mean upper limb strength showed a significant increase of 15.6%, lower limb strength significantly increased by 27.1%, and trunk strength showed a significant increase of 27.1% (Fig. 5). It was also observed that 44 patients started standing, and 10 patients showed improved gait.

Improvement in DMD patients after stem cell therapy. A statistical analytical graph representing significant improvement (p < 0.05) in DMD patients with respect to upper limb, lower limb, and trunk strength posttherapy. y-axis = statistical mean value of muscle strength before and after stem cell therapy.
Statistical Analysis of the Effect of Stem Cells in Duchenne Muscular Dystrophy (DMD) Using Student's Paired t Test
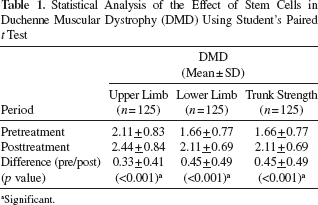
aSignificant.
In LGMD patients, 16 (80%) of 20 cases showed improvement (Fig. 6), with 16 (80%) cases showing improved lower limb strength and 12 (60%) cases showing improved upper limb strength (Fig. 7). Serum CPK reduction was recorded in 10 patients (50%). Improved FIM score was noticed in 13 patients (65%). EMG changes were observed in 4 cases (20%), whereas 3 cases (15%) showed improvement in musculoskeletal MRI (Figs. 8–10). The statistical data showed significant improvement in upper limb, lower limb, and trunk strength. According to the statistical analysis, as shown in Table 2, in LGMD, the mean pretreatment upper limb strength was 2.44, lower limb strength was 1.98, and trunk strength was 1.92. After treatment, mean upper limb strength showed an increase of 18%, lower limb strength showed an increase of 27.3%, and trunk strength showed an increase of 25% (Fig. 11). It was also observed that 7 patients started standing and 4 patients showed improved gait.

Graph showing improvements in limb-girdle muscular dystrophy (LGMD) patients after stem cell therapy. y-axis = number of patients (n = 20).

Graph showing symptomwise improvements in LGMD patients after stem cell therapy. Number of patients showing improvements with respect to trunk strength, upper limb (UL) strength, lower limb (LL) strength, gait, and standing are shown. y-axis = number of patients (n = 20).

Pre- and posttreatment magnetic resonance imaging (MRI) of peroneus longus and brevis muscles. (A) MRI scan of peroneus longus and brevis muscles (arrow) before stem cell therapy. (B) MRI scan of peroneus longus and brevis muscles (arrow) after stem cell therapy showing muscle regeneration.

Pre- and posttreatment MRI of lateral head of gastrocnemius muscle. MRI scan of lateral head of gastrocnemius (arrow) before stem cell therapy. (B) MRI scan of lateral head of gastrocnemius (arrow) after stem cell therapy showing muscle regeneration.
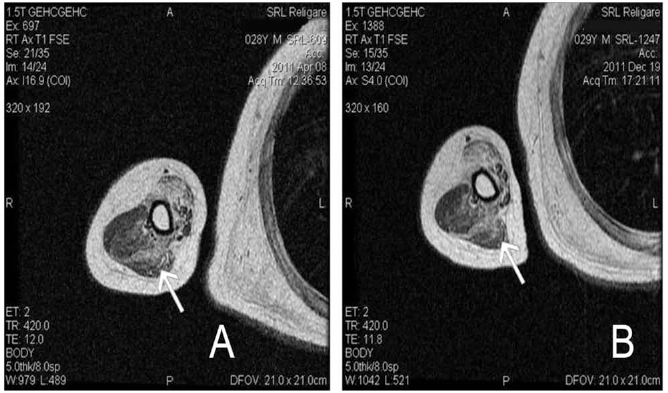
Pre- and posttreatment MRI of long, medial, and lateral head of triceps muscle. MRI scan of long, medial, and lateral head of triceps (arrow) before stem cell therapy. (B) MRI scan of long, medial, and lateral head of triceps (arrow) after stem cell therapy showing muscle regeneration.

Improvement in DMD patients after stem cell therapy. A statistical analytical graph representing significant improvement (p < 0.05) in LGMD patients with respect to upper limb, lower limb, and trunk strength posttherapy. y-axis = statistical mean value of muscle strength before and after stem cell therapy.

Graph showing improvements in Becker muscular dystrophy (BMD) patients after stem cell therapy. y-axis = number of patients (n = 5).

Graph showing symptomwise improvements in BMD patients after stem cell therapy. y-axis = number of patients (n = 5).
Statistical Analysis of the Effect of Stem Cells in Limb-Girdle Muscular Dystrophy (LGMD) Using Student's Paired t Test

aSignificant.
Among 5 BMD cases, 3 (60%) showed improvement with respect to upper and lower limb strength as well as gait (Figs. 12 and 13). Two cases showed changes in FIM score and reduction in serum CPK.
Discussion
No definitive treatment is currently available for control of disease progression or improvement of muscle strength in disorders such as muscular dystrophy. Stem cell transplantation has been proposed as a treatment for such disorders. Bone marrow-derived stem cells are easily obtainable in adult tissue and, with the appropriate microenvironment, are known to differentiate into nonhematopoietic tissues including skeletal muscle, brain, epithelium, and heart after transplantation using fusion and reprogramming (1,12).
Studies have also proved the safety and feasibility of autologous bone marrow-derived MNC transplantation along with their potential to improve the quality of life for patients with neurological disorders (17,24). The objective of our study was to evaluate the clinical effects of autologous bone marrow-derived MNCs administered intrathecally and intramuscularly in muscular dystrophy patients. The MNCs comprise a variety of cells such as hematopoietic stem cells, tissue-specific progenitor cells, stromal cells, and specialized blood cells in different stages of development. These cells have the capacity to mobilize and exert their reparative effects at the site of injury. They are known to enhance angiogenesis and contribute to neovascularization by producing signaling molecules such as vascular endothelial growth factors and fibroblast growth factors (FGF2) (19). Along with an increase in angiogenesis, they also promote tissue remodeling, prevent apoptosis, decrease inflammation, release growth factors, and activate the satellite cells (16).
In an animal study, it has been demonstrated that intramuscular injection of myoblasts into muscular dystrophy (mdx) mice, which lack dystrophin, resulted in fusion with host fibers and extensive dystrophin production (15). It is reported that the brain shows most abundant expression of dystrophin after the muscles. Mutations in the distal part of the dystrophin gene also correlate well with the cognitive impairment in DMD patients (18). Hence, the MNCs were administered intrathecally as well as intramuscularly. MNC transplantation was followed by an individualized intensive rehabilitation program. Experimental studies show that a combination of various therapies such as cellular therapies (stem cells) and exercise (neurorehabilitation and neurofacilitation) together yield a better outcome as opposed to single strategies used independently (21).
In our study, with a mean follow-up of 12 ± 1 months, with evaluation done by the same physiotherapist, 86.67% of cases showed symptomatic and functional improvements. No significant adverse events were noted. The improvements in muscle strength shown by MMT along with gait improvements contributed toward a positive shift on assessment scales such as FIM and the Brooke and Vignos Scales.
At follow-up, a clinical assessment and investigational reports were analyzed to determine whether a second stem cell therapy was required. The patients in whom improvements were seen or deterioration had stalled could have a potential to improve further with the second procedure. However, the response to the second transplantation needs further evaluation.
In conclusion, autologous bone marrow-derived MNC transplantation followed by a comprehensive rehabilitation program is a safe treatment option that produces functional improvements, thereby enhancing quality of life in muscular dystrophy. The limitations of this study are that it is a single-center study with no control group and a mean follow-up of only 1 year. We recommend multiple large multicentric clinical trials for further analysis and to substantiate its efficacy.
Footnotes
Acknowledgment
The authors declare no conflicts of interest.
